Abstract
Congenital hepatic fibrosis with autosomal recessive or dominant inheritance has been described in humans, cats, piglets, and dogs. In horses, only two cases of congenital hepatic fibrosis have been previously reported. This retrospective study of records from the Institute for Animal Pathology, University of Berne, identified 30 foals with liver lesions compatible with congenital hepatic fibrosis. Anamnestic data revealed clinical signs of severe liver injury in most affected animals. Pathologic examination showed severely enlarged, firm livers with thin-walled cysts. Histologically, the livers showed diffuse porto-portal bridging fibrosis with many small, irregularly formed and sometimes cystic bile ducts. All foals belonged to the Swiss Freiberger breed. Pedigree analysis revealed that the diseased animals could be traced back to one stallion. These results strongly suggest that congenital hepatic fibrosis in Swiss Freiberger horses is a recessively inherited autosomal genetic defect.
Biliary cystic disease of the liver, either solitary or multiple, has been reported in various species. 2 7 9 A widely accepted classification in man 5 distinguishes between solitary cysts of the liver, polycystic disease of the liver (childhood type and adult type), congenital hepatic fibrosis, and congenital dilatation of bile ducts (type I–V).
There is strong evidence that congenital hepatic fibrosis and the childhood type of polycystic liver disease are identical. 3 Because they are often accompanied by cysts in kidneys and the pancreas, they are also referred to as polycystic disease (PD) or polycystic kidney disease (PKD). 1 The childhood type of polycystic disease is primarily characterised by porto-portal bridging fibrosis with abnormally structured and/or dilated bile ducts. 3 The kidneys sometimes contain cysts in the proximal tubules and collecting ducts but are most often normally structured. PKD is one of the most common genetic disorders in humans. The adult type of the disease follows an autosomal dominant trait of inheritance. 9 To date, three different genetic loci have been identified and two of them have been recently cloned. The childhood type is autosomal recessively inherited. 9 The genetic lesion has been localized on human chromosome 6 but has not yet been cloned. 4 9 10
A disease similar to the childhood type in man has been observed in one Swiss Freiberger foal 7 and in a 4-month-old female calf (Van den Ingh, unpublished observations). A disease similar to the adult type of polycystic liver disease in man is recognized in Persian cats and Persian crosses. 2 The disease in cats is characterized by rounded renal cysts, and in about 60% of cases, it is associated with biliary cysts and/or congenital hepatic fibrosis. The Swiss Freiberger is an indigenous horse breed from the western part of Switzerland with a rather high degree of inbreeding. Within the last few years, several foals were seen with a clinical history of liver failure and liver lesions similar to congenital hepatic fibrosis. To confirm an assumed hereditary trait and to investigate the prevalence of this disease in our files, we undertook a retrospective study.
The records of the Institute for Animal Pathology of the University of Berne between 1984 and 1999 were used to identify foals with liver lesions compatible with congenital hepatic fibrosis. Macroscopic findings, prevalence, and clinical pathology data reported here were derived from necropsy reports in the relevant case records. Tissue samples, fixed in neutral buffered formalin and stained with haematoxylin and eosin (HE), were subjected to histologic assessment. Pedigrees of affected foals were, where possible, analyzed.
In our case records, we found 30 foals with liver lesions compatible with congenital hepatic fibrosis. Twenty-one of these foals were female, seven were male, and in two animals, the sex could not be identified because of lack of data. The age of the diseased foals ranged between 1 and 12 months, with an average of 3.7 months. All foals belonged to the Swiss Freiberger breed. In 18 cases, a complete postmortem examination had been performed. In 12 cases, only the liver was submitted for examination.
In most cases, the onset of clinical signs occurred suddenly. Only some foals showed reduced body condition. In the majority of cases, the foals showed signs of severe liver damage. These included jaundice, neurological disorders, and abdominal distension with colic symptoms. Increased respiratory frequency and pyrexia were also common. In some cases, other clinical problems were superimposed. These included pneumonia and, in one case, osteomyelitis. Laboratory findings included increased leukocyte numbers and elevated gamma-glutamyltranspeptidase, alkaline phosphatase, and lactate dehydrogenase values.
Macroscopic examination revealed a severely enlarged liver with a relative weight between 3.4 and 7.8% in all affected foals. The normal relative weight of the liver in horses is 1.2–1.5%. Livers were pale, yellow to grey with a slightly nodular surface, had a prominent reticular pattern on the surface and cut section, and were of firm consistency. In most cases, multiple irregular cysts from <1 mm up to 20 cm in diameter, filled with bile-like fluid, were macroscopically visible (Fig. 1). The hepatic lymph nodes were often enlarged. Apart from enlargement of both kidneys in one foal, no renal lesions were observed and there were no consistent macroscopic changes in other organs. Significant histological changes were seen in all livers, in 10 of 12 examined brains, and in the kidneys of one foal. All livers showed diffuse, marked porto-portal bridging fibrosis without destruction of the limiting plate, thus accentuating the normal lobular pattern (Fig. 2). Within the fibrous tissue, there was proliferation of slightly dilated bile ductules forming a complex branching network and sometimes large biliary cysts lined with cuboidal to low columnar epithelial cells with regularly spaced, basally located nuclei. Some cysts contained sloughed epithelial cells and granular, lightly basophilic amorphous material. Furthermore, there was focal, moderate infiltration with mononuclear and polymorphonuclear leukocytes within the fibrous tissue (Fig. 3). In most cases, the hepatocytes showed evidence of cholestasis and moderate vacuolar changes. In the affected brains, there were small groups of astrocytes with clear swollen nuclei, consistent with Alzheimer type 2 cells. Furthermore, gliosis and slight spongiosis of the thalamus and white matter of the cerebral cortex were present. These lesions are compatible with hepatic encephalopathy. 8 In the animal with the enlarged kidneys, small cysts were observed in the proximal tubules and collecting ducts.
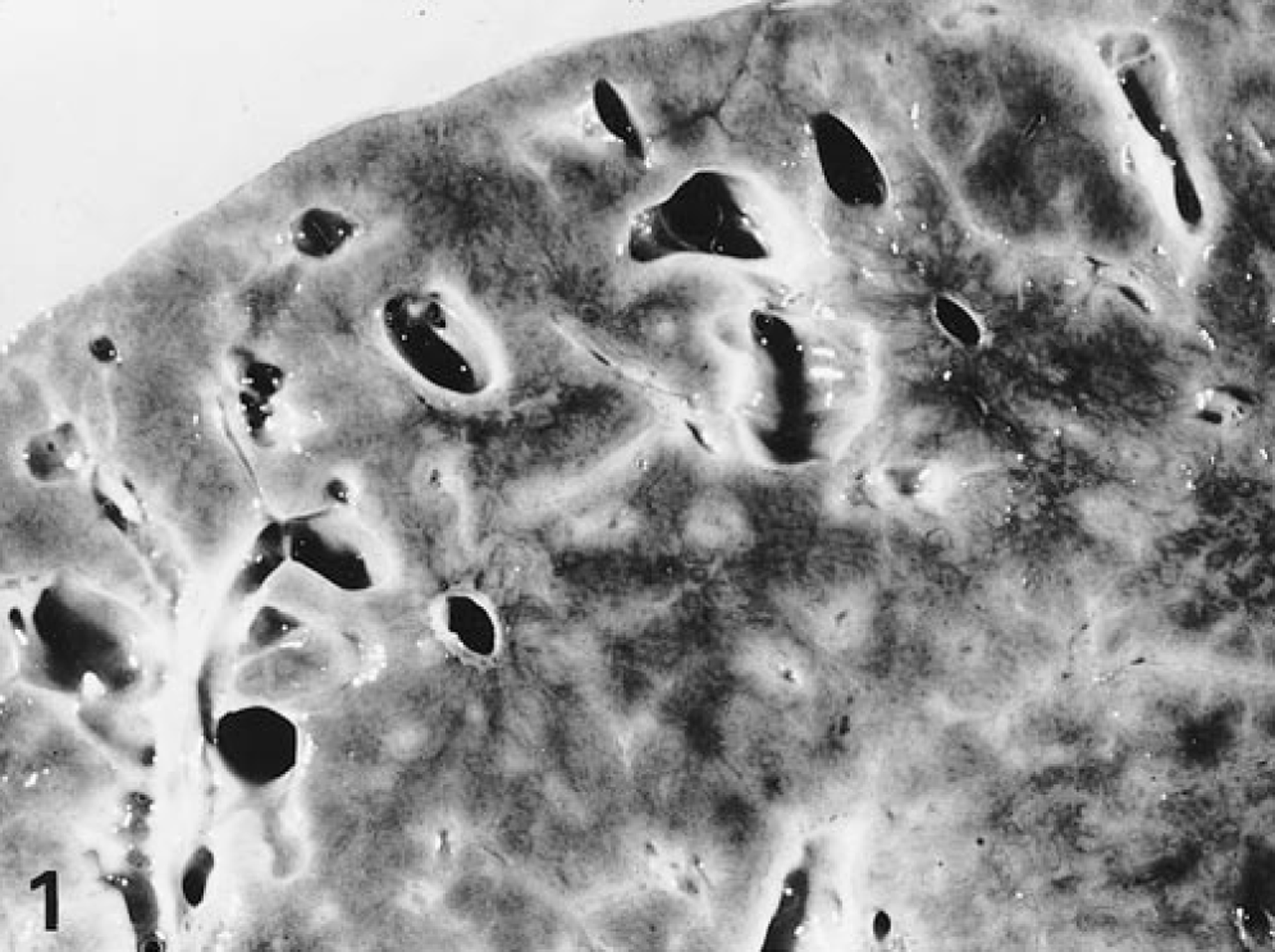
Liver; horse No. 30. Congenital hepatic fibrosis. The cut surface shows a prominent reticular pattern and multiple macroscopically visible, irregular cysts.
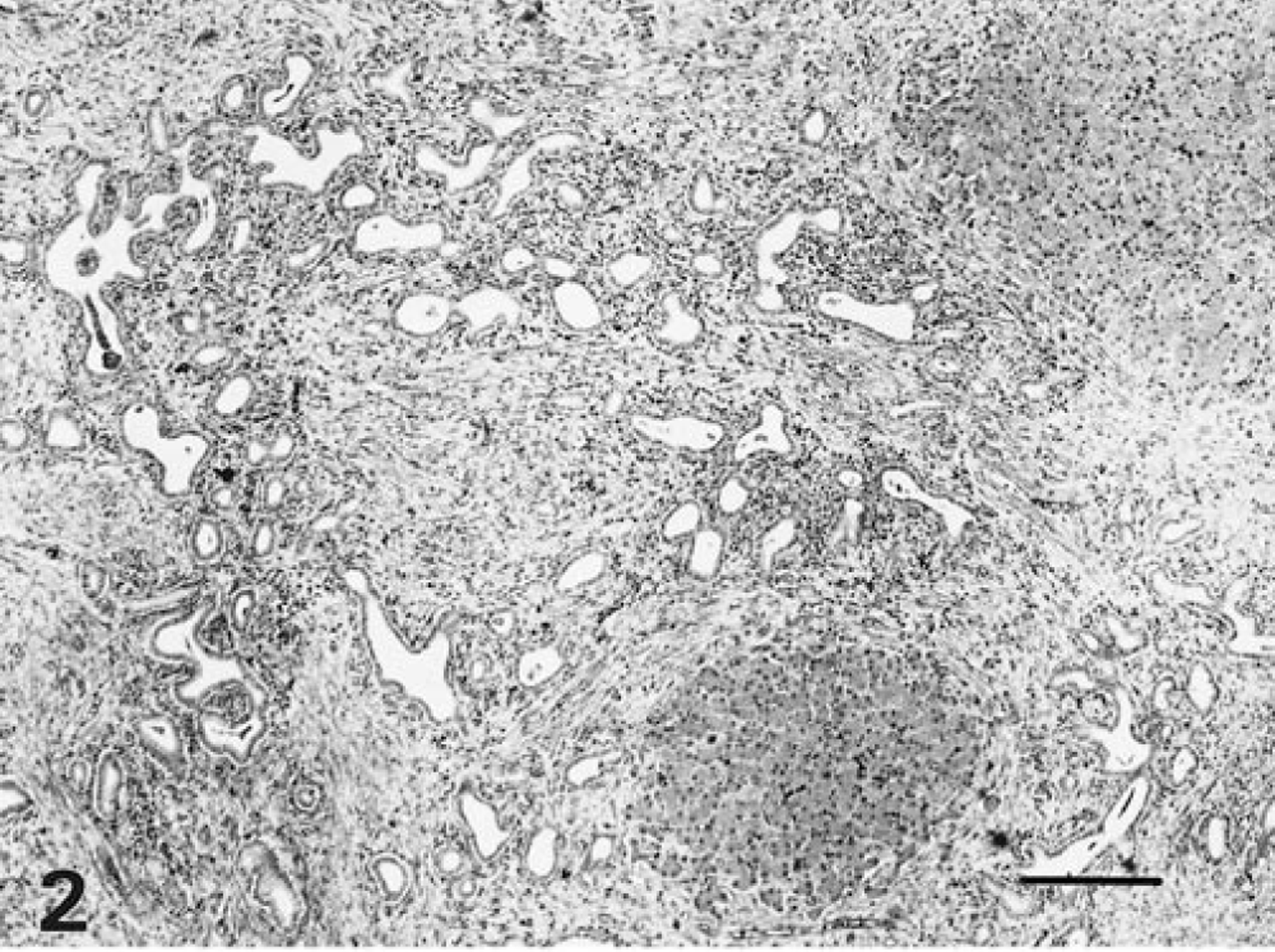
Liver; horse No. 30. Congenital hepatic fibrosis. Note the increased amount of fibrous connective tissue in the periportal area with irregular proliferation of biliary ductules. HE. Bar = 300 µm.
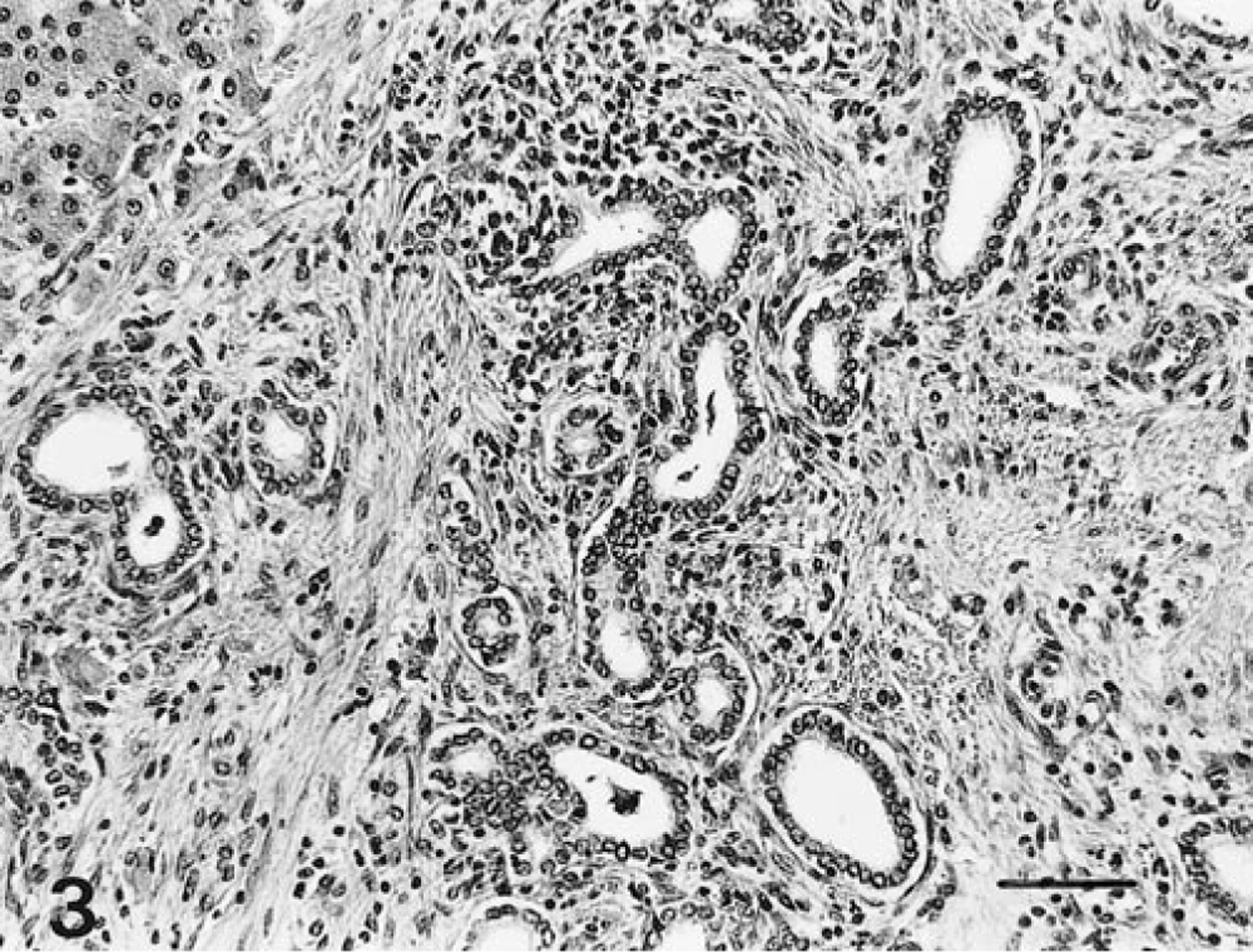
Liver; horse No. 30. Congenital hepatic fibrosis. Note that the cysts are lined by cuboidal epithelium and the moderate infiltration with mononuclear and polymorphonuclear leukocytes. HE. Bar = 100 µm.
Certified pedigrees were available for 23 horses. Ancestry was traced back for up to nine generations. Two stallions featured prominently in the pedigree of all foals. In 18 foals, sire and dam can be traced back to the stallion Elu. In five cases, the sire can be traced back to Elu and the dam can be traced back to Héroique. Both Elu and Héroique can be traced back to Wigar. Twice, siblings from the same sire–dam pairing were affected. Three mares had a history of abnormal offspring from previous pregnancies.
The onset of clinical signs and macroscopic and histologic findings, as well as the results of pedigree analysis suggest that the described disease in Swiss Freiberger foals closely resembles congenital hepatic fibrosis in man. As in man, 6 the liver seems primarily affected and the kidneys only exceptionally show cystic changes. The hepatic lesions in the Swiss Freiberger foal reported earlier 7 were identical to the lesions seen in our foals.
The etiology and pathogenesis of congenital hepatic fibrosis and the precise nature of the liver lesion at the time of birth remain unknown so far. The same stallion occurred within nine generations at maximum on both the dam and the sire side of the pedigree of all affected foals where these data were available. Such information is taken as evidence to support an autosomal recessive pattern of inheritance. The trait of genetic lesions is apparently not sex linked since male and female siblings presented identical liver lesions. The overrepresentation of females in our study is most likely due to the fact that male horses of the Swiss Freiberger breed have less value for the breeder and therefore are not submitted for postmortem examination.
The characterization of the genes responsible for congenital hepatic fibrosis in horses would be the most effective way to prove the genetic basis of the disease but must await a systemic analysis of the equine genome. Although this project is currently in progress, access to a gene map for the horse is not yet possible. At the present stage, a genetic defect can only be evaluated through pedigree analysis, and a decrease in the incidence of a genetic defect can only be affected through pedigree selection. In our case, it is evident that inbreeding among the affected two lines should be avoided through genetic advice to the owners, the veterinarian, and the breeding association.
Footnotes
Acknowledgements
We thank all referring veterinarians, the breeding association of Swiss Freiberger horses, and the breeders who submitted pedigrees for their cooperation.
