Abstract
Congenital hepatic fibrosis is a disorder of biliary system development histologically characterized by diffuse periportal to bridging fibrosis with numerous small often-irregular bile ducts and reduction in the number of portal vein branches. The condition results from abnormal development of the ductal plate, the embryonic precursor to the interlobular bile ducts. It has rarely been reported in veterinary species, and it has never been reported in dogs. This article describes 5 cases of a ductal plate malformation in dogs consistent with congenital hepatic fibrosis. On light microscopy, all 5 livers had severe bridging fibrosis with a marked increase in the number of small bile ducts, which often had irregular, dilated profiles reminiscent of the developing ductal plate. In addition, 80% (4 of 5) of cases lacked typical portal vein profiles. Cytokeratin 7 and proliferating cell nuclear antigen immunohistochemistry was performed on the 3 cases for which paraffin-embedded tissue was available. The bile duct profiles were strongly positive for cytokeratin 7 in all 3 cases, and they were negative for proliferating cell nuclear antigen or only had rare positive cells. All 5 dogs presented with clinical signs of portal hypertension. Congenital hepatic fibrosis should be considered in the differential diagnosis in young dogs that present with portal hypertension and lesions that may have been interpreted as bridging biliary hyperplasia or extrahepatic biliary obstruction.
The ductal plate is an embryonic structure that consists of a 2 cell–thick sleeve of hepatoblasts that abut the mesenchyme surrounding the embryonic portal vein. 14 These cells can be distinguished from other hepatoblasts by the presence of cytokeratins 7 and 19, which are normally restricted to adult biliary epithelium. 1 The ductal plate undergoes remodeling in utero, first forming a double-layered, often-circumferential tubular structure and subsequently remodeling into only a few persistent ductules, which are incorporated into the connective tissue of the developing portal tract to form the mature intrahepatic biliary tree. Incorporation of the maturing bile ducts into the portal tract is likely related to the development of the hepatic arterial branches because incorporation always follows the emergence of the arterial branches.11 Incomplete remodeling results in persistence of excessive embryonic bile duct structures, termed ductal plate malformation. 5 Several types of ductal plate malformations have been described in humans, each occurring during a specific time in embryonic development. Malformations that occur late in development affect the small interlobular bile ducts.
Congenital hepatic fibrosis is a type of ductal plate malformation that affects the small interlobular bile ducts. 6,7 It is an inherited disorder in humans, but it is not well described in veterinary species. Humans with congenital hepatic fibrosis develop bridging bands of fibrous connective tissue that contain numerous small bile ducts, often characterized by irregular contours, contact of the ducts with hepatocytes, and ball-like protrusion of the duct wall into the lumen. 5,10 The fibrosis may be progressive. Typically, inflammation and cholestasis are absent. Portal hypertension with acquired portosystemic shunts and ascites is common and may be due to the effects of progressive fibrosis or to the typical insufficiency of portal veins; portal vein profiles are often hypoplastic with an abnormal branching pattern. 5,6 The liver is usually firm. There may be macroscopic or microscopic biliary cysts and/or acquired portosystemic shunts. Surprisingly, hepatic enzymes and function tests may be normal with no regenerative hyperplasia present—in contrast to end-stage liver, or cirrhosis. 6,7
The few descriptions of congenital hepatic fibrosis in veterinary species include a report in cats with polycystic kidney disease, 18 a report in 30 related Swiss Freiberger foals, 9 rare reports in bovine fetuses or calves, 2,17 and a recent report in an African green monkey. 16 There are also rare reports of congenital cystic liver disease in dogs, including a case series in 7 dogs, 15 single reports of polycystic liver and kidney disease in Cairn Terriers 13 and West Highland White Terriers, 12 and a report of Caroli disease in young dogs. 8 None of these dogs were definitively diagnosed with congenital hepatic fibrosis, however, and all had macroscopically visible cystic lesions, in contrast to the currently presented cases.
The following case series describes ductal plate malformation in 5 dogs with microscopic lesions most consistent with congenital hepatic fibrosis. Histologically, all examined livers had extensive bands of bridging fibrosis containing numerous irregular bile ducts with no evidence of nodular regeneration. Four cases also had absent or hypoplastic portal veins, which is a characteristic finding in congenital hepatic fibrosis.
Materials and Methods
Two of the 5 dogs were treated at the Veterinary Teaching Hospital at North Carolina State University; hepatic biopsy specimens were submitted to the pathology service. Tissue sections from the remaining 3 dogs were submitted from other institutions to Dr John Cullen at North Carolina State University for consultation; 2 were liver biopsy specimens and 1 was a necropsy specimen. Serum chemistry was performed in all 5 cases; bile acids tolerance test was performed in 3 cases. Four cases had abdominal ultrasound.
Tissues were fixed in neutral-buffered formalin, routinely processed, and embedded in paraffin. Sections were cut at 5 μm and stained with hematoxylin and eosin (HE). Histologic sections from each case were examined by at least 2 pathologists.
Immunohistochemistry for cytokeratin 7 and proliferating cell nuclear antigen (PCNA) was performed on the cases for which paraffin-embedded tissue was available (case Nos. 1, 3, 5). Before incubation with primary antibodies, sections of paraffin-embedded liver were placed in a 60°C oven for 25 minutes, deparaffinized, and rehydrated with running tap water.
For cytokeratin 7 immunohistochemistry, commercially prediluted monoclonal mouse anti-human antibody clone OV-TL 12/30 (Dako, Carpinteria, CA) was used. Antigen retrieval was performed using proteinase K (Dako) for 40 minutes. The slide was then incubated with the primary antibody for 60 minutes, with the Mouse Envision polymer (Dako) for 20 minutes, with 3,3′-diaminobenzidine (DAB) for 5 minutes, and with dilute hematoxylin (counterstain) for 5 minutes.
For PNCA staining, commercially prediluted PNCA Clone PC10 (Dako) was used; no antigen retrieval was performed. The slide was incubated with the primary antibody for 10 minutes, with Dako LSAB2 secondary and tertiary reagents for 10 minutes each, with DAB for 5 minutes, and with dilute hematoxylin for 5 minutes.
All immunohistochemistry was performed on an autostainer (Dako). For all immunohistochemical reactions, negative controls using nonimmune serum were treated under the same conditions. Internal positive controls included the normal bile ducts in liver sections for cytokeratin 7 staining. Controls for PCNA included normal liver (with few cells labeled) and brain (no labeled cells).
Results
Clinical History and Antemortem Diagnostic Testing
Table 1 lists signalment, clinical pathology results, and method of diagnostic imaging (ultrasound, biopsy or necropsy). All 5 dogs presented with clinical signs of liver disease, including ascites (3 dogs), vomiting (1 dog) or seizures (2 dogs). Clinical pathology data for the 5 cases revealed mildly to moderately elevated alkaline phosphatase, gamma glutamyl transferase, and alanine aminotransferase. Bile acids were elevated in the 3 cases in which they were measured. Two cases had ammonium biurate crystals in the urine and 3 cases had hypoalbuminemia. Two cases had low blood urea nitrogen and 1 case had elevated bilirubin.
Signalment, clinical pathologic data, and diagnostic imaging in 5 dogs with congenital hepatic fibrosis. a
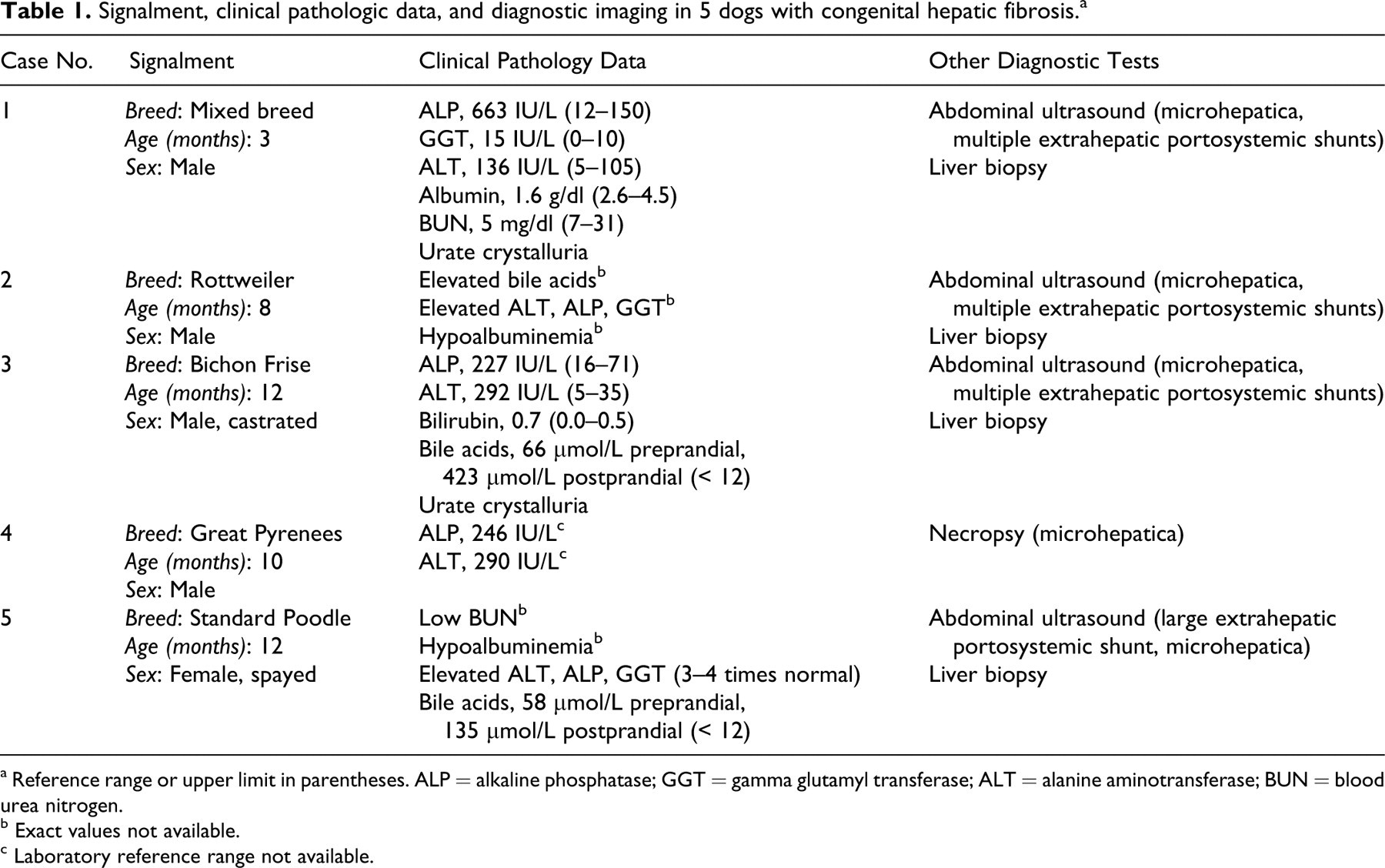
a Reference range or upper limit in parentheses. ALP = alkaline phosphatase; GGT = gamma glutamyl transferase; ALT = alanine aminotransferase; BUN = blood urea nitrogen.
b Exact values not available.
c Laboratory reference range not available.
Abdominal ultrasound was performed on 4 cases. In all cases, microhepatica was appreciated. Four cases had multiple extrahepatic portosystemic shunt vessels; 1 case (No. 5) had a single extrahepatic portosystemic shunt vessel. In case No. 4, microhepatica was observed postmortem.
Histologic Findings
In all cases, the hepatic parenchyma was divided by portal-to-portal bridging bands of dense fibrous tissue with numerous small irregular bile duct profiles (Fig. 1). The bile duct profiles had irregular contours with irregular to absent lumina, and they were lined by oval to cuboidal epithelial cells with finely stippled chromatin and a variably prominent nucleolus (Figs. 2, 3). The ductules were tortuous, with frequent branching (all 5 cases) and ball-like protrusion of the wall into the lumen (3 of 5 cases). Ductular profiles were often present along the edge of the limiting plate, in direct contact with periportal hepatocytes (Fig. 2). Hyperplastic bile duct profiles were accompanied by mild arteriolar hyperplasia in 4 cases, and portal vein profiles were hypoplastic to absent in 4 cases (Fig. 2). Nodular regeneration was absent in all 5 cases, and inflammation was absent to minimal.
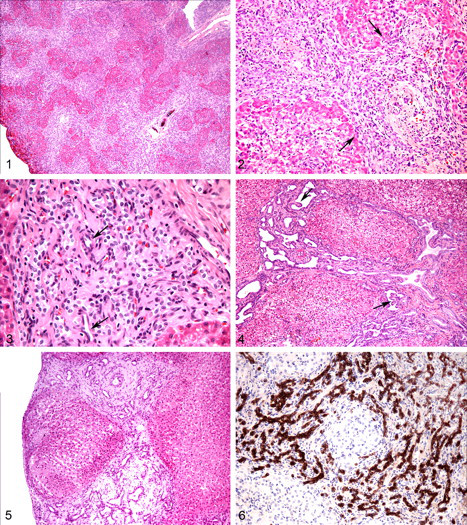
One case (No. 1) had multiple foci of swollen hepatocytes containing one or more clear cytoplasmic vacuoles (Fig. 2). In case No. 3, the immature bile duct profiles were often mildly to moderately ectatic, with prominent ball-like protrusions into the lumen (Fig. 4). One case (No. 4) had more extensive fibrosis, with multifocal disruption of the limiting plate and dissection of periportal hepatocytes. The fibrous matrix in case No. 5 was loose, myxomatous, and quite abundant, with multifocal effacement of the normal lobular architecture (Fig. 5).
The histologic diagnosis was made at necropsy in 1 dog and by examination of a one-time hepatic biopsy specimen in the remaining 4 dogs. These dogs were subsequently lost to follow-up, so the outcome of the disease is unknown.
Immunohistochemistry
To identify the immature structures as biliary in origin, rather than as arterioles or progenitor cells, cytokeratin 7 immunohistochemistry was performed on the cases in which paraffin blocks were available (Nos. 1, 3, 4). All ductular profiles within the fibrous tissue were moderately to strongly positive for cytokeratin 7 (Fig. 6).
Immunohistochemistry for PCNA was also performed on the same 3 cases for which paraffin blocks were available. The ductular epithelial cells were diffusely negative in case No. 4, and only rare cells were positive in case Nos. 1 and 3, indicating that the bile duct epithelial cells had a low rate of replication.
Discussion
Congenital hepatic fibrosis is a disorder of biliary system development resulting from ductal plate malformation of interlobular bile ducts. The disorder is inherited in an autosomal fashion in humans, and the previous report in related Swiss Freiberger horses may indicate a hereditary basis for this disorder in animals as well. 9 Information on littermates was not available in the current canine cases, so a hereditary basis could not be investigated.
All the dogs had bridging hepatic fibrosis with numerous irregular bile duct profiles in ductal plate configuration and a lack of nodular regeneration; these features closely resemble the histologic appearance of human congenital hepatic fibrosis. Clinical signs of portal hypertension commonly appear during childhood or adolescence; however, progression of the human disease is variable, and people can present with hepatic disease at any age, including adulthood. 6,7 All the dogs in this report presented with clinical signs of liver disease and portal hypertension by 1 year of age, supporting a congenital basis.
During embryonic development, primitive bipotential progenitor cells in the liver express cytokeratins 8, 18, and 19. During gestation, those progenitor cells that are committed to form parenchymal cells lose cytokeratin 19 expression, whereas those destined to form bile ducts retain it and gain expression of cytokeratin 7. Previous studies of ductal plate malformations in humans have documented expression of cytokeratins 7, 8, and 19 in the ductal plate cells. 1 In the dogs of this report for which tissue was available for immunohistochemistry, diffuse positive cytokeratin 7 expression was observed in all the immature ductular structures, identifying them as bile ducts rather than arterioles or progenitor cells. Only rare bile duct epithelial cells were positive for PCNA. These results are consistent with a congenital anomaly rather than a ductal reaction to hepatic injury (biliary hyperplasia), which would contain numerous PCNA-positive cells.
A percentage of humans with congenital hepatic fibrosis have concurrent renal cystic disease. 3,5,6 Of the 2 dogs in this series in which renal tissue was available for evaluation, neither had evidence of renal disease. This is in contrast to several previous reports of congenital biliary disease in veterinary species, including dogs. 8,9,12,13,15,17,18 Cystic bile ducts are also common in humans with congenital hepatic fibrosis, and they were present in the cases reported in cats and Swiss Freiberger horses, as well as in the previous reports of congenital biliary disease in dogs. 8,9,12,13,15,18 Only 1 of the current cases (No. 3) had ectatic to mildly cystic bile ducts, which were not visible macroscopically.
Four of 5 dogs had hypoplastic to absent portal veins. Portal vein hypoplasia is a common concurrent histologic finding in humans with congenital hepatic fibrosis. During organogenesis, the development of the biliary system mirrors that of the portal venous system, and ductal plate malformations are often accompanied by portal vein malformations 11,14 that have a “pollard willow” pattern, in which large venous branches abruptly give rise to numerous small branches, without intervening intermediate-sized portal veins. 5,6
There is significant histologic variation in human ductal plate malformations, with congenital hepatic fibrosis often coexisting with other ductal plate malformations, such as Caroli disease and von Meyenburg complexes (intrahepatic bile duct hamartomas), as well as with polycystic kidney disease. 5,6 Similarly, there are subtle histologic differences in some of the canine cases presented here. For example, the liver from the standard Poodle (No. 5) had a more myxomatous stroma than that of the other dogs. Myxomatous stroma is present in another type of ductal plate malformation, termed mesenchymal hamartoma. 3,5 As with congenital hepatic fibrosis, mesenchymal hamartoma is thought to be due to a malformation late in gestation, affecting the small interlobular bile ducts. Therefore, it is possible that case No. 5 represents a ductal plate malformation of the interlobular ducts with characteristics of both congenital hepatic fibrosis and mesenchymal hamartoma. The only reported case of mesenchymal hamartoma in a veterinary species was in an equine fetus. 3 Some of the dogs in the current study had more fibrosis than others, with 1 dog (No. 4) having scattered areas of fibrous tissue that dissected through the hepatic parenchyma.
Despite these subtle differences, all cases had bridging fibrosis with a marked increase of small or irregular bile duct profiles, minimal to absent inflammation, and a lack of nodular regeneration or other histologic evidence of chronic hepatitis. These histologic findings satisfy the criteria for diagnosis of ductal plate malformation, most closely resembling congenital hepatic fibrosis. Ductal plate malformations are not well defined in veterinary species, and it is likely that these disorders in animals do not manifest exactly as they do in humans, because there are subtle interspecies differences in biliary development.
Because of the paucity of portal vein profiles, congenital portosystemic shunt or portal vein hypoplasia might be considered in the differential diagnosis. In congenital portosystemic shunts, however, portal hypertension does not occur, because portal blood is shunted into the lower-pressure vena cava. Furthermore, extensive hepatic fibrosis with numerous bile duct profiles is not a feature of congenital portosystemic shunts. Interestingly, dog No. 5 had a single large extrahepatic shunt identified on abdominal ultrasound, consistent with a congenital portosystemic shunt. Histologically, however, the liver of dog No. 5 had complete lack of lobular architecture with bridging myxomatous to fibrous connective tissue containing many irregular bile duct profiles. It is possible that this case had both a congenital ductal plate malformation and a congenital portosystemic shunt. Portal vein hypoplasia, the preferred term to microvascular dysplasia, is also characterized by a lack of portal vein profiles and can lead to portal hypertension, given that congenital shunting is not part of this disease. 4 Although some cases of portal vein hypoplasia can exhibit bridging fibrosis, this disorder lacks the prominent increase in bile duct profiles seen with congenital hepatic fibrosis.
The prominent biliary proliferation in these cases of congenital hepatic fibrosis might prompt consideration of chronic extrahepatic biliary obstruction. Congenital hepatic fibrosis can be distinguished from chronic extrahepatic biliary obstruction by the typical lack of inflammation or evidence of cholestasis, but in some cases the history and clinical and imaging data will need to be evaluated carefully.
Congenital hepatic fibrosis should be considered in the differential diagnosis for dogs—especially, young dogs—with signs of portal hypertension. The presenting clinical signs of congenital hepatic fibrosis mimic those of chronic end-stage liver disease, although the pathogenesis is very different. The histologic changes also differ from those of end-stage liver disease in that (1) there is no evidence of nodular regeneration despite the presence of bridging fibrosis and (2) the bands of fibrous tissue contain numerous bile duct profiles in ductal plate configuration.
Footnotes
The authors declared that they had no conflicts of interests with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
Acknowledgements
We would like to thank Sandra Horton for her assistance with immunohistochemical staining. Dr. Brown was partially supported by a fellowship from sanofi-aventis, organized by the American College of Veterinary Pathologists and Society of Toxicologic Pathology Coalition for Veterinary Pathology Fellows.
