Abstract
Three bald eagles (Haliaeetus leucocephalus) and 1 golden eagle (Aquila chrysaetos) were admitted to rehabilitation facilities with emaciation, lethargy, and an inability to fly. Intravascular schizonts and merozoites were present in 2 bald eagles, mainly in the lung tissue, whereas the third bald eagle and the golden eagle had lymphohistiocytic encephalitis with intralesional schizonts and merozoites. In all eagles, protozoal tissue cysts were present in skeletal musculature or heart. The protozoal organisms were morphologically compatible with a Sarcocystis sp. By immunohistochemistry, the protozoal merozoites were positive for Sarcocystis falcatula antigen in all cases when using polyclonal antisera. Furthermore, the protozoa were confirmed to be most similar to S. falcatula by polymerase chain reaction in 3 of the 4 cases. To the authors' knowledge, this report presents the first cases of natural infection in eagles with S. falcatula as a cause of mortality.
Sarcocystis falcatula is a protozoan that inhabits the intestine of the Virginia opossum (Didelphis virginiana), which is its definitive host in North America. Thus far, members of 4 orders of birds, namely Psittaciformes, Passeriformes, Columbiformes, and most recently Strigiformes have been recognized as intermediate hosts for this parasite. 3,4,6,11,22,24,26 Neuronal infection by Sarcocystis organisms usually is attributed to Sarcocystis neurona, another intestinal sarcocyst of the Virginia opossum. However, S. falcatula also is capable of infecting neurons in birds. 26 This study documents, for the first time, that golden eagles and bald eagles (order Falconiformes) may serve as intermediate hosts for S. falcatula and that S. falcatula can cause clinical disease and death in these species from encephalitis with neuronal infection or from widespread pulmonary endothelial cell infection.
Case 1 involved an adult female golden eagle (Aquila chrysaetos). Cases 2, 3, and 4 involved an adult female bald eagle (Haliaeetus leucocephalus), an adult male bald eagle, and a second-year male bald eagle, respectively. The birds were unable to fly and were admitted to rehabilitation facilities in Virginia, Indiana, and Minnesota. The golden eagle and 2 bald eagles (cases 1, 2, and 3) were hospitalized for 3–4 days before they died despite supportive care. One bald eagle (case 4) was euthanized at admission because of an inoperable wound with metallic projectile fragments exposing the joint surfaces of the tarso-metatarsal joint despite being bright, alert, and responsive. All hospitalized birds were emaciated, dehydrated, anemic, and hypoproteinemic (Table 1). The golden eagle exhibited miotic pupils, and consensual reflexes were absent. Placing deficits were present in both limbs, and grasp reflexes were markedly weakened bilaterally. Toe pinch reflex and wing reflexes were diminished. The eagle had intention tremors when attempting to apprehend food. The bird exhibited labored breathing and died. One bald eagle (case 2) was hock sitting on initial presentation, with a mild bilateral wing droop and mild ventroflexion of the neck. Within 48 hr of admission, the patient began exhibiting a normal stance but became dyspneic and would lie in ventral recumbency after handling for supportive care. At 96 hr after admission, the patient was found nonresponsive to stimulus and lying in ventral recumbency with extended head and wings, severe dyspnea, and open-mouth breathing. The patient subsequently died. One bald eagle (case 3) exhibited generalized weakness and lethargy on admission. Twelve hours after admission, the patient remained weak, resting in sternal recumbency, with moderate dyspnea. Thirty-six hours after admission, the patient remained weak and was standing with the neck in ventroflexion. Sixty hours after admission, the patient was found in ventral recumbency and unable to lift its head, and the eagle died shortly thereafter. Complete blood cell counts revealed leukocytosis with heterophilia in the 3 hospitalized birds and lymphocytosis in both hospitalized bald eagles (Table 1).
Whole-body radiographs of the golden eagle (case 1) revealed numerous metallic projectiles in the subcutaneous tissue, near 1 kidney, and embedded in the atlas (C1). The bird had a right ulnar fracture with callus formation and several metallic fragments approximately 5 cm distal to the elbow. Radiographic findings in a bald eagle (case 3) included a fracture of the left coracoid immediately lateral to the articulation, with the sternal coracoidal facet and a small metallic fleck in the soft tissue of the caudoventral abdomen.
Body weight, hematocrit, total protein, and blood cell count in cases 1–3 with reference values for golden eagles (Aquila chrysaetos) and bald eagles (Haliaeetus leucocephalus). *
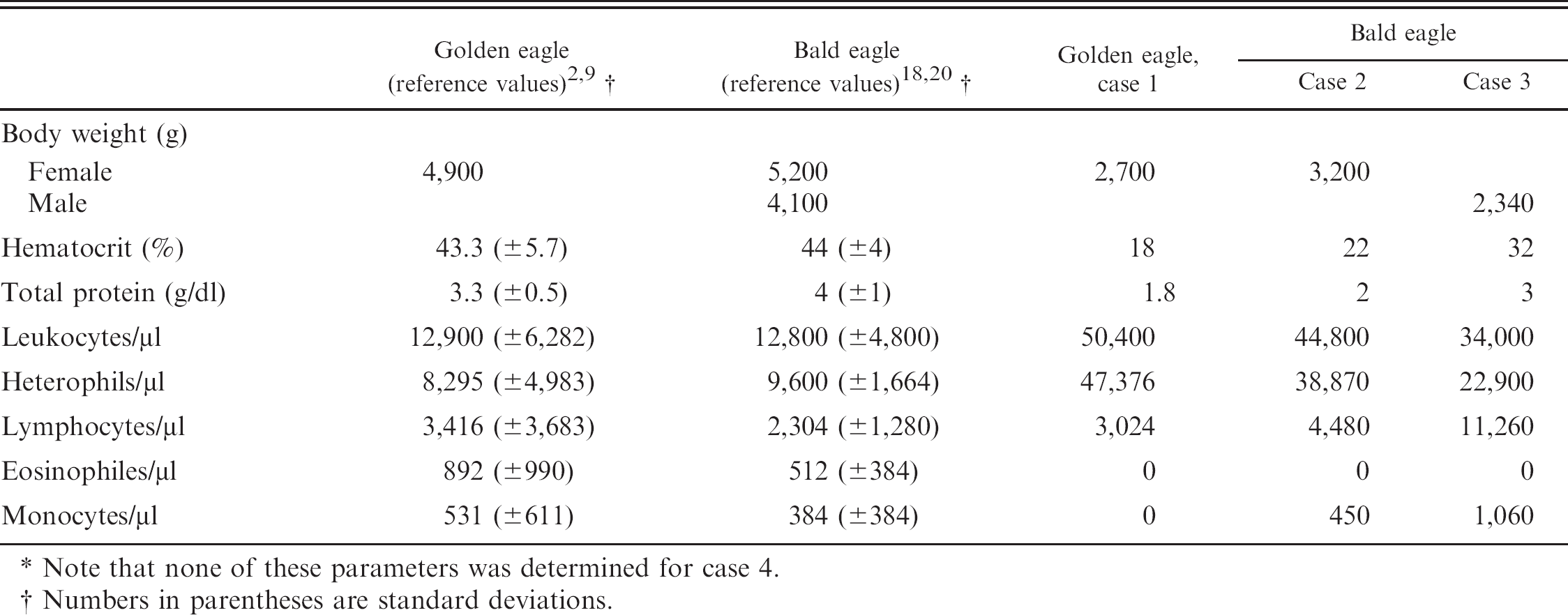
Note that none of these parameters was determined for case 4.
Numbers in parentheses are standard deviations.
The blood-lead level was below the detection level in the golden eagle. Blood-lead levels were mildly to moderately elevated at 0.35 ppm and 0.25 ppm in 2 cases (cases 2 and 4), consistent with subclinical cases of lead intoxication, and was greater than 0.65 ppm in the third bald eagle (case 3), consistent with a case of lead toxicity.
Necropsy confirmed emaciation in all the animals and confirmed the presence of the reported fractures in cases 1, 3, and 4. In addition, the lungs of 2 bald eagles (cases 2 and 3) had areas of dark-red discoloration. The pericardial sacs of these 2 birds contained approximately 2 ml of a clear, amber-colored fluid. Heart, liver, spleen, kidney, brain (cases 1, 2, 4), lung (cases 1–3), and skeletal muscle (cases 1 and 4) were collected for histological examination.
Parasite-related histopathological findings were restricted to brain, lungs, heart, and skeletal muscle of the eagles. Liver, spleen, and kidney did not have significant lesions in any of the 4 cases. Central nervous system lesions were present in the golden eagle and a bald eagle (cases 1 and 4). The neuropil of the ventral brainstem of the golden eagle was characterized by the presence of numerous clear spaces (“spongiform state”) and multifocal glial nodules. Smaller foci of gliosis were also present in the cerebellar cortex of this bird, often extending from the molecular layer into the granular cell layer. Occasional blood vessels were surrounded by a few macrophages, lymphocytes, and plasma cells. A mild infiltrate of similar composition was present multifocally in the meninges. Multiple profiles of protozoal organisms compatible with a Sarcocystis sp. were present within both the ventral brainstem and cerebellar lesions, and consisted of schizonts in various stages of development that ranged from immature schizonts with large central round to oval to multilobulated nuclei and peripheral pale amphophilic cytoplasm, to mature schiz-onts that contained clusters of small elliptical merozoites, roughly 1–2 × 4–6 μm, with central nuclei. Merozoites within the mature schizonts were clustered randomly (Fig. 1A) or occasionally arranged in a rosette pattern around central pale residual bodies. The organisms were present within neurons (e.g., Purkinje cells) and extracellularly in the neuropil. The affected bald eagle (case 4) had a moderate widespread perivascular infiltration, with plasma cells, lymphocytes, and macrophages in cerebrum, mesencephalon, and cerebellum. Numerous microglial nodules were present in the molecular layer of the cerebellum (Fig. 1B). Very infrequent protozoal organisms were found within or adjacent to some of these microglial nodules. The organisms consisted primarily of single immature schizonts with large round to lobated basophilic nuclei surrounded by pale amphophilic cytoplasm with rare clusters of merozoites (mature schizonts) or clusters of free merozoites, the latter seen within foci of inflammatory cell necrosis. The lungs were the site of significant protozoa-associated findings in cases 2 and 3. The lungs of these bald eagles had random capillary thrombi and intra-endothelial protozoal schizonts and merozoites. The protozoal schizonts were compressed within the pulmonary capillaries, which resulted in elongated serpentine profiles. Some of these schizonts were mature and contained numerous well-formed merozoites whereas others were immature and had large basophilic serpentine nuclei (Fig. 1C). The affected lungs had a mild multifocal lymphoplasmacytic, histiocytic and polymorphonuclear infiltration of the interstitium.
Protozoal tissue cysts without an inflammatory response were present in the skeletal muscle of both eagles with encephalitis (cases 1 and 4) and in the hearts of both eagles with pneumonia (cases 2 and 3). Sections of multiple skeletal muscles, including pectoral muscle, ocular muscle, and longissimus muscle, contained several protozoal tissue cysts in the golden eagle. These cysts were elongated in shape that conformed to the host myofibers and were up to 30–40 μm in diameter and 250 μm in length. The tissue-cyst walls were up to 3–4 μm thick and had fine villous projections (Fig. 1D). Numerous bradyzoites were visible within the cysts, which lacked any visible septation. Rare protozoal tissue cysts were also present in the skeletal muscle of the bald eagle (case 4). They ranged from 21 to 57 μm × 65 to 547 μm, with visible internal septa, metrocytes, and closely packed bradyzoites. The tissue-cyst walls ranged from 1 to 4 μm in width, with distinct long, thin, villous-like projections in those cysts with thicker walls. Tissue cysts and protozoal schizonts were detected in the hearts of bald eagle cases 2 and 3. There was a mild-to-moderate plasmacytic myocarditis, with rare protozoal zoites and few protozoal tissue cysts in 1 animal (case 2) and a moderate multifocal histiocytic and polymorphonu-clear myocarditis with cardiomyocyte degeneration and rare large protozoal tissue cysts in the other animal (case 3). The tissue cysts in case 2 ranged from 21 to 72 μm × 23 to 163 μm. The cyst wall ranged from 1 to 3 μm in width and had distinct fine, thin, villous-like projections. These cysts contained numerous tightly packed bradyzoites with internal septation and metrocytes along the periphery and angles of the internal septa. Two tissue cysts examined in the heart of case 3 ranged from 44 to 99 μm × 124 to 209 μm. These cysts had a thin wall approximately 1 μm in width that had fine striations suggestive of villous projections. Bradyzoites were tightly packed within the cysts, which had distinct thin internal septa.
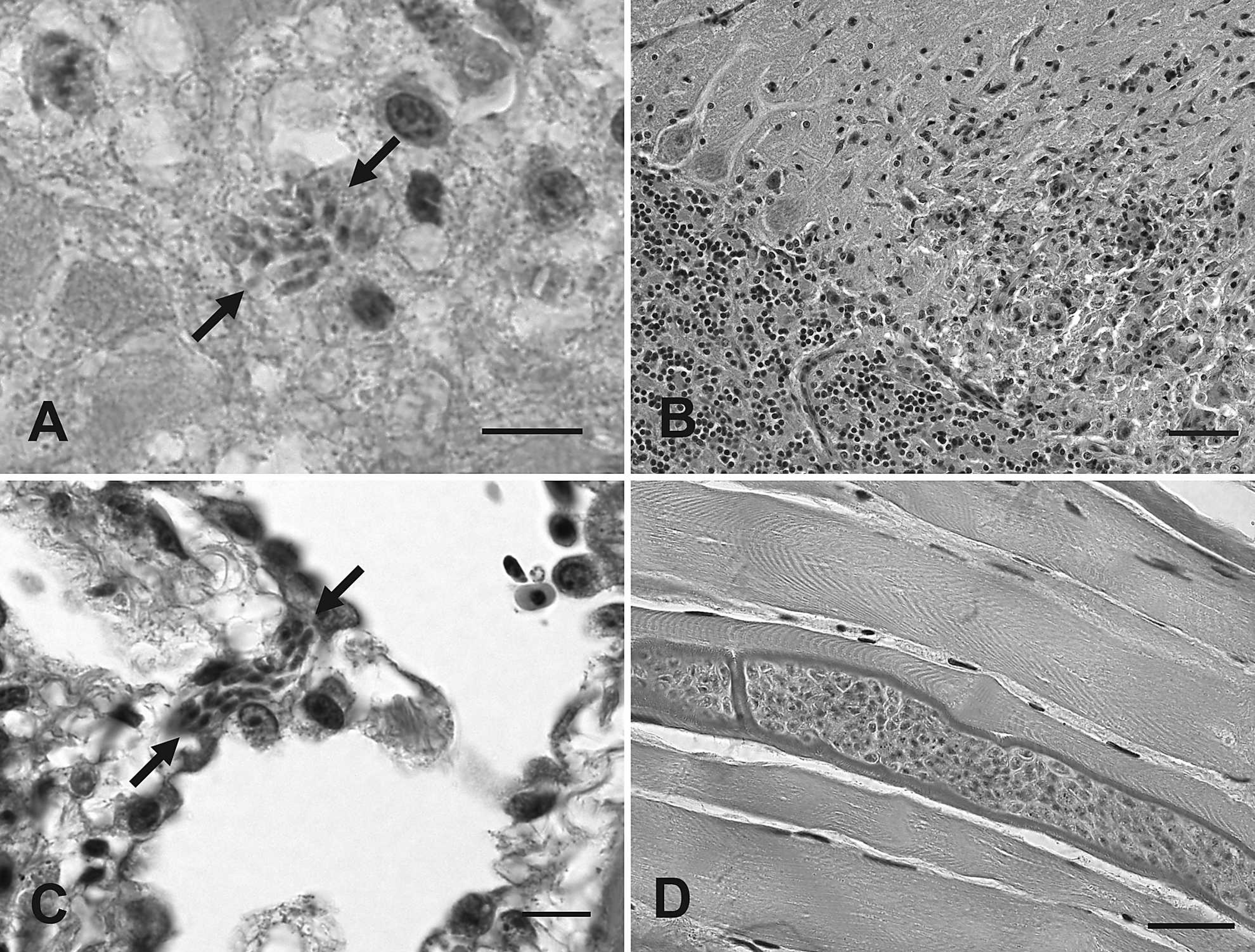
Photomicrograph. A, brain stem, case 1: a cluster of protozoal merozoites is lying freely within the neuropil (arrows). Hematoxylin and eosin (HE) stain. Bar = 10 μm.
The immunohistochemistry (IHC) methods were identical to those previously described. 26 By IHC, merozoites and schizonts detected in the samples of brain (cases 1 and 4), lung (cases 2 and 3), heart (cases 2 and 3), spleen (case 2), and skeletal muscle (case 4) all reacted strongly positive with polyclonal antiserum raised in rabbits against tissue-culture–derived whole S. falcatula merozoites (Fig. 2A–C). A small subset of the merozoites in the brain of 1 eagle (case 1) also reacted weakly positive to polyclonal antiserum against S. neurona, whereas small numbers of merozoites in the lung (case 3) reacted weakly with the monoclonal antibody raised against S. neurona but did not react to the polyclonal S. neurona antiserum. The IHC reactivity of the tissue cysts varied. In the golden eagle (case 1), the walls of the muscle cysts generally reacted positive with the polyclonal S. neurona antisera, whereas the bradyzoites reacted weakly positive with the S. falcatula polyclonal antisera (Fig. 2D, E). In the remaining birds, the majority of tissue cysts did not react to either of these antisera. None of the merozoites or tissue cysts reacted with polyclonal antisera to Toxoplasma gondii or Neospora caninum.
Sections of deparaffinized skeletal muscle from the golden eagle (case 1) that had the highest density of muscle-tissue cysts were processed for transmission electron microscopy (TEM) as previously described. 25 A total of 3 tissue-cyst profiles were located by TEM and measured approximately 11 μm × 23 μm, 18 μm × 22 μm, and 19 μm × 19 μm. Details of the bradyzoite and metrocyte morphology were obscured because of postmortem tissue deterioration and the processing of formalin-fixed deparaffinized material. The tissue cysts contained approximately 21–45 individual zoites, which were 2.0–2.3 μm in width, with a single zoite that was 4.8 μm in length. No internal septa were visible. The cyst walls had long narrow projecting villous-like structures. The smallest cyst, considered to be an immature cyst, had a wall with villous-like projections that ranged from 0.17 to 0.23 μm in width and 0.81 to 0.86 μm in length. The remaining 2 cysts, considered to be more mature, had longer villous-like projections that were 0.22–0.40 μm wide and 1.52–2.23-μm long (Fig. 2F). Features common to all of the villous-like projections included an electron dense 0.03–0.54 μm peripheral layer and long, thin, dense, filamentous structures that extended down through the centers of the villous structures that varied in number from 1 or 2 filamentous structures in the smaller, more immature cyst to 6–10 filamentous structures in the longer villi of the more mature cysts (Fig. 2F). The surface of the longer villi of the more mature cysts also had a distinct, regular undulating or “hobnailed” surface (Fig. 2F).
Blocks of paraffin-embedded brain and skeletal muscle (cases 1 and 4), lung and heart muscle (case 2), and lung tissue (case 3) were examined by polymerase chain reaction (PCR) for the presence of protozoal DNA. The methods of DNA extraction, PCR conditions, and reaction times were identical to those previously described. 26 Apicomplexan panspecific primers that target the first internal transcribed spacer (ITS1) region were used to amplify extracted DNA in a nested PCR reaction. The outer (ITS1DF 5'-TACCGATTGAGTGTTCCGGTG-3’, ITS1DR 5'-GCAATTCACATTGCGTTTCGC-3') and inner (ITS1-diF 5'-CGTAACAAGGTTTCCGTAGG-3', ITS1diR 5'-TTCATCGTTGCGCGAGCCAAG-3') primers were designed based on the primers reported in 2 studies (Miller RH, Nandra AK, Stitt T, et al.: Unexpected diversity in tissue-cyst coccidia [Toxoplasma gondii and Sarcocystis spp.] infecting wild birds in Western Canada. Submitted for publication). 23 The PCR products from positive bands were cleaned by using a commercial PCR clean-up system a and sequenced at the DBS sequencing facility at the University of California, Davis, California. A BLAST search was used to compare the resulting sequences to similar sequences available in GenBank. The PCR analyses of each tissue examined from cases 1–3 revealed a single (approximately 1,000 bp) amplification product that was consistent with the predicted amplification product size of S. falcatula, whereas no PCR amplification products were detected from the brain or muscle tissue of 1 bald eagle (case 4). Subsequent sequencing results from positive bands (cases 1–3) were highly similar to S. falcatula and differed from S. neurona. The resulting ITS1 sequences from the brain and skeletal muscle (GenBank accession no. FJ904650) of case 1 as well as lung (GenBank accession no. FJ904651) and heart muscle of case 2, were 98% similar to several S. falcatula sequences in GenBank. The sequences were 96% similar to the most closely related S. neurona sequences and only 92% similar to a Sarcocystis lindsayi ITS1 sequence. The ITS1 sequence from lung of case 3 was 99% similar to several S. falcatula sequences, 97% similar to the most closely related S. neurona sequences, and only 91% similar to an S. lindsayi sequence. Sequences from all 3 cases were aligned with multiple other Sarcocystis ITS1 sequences and an unweighted pair group method with arithmetic mean (UPGMA) phylogenetic tree was generated by using the program ClustalX (version 2.0.8). 10 A UPGMA tree showed that the ITS1 sequences from cases 1–3 cluster with several other previously described S. falcatula ITS1 sequences and are more distantly related to either S. neurona or S. lindsayi (Fig. 3).
The histopathological findings, IHC immunore-activity, and PCR results of the protozoal schizonts and merozoites in the brain and lungs in cases 1–3 confirm that these eagles were infected with a parasite that was highly similar and most likely identical to S. falcatula. The sequencing of the ITS1 region of multiple protozoa of the eagles of this study produced a 98% to 99% match with ITS sequences from S. falcatula in GenBank. Compared with S. neurona, there appears to be more natural sequence variation among different S. falcatula strains across the ITS1 region. Therefore, it is not surprising that the S. falcatula sequences identified in the present study were not 100% identical to each other or to previously published S. falcatula ITS1 sequences. However, the ITS1 region has been shown to differentiate S. falcatula from S. neurona. 12,23
Of the 4 eagles examined, only the Sarcocystis tissue cysts in the muscle of the golden eagle reacted with the antisera as previously reported for S. falcatula tissue cysts, whereas the immunoreactivity of tissue cysts in the other birds was negative. 6 These results are consistent with previous findings that indicate that IHC with polyclonal antisera is useful for the detection of both S. falcatula and S. neurona schizont and merozoite stages in known susceptible host species. It is also useful for preliminary detection of S. falcatula, S. neurona, or closely related protozoans in unknown host species but is neither consistent nor specific for detection of the tissue-cyst stage of these protozoans. 6,14,21 The morphologic features support the PCR results that the muscle of the golden eagle harbored S. falcatula tissue cysts, but the identity of the tissue cysts in the muscle of the remaining birds was not determined. In the golden eagle, there was a relatively large number of muscle-tissue cysts that had walls consistent with S. falcatula, based on ultrastructural characteristics such as shape and size of the projections with central filamentous structures and the peripheral electron dense layer with a “hobnailed” contour. 4,15 The muscle of the golden eagle did not have contaminating protozoal schizonts based on IHC, so that the amplified gene sequence can be attributed to the tissue cyst.
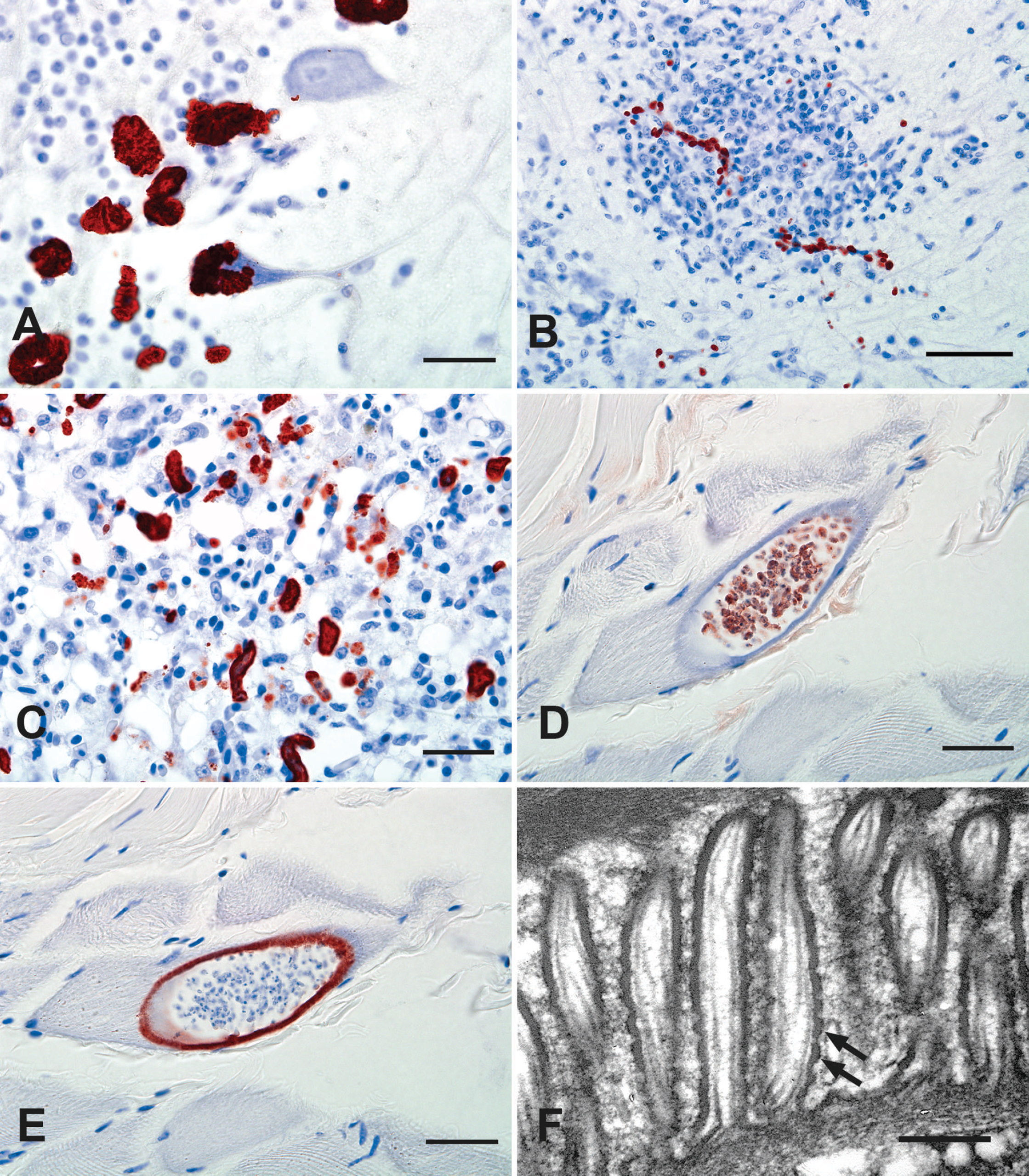
Photomicrograph.
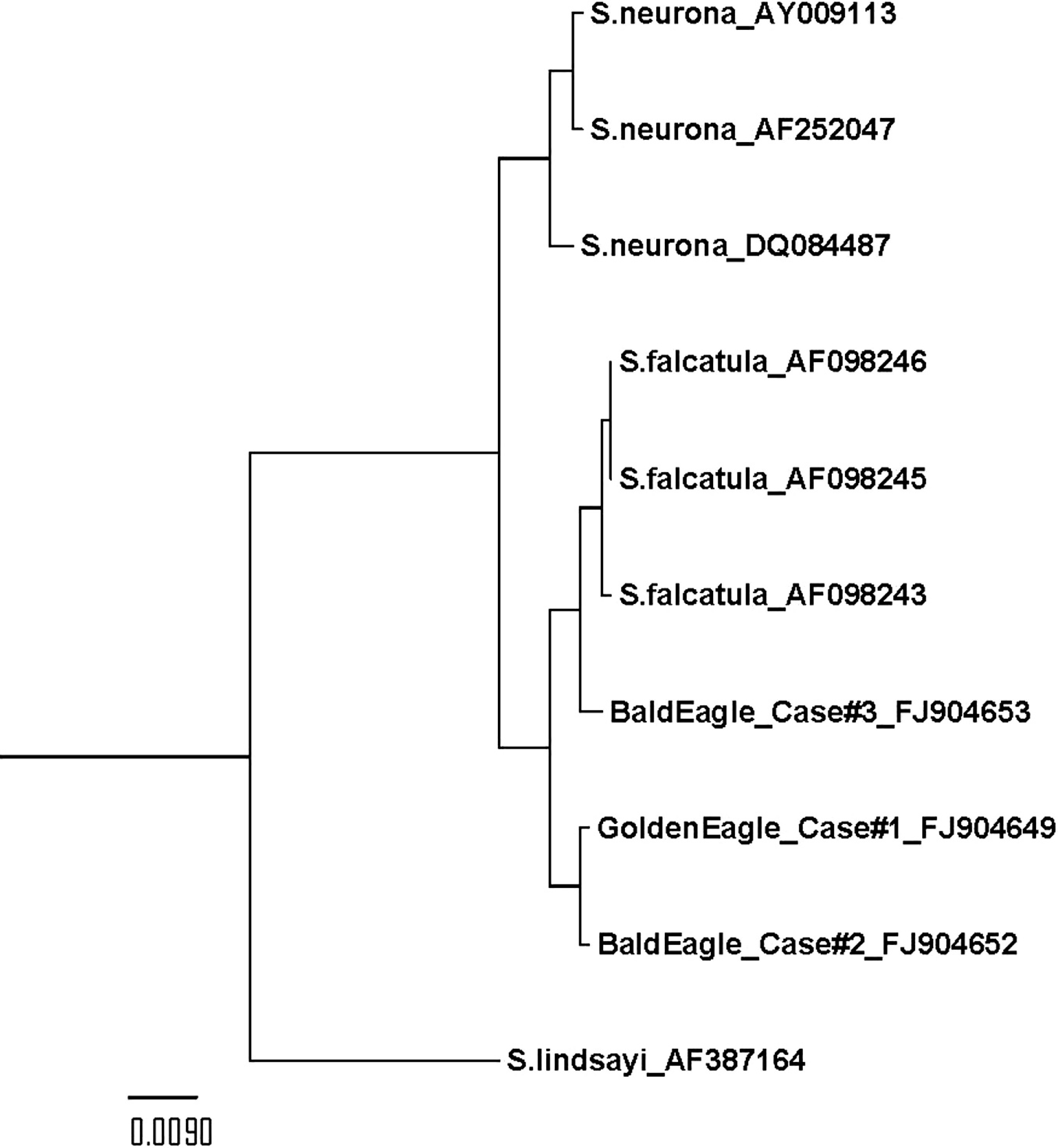
An unweighted pair group method with arithmetic mean (UPGMA) phylogenetic tree, showing the relationship between first internal transcribed spacer (ITS1) sequences from eagle cases 1–3 with previously published Sarcocystis falcatula, Sarcocystis neurona, and Sarcocystis lindsayi ITS1 sequences with their respective GenBank accession numbers. The UPGMA tree was created by using the program ClustalX (version 2.0.8).
Although protozoa in the brain of the bald eagle in case 4 reacted to S. falcatula antiserum in an identical fashion to that in cases 1–3, PCR of both brain and muscle were negative for Sarcocystis spp. This negative PCR result may reflect the low density of protozoa present in the brain and muscle of this bald eagle, because the PCR procedure was designed to detect a wide range of Sarcocystis spp.
The presence of replicative schizonts in the tissues of golden and bald eagles confirmed that these species are intermediate hosts of S. falcatula. The detection of S. falcatula tissue cysts in the golden eagle suggests that falconiformes are true, rather than aberrant intermediate hosts. Before this report, susceptible intermediate hosts of S. falcatula were thought to be limited to birds within the 4 phylogenetic orders: Psittaciformes, Passeriformes, Columbiformes, and Strigiformes. 3,4,6,11,22,24,26
North and South American opossums are recognized as the definitive host for S. falcatula. 3–5,7 Among birds of prey, unidentified Sarcocystis spp. were previously reported as a cause of encephalitis in a bald eagle, golden eagle, and northern goshawk (Accipiter gentilis). 1,8,17
Clinical disease occurs in intermediate hosts of S. falcatula in association with tissue destruction and inflammation associated with asexual protozoal multiplication (i.e., schizogony) after an initial oral infection. Among the 4 birds described in this study, 2 had proliferation of merozoites in pulmonary endothelium and 2 had encephalitis, with proliferation of merozoites within neurons and glial cells. Dyspnea and death associated with the rupture of infected pulmonary endothelial cells is the most common form of clinical disease seen in psittacines following ingestion of S. falcatula sporozoites, although primary encephalitis from either rupture and necrosis of brain endothelial cells or within the brain parenchyma also has been reported to occur due to S. falcatula infections. 19,24,26 Intermediate hosts, such as the eagles in the present study, may typically become infected with S. falcatula via oral ingestion of feed contaminated with opossum feces that contained sporocysts. It is also possible that transmission to the birds described in the current study may have occurred through direct ingestion of opossums that harbored S. falcatula sporocysts within their intestinal tract, because both golden eagle and bald eagle are known to occasionally prey on opossums. 13,16
In conclusion, eagles may serve as intermediate hosts for S. falcatula. This parasite should be considered in the differential diagnoses for eagles with chronic disease, neurological disease, and/or rapidly progressive respiratory disease. Polyclonal antisera are useful diagnostic tools that aid in the detection of S. falcatula or S. falcatula–like schizonts and merozoites in tissues by IHC. However, confirmation of a S. falcatula infection by PCR, culture, or the detection of specific ultrastructural features in conjunction with IHC is warranted for unequivocal identification of S. falcatula.
Acknowledgements. The authors thank Zach Walker of the Dwight Chamberlain Rehabilitation Center (Hardy Lake Reserve) as well as Drs. Mark Ruder and Dave McRuer for veterinary care and review of the manuscript. The authors are also indebted to the CAHFS histology laboratory for technical support on IHC, to Bob Nordhausen for his electron microscopy expertise, and to Amandee Nandra and Michael Grigg for sharing unpublished reagents and for their assistance developing the nested primers across the ITS1 region.
Footnotes
a.
ExoSAP-IT®, USB Corp., Cleveland, OH.
