Abstract
During necropsy of a 6-year-old, male African Green monkey (Cercopithecus aethiops), multifocal pale, white to tan areas, 1–2 cm in diameter, were noted in the right lateral lobe, medial lobe, and capsular surface of the liver. Light microscopic examination of the liver revealed numerous spindle cells dispersed within interconnecting, broad bands of collagen, up to 200 μm in width, which often spanned and connected portal areas. A diagnosis of hepatic ductal plate malformation, most closely resembling congenital hepatic fibrosis with von Meyenburg complexes, was made. Few cases of ductal plate malformations in domestic animals are reported in the literature. To our knowledge, this is the first case reported in a nonhuman primate.
Fetal ductal plate malformations represent disruption or failure of the epithelial-mesenchymal interaction during fetal development of the liver that results in expansion of portal areas by an excess of embryonic bile ducts and fibrous tissue. Ductal plate malformations comprise the core of various hepatic diseases in humans. These diseases include but are not limited to congenital hepatic fibrosis (CHF), Caroli disease, von Meyenburg complexes, Caroli syndrome, autosomal recessive polycystic kidney disease (ARPKD), and autosomal dominant polycystic kidney disease (ADPKD). Few such malformations of domestic animals have been reported in the literature aside from the polycystic kidney (pck) mouse model. The few cases reported in the literature include Caroli's disease in young dogs, 5 biliary atresia in a dog and in a foal, Caroli syndrome in a rat, 2 and congenital hepatic fibrosis in a newborn calf. 9 To the best of our knowledge, ductal plate malformations have not been reported in nonhuman primates.
During necropsy of a 6-year-old, male African Green monkey (Cercopithecus aethiops), multifocal pale, white to tan areas, 1–2 cm in diameter, were noted in the right lateral lobe, medial lobe, and on the capsular surface of the liver. All other tissues were considered grossly and histologically normal. A section of formalin-fixed, paraffin-embedded liver was examined by light microscopy. Affecting approximately 85% of the section, numerous spindle cells were dispersed within interconnecting, broad bands of collagen, up to 200 µm in width, which often spanned and connected portal areas (Fig. 1). Spindle cells had indistinct cell borders, scant eosinophilic fibrillar cytoplasm, oval nuclei with finely stippled chromatin and one variably distinct nucleoli. Throughout these bands of spindle cells were increased numbers of small bile duct profiles that were occasionally ectatic. Few individualized fibrous plates served as a central axis for surrounding long, continuous, dilated bile duct profiles. Remaining fibrous plates were completely surrounded by streams of spindle cells. Portal areas were often closely opposed and expanded by abundant, mature collagen. A few small, non-portal-associated bile ducts and spindle cells were dispersed throughout the hepatocellular parenchyma. Few to moderate numbers of lymphocytes and plasma cells with few Kupffer cells infiltrated collagenous bands and portal areas (Fig. 2).
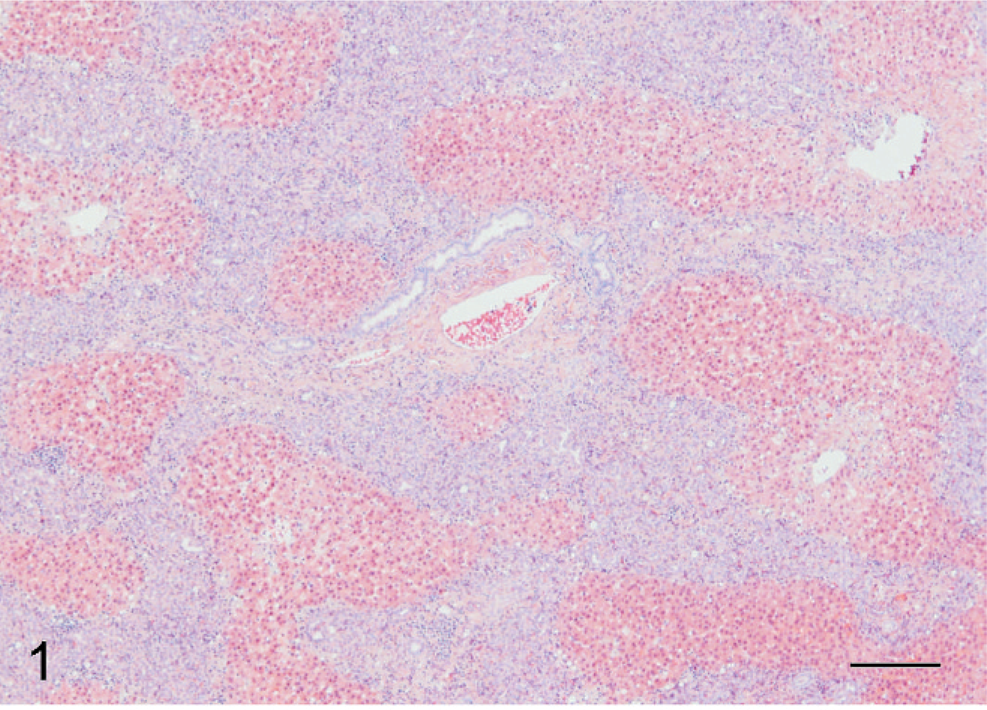
Liver; African green monkey. Numerous spindle cells arranged in broad bands often with central plate of collagen and semicircularly arranged dilated bile ducts. HE. Bar = 200 µm.
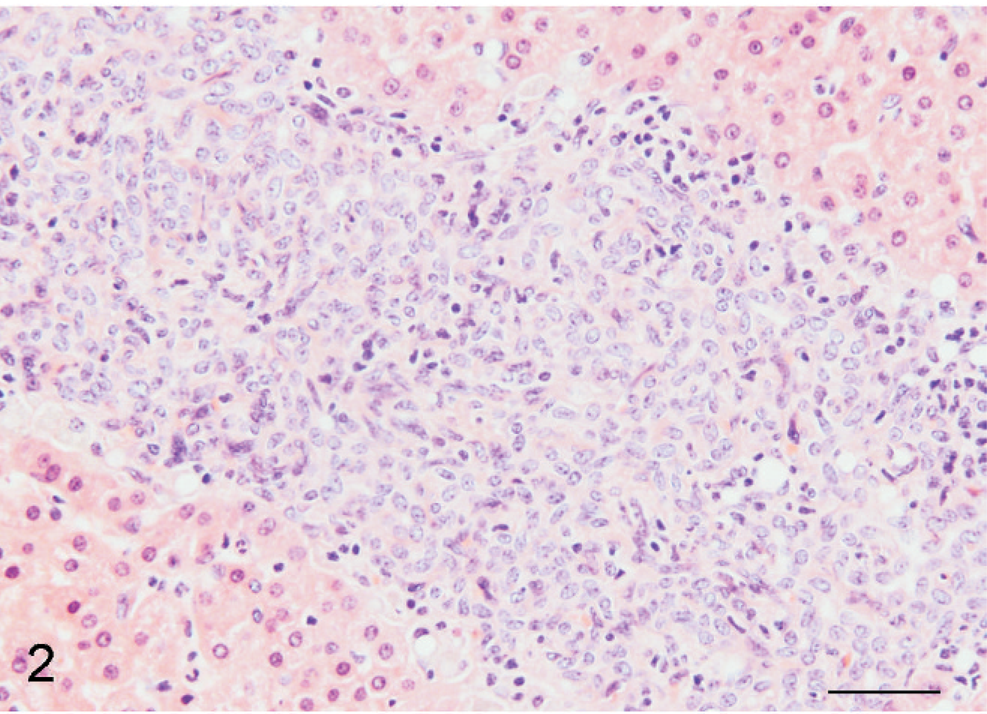
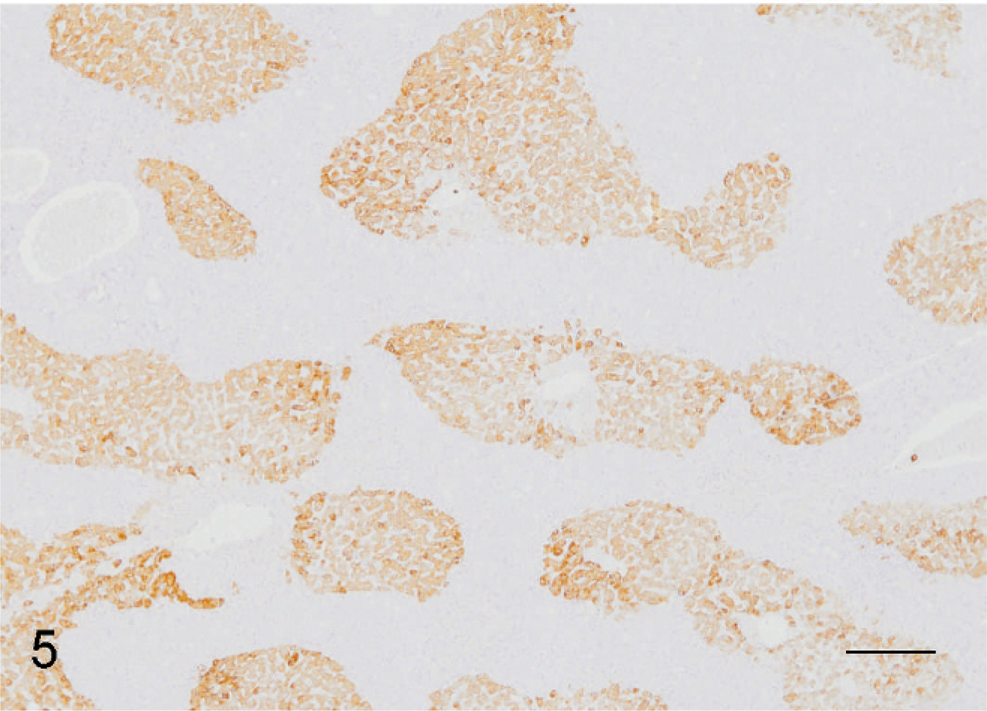
Liver; African green monkey. Numerous spindle cells arranged in broad bands and interconnecting streams mixed with few plasma cells and lymphocytes. HE. Bar = 50 µm.
Primary antibodies utilized for immunohistochemistry were CD34 (Dako, Carpinteria, CA; 1 : 200 dilution); cytokeratin (CK) 7 (Dako; 1 : 400 dilution); CK 19 (Dako; 1 : 200 dilution); CK 8 (Neomarker, Fremont, CA; 1 : 400 dilution); hepatocyte specific antigen (Dako; 1 : 2,000 dilution); and epithelial membrane antigen and collagen IV (prediluted product EZ prep, Ventana, Tucson, AZ). All sections were counterstained with Ventana hematoxylin (Ventana) for nuclear counterstaining visualization.
Spindle cells, presumed to be precursor/bile duct epithelial cells, and preexisting bile duct epithelium were diffusely and strongly intracytoplasmically immunoreactive for CK 19, CK 7, and CK 8. Spindle cells demonstrated strongest reactivity to CK 19 (Fig. 3). Scattered individual CK7+ and CK19+ spindled cells, not associated with portal areas, were noted within more normal hepatic lobules. Hepatocytes demonstrated generally diffuse, strong intracytoplasmic reactivity for hepatocyte specific antigen (SA) (Fig. 5). Rarely, individual cells within the bands of spindle cells also demonstrated hepatocyte SA reactivity. These cells were most likely entrapped individual mature hepatocytes. Collagen bundles immediately surrounding bile ducts within portal areas demonstrated prominent reactivity to collagen IV antibody (Fig. 4). Interestingly, remaining surrounding portal collagen was nonreactive to this antibody. Collagen IV is present in mesenchyme surrounding ductal plates early in gestation; persistent collagen IV immunoreactivity, here, supports the diagnosis of a ductal plate malformation.
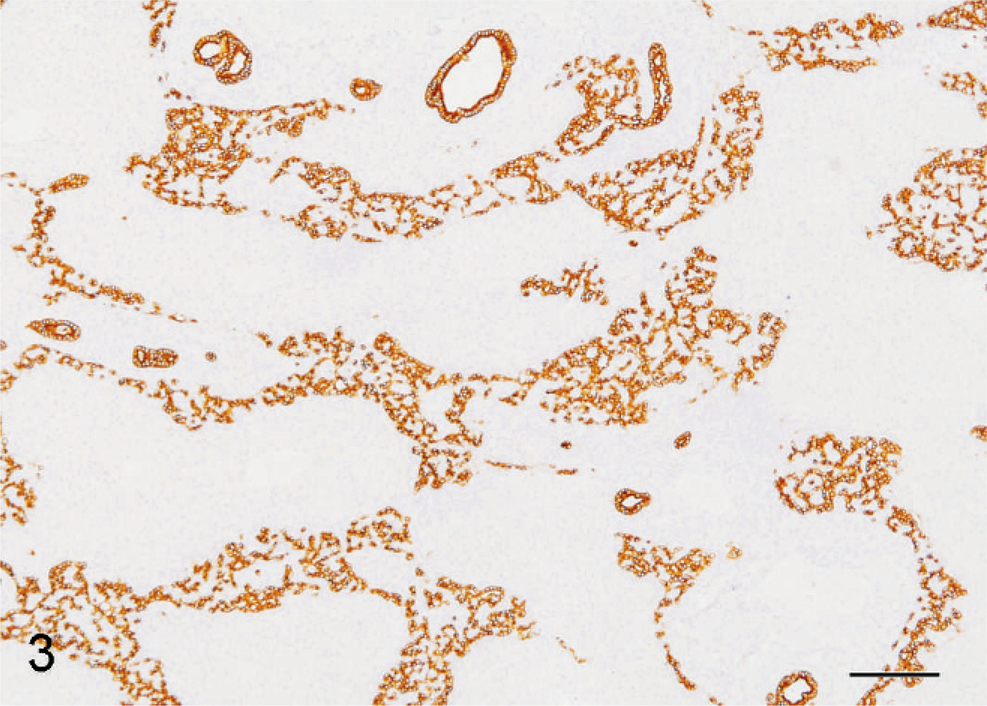
Liver; African green monkey. Spindle cells and bile duct epithelium are diffusely and strongly intracytoplasmically immunoreactive to CK 19. Bar = 200 µm.
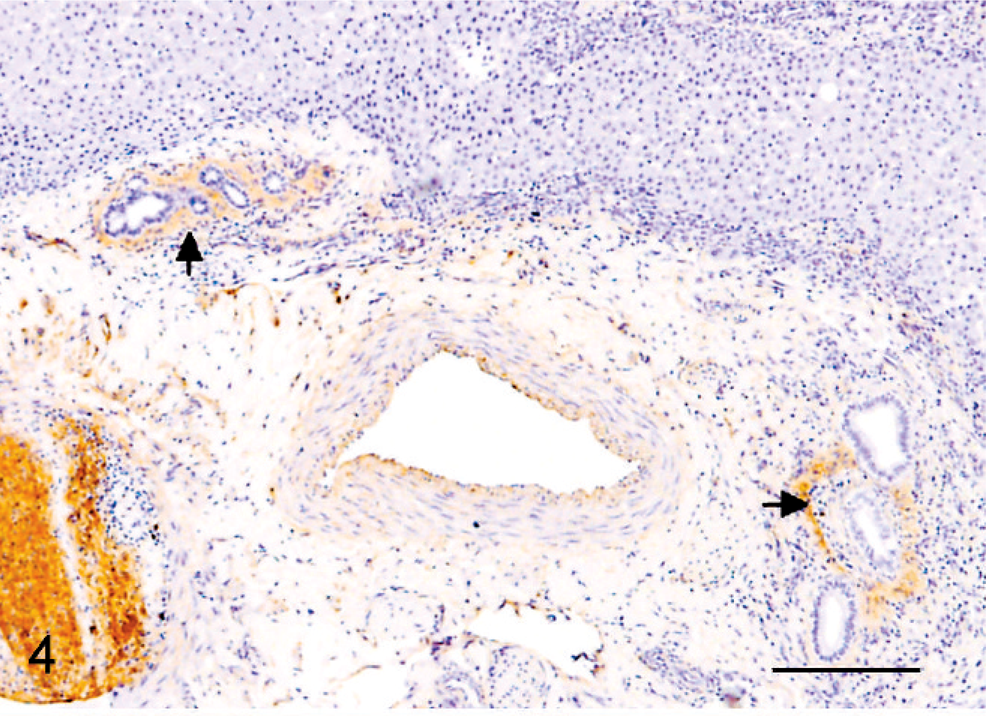
Liver; African green monkey. Collagen immediately surrounding bile ductules is most strongly immunoreactive to collagen IV (arrow). Bar = 200 µm.
Liver; African green monkey. Strong reactivity to hepatocyte antigen accentuates the hepatic fibrosis. Bar = 200 µm.
Livers from 3-day-old, 1-year-old, and 6-year-old African green monkeys were used as morphologic and immunohistochemical controls. Livers from all controls demonstrated few portal lymphoplasmacytic aggregates, considered histologically to be within normal limits. Bile duct epithelium from 1-year-old and 6-year-old control livers demonstrated moderate to strong intracytoplasmic immunoreactivity for CK 7, 8, and 19 and were nonreactive for epithelial membrane antigen. Diffusely, collagen bundles were nonreactive for collagen type IV antibody.
Comparatively, the liver from the 3-day-old monkey demonstrated increased portal collagen and spindle cells, interpreted as precursor cells. Definitive ductal plate structures were not observed. Faint multifocal reactivity to collagen type IV was noted in portal areas, subendothelially. Continued collagen IV immunoreactivity is not surprising considering the age of this animal. These findings coincide with the immunoreactivity of the index case and the lack of reactivity in the older control animals. Interestingly, both hepatocytes and bile duct epithelium from that neonate demonstrated diffuse, strong intracytoplasmic reactivity to CK 8, in contrast to the affected primate, the 1-year-old primate, and 6-year-old primate. The neonate's liver reacted similarly to all other immunohistochemical stains as did the other control animals' livers.
In mammals, the liver is a derivative of the embryonic gut and arises as a ventral outgrowth from the foregut endoderm. This diverticulum develops into 2 outgrowths, a pars hepatica that will form the liver parenchyma and a pars cystica that will form the gall bladder. 8 This development process occurs similarly in humans. Early in gestation, hepatocyte precursor cells surround the mesenchyme of the portal vein and progressively contract to a condensation of cells forming the ductal plate. Ductal plates are selectively deleted or retained with remaining precursor cells forming future intrahepatic bile ducts.
Research performed on human tissue has elucidated cytokeratin and hepatocyte SA expression in the developing precursor cells and differentiating bile duct epithelium and hepatocytes. Primitive hepatocytes or precursor cells express CK 8, 18, and 19. 1 Early in human gestation, primitive hepatocytes committed to form liver parenchymal cells lose CK 19 expression, while those destined to form intrahepatic bile ducts remain strongly reactive for these and acquire, additionally, CK 7 later in gestation. 10 CK 19 is also localized in oval cells in rats during carcinogenesis and liver regeneration. 10 Early hepatic progenitor cells additionally express HepPar1 antigen/hepatocyte SA. As progenitor cells differentiate, those cells programmed to become hepatocytes retain hepatocyte SA reactivity, while those that differentiate to bile duct epithelium lose this reactivity. CD34, a hematopoietic stem cell marker, is expressed by ductal plate cells up to 22 weeks' gestation and by vascular endothelium. 1 Reactivity to CD34 would help support the persistence of hepatocyte precursor cells. CD34 was not useful in this African green monkey; cross-reactivity of the chosen antibody clone was not demonstrated in liver sections examined.
Ductal plate malformation is a basic feature of bile duct disorders such as ADPKD, ARPKD, Caroli disease, Caroli syndrome, CHF, and von Meyenburg complexes. 3 These can be divided into diseases characterized by biliary dilation or ectasia (ADPKD, ARPKD, and Caroli disease) or those characterized by involution or destructive cholangitis (von Meyenburg complexes, CHF, and Caroli syndrome).
In humans, CHF can be characterized by fibrous enlargement of portal tracts with abnormally shaped bile ducts or can be demonstrated as bands of fibrous connective tissue with increased numbers of bile ducts that are in continuity with the rest of the biliary system. 3 A proposed pathogenesis of CHF is that it represents a progressive, destructive cholangiopathy in a fetal liver that has arrested or incompletely remodeled its interlobular ducts. 4 The cholangiopathy may vary in its progression after a few years. CHF in humans is most often associated with renal cystic disease. However, severity of kidney lesions may vary with the age of the individual. Kidneys of perinates and neonates are made up almost entirely of cystic tubules; juvenile disease can present as more irregularly distributed cysts, tubular atrophy, and interstitial fibrosis; older individuals can demonstrate tubule damage without cyst formation. 4
The von Meyenburg complexes comprise a variable number of dilated bile ducts embedded in a fibrous, sometimes hyalinized stroma. 3 It is hypothesized that the lesion is a remnant of ductal plate malformations of the smaller, peripheral branches of the intrahepatic biliary tree.
The lesion described in this African green monkey most closely resembles congenital hepatic fibrosis with von Meyenburg complexes. In humans, CHF is an autosomal recessive disorder often observed with other cystic-forming syndromes, such as ARKPD, Meckel-Gruber syndrome, and Jeune syndrome. Additional lesions such as polycystic kidneys and polydactyly and central nervous system defects were not identified in this primate; therefore, a comparison with other human diseases with multiorgan involvement, such as Meckel-Gruber syndrome, could not be made. Because of the age of the animal and the progressive proposed pathogenesis of CHF and renal lesions, it is possible that renal lesions may have existed, were not evident grossly, and were not identified in the renal section examined. Another hypothesis for the lack of kidney involvement in this primate is that it may resemble the Pkhd1ex40 rodent model more closely than the human disease. Pkhd1ex40 mice express a modified Pkhd1 transcript and develop severe malformations of intrahepatic bile ducts but develop morphologically and functionally normal kidneys. 7 To our knowledge, this is the first reported case of a hepatic ductal plate malformation in a nonhuman primate.
Footnotes
S. M. Wallace is a Major and T. W. Blanchard is a Colonel in the US Army. The opinions or assertions herein are those of the authors and do not necessarily reflect the view of the Department of the Army or the Department of Defense.
