Abstract
This report describes the characterization of a Bartonella henselae abortion in an equine fetus by gross, histologic, immunohistochemical, ultrastructural, and molecular methods. Bartonella henselae can cause cat scratch disease, bacillary angiomatosis, bacillary peliosis, and endocarditis in humans and other animals. The bacterium has been isolated from several mammalian species but only recently from equids; however, it has not been linked to abortion in equids. An aborted equine fetus exhibited necrosis and vasculitis in multiple tissues, with intralesional Gram-negative short-to-spirillar bacteria. Nucleotide sequence analysis of the bacterial 16S rRNA gene amplified from the DNA extracted from fetal tissues revealed 99.9% homology to that of B. henselae. The presence of B. henselae in the fetal tissues was further confirmed by polymerase chain reaction amplification and nucleotide sequence analysis of other Bartonella species-specific genes. Microorganisms were immunohistochemically labeled with a monoclonal antibody to B. henselae and were ultrastructurally characterized. Attempts to detect known causative agents of equine abortion were unsuccessful. Given the severity of vasculitis and the presence of intralesional bacteria, we concluded that B. henselae infection caused the abortion of this foal.
Keywords
An aborted female Quarterhorse fetus of approximately 240–270 days' gestation was submitted for necropsy. The aborting mare had no history of clinical signs or prior abortions. Seven other mares on the farm had carried foals to term and exhibited no clinical signs. The portions of placental membrane available for evaluation were grossly unremarkable. On gross examination of the fetus, there were multiple, tan, 1–3 mm in diameter foci scattered randomly throughout the liver (Fig. 1), kidneys (Fig. 2), lungs, and adrenal glands. On histologic examination, these foci corresponded to areas of necrosis and vasculitis. Subacute-to-chronic vasculitis was characterized by expansion of the tunica media and adventitia of small-to-medium caliber arteries and arterioles by leukocytes and loose connective tissue. The inflammatory infiltrate associated with vessels and areas of necrosis was composed of a mixture of lymphocytes, macrophages, and neutrophils. Similar lesions were also apparent in the placenta (chorioallantoic vasculitis), spleen, ovary, brain (gray and white matter of the cerebrum and cerebellum), heart, and pancreas. Colonies of Gram-negative bacteria were present within necrotic areas and vessel lumens of all tissues examined. These bacteria were short or spirillar rods, up to 5-µm long, in Warthin-Starry silver stained sections (Figs. 3 and 4). In addition, hepatocytes varied in size and formed irregular hepatic cords. Many hepatocytes were large and contained multiple, up to 10, nuclei. This morphologic appearance was compatible with that reported for giant-cell hepatopathy. 4, 18
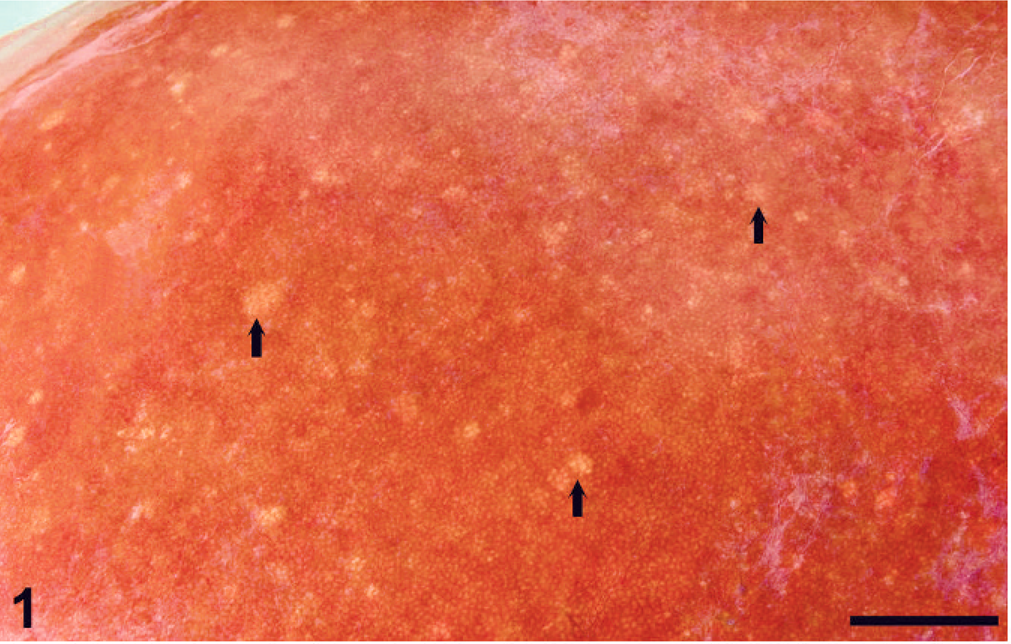
Liver; equine fetus. Multifocal necrosis (arrows) randomly distributed throughout the hepatic parenchyma. Bar = 1.0 cm.

Kidney; equine fetus. Multiple, pale foci (arrows) are distributed throughout the renal cortex. These foci correspond histologically to necrosis and vasculitis, with intralesional bacteria. Bar = 1.0 cm.
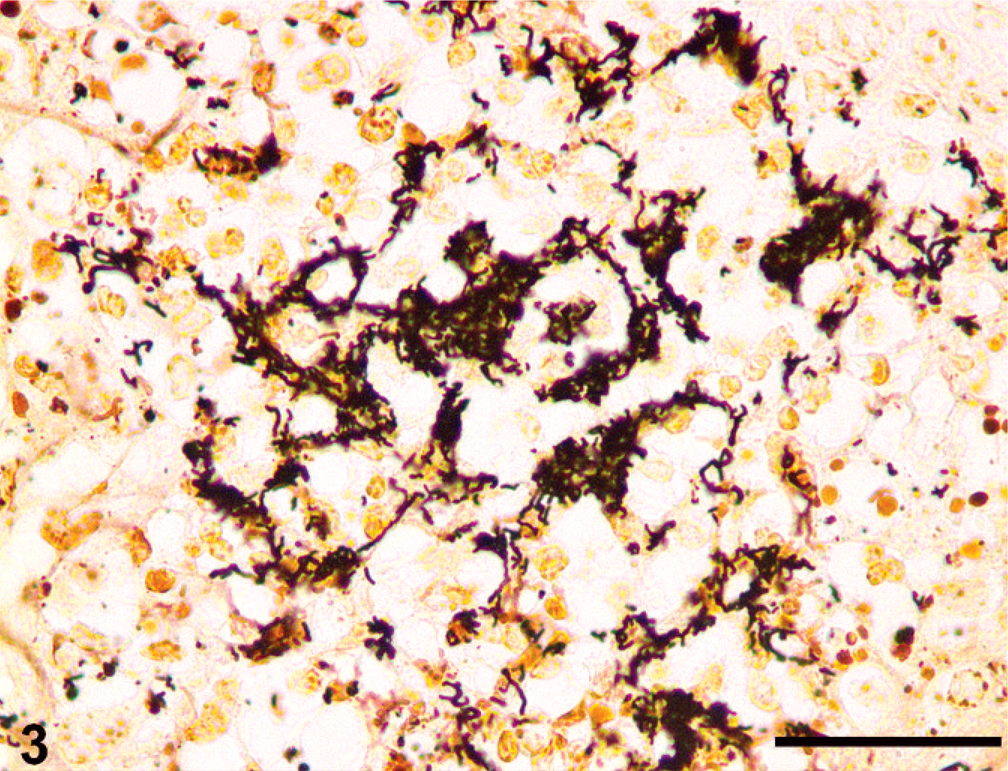
Lung; equine fetus. Dense colonies of bacteria are present within an area of necrosis. Bacteria vary from short to long, wavy rods. Warthin-Starry silver stain. Bar = 125 µm.
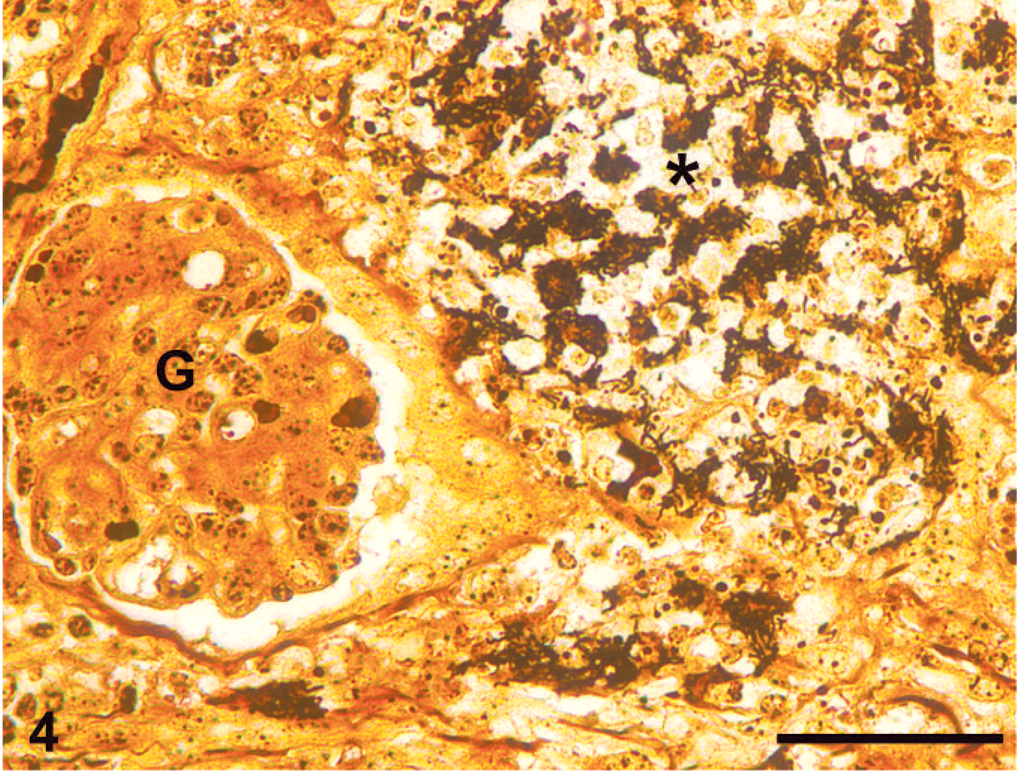
Kidney; equine fetus. Demonstration of bacteria (∗) corresponding to anti-B. henselae labeling in a sequential section (Fig. 5a). Warthin-Starry silver stain. Bar = 50 µm. Glomerulus (G).
Differential diagnoses for the gross and histologic lesions included Actinobacillus equuli, leptospirosis, borreliosis, salmonellosis, Actinomyces sp., equine herpesvirus-1, and equine viral arteritis virus. 3, 4, 17 Fluorescent antibody testing did not identify herpesvirus or Leptospira spp. antigen. Virus isolation was negative for herpesvirus and equine arteritis virus. There was no growth of bacteria on aerobic cultures of fresh and frozen tissues, including specific conditions for Salmonella sp. and Heamophilus/Actinobacillus spp.
DNA from frozen lung, kidney, and spleen samples of the foal was extracted by using a commercial kit (DNeasy Tissue Kit, Qiagen, Valencia, CA). The extracted DNA was used as a template in various polymerase chain reaction (PCR) assays to ascertain the identity of the bacterial agent present in these tissues (Table 1). When primers specific for amplifying the conserved 16S rRNA gene sequences from all eubacteria were used, successful amplification of the expected-size DNA fragments were obtained with both partial and full-length gene primer-pairs. The amplified fragments were cloned into pGEM-T vector, and both strands of inserts in 4–6 independent clones were sequenced, and the nucleotide sequences were compared with those available in the databases at the National Center for Biotechnology Information (http://www.ncbi.nlm.nih.gov) by using BLAST programs. The amplified products were 99.9% identical to the corresponding sequences of the 16S rRNA gene of Bartonella henselae. To further confirm the presence of B. henselae, PCR was performed with primers specific to the pap31 gene and the 16S–23S intergenic transcribed spacer (ITS) region. In both cases, there was specific amplification of expected-size DNA fragments. Nucleotide sequence analysis of the ITS and pap31 amplified products showed 100% and 99.8% homology, respectively, with the corresponding sequences of B. henselae. All the tissues tested negative for pathogenic Leptospira species by a previously described real-time PCR assay. 14 No amplification of Borrelia-specific DNA was obtained from any of the tissues tested. 7
Summary of genomic analyses used to diagnose Bartonella henselae.

Forward primer.
Ref. 9.
Reverse primer.
Ref. 11.
Ref. 13.
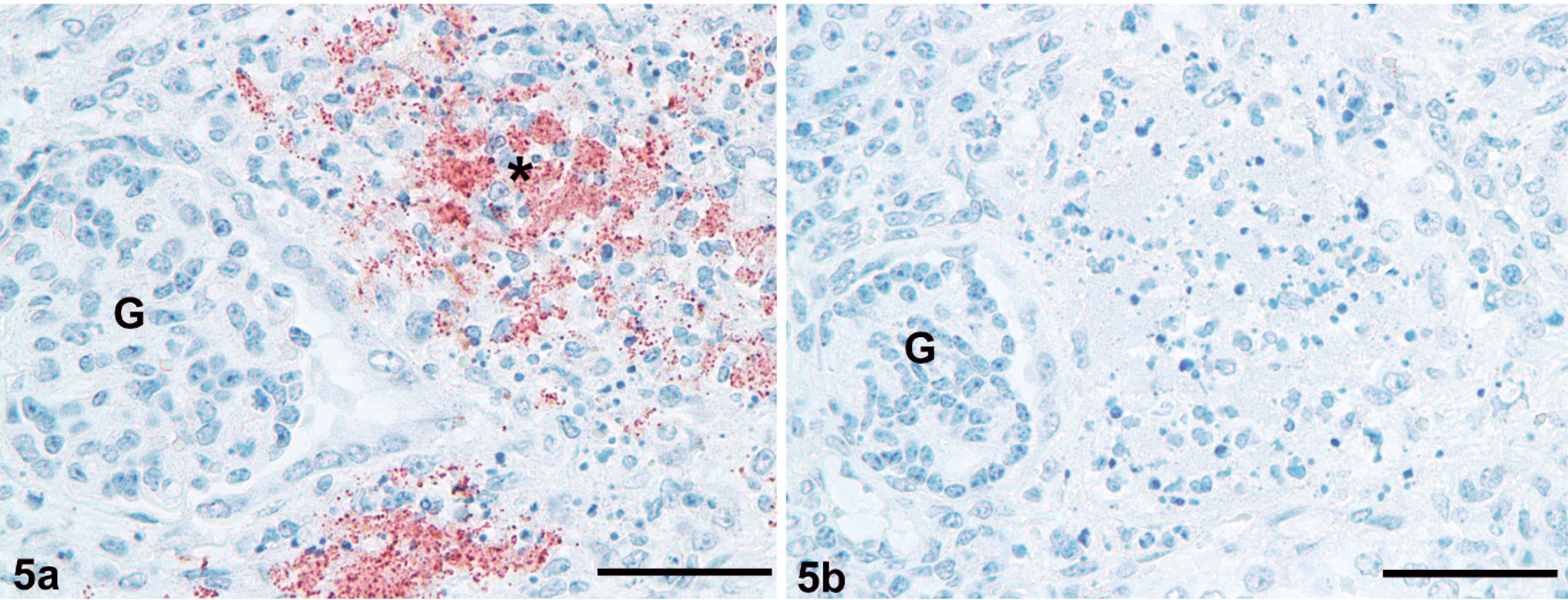
Sections of kidney were labeled with mouse monoclonal anti-B. henselae antibodies (Abcam Inc, Cambridge, MA). There was strong labeling of bacteria in the kidney (Fig. 5a). Commercially available B. henselae–inoculated rat lung (Newcomer Supply, Middleton, WI) served as a positive control. Sections of kidney were also positively labeled by rabbit polyclonal anti–Borrelia burgdorferi antibody (Biodesign, Saco, ME). This labeling was confirmed as a false positive by demonstrating cross-reactivity of the polyclonal anti–Borrelia burgdorferi antibody with the previously described B. henselae control.
Transmission electron microscopy of kidney revealed numerous aggregates of wavy or rod-shaped bacilli approximately 0.3 × 2.1 µm (Fig. 6). These organisms had a tri-laminar cell wall characteristic of Gram-negative bacteria. There were few electron-dense clusters within the cytoplasm and occasional, centrally located, intracellular, linear, rod-like structures. These rod-like structures were of mixed electron density, appeared to have longitudinal striations, and did not follow the contour of the organisms. Bacteria did not exhibit a regular spiral wavelength or periplasmic flagella, further ruling out spirochetes such as Leptospira sp. or Borrelia sp.
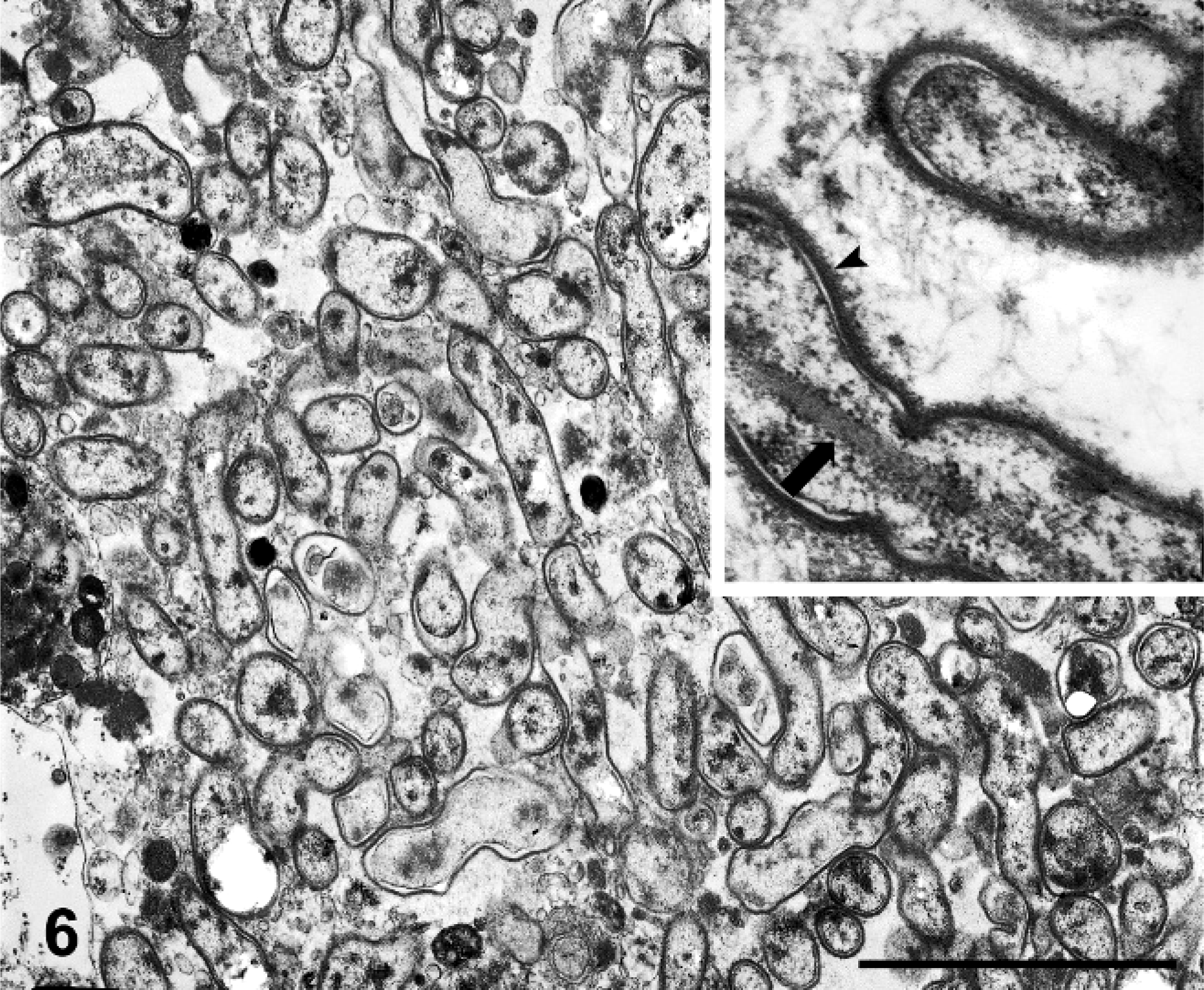
Kidney, equine fetus. Wavy bacilli. Inset: Organisms have a tri-laminar cell wall (arrow head) typical of Gram-negative bacteria, and they lack a periodic wavelength or periplasmic flagella. Many bacilli contain a linear, striated, intracellular rod (arrow). Transmission electron microscopy. Bar = 1 µm.
The presence of bacteria, associated with inflammation, in many tissues indicates that these organisms were the likely cause of abortion of this foal. PCR amplification and nucleotide sequence analysis of 3 different target DNA sequences from multiple tissues provided compelling evidence for the presence of genomic material of B. henselae in the tissues of this aborted fetus. B. henselae has only recently been detected in the blood of horses, but our report represents the first documentation of B. henselae causing equine abortion. 10 Although the morphology is not typical for B. henselae, the immunohistochemical labeling with B. henselae antibody correlates with observed organisms. The ultrastructural morphology is divergent from the reported 1-µm bacillus form of B. henselae. 1 Variation of the ultrastructural morphology of the B. henselae, however, was documented in the literature. 6, 16 After multiple passages of the organism through mice, B. henselae had increasing irregular contours and decreased fimbriae. 16 In an in vitro cell culture with human umbilical vein endothelial cells, B. henselae had a wavy morphology, with similar dimensions (0.25 µm × 2 µm) as the organism observed in this case report. 6 Interestingly, both of these reports suggested that alterations in morphology may be associated with increased pathogenicity. The identity and significance of the intracellular, rod-like structure observed ultrastructurally is unknown. The structure may represent a component of the bacterial cytoskeleton. In the absence of a previous description of a similar finding in B. henselae, it is possible that the structure is an artifactual change, such as polymerization of intracellular genomic material or protein. 12
We found no reports of abortion caused by B. henselae in any species. Transplacental transmission, increased fetal death, and placental vasculitis were documented in mice infected with Bartonella birtlesii, but a similar phenomenon has not been observed with B. henselae. 2 Experimental infection with B. henselae produces reproductive failure in cats, characterized by a decreased conception rate and increased time to become pregnant; however, perinatal transmission could not be demonstrated. 8 The molecular studies performed clearly differentiated B. henselae from other Bartonella sp., thus the observation of fetal colonization is a novel finding for B. henselae.
It is unclear if the angiocentric lesions observed in this fetus are simply a function of intravascular dissemination of the organism or if this pattern draws parallels to the unique interaction of B. henselae with vascular endothelial cells in human bacillary angiomatosis and bacillary peliosis. 6 Detection of B. henselae in horses has been associated with purpura hemorrhagica and arthropathy, but a causal link was not established. 10 The vasculitis observed in this fetus may lend support for a role of B. henselae in other vasculitis-associated diseases of the horse, such as purpura hemorrhagica.
Giant-cell hepatopathy has been associated with leptospirosis in horses, but a similar human condition is considered a nonspecific response to fetal hepatic injury of a variety of causes. 5, 18 Leptospirosis was ruled out in this fetus by multiple methods, thus, disputing a causative relationship between equine giant-cell hepatopathy and Leptospira sp. infection.
Attempts to culture B. henselae from frozen tissue, by using chocolate agar and horse blood agar, was unsuccessful. This is most likely because of poor bacterial survival after freezing and the prolonged time period between collection of necropsy specimens and culture attempts. The aborting mare in this case was lost to follow-up before appropriate samples could be collected for serologic evaluation and bacterial culture. This information may have been helpful in elucidating the pathogenesis of infection. Clinical infections with B. henselae in humans are most commonly found in immunosuppressed patients with HIV-AIDS. 1 It is possible that pregnancy-associated immunosuppression of the mare could have increased susceptibility to fetal infection. 15
In summary, based on histologic, immunohistochemical, and molecular diagnostic test results, we concluded that the examined fetus was infected with B. henselae. Bartonella infection has not been reported previously in horses and should be added to the list of possible causes of equine abortion.
Footnotes
Acknowledgements
Dr. Johnson was supported by a fellowship from Pfizer organized by the American College of Veterinary Pathologists and Society of Toxicologic Pathology Coalition for Veterinary Pathology Fellows. The authors appreciate the help and advice provided by Dr. Wu (Purdue University Animal Disease Diagnostic Laboratory) and Dr. Guptill (Purdue University, Department of Veterinary Clinical Sciences). Many thanks to the Minnesota Veterinary Diagnostic Laboratory for performing the Bartonella and Borrelia immunohistochemistry, and to the molecular diagnostic laboratory and histology laboratory of the Purdue University Animal Disease Diagnostic Laboratory.
