Abstract
A case of fatal nonneurological equine herpesvirus 1 (EHV-1) infection in a yearling filly is described. Gross lesions included extensive pulmonary edema, prominent laryngeal lymphoid follicles, and congestion and edema of the dorsal third ventricle choroid plexus. Histologically, there was vasculitis, hemorrhage, and edema in the lungs and dorsal third ventricle choroid plexus as well as mild intestinal crypt necrosis with occasional intranuclear inclusion bodies. The perivascular and vascular inflammatory infiltrates were comprised mainly of T lymphocytes and macrophages. EHV-1 antigen was identified within the nucleus and cytoplasm of endothelial cells, dendritic-like cells of the pharyngeal lymphoid follicles, pharyngeal glandular epithelium, crypt enterocytes, and monocytes. Attempted virus isolation was negative. Weak seroconversion for EHV-1 was observed. Herpesvirus-like particles were identified within pharyngeal endothelial cells by transmission electron microscopy. Polymerase chain reaction amplified 369 and 188 base-pair fragments specific for EHV-1. The scarcity of pathognomonic viral inclusions and lesions in this case suggests that this disease may not be recognized, particularly in situations when ancillary laboratory procedures are limited.
Equine herpesvirus (EHV-1) causes abortion, stillbirth, respiratory disease, and encephalomyelopathy secondary to vasculitis in horses. 4 5 10 18 The infection is almost uniformly fatal in newborn foals. The outcome of the neurological form is variable, with some cases leading to paraplegia, quadriplegia, recumbency, and death or euthanasia due to the critical condition of the patient. We describe a fatal, nonneurological EHV-1 infection in a yearling filly associated with multisystemic vasculitis, severe pulmonary edema and hemorrhage, and mild enterotyphlocolitis, with particular emphasis on the distribution of the EHV-1 antigen (EHV-1Ag) identified with the avidin-biotin complex (ABC) indirect immunoperoxidase histochemical technique on affected tissues.
A yearling Thoroughbred filly presented with a history of fever (up to 105 F), depression, and lethargy of a few days duration. Several other horses on the property were also febrile. These animals had not been vaccinated for equine respiratory viral pathogens. The filly was treated for pyrexia with nonsteroidal anti-inflammatory agents. She was found dead in her stall the morning of presentation.
On necropsy, the mucous membranes were severely congested and cyanotic. There were approximately 5 liters of clear yellow-orange watery fluid in the thoracic cavity (Fig. 1). Similar fluid infiltrated and expanded the interlobular septa and visceral pleura of the lungs, which were very heavy. The trachea contained abundant froth, which extended to bronchi and bronchioli in association with fibrin. The lymphoid follicles around the epiglottis were prominent. The meningeal blood vessels were moderately hyperemic and the dorsal third ventricle choroid plexus was severely edematous and congested.
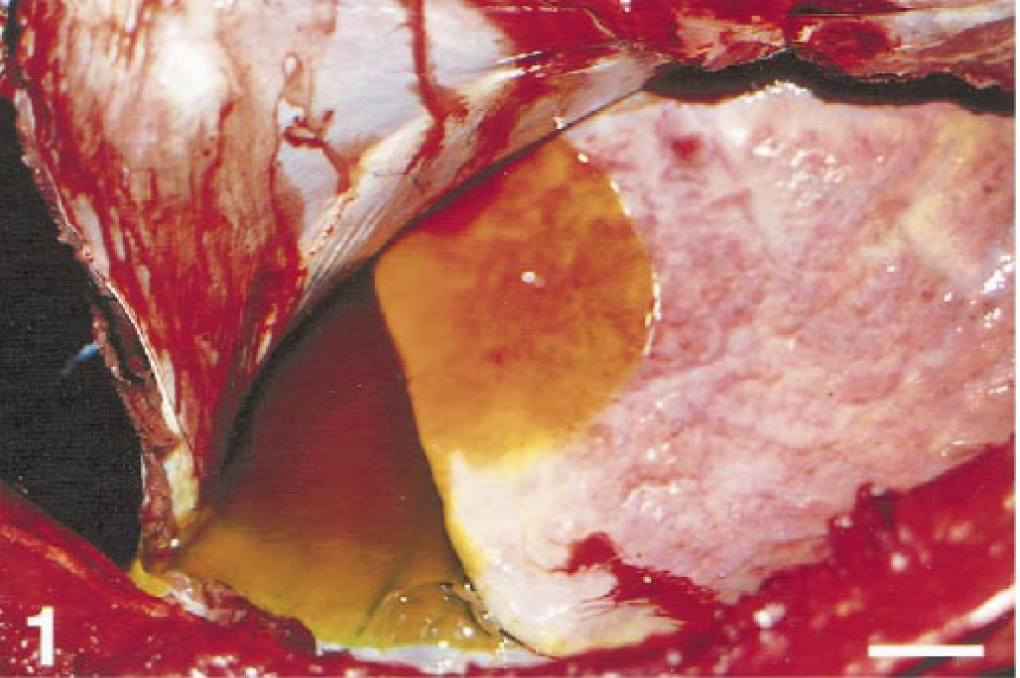
Filly, lung (L). Note the severe pulmonary edema and hydrothorax indicated by prominent interlobular septa, pleural opacity, and yellowish-orange fluid within the thoracic cavity. Bar = 5 cm.
The primary histological lesion was vasculitis, which was particularly severe in the lungs and dorsal third ventricle choroid plexus. Within the pulmonary parenchyma, there was extensive vasculitis with diffuse, acute pulmonary edema and perivascular hemorrhage (Fig. 2). Alveoli, alveolar ducts, and bronchioli contained proteinaceous fluid, fibrin, and detached but morphologically intact epithelial cells as well as infrequent macrophages engaged in erythrophagocytosis and scattered hypertrophied type 2 pneumocytes. Edema, hemorrhage, and inflammation, composed of monocytes and lymphocytes, were present around blood vessels and extended into septa and subpleural connective tissue. Severe congestion of blood vessels, especially involving the capillary network, rare ruptured alveolar septa, endothelial swelling and detachment, margination of leukocytes, and edema of the tunica media were also observed. Lymphatics were dilated and contained fluid, fibrin, and occasional leukocytes. The zona glomerulosa of the adrenal glands contained a few necrotic cells characterized by nuclear pyknosis or chromatin margination. Although lymphoid follicles were prominent, germinal centers were depleted, particularly within the intestine. Mild, multifocal intestinal crypt necrosis, associated with mild to moderate numbers of lymphocytes, macrophages, and few plasma cells, was identified. Within the liver, there was mild, portal, lymphocytic infiltration and congestion of the sinusoids. There was meningeal and parenchymal congestion of blood vessels, which contained a few leukocytes. The dorsal third ventricle choroid plexus stroma was severely edematous and congested (Fig. 3) with moderate, multifocal, acute hemorrhages and vasculitis characterized by abundant intravascular, perivascular, and stromal lymphocytes and macrophages (Fig. 4). Blood vessels in several organs were lined by swollen endothelial cells and contained intraluminal monocytes. Intranuclear acidophilic Cowdry type A inclusions were not frequent and were occasionally detected within endothelial cells of the choroid plexus and enterocytes (Fig. 5). Histochemical stains to identify bacteria, protozoa, and fungi were negative.
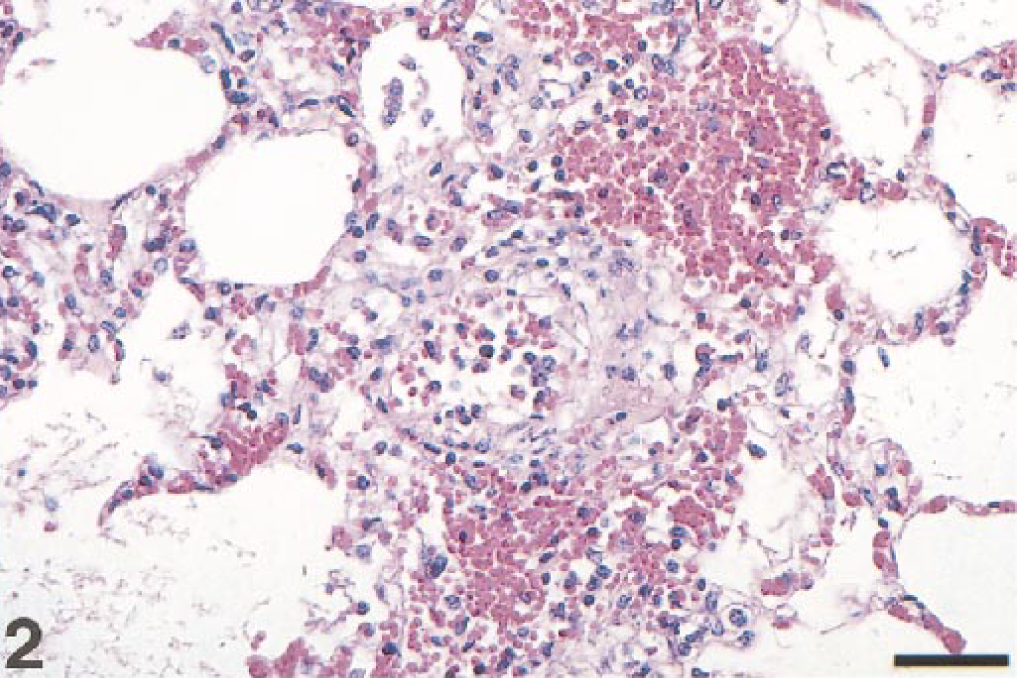
Filly, lung. Vascular necrosis, edema, and hemorrhage with near complete endothelial loss, effacement of the tunica media and adventitia, and peripheral intra-alveolar erythrocytes and fibrin. HE. Bar = 80 µm.
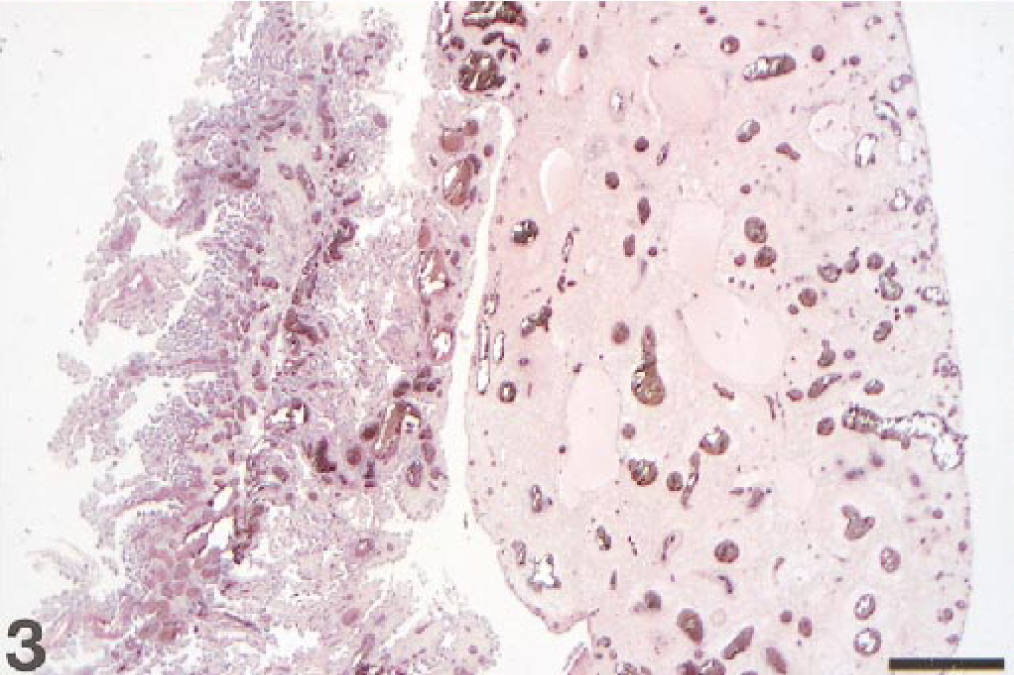
Filly, dorsal cerebral choroid plexus. Severe congestion and edema of the dorsal cerebral choroid plexus (right). Normal cerebellar choroid plexus (left). HE. Bar = 500 µm.
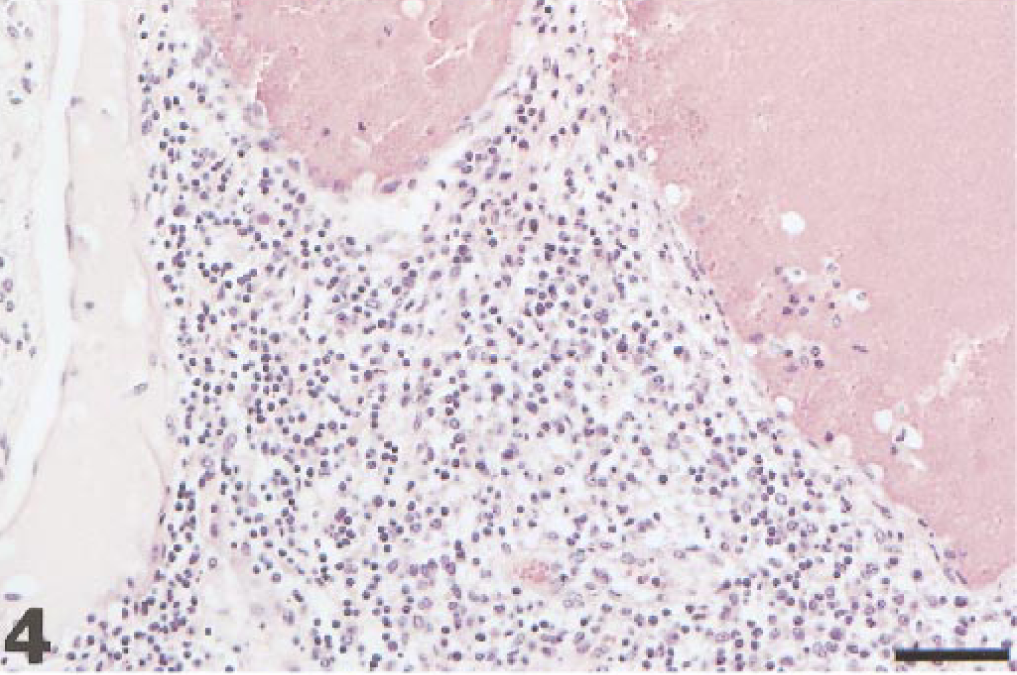
Filly, dorsal third ventricle choroid plexus. Severe inflammation comprised of lymphocytes and macrophages, with vascular congestion and intravascular leukocytosis. HE. Bar = 100 µm.
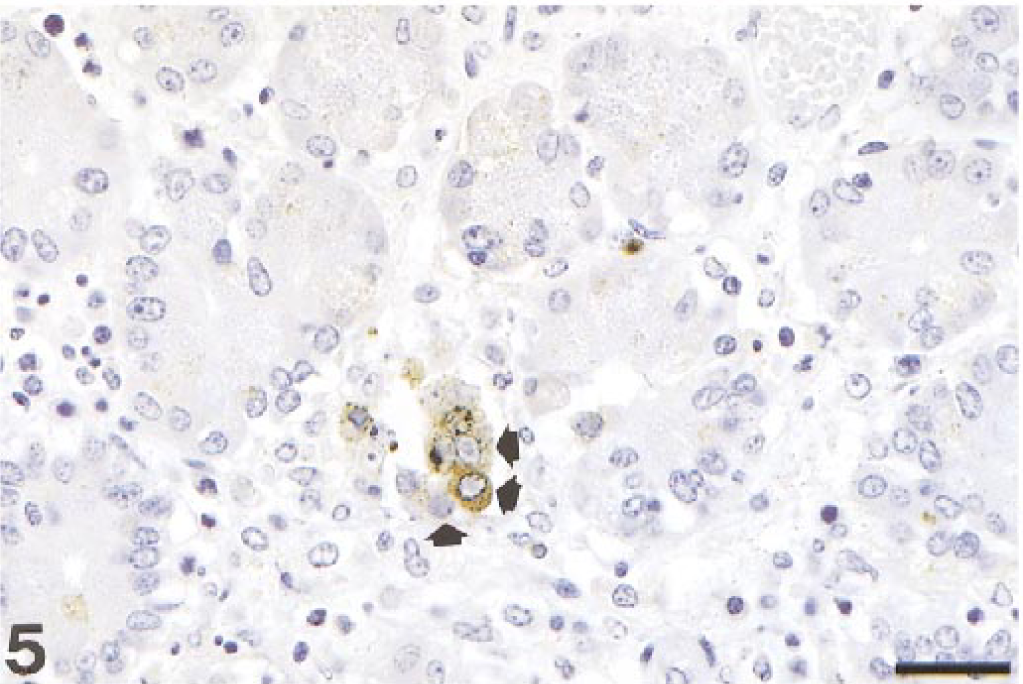
Filly, small intestine. Necrosis of the crypt glandular epithelium with intracellular EHV-1 antigen and intranuclear inclusions (arrows). Bar = 70 µm. ABC immunoperoxidase histochemistry and hematoxylin (IHCH).
The leukocyte populations infiltrating the choroid plexus, lung, and pharynx were examined using antibodies recognizing CD3 of T cells (murine monoclonal antibody (mMab), dilution 1:100; DAKO Corp., Carpinteria, CA, USA), BLA36 of B cells (mMAb, dilution 1:20; DAKO), and MAC 387 of macrophages (mMAb, dilution 1:1,000; DAKO) with contemporaneous evaluation of cell morphology. Positive controls were tissues from normal horses containing the cell types mentioned above. The intravascular leukocytes were comprised of numerous, sometimes clustering macrophages and slightly fewer T cells. Both cell types were often adjacent or adhering to the endothelium. The perivascular infiltrates were mainly comprised of T cells with fewer macrophages and occasional B cells.
EHV-1Ag was identified using EHV-1-specific murine monoclonal antibody 26A5 IgG2a isotype, which recognizes the EHV-1 envelope glycoprotein C, and the ABC immunoperoxidase automated technique. EHV-1Ag was intranuclear and intracytoplasmic, localized in various organs and cell types, and had a granular or clumped appearance. Sometimes the staining was so diffuse and abundant that it obscured cytological details. Within the lung, EHV-1Ag was detected in occasional endothelial cells. Multifocal EHV-1Ag staining was observed within the dendritic-like cells of the pharyngeal lymphoid follicles and glandular epithelium. EHV-1Ag was detected in greatest abundance in the pharyngeal mucosal epithelium (Fig. 6). In the intestine, EHV-1Ag was contained multifocally within the crypt enterocytes (Fig. 5). A few scattered endothelial and epithelial cells of the adrenal zona glomerulosa contained EHV-1Ag. Blood vessels in lung, intestine, pharynx, trigeminal ganglion, adrenal glands, skin, kidney, and nasal turbinates contained EHV-1Ag within morphologically normal and swollen endothelial cells, as well as intravascular monocytes (Fig. 7). EHV-1Ag was also identified in frozen sections with a caprine polyclonal antibody (dilution 1:50, American Bioresearch, Severville, TN, USA), which recognizes several viral epitopes shared by EHV-1 and EHV-4. Tissues were also tested with a murine monoclonal antibody, which recognizes a 30 kDa membrane protein of equine arteritis virus 6 and resulted negative for this viral antigen. Equine fetal and neonatal tissues, containing lesions and EHV-1 and equine arteritis virus, respectively, were used as positive controls. Negative controls included equine and nonequine tissues with no histological and virological evidence of infection.
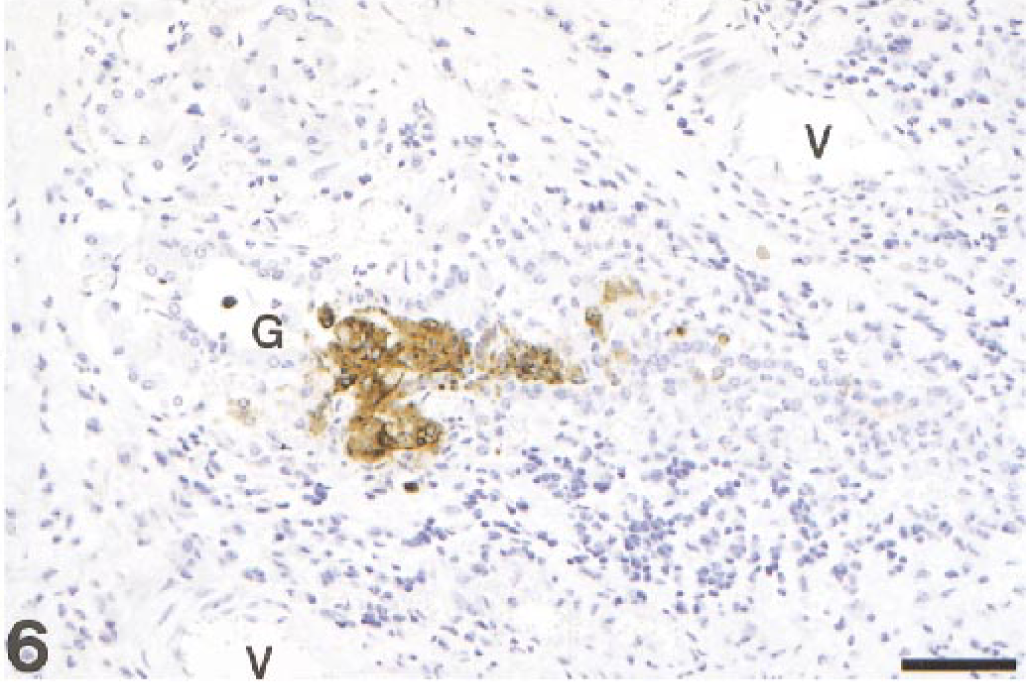
Filly, pharynx. The glandular epithelium contains abundant intranuclear and intracytoplasmic EHV-1 antigen. Lymphocytic infiltrate extends below the gland. (G = gland, V = vessels). IHCH. Bar = 100 µm.
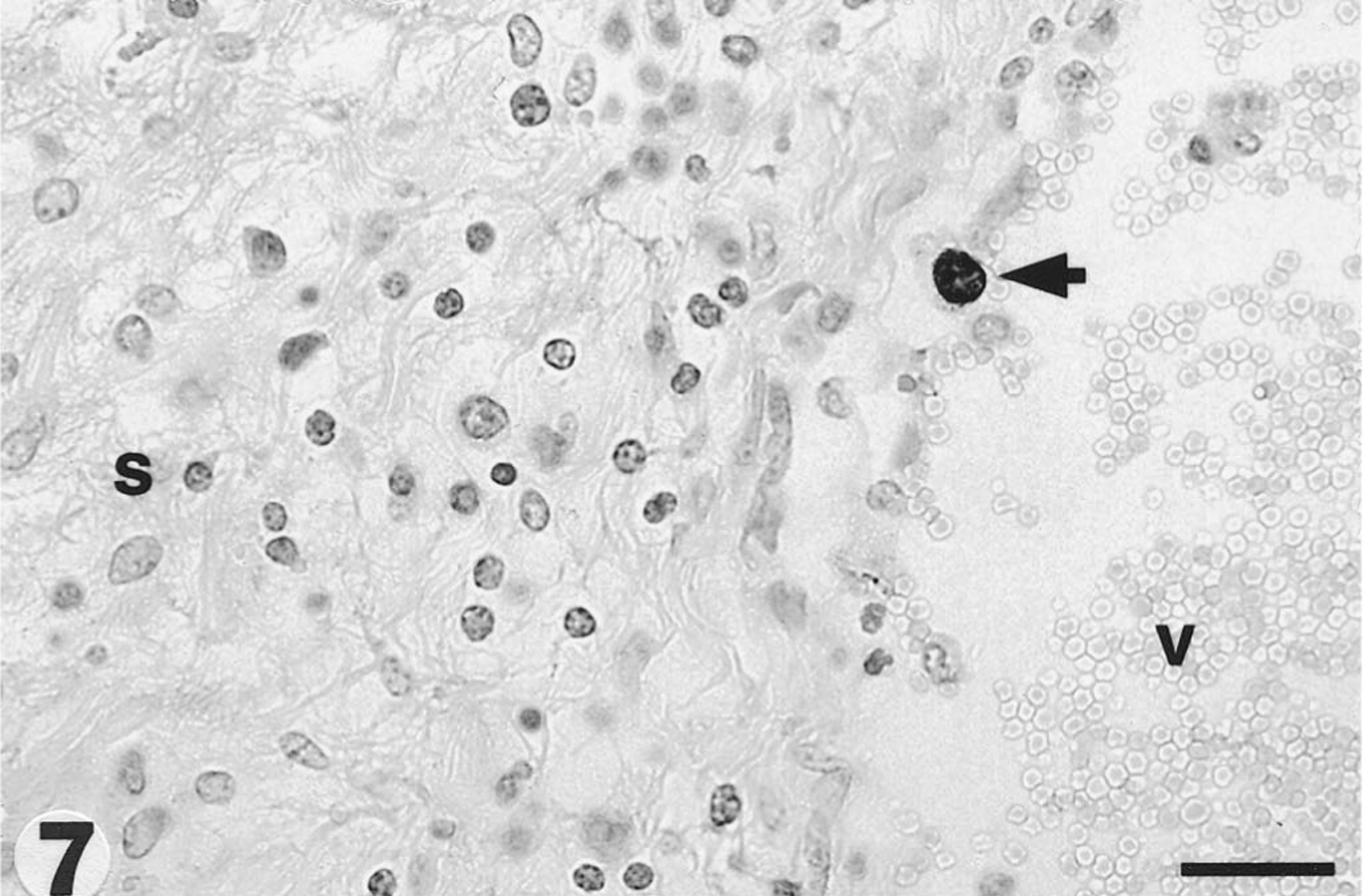
Filly, dorsal choroid plexus. Circulating monocyte (arrow) containing EHV-1 antigen. The endothelial lining of the vessel is missing and the stroma (S) is severely edematous and contains lymphoid cells (V = vessel lumen). IHC H. Bar = 60 µm.
Ultrastructurally (Fig. 8), the nucleus of endothelial cells in the pharynx had a nuclear envelope composed of two membranes separated by a perinuclear cistern adjacent to marginated, electron-dense, clumped chromatin. The nucleus contained aggregates of fuzzy, granular material (nucleoplasm), dark, intertwining material (vermicellar bodies), and herpesvirus-like particles. The virus particles either had solid, dense cores or hollow cores, were stippled (peplomers), or empty with no core. In addition, there were aggregates of incomplete capsids toward the nuclear periphery.
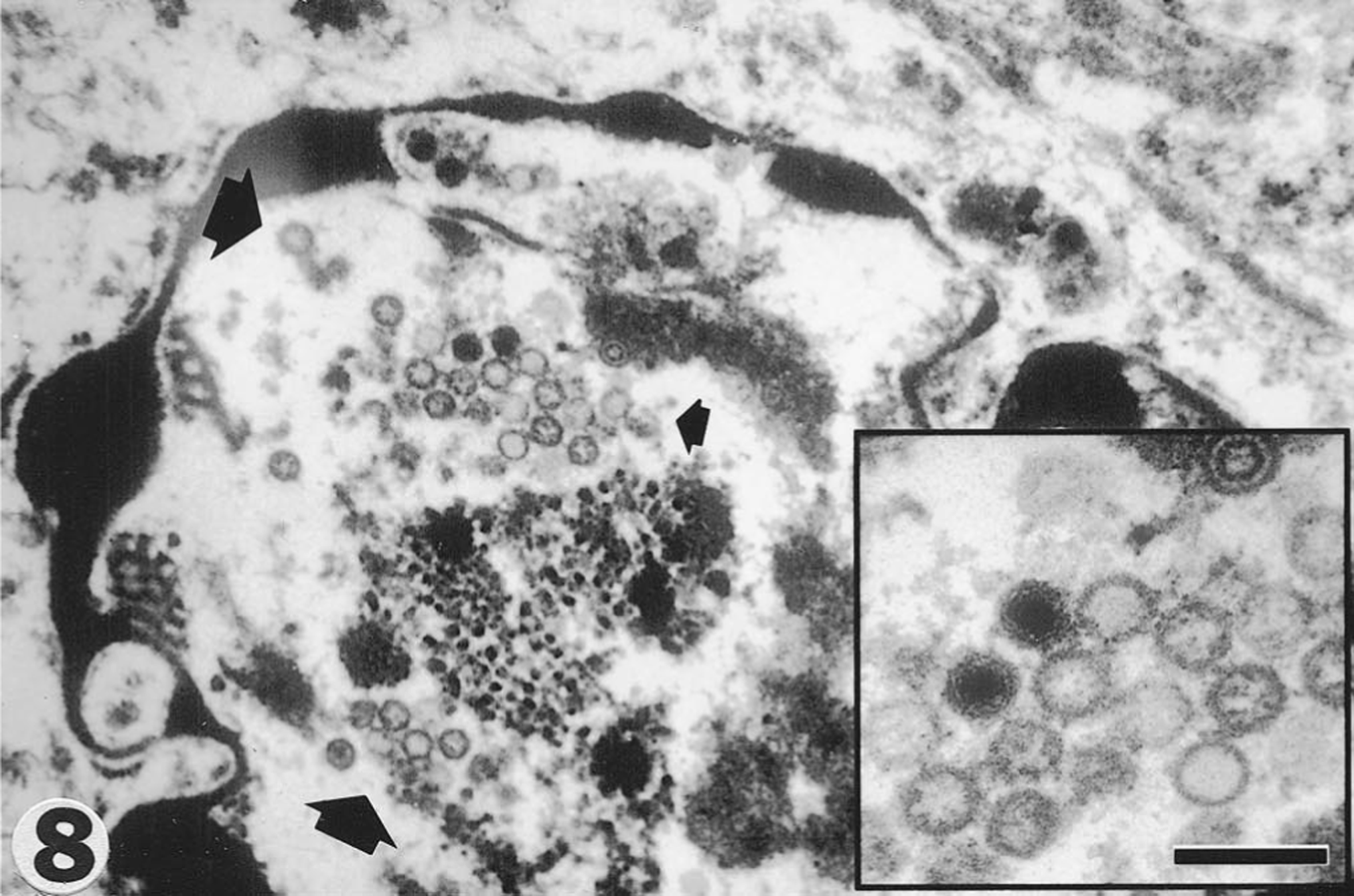
Filly, pharynx. Endothelial cell nucleus with marginating chromatin containing numerous virions (circumscribed by the three arrows).
Virus isolation was attempted using RK-13 cells (ATCC CCL 37) and equine lung cells from fresh and frozen pooled tissues. No virus was detected after two blind passages in cell culture.
Streptococcus Lancefield type C was isolated from the lung and thoracic fluid.
Sera had been collected from all the horses on the premises approximately 2 weeks prior to onset of illness in this filly and were tested with serum neutralization for the presence of EHV-1 antibodies. Of the 10 horses tested for EHV-1 antibodies, 5 had a titer of 256, 2 a titer of 64, 1 a titer of 32, and 2 horses, including the filly reported here, resulted seronegative. Thoracic effusion fluid collected from the filly that died had an antibody titer of eight for EHV-1.
The polymerase chain reaction (PCR) was performed on DNA extracted from paraffin blocks of lung using two sets of primers (data not shown). Primer set 1 15 amplifies a 369-bp product from bp numbers 1025–1393 of the EHV-1 glycoprotein 13 (gp13) gene. Primer set 2 12 is a set of three primers that amplifies both EHV-1 and EHV-4 simultaneously, with the EHV-1 amplification generating an 188-bp band and EHV-4 generating a 677-bp band. Primer set 2 amplifies a sequence from position 2699–2886 of the EHV-1 glycoprotein B (gB) gene. Amplification products were consistent with EHV-1 sequences and the published results from the original designers of the primer sets. Primer set 2 did not amplify the EHV-4 sequence. Amplifications were performed several times and the data reproduced appropriately. Positive controls were formalin-fixed, paraffin-embedded, virus-infected fetal tissues. Negative controls were tissues belonging to several species with no evidence of lesions or any viral infection.
Fatal nonneurological EHV-1 infection in young adult equids appears to be a rather rare event. Blunden and collaborators 1 provided a detailed description of a severe, multisystemic form of EHV-1 infection in an adult zebra stallion, associated with a multifocal, necrotizing rhinitis, marked hydrothorax and pulmonary edema, and viral antigen detectable in epithelial cells, local endothelial cells, and intravascular leukocytes of the nasal mucosa, lung, epididymes, Leydig cells, and testicular germinal epithelium. A second report, which emphasized distribution of lesions, described a condition similar to our case in a 2-year-old horse. 8 In both cases, virus isolation was successful.
The typical necrotizing bronchiolitis frequently associated with EHV-1-related perinatal death in foals and the severe multifocal coagulative necrosis in parenchymal organs 4 14 18 were not observed in this case except for some mild adrenal cortical necrosis.
The pulmonary edema and hemorrhage associated with vasculitis observed in this filly were diffuse, severe, and resembled fulminant equine viral arteritis.
6
13
There were also some similarities with acute equine respiratory syndrome due to Hendra virus infection,
9
African horse sickness,
19
purpura hemorrhagica, and shock.
11
Tissues from this filly did not contain EAV antigen. Several cell types were examined ultrastructurally with no evidence of other virions. Virus isolation using RK-13 and equine lung cells resulted negative. Distribution of lesions correlated with EHV-1 antigen. Endothelia syncytia, which may be suggestive of Hendra virus infection, were not seen. No products that could produce anaphylactic or endotoxic shock had been administered to this filly. Streptococci Lancefield type C were also isolated from the thoracic fluid. We believe this bacterium did not play a role in this horse demise but is the result of a postmortem growth of a resident bacterial population of the respiratory tract. This filly did not have evidence of strangles, had not received a vaccine to prevent the disease, and did not present with a cutaneous vasculitis with neutrophils, which is one of the features of
Immunoperoxidase histochemistry enabled identification of the etiologic agent, especially in light of negative virus isolation. PCR on DNA from paraffin blocks allowed the identification of fragments specific for EHV-1 and permitted identification of viral DNA in tissues, such as the lung, with a small amount of viral antigen. The presence of EHV-1Ag within several cell types, such as endothelium, epithelium, and macrophages, indicates its cellular polytropism. 4 5 EHV-1Ag localization within endothelium and circulating monocytes in multiple organs morphologically demonstrated monocyte-associated viremia and diapedesis of infected monocytes within tissues. No EHV-1 was detected within other leukocytes. Although it is still possible that small quantities of EHV-1 antigen were contained within lymphoid cells, these findings indicate the circulating monocyte may be the primary cell supporting the EHV-1 viremia. The inflammation associated with vascular lesions was comprised of T cells and macrophages, with a few neutrophils and occasional B cells. In cases of EHV-1 infection in newborn foals with pneumonia and adults with encephalomyelopathy associated with EHV-1 vasculitis, the predominant perivascular inflammatory cell type is the T lymphocyte (F. Del Piero, unpublished data). T lymphocytes may have a significant cytotoxic effect on EHV-1 infected cells, contributing to the severity of cytolysis within the vascular lesion. This cytolytic activity may be mediated by class I major histocompatibility complex-restricted CD8+ T cells, as previously described in EHV-1-infected CBA (H-2 (k)) mice. 16 Several small pulmonary vessels had severe endothelial loss associated with small amounts of EHV-1Ag. It is possible that we observed a phase of the infection where peak endothelial viral replication, and consequent cytopathic effect, already occurred. In addition, the severe endothelial damage may have induced hypoxia and necrosis of the tunica media. 7
Colitis has previously been associated with systemic EHV-1 infection in a horse with neurological signs and lesions suggestive of EHV-1 infection. 2 EHV-1 intestinal crypt necrosis with intraepithelial viral antigen is not an unusual finding in EHV-1 neonatal death. 4
There were no lesions within the central and peripheral nervous system other than a mild, focal, perivascular infiltrate within one of the trigeminal ganglia, which is likely unrelated to the virus infection or may have been induced by sporadic viral endothelial colonization in the ganglion. The lack of evidence of infection of the central and peripheral nervous system in this filly is intriguing. The fact that the endothelium of the nervous system was spared from infection may be explained by differences in virus strain and viral affinity to central nervous system endothelial receptors. In addition, both the chronology and route of infection can potentially influence the distribution of a virus in tissues. 3 6 17
The antibody titers identified in the horses on this farm indicate previous vaccination or natural exposure to EHV-1 and/or EHV-4. The weak seroconversion observed in this filly may be related to the short period of illness or her inability to produce a stronger antibody response. The failure to isolate EHV-1 from several tissues could be related to the presence of very low concentrations of virus within the parenchyma of the tissues submitted for virus isolation. In some lung sections, e.g., a small amount of EHV-1 was identified with immunohistochemistry and PCR.
This case of EHV-1 infection had an atypical presentation. This could be related to a particularly virulent endotheliotropic strain able to bypass the immunitary defenses of this filly, which also presented with a rather inconspicuous humoral response for EHV-1.
In conclusion, EHV-1 can induce nonneurological fatal disease in young equine adults characterized by prominent vasculotropism. This previously unidentified clinical and pathological presentation should always be considered and included in the possible EHV-1 manifestations. Lesions may resemble equine viral arteritis, African horse sickness, acute equine respiratory syndrome due to Hendra virus infection, purpura hemorrhagica, and shock. Considering the scarcity of pathognomonic viral inclusions and lesions in this case, it is possible that similar cases may be overlooked, particularly where ancillary diagnostic laboratory testing is limited. Identification of viral antigen in tissue sections with monoclonal-specific antibodies was the most convenient and reliable technique used to obtain a diagnosis in this case.
Footnotes
Acknowledgements
We would like to thank Dr. George Allen of the Gluck Equine Research Center, University of Kentucky, Lexington, and Dr. Amy Glaser, Cornell University, New York State Diagnostic Laboratory for providing the EHV-1 and EAV monoclonal antibodies reagents respectively, Dr. Mary Lynne Vickers of the Livestock Disease Diagnostic Center, University of Kentucky, for conducting the serological testing, and Dr. Anthony Castro of Pennsylvania State University for carrying out the immunofluorescent examination of frozen sections and attempted virus isolation. We are grateful to Jacqueline Ferracone and Suzanne Hindman for excellent technical assistance and to Mrs. Alexis Wensky-Roberts (Image Lab®) for photography.
