Abstract
Chromobacterium violaceum infections are highly fatal and rarely reported in domestic animals. This report describes a fatal case of C. violaceum septicemia in a 7-day-old female beef calf. The calf had necrosuppurative omphalophlebitis, necrotizing interstitial pneumonia, necrosuppurative hepatitis and splenitis, anterior uveitis with hypopyon, suppurative polyarthritis, and disseminated hemorrhagic meningitis with multifocal necrotizing encephalitis. Histologically, clusters of gram-negative bacilli were found in many of the lesions. C. violaceum was isolated in high numbers from the lungs, liver, spleen, carpus, and in pure culture from the cerebrospinal fluid. To our knowledge, this is the first case report of chromobacteriosis in a calf.
Keywords
Chromobacterium violaceum, a gram-negative saprophytic bacillus, is an opportunistic pathogen of mammals. 20 It is found in soil and water of tropical and subtropical regions around the world. Infections by C. violaceum are uncommon and frequently associated with high mortality. In the USA, fatal cases have been reported in humans, 16, 19, 24 dogs, 8 pigs, 22 sheep, 6 gibbons, 13 colobus monkeys, 14 a macaque, 18 and a red panda. 9 Lesions documented in animals include visceral abscesses and necrosis, pleuropneumonia, and pericarditis. Natural or experimental C. violaceum infection has not been reported in cattle. This report describes a case of acute, fatal septicemia associated with C. violaceum infection in a calf.
A 7-day-old, female, mixed-breed beef calf was presented to the Texas A&M University Veterinary Medical Teaching Hospital with clinical signs of lateral recumbency, dehydration, scleral injection, anterior uveitis with hypopyon in the right eye (Fig. 1), dyspnea, and navel enlargement. The calf had a packed cell volume of 20% (reference values, 24–46%) and serum total protein of 3.2 g/L (reference values, 5.95–8.0 g/L). The calf died despite treatment including blood transfusion, respiratory support, intravenous fluids, ceftiofur, and flunixin meglumine administration.
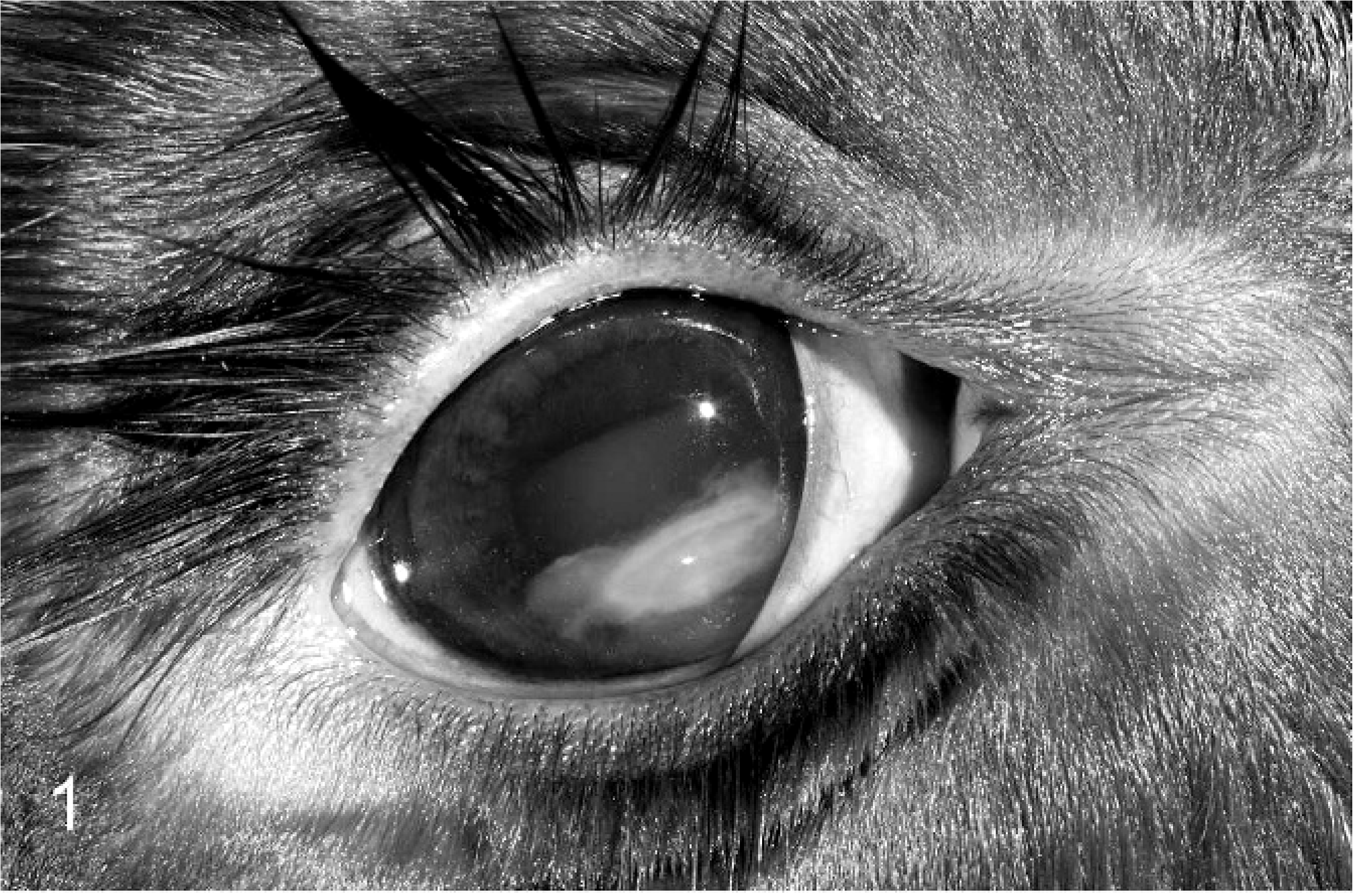
Eye; calf. Suppurative anterior uveitis in the right eye with purulent exudate filling the anterior chamber (hypopyon).
At necropsy, the nasal mucosa and conjunctiva were dark red and swollen bilaterally. The mucosa in the anterior nasal cavity was covered with a diphtheritic membrane that occluded the passage. The lungs were dark red, uncollapsed, heavy, and firm. The lung exuded a clear frothy fluid on cut section (Fig. 2). The pericardium was filled with serosanguineous fluid. The synoviae of the carpal, stifle, and tarsal joints were thickened, and these joints contained orange to red, watery synovial fluid with tan coagula. The meninges were petechiated (Fig. 3), and tan exudate covered the ventral aspect of the brainstem. Cerebrospinal fluid was turbid, brown, and contained tan, fibrinous strands. The umbilicus was edematous and had an accentuated, congested vasculature. Samples of lung, liver, spleen, brain, a swab from a carpus, and cerebrospinal fluid (CSF) were submitted for bacterial culture.
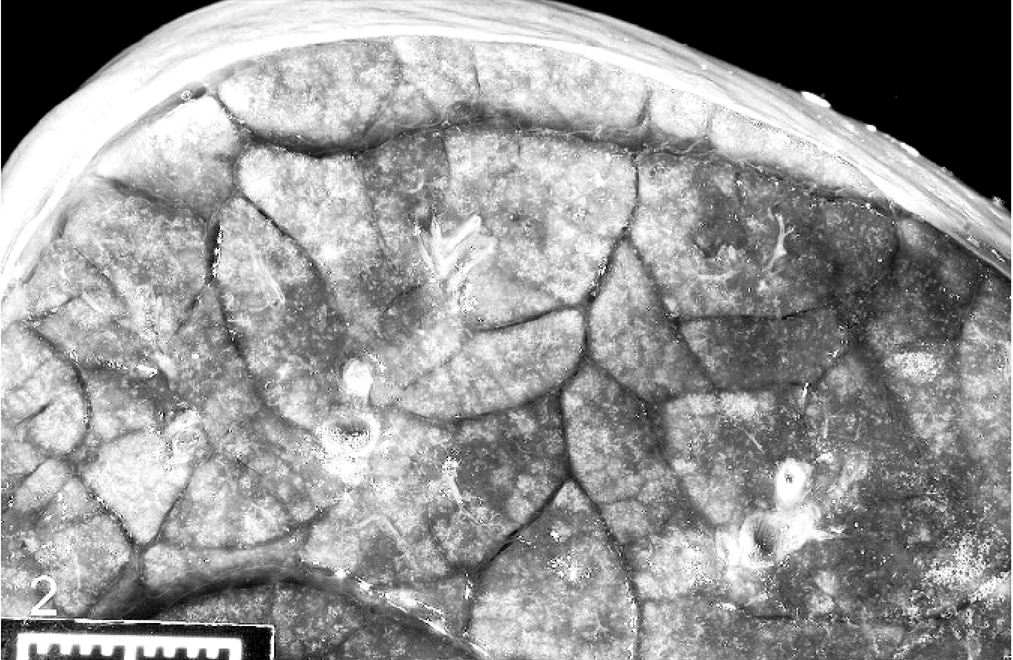
Lung; calf. Cut surface of the lung, showing expansion of the interlobular septa. Bar = 1 cm.
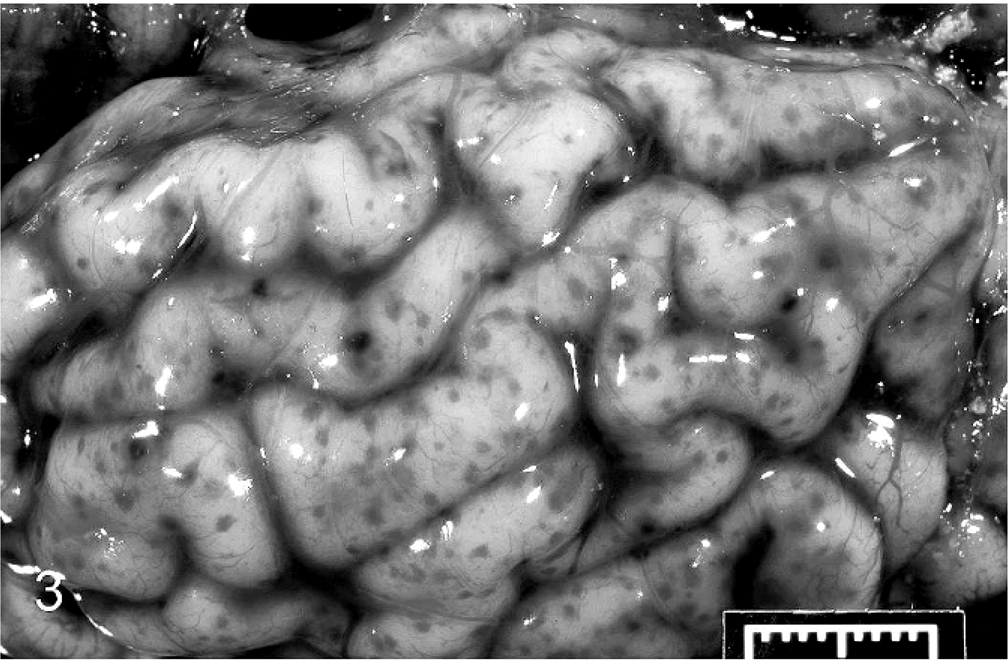
Brain; calf. Petechiae and ecchymoses on the meninges of the cerebrum. Bar = 1 cm.
Tissue samples were fixed in 10% formalin, routinely processed, and stained with hematoxylin and eosin for histopathologic evaluation. Tissue sections were also stained with Gram's stain to investigate the presence of bacteria. Microscopically, diffuse interstitial pneumonia was evident in the lungs (Fig. 4). Alveolar septa were hyperemic, expanded by edema, and contained an increased number of macrophages and lymphocytes. Alveolar spaces contained erythrocytes, fibrin, macrophages, and neutrophils. Interlobular septa and subpleural spaces were expanded by edema, hemorrhage, and abundant fibrin accumulation. Random, multifocal areas of necrosis containing degenerate neutrophils were scattered throughout the lung parenchyma. Throughout the brain, the meninges contained multifocal areas of hemorrhage and accumulations of degenerate neutrophils, often accompanied by accumulations of fibrin. Rare, small foci of parenchymal necrosis containing degenerate neutrophils were in the molecular layer of the cerebellum (Fig. 5). The umbilicus was thickened by edema, and vascular lumina were narrowed by thick fibrinocellular thrombi containing degenerate neutrophils and necrotic debris. Random multifocal areas of necrosis, often containing accumulations of degenerate neutrophils, were in the liver (Fig. 6) and spleen. The renal glomeruli contained numerous fibrin thrombi. Small clusters of gram-negative bacilli were evident within the necrotic foci in the lung, liver, spleen, brain, and umbilicus. Additionally, necrosuppurative rhinitis with necrotizing vasculitis was present in the grossly evident nasal cavity lesion. Fungal hyphae morphologically consistent with Aspergillus spp. were present within the lesions. Fungal hyphae were not found in other tissues.
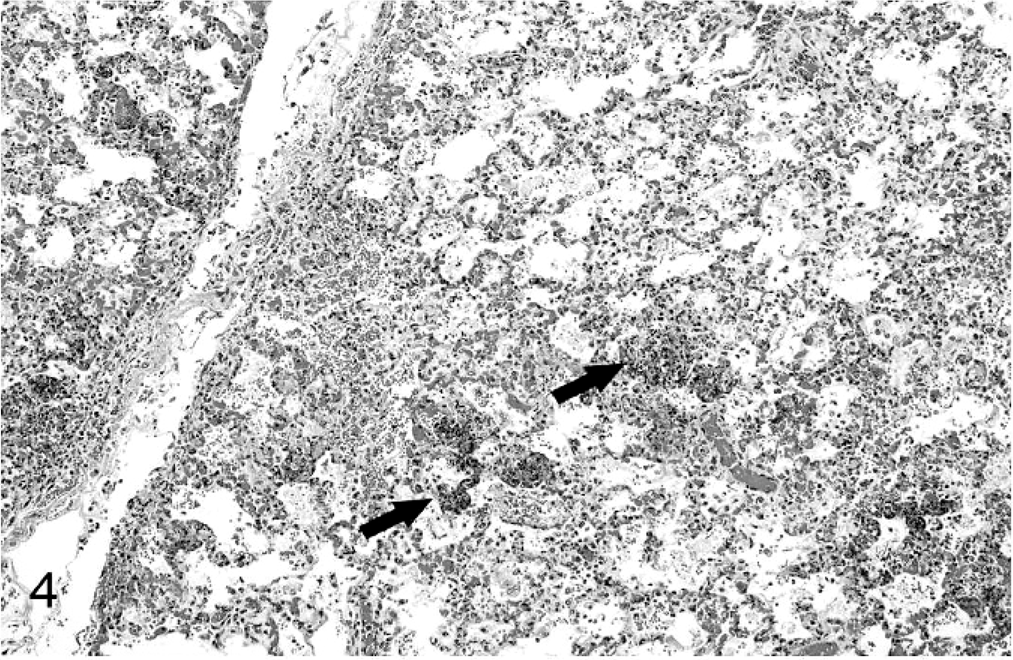
Lung; calf. Diffuse interstitial pneumonia with multifocal areas of embolic necrosis (arrows). HE.
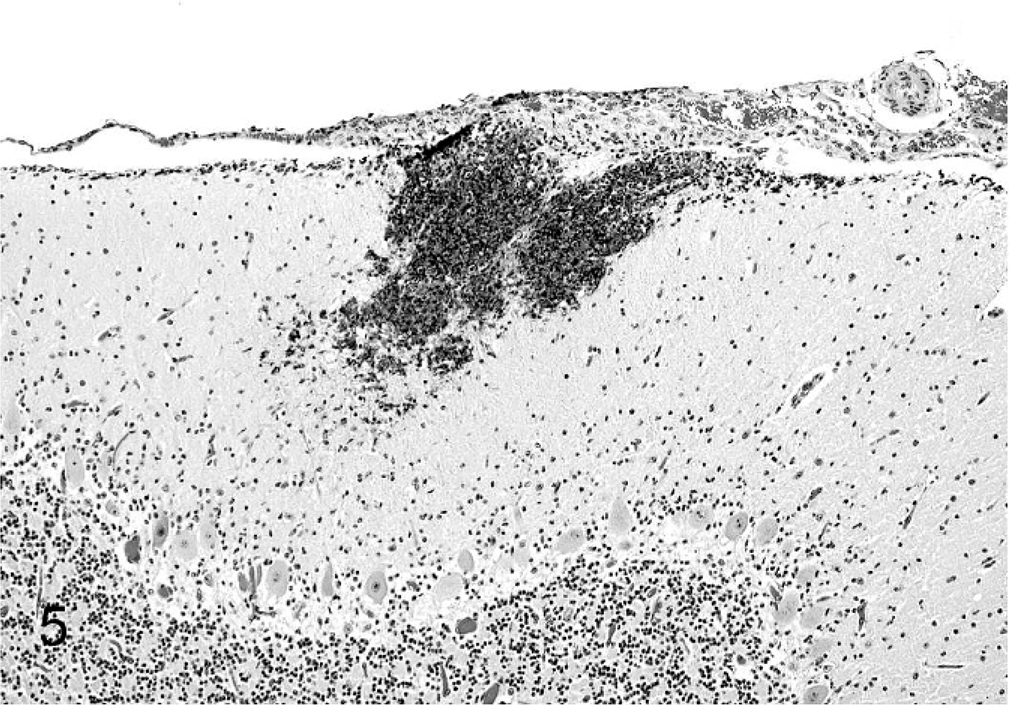
Cerebellum; calf. A focal area of necrosis and neutrophilic encephalitis in the molecular layer. HE.
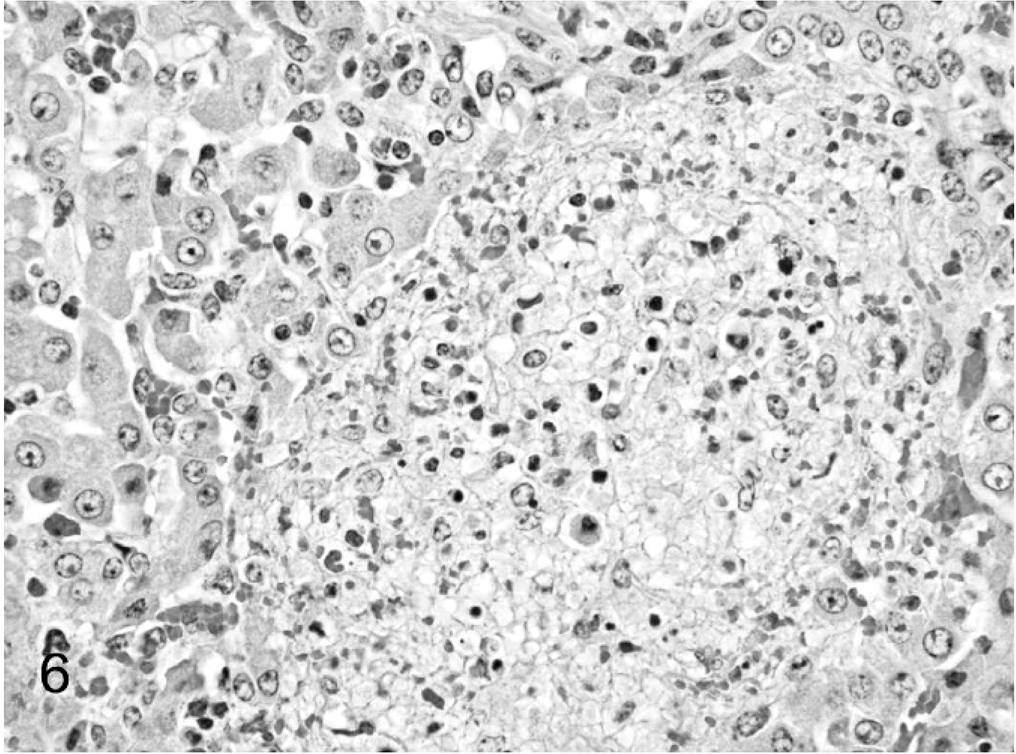
Liver; calf. A focal area of random hepatocellular necrosis. HE.
C. violaceum was isolated in high numbers from the lungs, liver, spleen, carpus, and in pure culture from the CSF. Both Streptococcus bovis and Escherichia coli were isolated from the lung, liver, spleen, and carpus. Staphylococcus spp. was isolated from the lung and carpus. It was concluded that C. violaceum was the primary cause of the septicemia and meningoencephalitis with secondary involvement of other bacteria.
C. violaceum infection of animals was first documented in water buffaloes. 25 Since then, infection has been reported in pigs, 15, 22 sheep, 6 gibbons, 13 a macaque, 18 a red panda, 9 colobus monkeys, 14 and dogs. 8, 11 The lesions of C. violaceum infection are typically those of septicemia with necrotizing lesions in multiple organs. C. violaceum has been reported to cause abscesses and necrosis in the lungs, liver, spleen, subcutis, tonsillar tissue, and lymph nodes. 6, 9, 11, 13, 14, 18, 22 Other lesions reported in pigs include pleuropneumonia and mild nonsuppurative meningoencephalitis. 15, 22 Recently, effusive pericarditis and pericardial fibrosis associated with C. violaceum infection was reported in a dog. 8 Manifestations of human chromobacteriosis include cellulitis at a site of local trauma, septicemia, and abscesses involving the liver, kidney, and lung. 16, 17, 19, 24
The clinical course and lesions in this case were similar to those described in previous animal reports of chromobacteriosis. The infection was acute, with the calf dying 3 days after the appearance of clinical signs. The route of infection in animals is usually either oral or skin abrasions. The presence of omphalophlebitis in this case indicates that the umbilicus was the likely point of entry, as is common in neonatal calf septicemias. The low plasma protein suggests that failure of passive transfer (FPT) may have been involved. The lack of grossly visible areas of necrosis, usually described as abscesses in most reports, might be explained by the fulminant clinical course. Commonly, neonatal bacterial suppurative meningitis (NBSM) in calves is associated with E. coli, 12 and other implicated bacteria include Salmonella spp., Streptococcus spp., Mycoplasma spp., Pasteurella spp., and Mannheimia spp. Isolation of a pure culture of C. violaceum from the CSF suggests that C. violaceum was solely responsible for the central nervous system lesions. Brain involvement appears to be uncommon in both animal and human cases of chromobacteriosis, and lesions are generally restricted to meningitis. This calf had mild parenchymal involvement in the cerebellum. The morphology of the fungal hyphae observed in the nasal mucosa closely resembles species of Aspergillus and Fusarium, which are common causes of opportunistic fungal infections in cattle.
C. violaceum infections may be underdiagnosed for several reasons. Acute death often precludes the antemortem diagnosis of C. violaceum infection, and animals dying with obvious lesions of septicemia may not be cultured. C. violaceum is a common saprophyte found in water and soil, and isolation from animals at necropsy may be interpreted as a contaminant. Polymicrobial infections, frequently found in septicemic calves, may further confound accurate diagnosis of chromobacteriosis. 12
The diagnosis in human and animal C. violaceum infections is based on culture of blood samples, tissues, abscess fluid, and exudates, and routine biochemical tests to identify the organism. Recently, the genome of C. violaceum has been sequenced, 3 and a multiplex polymerase chain reaction based on genes of a type III secretion system has been developed for rapid detection of C. violaceum from various sources, including humans, animals, and the environment. 21 Contaminated soil and water are common sources of infection for mammals. Recently, C. violaceum was isolated from the Ixodes ricinus tick, which may represent another potential source of infection. 23
Multiple potential virulence factors have been identified in the C. violaceum genome. C. violaceum encodes for type IV pili, which have been shown to be involved in host cell adhesion, twitching motility, and DNA transfer in several pathogenic gram-negative bacteria. 4, 7 C. violaceum uses a type III secretion system to deliver effector proteins into the cytoplasm of host cells. Genes that encode this type III system have high similarity to genes from Salmonella spp. and Yersinia spp. 2, 4 It is well known that lipopolysaccharide (LPS) of gram-negative bacteria activates host cells to produce high levels of inflammatory cytokines, resulting in septic shock. C. violaceum also codes for LPS and chromobacteriosis in mammals usually results in septicemia. LPS genes of C. violaceum show high similarity to those of Neisseria meningitides, an important cause of human meningitis. 4 Recently, hemolysin and transporter genes from C. violaceum, similar to those described in Serratia marcescens, have been cloned and characterized. 5 The hemolysin produced by C. violaceum likely has a variety of cytotoxic effects and may play an important role in the virulence of the organism. C. violaceum strains can be classified as pigmented and nonpigmented. The pigmented strains produce violacein, a pigment that gives culture media a characteristic purple color. A notable feature of C. violaceum is that it contains multiple drug resistance genes, including beta-lactam antibiotic resistance genes and drug efflux pump genes, 10 and has been found to be resistant to a wide range of antibiotics such as ampicillin, rifampin, vancomycin, and cephalosporins. 1, 17
In conclusion, this report suggests that C. violaceum can infect calves and cause fatal septicemia. This is apparently the first detailed report of a C. violaceum infection in a calf. Because C. violaceum contains multiple drug and antibiotic resistance genes, treatment of the infection in both humans and animals is challenging. Although infections are rare, C. violaceum should be included in the differential diagnosis of septicemia in calves.
