Abstract
A 2-year-old gelding presented with a history of lethargy and anorexia. Physical examination revealed pleural and abdominal fluid, as well as several masses in the scrotum. The horse became acutely dyspneic despite 7 days of supportive care. Because of the poor prognosis, the owners elected euthanasia. Gross necropsy findings included multiple masses in the scrotum and inguinal canals and along the dorsal peritoneal cavity. The neoplasm infiltrated the kidneys, liver, spleen, mesenteric lymph nodes, mesentery, and abdominal surface of the diaphragm. Histologically, the neoplasm is composed of spindle to round cells arranged in densely cellular areas, vague streams, and rare rosettes. Neoplastic cells were immunoreactive for S-100 protein, glial fibrillary acidic protein, neuron-specific enolase, neurofilament protein, and synaptophysin. Based on gross, histological, and immunohistochemical findings, a diagnosis of peripheral primitive neuroectodermal tumor was made. Primitive neuroectodermal tumors are rarely described in horses that were associated with the eyes.
Primitive neuroectodermal tumors (PNETs) are small, round-cell tumors of neural crest origin. Such tumors were originally characterized in the central nervous system, but have more recently been described in the periphery. Peripherally located PNETs (pPNETs) are members of the Ewing sarcoma family of tumors. Currently, this family of tumors also includes the intraosseous pPNET (Ewing sarcoma), the extraosseous Ewing sarcoma, and the thoracopulmonary Askin tumor.4,9,17
In human beings, pPNETs are most commonly diagnosed in children in their second decade of life and show a slight male predominance. Soft tissue pPNETs are often found in the chest wall, in paravertebral extradural locations, or in the retroperitoneum or associated with large peripheral nerves. 9 However, there have been several case reports of pPNETs arising in the kidney. 1 Regardless of the point of origin, these tumors are highly aggressive, often quickly metastasizing to the lung and bone. 9 Historically, diagnosis of pPNET was based on histomorphological and ultrastructural findings; however, more recently, immunohistochemical and cytogenetic analysis have become the standard.
A 2-year-old gelding, which was castrated 6 months previously, presented with a history of lethargy and anorexia. Blood work revealed hyperproteinemia, hyperglobulinemia, and hypoalbuminemia. Supportive care was initiated; however, 7 days later, the horse became acutely dyspneic. On examination, pleural and abdominal fluid was noted as well as masses in both cavities of the scrotum. Because of the poor prognosis, the owners elected euthanasia.
Gross necropsy findings included several large lobulated white masses that filled both scrotal cavities (approximately 15 cm) and extended through both inguinal canals and along the dorsal aspect of the peritoneal cavity. The neoplasm infiltrated both kidneys (Fig. 1), with the left kidney being almost entirely obliterated, as well as the liver, spleen, mesenteric lymph nodes, mesentery, and abdominal surface of the diaphragm; however, the gastrointestinal tract was not affected. In addition, masses were noted in the tracheobronchial lymph nodes; however, the lungs, heart, and pleural cavity were not affected. Five liters of serosanginous fluid was present in the peritoneal cavity and 3 liters in the thoracic cavity. The initial differential diagnosis based on gross necropsy findings included lymphoma, mesothelioma, and carcinomatosis.
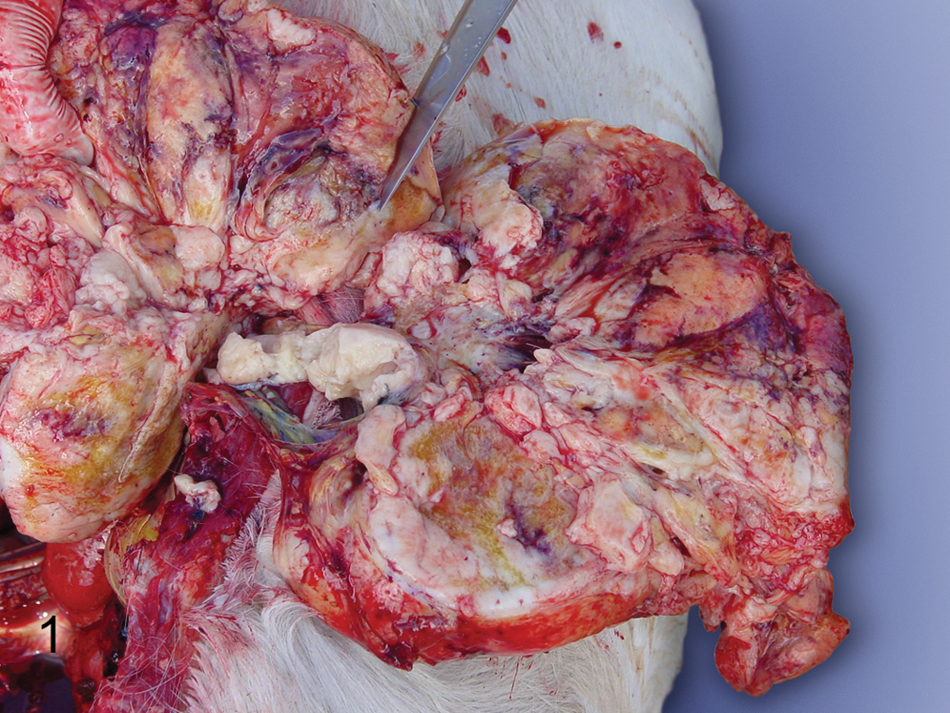
Right kidney; horse. Multiple tan nodules replace renal parenchyma throughout the cortex and extend into the medulla.
Representative tissues samples were fixed in 10% neutral buffered formalin, embedded in paraffin, sectioned at 4–6 μm, and stained with hematoxylin and eosin. Immunohistochemical analysis was performed on selected tissue sections using an autostainer a using protocols with or without antigen retrieval, as summarized in Table 1.
Immunohistochemical antibodies and protocol used in a peripheral primitive neuroectodermal tumor in a horse.*
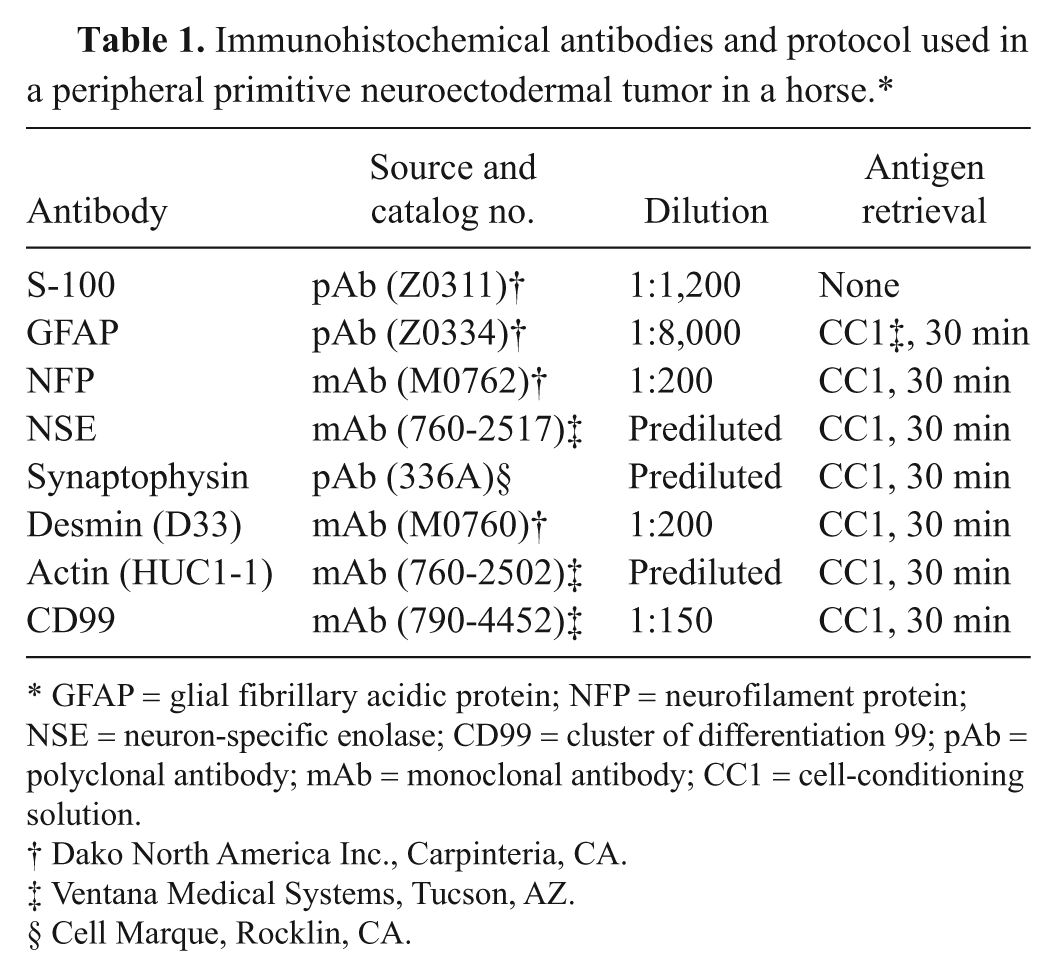
GFAP = glial fibrillary acidic protein; NFP = neurofilament protein; NSE = neuron-specific enolase; CD99 = cluster of differentiation 99; pAb = polyclonal antibody; mAb = monoclonal antibody; CC1 = cell-conditioning solution.
Dako North America Inc., Carpinteria, CA.
Ventana Medical Systems, Tucson, AZ.
Cell Marque, Rocklin, CA.
Histologically, in all locations, the neoplasm is composed of spindle to round cells that are arranged in densely cellular areas and short streams, and rarely form vague rosettes, separated by a fine fibrovascular stroma (Fig. 2). Cells have indistinct cell borders and small amounts of eosinophilic cytoplasm. Nuclei are oval to elongate and rarely triangular (carrot shaped) with moderately stippled chromatin and 1–2 distinct nucleoli. Mitotic figures average 2 per high power field, and there are rare multinucleated neoplastic cells. In addition, there are large areas of necrosis as well as areas of fibrosis. The diagnosis of sarcoma with a differential diagnosis of peripheral nerve sheath tumor, PNET, myofibroblastoma, and fibrosarcoma was made. Immunohistochemistry was performed for further classification.
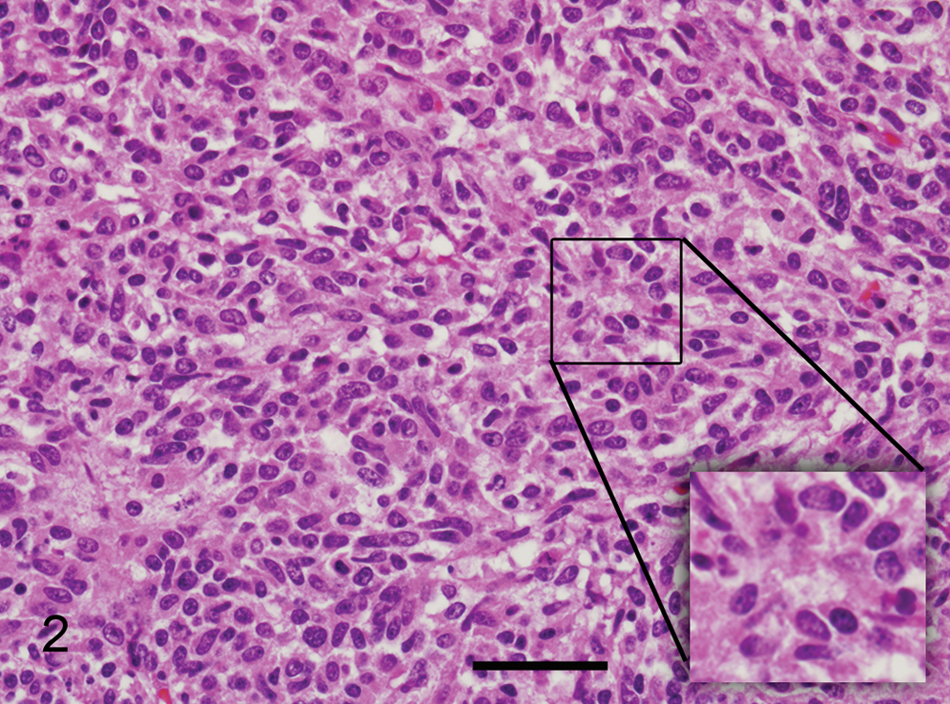
Peripheral primitive neuroectodermal tumor; horse. Densely cellular neoplasm composed of spindle to round cells with indistinct cell borders, scant amount of eosinophilic cytoplasm, with oval to elongated nuclei and finely stippled chromatin. Hematoxylin and eosin. Inset: vague rosette formation. Bar = 44 µm.
Immunohistochemically, the neoplastic cells were positive for synaptophysin (Fig. 3), S-100 protein, glial fibrillary acidic protein (GFAP), neuron-specific enolase (NSE), and neurofilament protein (NFP). Neoplastic cells were negative for muscle-specific actin. Desmin and cluster of differentiation (CD)99 could not be utilized in the diagnosis because of the lack of immunoreactivity. Histomorphological features of the neoplasm and immunohistochemical results were consistent with the diagnosis of pPNET.
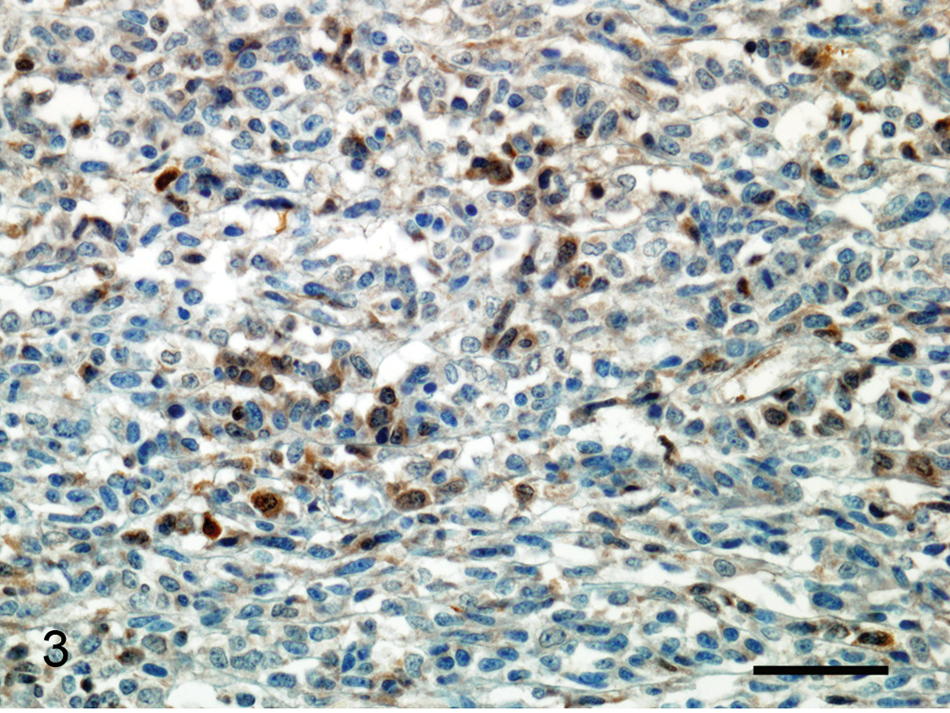
Peripheral primitive neuroectodermal tumor; horse. Multifocal immunoreactivity for synaptophysin in neoplastic cells. Immunohistochemistry, hematoxylin counterstain. Bar = 44 µm.
Abdominal tumors are rare in horses, with lymphoma being the most common; however, there are reports of adenocarcinomas, leiomyomas, leiomyosarcomas, myxosarcomas, ganglioneuomatosis, nerve sheath tumors, and carcinoids in the veterinary literature.6,10,11,14,18 PNETs are uncommonly found in young animals and children. There are 2 cases of PNETs in horses, both of which were intraocular tumors, 1 reported as a retinoblastoma and the other as a neuroepithelial tumor of the optic nerve.2,7
Peripherally located PNETs are members of the Ewing sarcoma family of tumors. In human beings, pPNETs are typically diagnosed based on a combination of histomorphological, immunohistochemical, ultrastructural, and cytogenetic findings. Peripheral PNETs are classically described as solidly cellular neoplasms composed of small round hyperchromic cells that at least occasionally form rosettes. Electron microscopic examination was used more frequently before widespread use of immunohistochemistry, and it is still useful today, especially when immunohistochemical findings are inconclusive. Ultrastructurally, pPNETs often have interdigitating cellular processes with filaments and microtubules, which can help differentiate those cases in which lymphoma is a key differential diagnosis. 4 Electron microscopic features of PNETs have previously been described in horses.2,7 Immunohistochemically, the cells are often positive for 1 or more neural markers, including NSE, S-100 protein, and NFP,4,15 and occasionally GFAP. 15 More recently, CD99 (p30/32), a cell surface glycoprotein product of the MIC2 gene, has been used as a reliable marker for this tumor family in human beings.9,13 Nonetheless, the genetic hallmark is the presence of a specific translocation, t(11;22)(q24;q12), which results in the EWS/FLI1 fusion gene and the expression of a chimeric protein, 17 and occurs in approximately 80% of the cases. 9 A more in-depth study of the expression of CD99 in tissues of various species will help determine its usefulness in veterinary pathology.
PNETs have been reported in the veterinary literature5,8,12,16; however, the majority of those reports more closely resembled the traditional Ewing sarcoma with significant bone involvement. In the present case, it is difficult to determine the point of origin of the tumor; however, no bony involvement was noted on gross examination. The left kidney is most likely the site of origin for this neoplasm, as the tumor almost completely obliterated the kidney and then appeared to spread by extension to the surrounding organs. Regardless of the point of origin, the neoplasm in all tissues examined was densely cellular and composed of undifferentiated spindle to round cells. Immunohistochemically, the cells were positive for more than 1 neural marker and were negative for muscle-specific actin, making a myogenic tumor less likely. Unfortunately, CD99 expression could not be determined because of the lack of immunoreactivity in control and sample tissues, which has also been reported in the dog. 3 Histomorphological and immunohistochemical findings were consistent with a pPNET, and electron microscopy was not pursued.
Footnotes
Acknowledgements
Paul R. Facemire is a Major and Shelley P. Honnold is a Lieutenant Colonel in the U.S. Army. Opinions, interpretations, conclusion, and recommendations are those of the authors and are not necessarily endorsed by the U.S. Army.
a.
Ventana Autostainers (Benchmark or XT platforms), Ventana Medical Systems, Tucson, AZ.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
