Abstract
Ductal plate malformations are abnormalities in the liver that arise from inappropriate or incomplete remodeling of the embryologic ductal plate. Various types of ductal plate malformations are reported in the human and veterinary literature, most commonly affecting domestic mammalian species but also fish. We investigated the occurrence and described the histopathologic features of ductal plate malformations in captive snakes. Malformations were identified in 18 snakes: 10 colubrids, 6 vipers, and 2 boids. There was no sex predilection, and the mean age was 17 years. The majority of lesions were incidental with most snakes having one or more comorbidities, most commonly neoplasia or systemic inflammation, that resulted in natural death or euthanasia. Ductal plate malformations in all livers were broadly characterized by a well-demarcated nodule of irregular bile ducts embedded within a varying amount of fibrous stroma. Malformations were further categorized based on the amount of fibrous stroma and dilation of the bile ducts as von Meyenburg complexes, cystic liver disease, and/or an intermediate hybrid subtype representative of cysts arising within von Meyenburg complexes. Histochemical and immunohistochemical staining, including Gomori’s trichome and pan-cytokeratin, respectively, were applied on select cases to confirm histologic features. Malignant transformation was not identified within this population.
Keywords
Ductal plate malformation (DPM) is a term used to describe a group of congenital diseases that result from inappropriate or incomplete remodeling of the embryonic ductal plate. Intrahepatic bile duct formation begins around the eighth week of gestation in humans and on embryonic day (E) 13.5 in mice. 8,34 Intrahepatic bile duct formation begins at the hilus and migrates along the portal branches toward the periphery. As branches of the portal vein grow smaller, so do the associated bile ducts. In the mouse, the hepatic parenchyma prior to E13.5 is composed of bipotential hepatoblasts. At E13.5, hepatoblasts within the mesenchyme and associated extracellular matrix immediately adjacent to portal veins and the portal sinus begin to differentiate into biliary precursor cells. 34 These cells begin to stain faintly positive with cytokeratins 7 and 19 while hepatoblasts in the adjacent parenchyma are only immunoreactive with cytokeratins 8 and 18. 8,34 At E14.5, biliary precursor cells successfully differentiate into biliary epithelial cells (BECs) and are strongly immunoreactive to cytokeratin 19. At E15.5, BECs form a single-layered sheet of cells within the portal mesenchyme termed the ductal plate. 34 The ductal plate becomes bilayered at E16.5 by duplicating the cuboidal BECs to form an inner portal layer and an outer lobular layer with a narrow lumen separating the two layers. 34 At E17.5, multiple dilations form between the two layers resulting in an array of tubules around portal veins. 34 Most undergo apoptosis, but the remaining tubules form bile ducts lined by functional cholangiocytes. 8,34
If there is complete failure or inappropriate formation of the ductal plate, the result is one or more benign hepatic lesions that are collectively termed ductal plate malformations (DPMs). In human and veterinary medicine, DPMs are often characterized based on (1) location—intra- versus extra-hepatic; (2) size of the affected bile ducts—small, medium, or large—which often correlates with location along the biliary tree; and (3) histological morphology. Five major DPMs are reported in the literature and include choledochal cysts, Caroli’s syndrome/disease, polycystic liver disease, congenital hepatic fibrosis, and von Meyenburg complexes. 7,8,35 Choledochal cysts arise from extrahepatic ducts and are histologically characterized as a single, large extrahepatic dilated duct with a fibrotic wall. 35 Caroli’s disease and Caroli’s syndrome affect large-caliber extra- and intrahepatic bile ducts resulting in segmental or isolated dilation of large-caliber bile ducts. Caroli’s syndrome, unlike Caroli’s disease, is often associated with congenital hepatic fibrosis and subsequently results in portal hypertension. 8 Congenital hepatic fibrosis affects the small-caliber bile ducts and is characterized by hepatic parenchymal loss with replacement by interlobular small bile duct proliferation. 5 Polycystic liver disease is characterized by multiple cysts affecting the medium-caliber bile ducts multifocally throughout the liver. This disease is associated with polycystic kidney disease and/or congenital hepatic fibrosis. 11 Last, von Meyenburg complexes, previously known as biliary hamartomas, are benign, incidental, typically subcapsular, well-demarcated nodules composed of dilated small bile ducts surrounded by fibrous stroma. 3,7,27 These have been associated with malignant transformation into cholangiocarcinoma as well as the hypothesized origin of cyst formation in some cases of adult-type polycystic liver disease due to progressive dilation and fluid filling of the bile ducts. 8,17,29,31
The purpose of this retrospective study was to investigate the occurrence and describe the histopathologic features in snakes with DPMs submitted to North Carolina State University College of Veterinary Medicine (NCSU CVM) from the years 2008 to 2018. Comorbidities in this population were also documented.
Materials and Methods
Study Population
Cases submitted to the surgical or necropsy service at NCSU CVM between 2008 and 2018 were considered for this study. A search of the database yielded 18 snakes out of a total of approximately 260 snake-specific tissue submissions, of which there were 204 necropsies and 56 biopsies. Cases were received from 4 facilities, including the North Carolina Zoo, North Carolina Museum of Natural Sciences, North Carolina Aquarium at Pine Knoll Shores, and the North Carolina Aquarium on Roanoke Island. Signalment, clinical histories, and supplemental diagnostic tests were not available for all cases.
Histopathology
Tissues were fixed in 10% neutral buffered formalin for a minimum of 48 hours prior to being processed. All paraffin-embedded tissues were sectioned at 6 µm and mounted on slides that were routinely stained with hematoxylin and eosin for histologic review. Four cases (cases 2, 5, 13, and 14) with representative lesions were further evaluated with Gomori’s trichrome. Findings were diagnosed according to the histomorphology of the malformed bile duct profiles and then further classified based on the degree of cystic dilation and the amount of associated fibrous connective tissue. Small subcapsular lesions composed of minimally ectatic small-caliber bile ducts embedded within a moderate to abundant dense stroma were classified as von Meyenburg complexes. Larger, often multifocal, lesions featuring moderate to markedly dilated bile ducts with flattened epithelium embedded within a scant amount of dense stroma were classified as cystic liver disease. The remaining lesions, which had features observed in both von Meyenburg complexes and cystic liver disease, were classified as an intermediate subtype of DPM representative of cystic liver disease arising within a von Meyenburg complex.
Immunohistochemistry
Immunohistochemistry was performed on formalin-fixed, paraffin-embedded tissues from selected cases (cases 5, 13, and 14) based on the desired subtype. Pan-cytokeratin immunohistochemistry was performed using an automated stainer (Autostainer Link 48, Dako). The tissues were deparaffinized in xylene and rehydrated through a graded series of ethanol to running water. The antigen retrieval of choice was ready-to-use proteinase K incubated for 10 minutes at room temperature. The remaining incubations were also done at room temperature. Endogenous peroxidase was blocked using 3% hydrogen peroxide for 10 minutes. Slides were immediately placed into Tris-buffered saline and this step was repeated after each sequential step. The primary antibody used was mouse monoclonal pan-cytokeratin (AE1/AE3; Dako) at a dilution of 1:50 and was incubated for 30 minutes. A tertiary component using Dako Envision + Mouse polymer was applied for 30 minutes. Negative controls were run by omitting the primary antibody. Cross sections of mammalian small intestine and internal control tissues (snake intestinal, renal and pulmonary epithelium, and normal bile ducts in unaffected sections of liver) with expected positive immunostaining for each snake were used as positive controls. 3,3′-Diaminobenzidine (DAB) chromogen was applied for 5 minutes. The slides were counterstained with hematoxylin for 5 minutes, rinsed, and then dehydrated.
Results
Study Population
DPMs were identified in 18 captive snakes submitted to NCSU CVM between 2008 and 2018 by 4 collaborative facilities. The North Carolina Zoo submitted 10 cases, followed by 5 cases from the North Carolina Museum of Natural Sciences, 2 cases from the North Carolina Aquarium at Pine Knoll Shores, and 1 case from the North Carolina Aquarium on Roanoke Island. Affected snakes included a variety of species, including 4 rattlesnakes (Crotalus sp.), 3 corn snakes (Pantherophis guttatus), 2 copperheads (Agkistrodon contortrix), 2 desert rosy boas (Lichanura trivirgata gracia), and 1 Huachuca mountain kingsnake (Lampropeltis pyromelana woodini), eastern milk snake (Lampropeltis triangulum triangulum), rough green snake (Opheodrys aestivus), scarlet kingsnake (Lampropeltis elapsoides), Sonoran gopher snake (Pituophis catenifer affinis), eastern kingsnake (Lampropeltis getula), and northern pine snake (Pituophis melanoleucus). There was no sex predilection (10 males and 8 females). The mean age was 17 years, with ages ranging from 10 to 23 years. All cases were submitted as necropsies. In 5 cases, the submission did not contain any descriptions of hepatic abnormalities. Of the remaining 13 snakes, all were described as having hepatic nodules corresponding histologically to DPMs. These varied grossly based on classification but were all well-demarcated. Cystic DPMs were large (up to 60 mm in diameter) intraparenchymal to subcapsular with a convex or concave thin-walled appearance and filled with clear green to yellow fluid. The von Meyenburg complexes were typically smaller (1-3 mm in diameter) subcapsular, slightly collapsed, and lighter brown compared to the adjacent parenchyma. The intermediate subtype varied widely from 1 to 30 mm. The signalment and clinicopathological findings from each case are summarized in Supplemental Table S1.
Microscopic Features
All lesions were characterized as fibrocystic diseases, specifically cystic liver disease, von Meyenburg complexes, or a hybrid intermediate subtype likely representing cystic liver disease arising from von Meyenburg complexes. DPMs were classified based on (1) the amount of fibrous stroma and (2) the variation in cystic dilation of the bile ducts. Of the 18 total snakes, 10 had multifocal DPMs and 8 had a focal lesion identified histologically. For snakes with multifocal DPMs, all lesions in a given snake were diagnosed as the same fibrocystic disease, except for case 8, which had evidence of both polycystic liver disease and the intermediate subtype of DPM.
Cystic DPMs representative of cystic liver disease were identified in 5 snakes (cases 4, 8, 11, 12, and 13); these were well-demarcated and compressive with marked cystically dilated bile duct profiles that were lined by flattened biliary epithelial cells surrounded by a minimal amount of dense fibrous connective tissue (Fig. 1). These lesions were predominately small, ranging from 2 to 4 mm in width. In contrast, DPMs with a more robust fibrous component similar to those described in von Meyenburg complexes were identified in 5 other snakes (cases 2, 3, 7, 9, and 15). These DPMs were characterized by numerous irregular small-caliber bile duct profiles that had minimal to no ductal ectasia and were lined by a single layer of well-differentiated cuboidal epithelial cells. These were embedded within a uniformly moderate to robust, dense fibrous connective tissue that did not compress the adjacent parenchyma (Fig. 2). They were typically found immediately beneath the liver capsule and generally were the smallest lesions. In von Meyenburg complexes, atrophied hepatocytes were observed within the lesions. The stroma of the cystic DPMs was often too compressed to detect intralesional hepatocytes. Additionally, cystic DPMs were more frequently surrounded by a thin rim of atrophied hepatocytes, whereas the smaller von Meyenburg complexes did not have compression of the adjacent parenchyma.
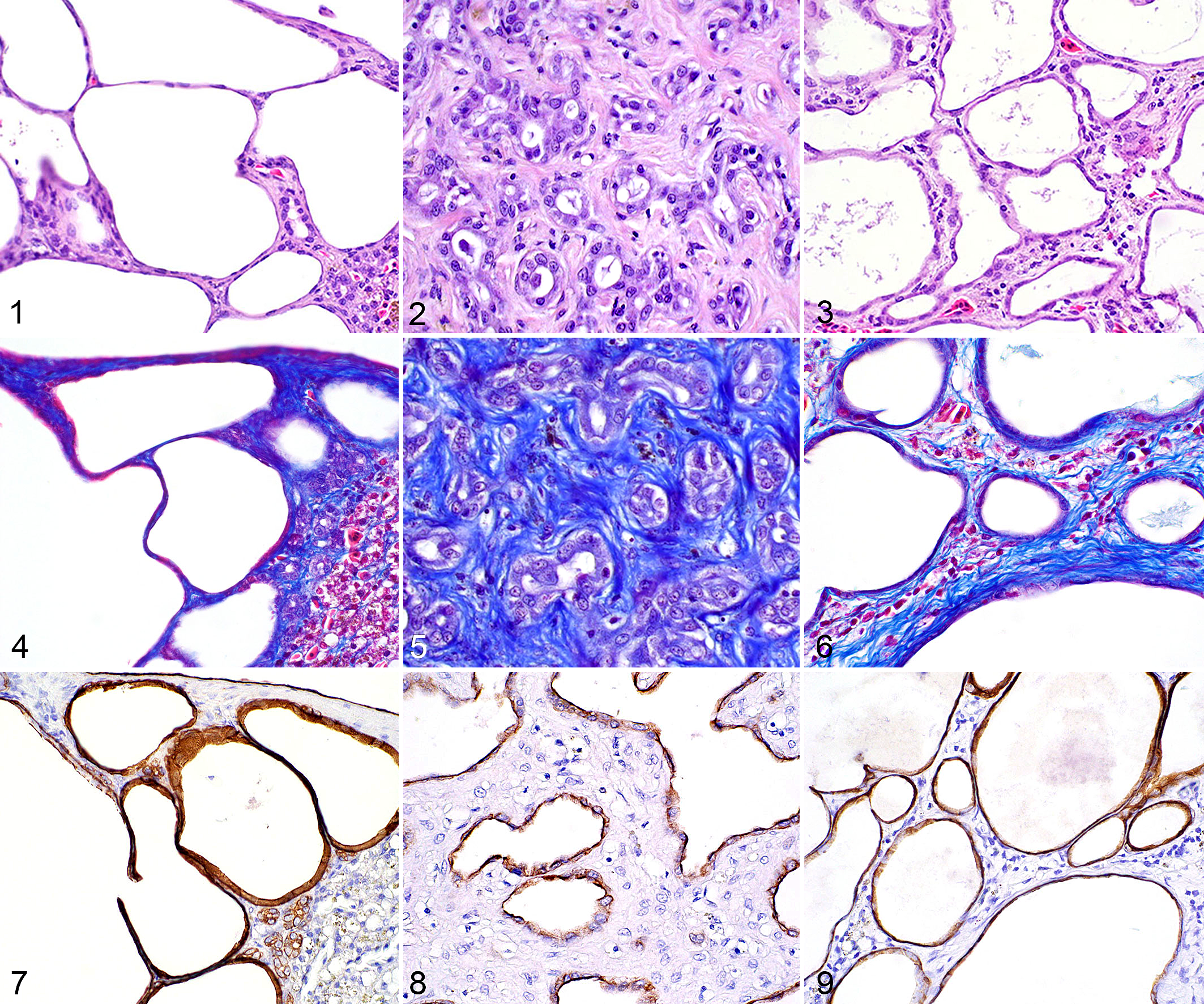
Ductal plate malformation (DPM), liver, snake. Cystic liver disease (Figs. 1, 4, 7), von Meyenburg complex (Figs. 2, 5), and intermediate subtype (Figs. 3, 6, 8, 9).
The majority of DPMs, observed in 9 snakes (cases 1, 5, 6, 8, 10, 14, 16, 17, and 18), were classified as intermediate, or a hybrid, subtype with features of cystic liver disease and von Meyenburg complexes. One snake, case 8, had 2 discrete types of DPMs: a cystic and an intermediate DPM. In our study population, the intermediate subtype DPMs had bile ducts that were often dilated and supported by varying degrees of stroma. The stromal component was more extensive than the cystic liver disease variants and ranged from scant and dense to abundant and loose or densely packed. In this subtype, the bile ducts were lined by attenuated to cuboidal epithelial cells and were moderate to markedly dilated to occasionally ectatic (Fig. 3). These DPMs ranged in size but were often larger than the previously described DPMs and sometimes contained variable areas of small bile ducts embedded within a dense fibrous stroma or ectatic bile ducts supported by a scant fibrillary stroma. Rarely, they were large enough to cause significant distortion of the hepatic architecture. Chronic inflammatory cells were occasionally observed within the stroma of the DPMs, but without a preponderance in any subtype. The type of DPM diagnosed for each snake is listed in Supplemental Table S1.
One case from each of the different subtypes were evaluated with additional histochemical and immunohistochemical stains. Gomori’s trichrome was applied to detect collagen fibers, allowing for precise visualization of the fibrous stroma within the lesions. As expected, cystic DPMs had minimal collagen deposition which was significantly compressed and predominately confined to the peripheral edge of the lesion (Fig. 4). In contrast, the predominating feature of von Meyenburg complexes was the collagenous component that surrounded fewer and less ectatic small-caliber bile duct profiles (Fig. 5). Intermediate subtypes had more collagen than the cystic subtype, but it was less dense than a von Meyenburg complex (Fig. 6). Pan-cytokeratin was applied to confirm the epithelial origin of the ductal structure lining cells. In all cases, attenuated to cuboidal epithelial cells had moderate to strong cytoplasmic staining, indicative of an epithelial origin (Figs. 7–9).
Comorbidities
The 3 most common comorbidities identified were neoplasia, localized or systemic inflammation, and renal abnormalities. Neoplasia was most frequent with 14 tumors identified in 9 snakes. Non-hepatic tumors included myelolipomas, sarcomas (fibrosarcoma, undifferentiated sarcoma, subcutaneous soft tissue sarcoma, histiocytic sarcoma), colonic adenocarcinoma, chromatophoroma, ovarian dysgerminoma, and a metastatic neuroendocrine tumor of unknown origin. Neoplasms originating from the hepatic parenchyma included hepatocellular adenoma (cases 8 and 9) and hepatocellular carcinoma (case 9). Occasionally, primary hepatic and metastatic neoplasms abutted DPMs; however, no evidence of malignant transformation of a DPM was observed in any case. In one case (case 1), a DPM abutting a neuroendocrine tumor of unknown origin contained multifocal areas of biliary dysplasia with irregular papillary projections into the lumens. In another snake (case 10), there was a focal cholesteatoma, a pseudoneoplastic lesion of the middle ear. Systemic or severe localized inflammatory lesions in various organs were present in 10 snakes and had discrete granulomas or focally extensive or multiorgan, ulcerative, granulomatous or granulocytic inflammation. Affected organs included kidney, heart, liver, lungs, cloaca, oviduct, intestines, subcutis, bone, thyroid gland, and blood vessels. Nonneoplastic cystic lesions, not arising in the liver, were found in 4 snakes with concurrent DPMs. Affected organs included kidney (cases 8, 15, and 17), testes (case 17), and epididymis (case 7). Additional renal lesions included renal tophi in 5 snakes and various forms of nephritis in 4 snakes (glomerulonephritis, granulocytic nephritis, tubulointerstitial nephritis, and interstitial nephritis). Additional findings were considered incidental and are listed in Supplemental Table S1.
Discussion
To our knowledge, this is the first case series documenting and classifying DPMs in captive snakes. In this 10-year study, 18 snakes had histomorphologic features supportive of solitary or multifocal DPMs. The DPMs were all diagnosed from necropsy submissions but were not considered a cause of death or morbidity in any of the cases. Rarely, they became large enough to cause significant distortion of the hepatic architecture. The 3 most common comorbidities were neoplasia, localized or systemic inflammation, and renal lesions. Histochemical and immunohistochemical stains were applied to select cases with representative lesions. Gomori’s trichrome was selected to document the amount of fibrous connective tissue. The stain enhanced the visualization of the connective tissue within the DPMs: an abundant amount of fibrous stroma in the smaller subcapsular DPMs and minimal, markedly compressed collagen in the cystic variants. Pan-cytokeratin, an immunohistochemical marker for epithelial cells, was selected to definitively confirm that the markedly attenuated cells lining the cystically dilated DPMs were truly epithelial. A limitation to this study is the use of pan-cytokeratin rather than a biliary specific epithelial marker, such as cytokeratin 19, known to be reactive in reptiles, which was not readily accessible. Although we cannot definitively confirm the epithelium is biliary, given the histomorphologic features, other nonbiliary differentials in the liver would be unlikely, so the use of pan-cytokeratin was reasonable for this study.
In human and veterinary medicine, DPMs are often characterized based on location, size of the affected bile ducts, and histological morphology. The 5 major DPMs reported in the literature include choledochal cysts, Caroli’s syndrome/disease, polycystic liver disease, congenital hepatic fibrosis, and von Meyenburg complexes. 8,35 In our study population, 5 snakes had well-demarcated, small DPMs located in the subcapsular region of the liver, which were predominately composed of a fibrous stroma surrounding small-caliber bile ducts with minimal to no ductular ectasia. Given the location and histomorphologic appearance of the lesion, these DPMs were consistent with von Meyenburg complexes described in the mammalian literature. Previously known as biliary hamartomas, von Meyenburg complexes are benign malformations of the hepatobiliary system characterized by well-demarcated nodules composed of small bile ducts surrounded by fibrous stroma. 3,7 They are typically located at the periphery of the liver (subcapsular) and are clinically asymptomatic, benign, congenital, incidental lesions typically found at surgery or postmortem examination. 7,27
Another type of DPM observed in 5 snakes was a cystic variant similar histologically to polycystic liver disease affecting the medium-caliber bile ducts in mammals and fish. This cystic variant was composed of markedly ectatic bile duct profiles lined by attenuated epithelial cells surrounded by a minimal amount of compressed fibrous connective tissue. Polycystic liver disease in humans is a hereditary disease characterized by multiple cysts affecting the medium-caliber bile ducts multifocally throughout the liver. The autosomal dominant form is associated with autosomal dominant polycystic kidney disease, also known as the adult form, which is often observed for the first time around puberty. The childhood form is associated with autosomal recessive polycystic kidney disease which is often associated with congenital hepatic fibrosis. 11 Persian cats and Bull terrier dogs are two domestic species that have autosomal dominant polycystic kidney disease associated with defects in PDK1 and PDK2 that encode polycystin 1 and 2, respectively. 12,22 In one study, 68% of cats with polycystic kidneys also had either uni- or multilocular hepatic cysts and/or congenital hepatic fibrosis; one cat also had pancreatic cysts. 4 Lesions resembling childhood-type polycystic disease, probably with autosomal recessive inheritance, have been described in Cairn Terrier and West Highland White Terrier puppies. 23,24 Polycystic liver disease is also reported in numerous nondomestic animal species, including multiple species of fish 18,26,28 and individual reports in alpacas, 10 golden hamsters, 30 and senile chamois. 13
Last, the most commonly observed DPM in our cohort was an intermediate subtype with features supportive of a hybrid variant of cystic disease arising from von Meyenburg complexes with progressive dilation due to lining cell secretion. Nine snakes had this hybrid intermediate subtype, one of which (case 8) also had a cystic variant of DPM so this snake was counted in both categories (cystic and intermediate variants). These DPMs had varying degrees of ductular ectasia and amounts of supporting stroma that often had features characteristic of both cystic liver disease and von Meyenburg complexes. Since these DPMs had overlapping features observed with cystic disease and von Meyenburg complexes, these lesions were categorized into their own classification as an intermediate, or hybrid subtype.
Histologic features characteristic for choledocal cysts, Caroli’s disease/syndrome, or congenital hepatic fibrosis were not observed in our population. Choledochal cysts arise from extrahepatic ducts and are histologically characterized as a single, large dilated duct with a fibrotic, often inflamed, wall. 35 There is debate among researchers in the human literature as to whether these lesions are true DPMs versus pancreaticobiliary ductal unions resulting in inflammation and dilation. 20,33 In the veterinary literature, these lesions are rare and most commonly reported in cats. 2,32 Caroli’s disease and Caroli’s syndrome are rare autosomal recessive congenital disorders in humans that affect large-caliber extra- and intrahepatic bile ducts. Classic Caroli’s disease is considered to be the rarer, simple form characterized by segmental or isolated dilation of large-caliber bile ducts. Caroli’s syndrome is a more common but complex form of Caroli’s disease that in addition to large-caliber bile duct dilation there is also dilation of smaller ducts. Caroli’s syndrome, unlike Caroli’s disease, is often associated with congenital hepatic fibrosis and subsequently results in portal hypertension. 8 In veterinary medicine, most cases involve young dogs presenting with polyuria/polydipsia, vomiting, and less frequently ascites, and often have elevated serum bile acids and ALP. 15,21 In one study, 8 dogs from various breeds had ultrasonographic evidence of cystic dilations consistent with Caroli’s disease. 15 Congenital hepatic fibrosis is an autosomal recessive disease in humans but genetic inheritance is not well documented in veterinary medicine. The disease affects the small-caliber bile ducts and is characterized by hepatic parenchymal loss with replacement by interlobular small bile duct proliferation, which can be minimally ectatic to cystically dilated, and periductal fibrosis. Portal hypertension, portosystemic shunts, and ascites are common and may be due to the effects of progressive fibrosis or to the insufficiency of portal veins. Portal vein hypoplasia is a common concurrent histologic finding in humans. 5 Congenital hepatic fibrosis has been reported in dogs, cats, calves, horses, and nonhuman primates but to the authors’ knowledge has not been reported in snakes. 5,9,16,25,36 –38
Historically, DPMs have been misdiagnosed as benign biliary neoplasms, such as biliary adenoma or biliary cystadenoma, because they share histopathologic features: tubules of well-differentiated biliary epithelium supported by moderate amounts of intervening stroma. The difficulty distinguishing DPMs from benign biliary neoplasms has made it difficult to assess the true incidence of the two processes in human and veterinary pathology. 3,6 Differentiation can be challenging; however, features favoring an adenoma are a single lesion with compression of adjacent tissue and lack of intralesional hepatocytes, because biliary adenomas grow by expansion, resulting in spherical masses that compress the adjacent parenchyma and create a grossly visible bulging appearance of the subcapsular parenchyma. A biliary adenoma should not have hepatocytes embedded within the supporting stroma. 6,14 In contrast, DPMs arise from incomplete or inappropriate formation of the ductal plate which during improper development can result in hepatocytes being embedded within the stroma surrounding the bile ducts. 6 In our snake population, von Meyenburg complexes were noncompressive and had rare hepatocytes within the stroma. The cystic DPMs had stroma that was often too compressed to definitively determine if hepatocytes were present; however, these lesions resembled cystic liver disease rather than a neoplastic process due to the often-multifocal nature, minimal compression of the adjacent parenchyma, lack of mitotic figures, and minimal cellular variation. Additionally, over 50% of the cases had multifocal DPMs and/or renal cysts, supportive of congenital malformations rather than neoplasia. No biliary neoplasms were observed in our snake population and furthermore neoplastic transformation of DPMs was not observed.
In general, neoplastic transformation of DPMs is not well documented in the veterinary literature. In the human literature, malignant transformation is most frequently reported in Caroli’s disease, with approximately 7% of cases ultimately developing cholangiocarcinoma; less frequently reported are cholangiocarcinomas arising within von Meyenburg complexes. 1,17,19,29,31 In humans, there is speculation that increased risk of malignancy in DPMs may be due to bile stasis and prolonged exposure of the parenchyma to potential carcinogens. 31 In our study, one case (case 1) had a DPM with areas of dysplasia, but no overt evidence of neoplastic transformation was observed. We suspect that areas of dysplasia may have progressed to neoplasia if the animal had survived.
Generally, the prevalence DPMs in snakes is low; approximately 7% of case submissions from relatively aged captive populations submitted to the surgical or necropsy service at NCSU CVM. In snakes, these are asymptomatic, incidental, benign lesions found at postmortem examination with variable histomorphologic features. The location of the DPM in the liver of snakes may not be as helpful in identifying the specific type of DPM as in other species. We found that subcapsular DPMs with small-caliber bile ducts embedded in a dense fibrous stroma most closely resemble von Meyenburg complexes and that the cystic DPMs are similar to cystic liver disease reported in other veterinary species. The majority of our DPMs were a hybrid intermediate subtype of the two previously described DPMs that resulted in cystic lesions arising from von Meyenburg complexes. Lesions resembling choledocal cysts, Caroli’s disease/syndrome or congenital hepatic fibrosis were not identified, and malignant transformation was not observed. In conclusion, DPMs should be considered as a nonneoplastic differential diagnosis for single or multifocal mass-like lesions in the liver of snakes.
Supplemental Material
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211011941 - Ductal Plate Malformations in Captive Snakes
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211011941 for Ductal Plate Malformations in Captive Snakes by Olivia M. Patania, Brigid V. Troan and John M. Cullen in Veterinary Pathology
Footnotes
Acknowledgements
We thank the four facilities that submitted cases. We also thank the NCSU CVM histology laboratory for processing of histologic preparation and IHC staining.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
