Abstract
A striped skunk with neurological signs was euthanized and examined via necropsy. Histologically, protozoa were found in multiple tissues. Protozoal schizonts measured 15 to 25 μm in diameter and contained 4 to 6 μm crescent-shaped merozoites. Protozoa were associated with necrosis and inflammation in the lung, brain, liver, and nasal epithelium. Immunohistochemistry labeled protozoa strongly positive for Sarcocystis neurona. Polymerase chain reaction–amplified products from the protozoan were 99.6% identical to the corresponding portion of the nuclear small subunit ribosomal RNA gene of S neurona. S neurona origin was further confirmed by amplifying a 451–base pair DNA fragment from the skunk lung, which differed by just 2 or 3 base pairs from the small subunit ribosomal RNA gene of S neurona. Striped skunks act as intermediate and aberrant hosts for S neurona; however, S neurona has rarely been found in extraneural tissues in any species, and systemic sarcocystosis has not been reported in skunks. Additionally, canine distemper virus infection was confirmed with histopathology and immunohistochemistry. Concurrent canine distemper suggests that immunosuppression may have played a role in S neurona infection in this skunk.
Keywords
A juvenile male striped skunk (Mephitis mephitis) was submitted to the Animal Disease Diagnostic Laboratory for necropsy. The skunk was euthanized by the submitter because of severe neurological signs, including immobility, aggression, and head tremors. Grossly, the skunk’s lip margins and periocular tissues were thickened and hyperkeratotic. The carcass was dehydrated, and multiple nematodes, consistent with Physaloptera sp, were in the stomach.
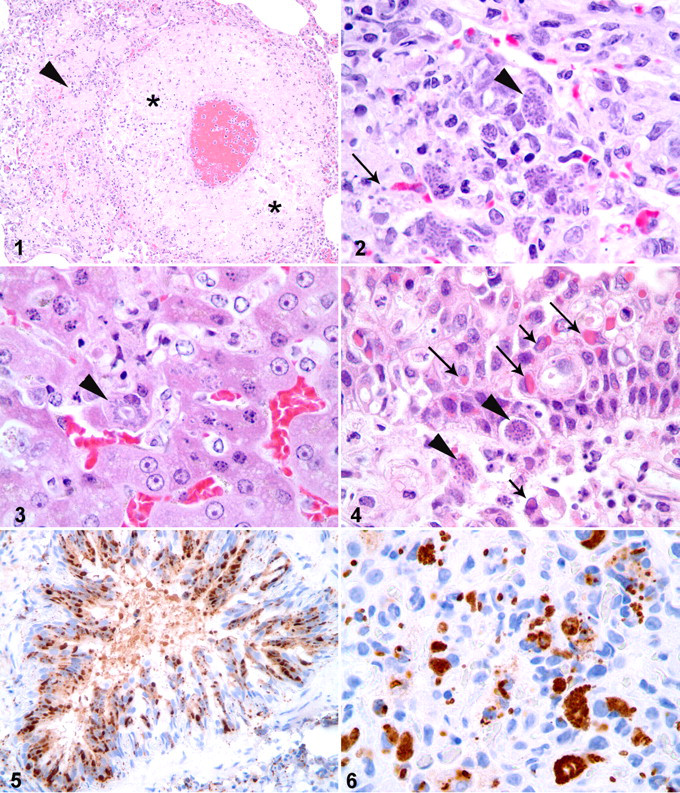
Histologically, a few pulmonary arteries throughout the lung had necrotic vessel walls, with loss of smooth myocytes and scattered cellular debris (Fig. 1 ). Surrounding pulmonary parenchyma was infiltrated by lymphocytes, histiocytes, and a few eosinophils. Several intracellular protozoal schizonts were within foci of vascular wall necrosis and the surrounding pulmonary parenchyma (Fig. 2 ). Schizonts measured 15 to 25 μm in diameter and contained 4 to 6 μm oval- to crescent-shaped merozoites. Rare schizonts contained merozoites arranged in a rosette-like pattern around a central clearing, indicative of endopolygeny. Similar protozoal schizonts and individual merozoites were associated with foci of hepatic necrosis (Fig. 3 ), areas of lymphocytic encephalitis, and foci of lymphohistiocytic rhinitis within the propria submucosa of the nasal cavity (Fig. 4 ). Rare protozoal schizonts were in the splenic parenchyma but were not associated with foci of necrosis or inflammation. Additionally, alveolar septa throughout the lungs were multifocally thickened by lymphocytes and histiocytes. Few bronchial and bronchiolar epithelial cells contained round, eosinophilic, cytoplasmic and intranuclear inclusions that measured from 4 to 7 μm in diameter. Similar inclusions were found in bile ductular epithelium, gastric epithelium, and nasal epithelium (Fig. 4).
Polymerase chain reaction amplification of apicomplexan-specific DNA sequences from the skunk lung. Numbers at left indicate the molecular size of the DNA marker bands in base pairs.
Polymerase chain reaction amplification of Sarcocystis neurona–specific DNA sequences from the skunk lung. Numbers at right indicate the molecular size of the DNA marker bands in base pairs.
Histologic lesions in this skunk—including interstitial pneumonia and multiple tissues with intraepithelial eosinophilic inclusion bodies—were consistent with canine distemper virus infection. Immunohistochemistry (IHC) was performed on sections of formalin-fixed, paraffin-embedded lung with a mouse monoclonal antibody to canine distemper virus (CMI, Sacramento, Calif), which was strongly positive in cells with either cytoplasmic or nuclear inclusions, as well as in some epithelial cells with no distinct inclusions (Fig. 5 ). Canine distemper virus causes immunosuppression in infected hosts, and secondary infections with opportunistic pathogens are possible. 2 Given that Toxoplasma gondii is an important opportunistic pathogen in immunosuppressed animals and humans, systemic toxoplasmosis was considered a likely concurrent infection in this skunk. However, protozoal schizonts in sections of lung exhibited endopolygeny, which is a histologic feature consistently attributed to Sarcocystis sp rather than T gondii. 7 IHC was performed on sections of formalin-fixed, paraffin-embedded lung with a mouse monoclonal antibody to T gondii (Millipore, Billerica, MA) and with a rabbit polyclonal antibody to Sarcocystis neurona (Dr Lindsay, Blacksburg, VA). Protozoa were weakly positive for T gondii and strongly positive for S neurona (Fig. 6 ).
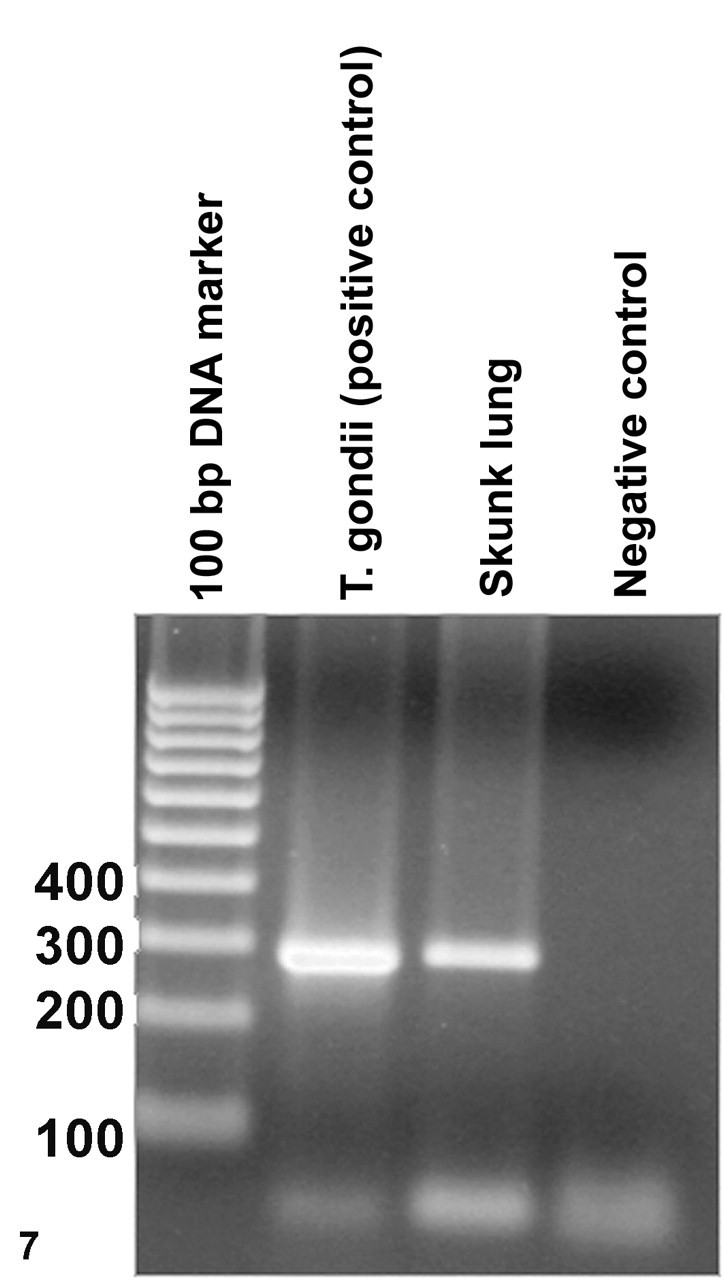
To further confirm the identity of the protozoal agent, polymerase chain reaction–based (PCR-based) amplification of specific DNA was followed by subsequent nucleotide sequence analysis of the amplified products. DNA from five 10-μm-thick sections of formalin-fixed, paraffin-embedded lung was extracted with a commercial kit (DNeasy Tissue Kit, Qiagen, Valencia, Calif) according to the manufacturer’s suggested procedure. DNA similarly extracted from tissue sections of a goat lung, confirmed to contain T gondii, was used as a positive coccidian control. A previously described PCR assay was performed on the extracted DNA to amplify a portion of the gene encoding nuclear small-subunit ribosomal RNA (nss-rRNA) from all coccidian parasites. 9 The PCR was performed in a total volume of 25 μl, with 5 μl of the extracted DNA as template and primers COC-1 (5′-AAGTATAAGCTTTTATACGGCT-3′) and COC-2 (5′-CACTGCCACGGTAGTCCAATAC-3′) according to published procedures. 9 Electrophoretic analysis of the amplified products on a 1.5% agarose gel revealed the amplification of an approximately 300–base pair (300-bp) DNA fragment from both the lung of the skunk and the positive T gondii control; the PCR product from the skunk tissues was slightly larger in size than that from the T gondii control (Fig. 7 ). The amplified DNA fragment from the skunk sample was cloned into pGEM-T vector (Promega, Madison, Wisc), and 2 separate clones were used to determine the nucleotide sequences of the cloned DNA fragments. Both cloned DNA fragments had identical nucleotide sequences of 299 bps. A BLAST search of all available databases at the National Center for Biotechnology Information (http://www.ncbi.nlm.nih.gov) revealed that the amplified products were 99.6% identical to the corresponding portion of the nss-rRNA gene of Sarcocystis falcatula strain Stiles (accession No. AY628220) and S neurona strain EPMUCD1 (accession No. U33149) and S neurona isolates from an opossum (accession No. AY628219), a harbor seal (accession No. AF252406), and a sea otter (accession No. AY009112). The other closely related Sarcocystis spp were Sarcocystis felis (AY190080) and Sarcocystis lacertae (AY015113) with 99.2% sequence identity. In comparison to the PCR product from the skunk tissue, the corresponding portion of the nss-rRNA gene of T gondii was 6 bps shorter and had only 93.7% sequence identity. Because Sarcocystis falcatula is primarily associated with avian species, the agent in the skunk tissues was considered to be S neurona or a S neurona–like parasite. 7
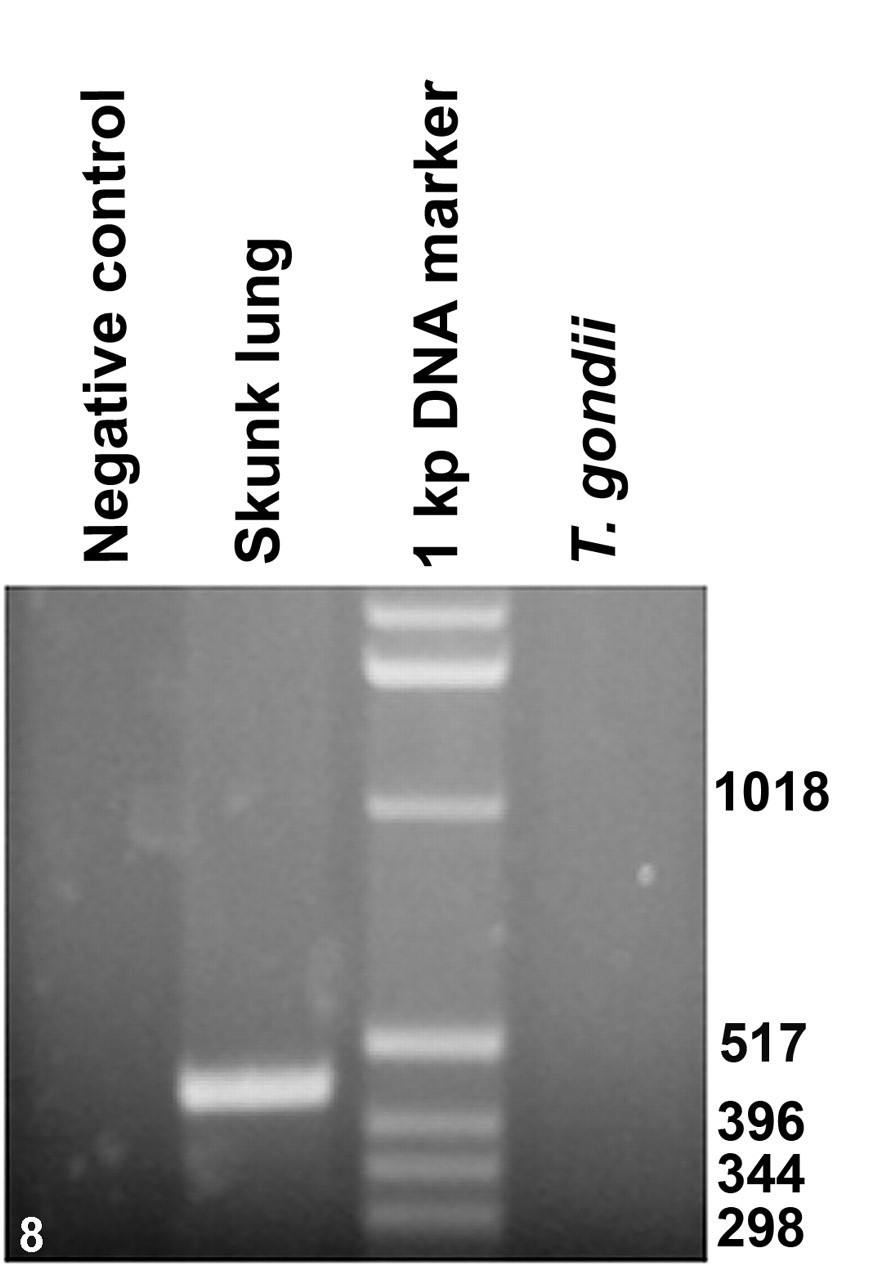
To further confirm the parasite identity, a recently described PCR assay was performed that can specifically amplify the small subunit ribosomal RNA gene sequences of S neurona but not S falcatula. 10 The PCR assay successfully amplified a 451-bp DNA fragment from the DNA extracted from the skunk lung tissue (Fig. 8 ). Nucleotide sequence analysis of the amplified fragment revealed that it differed just by 2 or 3 nucleotides from the S neurona small subunit ribosomal RNA gene sequences available at the GenBank databases (accession Nos. AY553981, AY518209, AY518210, AY518211, U07812).
Striped skunks serve as intermediate hosts for S neurona and develop sarcocysts within their skeletal muscles after oral inoculation with opossum-shed sporocysts. 3 Previous reports of skunks with encephalitis caused by protozoa morphologically similar to S neurona or positive for S neurona via IHC suggest that skunks can act as aberrant hosts for this parasite. 5,6 Thus, protozoal encephalitis in skunks has been associated with S neurona. However, systemic infection caused by S neurona or a S neurona–like protozoan has not been previously reported in skunks. Few reports have documented S neurona as a cause of disease in extraneural tissues in any species; however, S neurona–associated myocarditis has been documented in a raccoon, and S neurona–attributed pneumonia and hepatitis have been reported in a dog. 4,7,8 The current case demonstrates S neurona as a cause of histologic lesions in the brain, lung, liver, and nasal propria submucosa, confirming systemic infection. Systemic sarcocystosis has not been reported in the skunk.
Concurrent infection with canine distemper virus suggests that immunosuppression may have played a role in the development of systemic sarcocystosis in this skunk. Canine distemper causes immunosuppression in infected hosts, allowing concurrent infections with a range of opportunistic infectious agents, including protozoa. 2 The protozoan most commonly associated with immunosuppression in animals and humans is T gondii; however, infection with Sarcocystis sp has also been observed. Pulmonary sarcocystosis, suspected to be caused by Sarcocystis canis, was reported in a puppy also infected with canine distemper virus. 1 Encephalitis associated with Sarcocystis sp in a raccoon coinfected with canine distemper virus has also been documented. 12 Thus, S neurona may behave similarly to other Sarcocystis spp during episodes of immunosuppression. Research using Arabian horses with severe combined immunodeficiency found that such horses also infected with S neurona developed prolonged parasitemia and had dissemination of the protozoa to visceral tissues but did not develop disease associated with S neurona. 11 Horses deemed immunocompetent were also challenged with oral or intravenous S neurona. Immunocompetent horses did not develop parasitemia, and 4 of 6 immunocompetent horses developed neurologic disease associated with S neurona. 11 Thus, initial work suggests that immunocompetency is required for manifestation of the disease in horses. The current case suggests that immunosuppression contributed to disease associated with disseminated sarcocystosis in a skunk; thus, the pathogenesis of S neurona infection, as related to host immune status, may vary between equids and other species.
In summary, this is the first report of systemic S neurona infection in a skunk, as confirmed by IHC and PCR methods. Concurrent canine distemper was identified and may have contributed via immunosuppression to the severe systemic protozoal infection that led to the animal’s illness.
Footnotes
Acknowledgements
We would like to thank Joe Caudell and the Indiana Department of Natural Resources for submission of the case, Dee DuSold for technical assistance with immunohistochemistry, and Cecilia Santrich for technical assistance with polymerase chain reaction.
The authors declared that they had no conflicts of interests with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
