Abstract
Epithelioid cells are found in lymph nodes in cases of metastatic carcinoma; however, epithelioid cells with benign features have also been discovered incidentally in lymph nodes. Epithelioid cells were observed in mediastinal lymph nodes of cattle without cancer during routine diagnostic necropsy. To explain this finding, the authors evaluated the prevalence and histopathologic, immunohistochemical, and ultrastructural features retrospectively using mediastinal lymph nodes from 110 cattle and found that 66 of the lymph nodes contained epithelioid cells. In all 66 nodes, most of the epithelioid cells were individual or aggregated in nodal sinuses; in 21 nodes, some epithelioid cells formed tubular structures. The individual and aggregated epithelioid cells were mostly considered to be mesothelial in origin by immunohistochemical and ultrastructural examination; presumably, they entered the lymph nodes via lymphatic vessels, because they were found in afferent lymphatic vessels and nodal sinuses. Although the presence of epithelioid cells in lymph nodes prompts suspicion of metastatic carcinoma, the epithelioid cells in these bovine lymph nodes did not disrupt nodal architecture, lacked atypia or mitotic figures, and did not invade nodal parenchyma.
Lymph node anlagen are believed to arise from periarteriolar mesenchyme. At the confluence of lymphatic vessels and arterioles, lymphatics form cuffs enclosing the arterioles and outpouchings that form the capsule of the primordial lymph node. Reticular cells form a network that provides an appropriate environment for colonization by lymphocytes, then thymic and bone marrow–derived lymphocytes immigrate. 15 Thus, in normal development, there are no epithelial cells in lymph nodes. When epithelioid cells are observed in lymph nodes, it raises suspicion of metastatic carcinoma. Metastatic carcinoma cells have atypia, reduced E-cadherin expression, and increased expression of proteolytic enzymes. 8, 17 Generally, metastatic carcinoma in lymph nodes destroys the inherent tissue structure by proliferating and infiltrating nodal parenchyma. However, epithelioid cells with benign features have been discovered incidentally in human lymph nodes 4 and are known as benign epithelial inclusions (BEI), which must be differentiated from metastatic carcinoma. 3, 4, 7
Epithelioid cells were observed in the mediastinal lymph nodes of cattle without cancer in this study. Their prevalence and histopathologic, immunohistochemical, and ultrastructural features were evaluated in a retrospective study of mediastinal lymph nodes from 110 cattle.
Materials and Methods
Tissue Samples
Tissue samples of mediastinal lymph nodes from 110 cattle, which were submitted to the Laboratory of Veterinary Pathology of Rakuno Gakuen University for necropsy, were used. One cross section through the hilus of each mediastinal lymph node was examined histologically. The age of the cattle ranged from 3 months' gestation to 13 years of age; the age of 12 cattle was unknown. The sex distribution was 92 female and 18 male. The breed distribution was 92 Holstein Friesian, 13 Japanese Black, 3 hybrid types of Japanese beef cattle, 1 Jersey, and 1 Brown Swiss. There were a variety of clinical and pathologic diagnoses, but carcinoma was not observed in the pleural or abdominal cavity of any animal.
Histopathologic examination
Cross sections of mediastinal lymph nodes were fixed in 10% neutral buffered formalin, dehydrated in methanol, and embedded in paraffin wax. One paraffin block from each mediastinal lymph node was prepared, and 1 section from each paraffin block was cut at a thickness of 4 µm, then stained with HE.
Immunohistochemical examination
Serial unstained sections from paraffin-embedded tissues were prepared for immunohistochemical examination by the avidin-biotin-peroxidase complex (ABC) procedure (Vectastain Elite ABC Kit; Vector Laboratories, Burlingame, CA, USA). Primary antibodies (against cytokeratin, vimentin, factor VIII–related antigen, CD68, and E-cadherin) and pretreatments for antigen retrieval are listed in Table 1. After antigen retrieval, the sections were treated with 0.3% H2O2 in methanol for 10 minutes to suppress endogenous peroxidase activity, followed by incubation with 10% normal goat serum (Sigma Chemical Co., St Louis, MO, USA), then diluted in phosphate-buffered saline (PBS) (pH, 7.4). All sections were incubated with primary antibodies at 4°C for 16 hours. After washing with PBS, the sections were incubated with biotinylated mouse anti-goat IgG antibody or rabbit anti-goat IgG antibody for 30 minutes at room temperature, and with avidin peroxidase conjugate for 30 minutes. Finally, the sections were treated with chromogen (0.05% 3,3′-diaminobenzine tetrahydrochloride) and H2O2 and counterstained with hematoxylin. To evaluate immunohistochemical features of normal bovine mesothelial cells, visceral and parietal pleura were examined with each antibody. The following bovine tissues were used as controls: bronchial epithelium for cytokeratin and E-cadherin, vascular endothelial cells for vimentin and factor VIII–related antigen, macrophages for CD68, and basal lamina of vessels for laminin and fibronectin. For negative controls, the primary antibody was omitted.
Details of immunohistochemical immunofluorescent reagents.
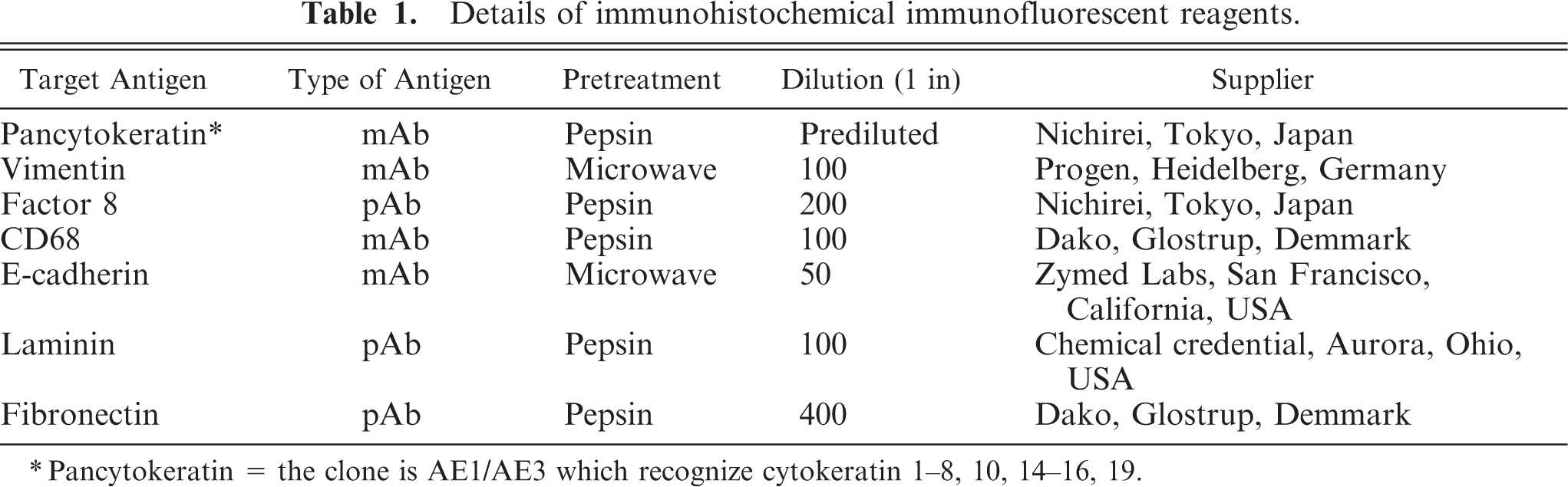
∗Pancytokeratin = the clone is AE1/AE3 which recognize cytokeratin 1-8, 10, 14-16, 19.
Immunofluorescent examination
Serial unstained sections from paraffin-embedded tissues were prepared for immunofluorescence examination by confocal laser-scanning microscopy. Primary antibodies (against cytokeratin, laminin, and fibronectin) and antigen-retrieval pretreatments are listed in Table 2. After pretreatment, the sections were incubated with 10% normal goat serum (Sigma Chemical Co., St Louis, MO, USA), then diluted in PBS (pH 7.4). To define the epithelioid cells, the sections were incubated with a mixture of cytokeratin and laminin antibody and cytokeratin and fibronectin antibody at 4°C for 16 hours. After washing with PBS, the sections were incubated for 30 minutes at room temperature with a mixture of the diluted (1 : 100) Alexa Fluor-488-labeled donkey anti-mouse IgG (Molecular probes Inc., Eugene, OR, USA) for detection of cytokeratin, the diluted (1 : 100) Alexa Fluor-555-labeled donkey anti-rabbit IgG (Molecular Probes Inc., Invitrogen) for detection of laminin and fibronectin, and the diluted (1 : 100) TO-PRO-3 (Molecular probes Inc.) as counterstain. After washing with PBS, the sections were examined with an Axiovert 200M confocal laser-scanning microscope (Carl Zeiss Inc., Tokyo, Japan). The following tissues were used as controls: bronchial epithelium for cytokeratin and basal lamina of vessels for laminin and fibronectin. For negative controls, sections were incubated without the primary antibodies.
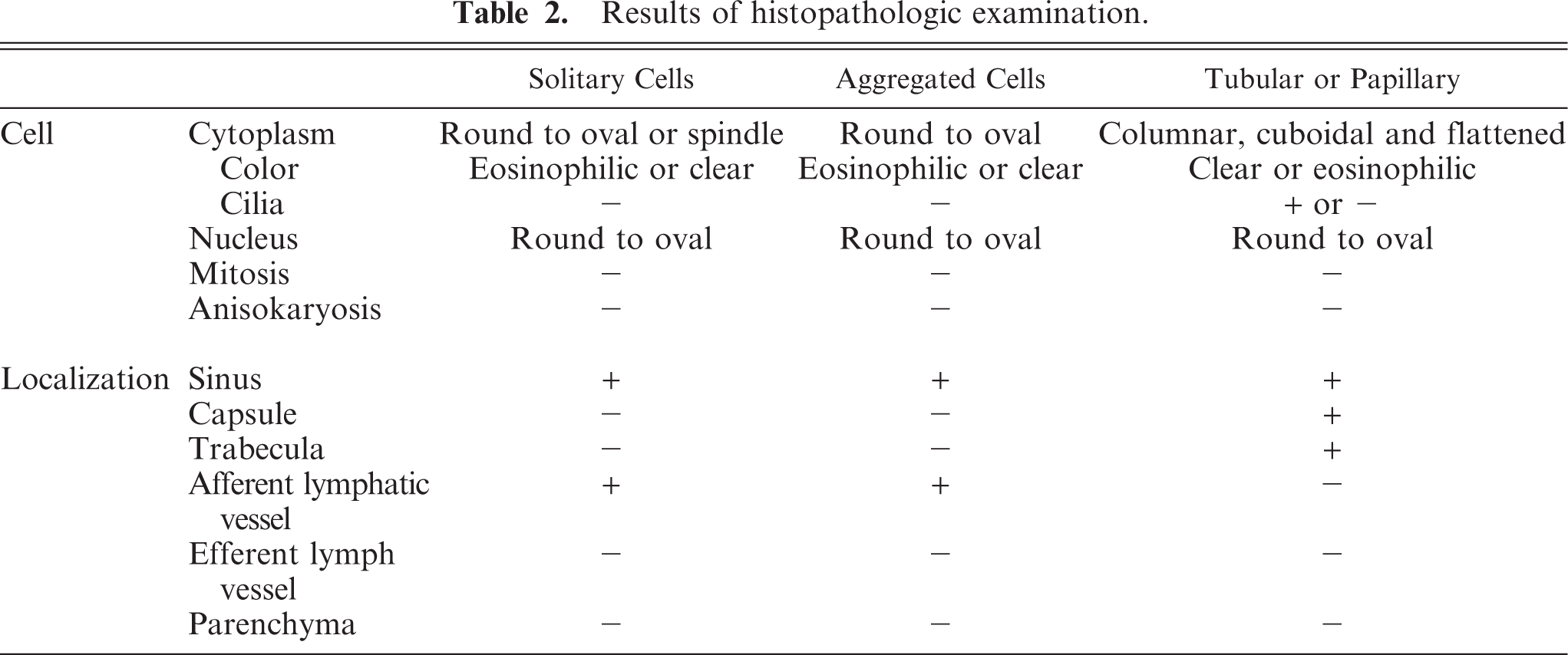
Results of histopathologic examination.
Ultrastructural examination
The 2.5% glutaraldehyde-fixed tissue in which abundant cytokeratin-positive cells were observed was cut into 1-mm cubes, post-fixed in 1% osmium tetroxide for 60 minutes at room temperature, and then dehydrated and embedded in Quetol 812. Ultrathin sections were stained with uranyl acetate and lead citrate and examined with a JEM-1220 transmission electron microscope calibrated with a standard measuring grid at an accelerating voltage of 80 kV.
Results
Prevalence
Of the 110 mediastinal lymph nodes examined in this study, 66 had epithelioid cells. Individual epithelioid cells were observed in all 66 nodes and were detected in all ages from fetal to adult cattle. Aggregated epithelioid cells were scattered among the individual cells and were detected at all ages. Epithelioid cells arranged in tubular or papillary structures were found in 21 of these 66 nodes; all affected cattle were older than 1 year. Age distribution and prevalence of nodal epithelioid cells are shown in Fig. 1. Only a few individual or aggregated epithelioid cells were observed in the fetuses; the number of these cells varied in other age groups. Epithelioid cells were found in 46 of 69 lymph nodes from cattle with pulmonary/respiratory disorders, whereas they were observed in only 20 or 41 nodes from cattle without respiratory disease.
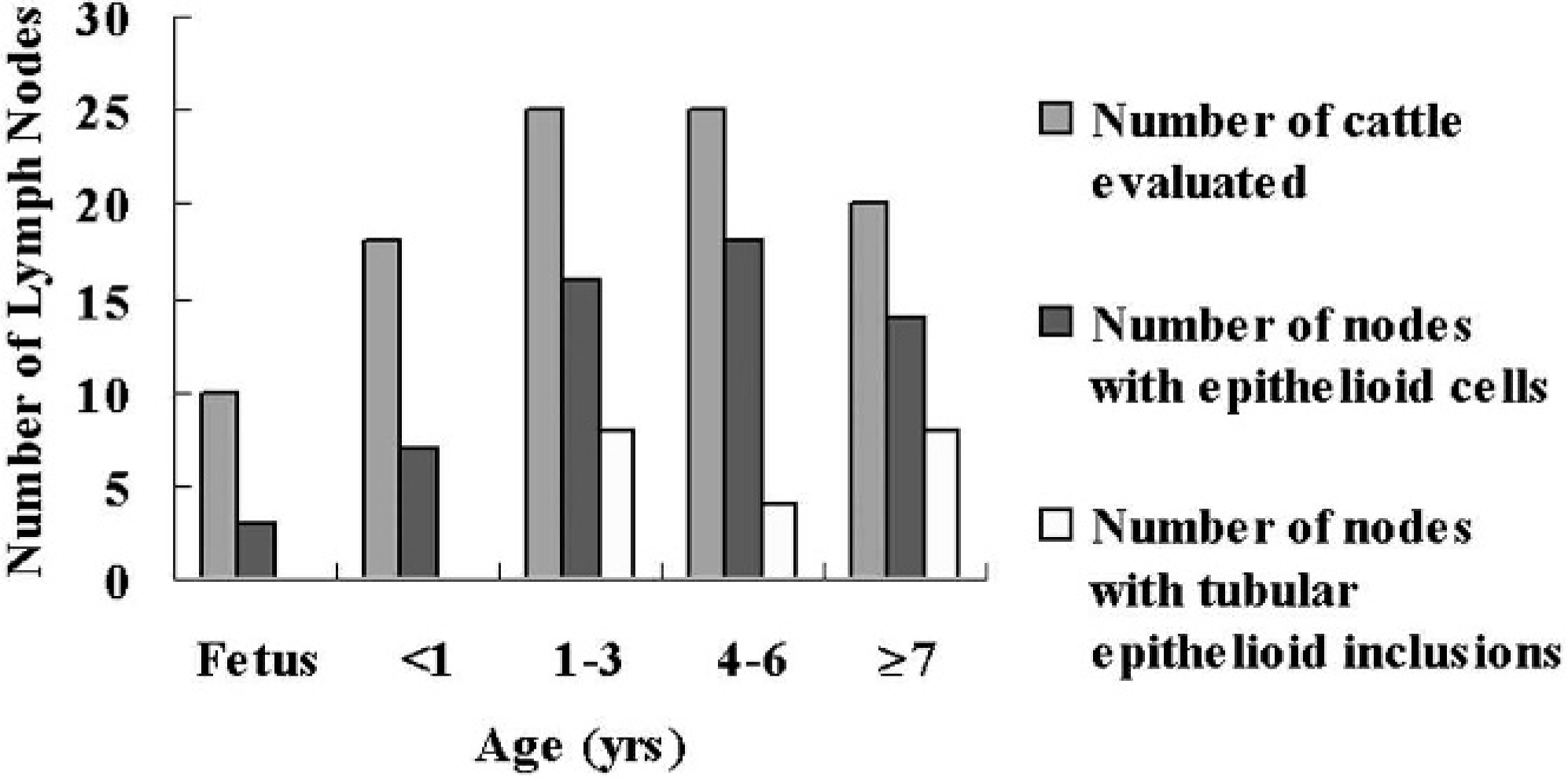
Prevalence of epithelioid cells in bovine mediastinal lymph nodes by age. One section of 1 lymph node was examined per animal. Individual or aggregated epithelioid cells were found in mediastinal lymph nodes of some cattle in all age groups. On the other hand, epithelioid cells arranged in tubular or papillary structures were found only in cattle older than 1 year.
Morphologic features
Individual epithelioid cells were mainly located in the dilated marginal to intermediate sinus and afferent lymphatic vessels but not in efferent lymphatic vessels. Epithelioid cells did not infiltrate nodal parenchyma. The number of individual epithelioid cells varied among nodes, and in some nodes with numerous epithelioid cells, neutrophils were observed in dilated sinuses. The number of epithelioid cells tended to decrease toward the efferent lymphatic vessels. Individual epithelioid cells had large round-to-oval nuclei and abundant eosinophilic or clear cytoplasm. They were free in the sinuses and morphologically similar to histiocytes (Fig. 2). Multinucleated cells or cells containing cytoplasmic vacuoles were rarely observed. Spindle cells were also observed free in nodal sinuses or lining the sinuses. Anisokaryosis or mitotic figures were not found.
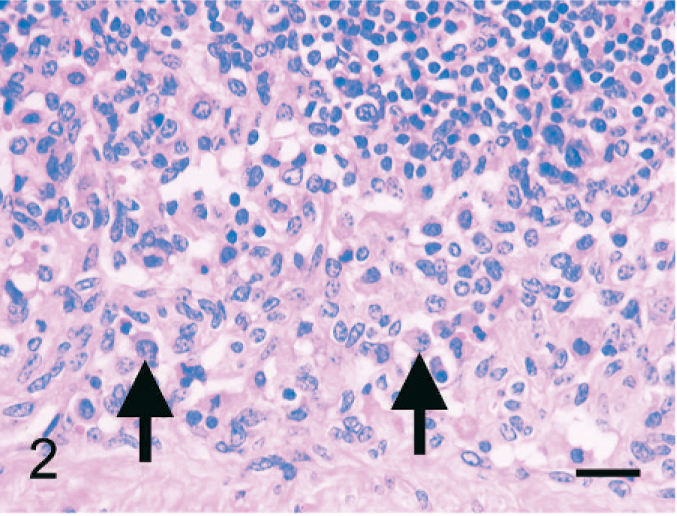
Bovine mediastinal lymph node; case No. 37. Individual epithelioid cells have a histiocytoid appearance with a large, round-to-oval nucleus and abundant eosinophilic cytoplasm (arrows). HE. Bar = 30 µm.
Aggregated epithelioid cells were free in the sinuses and were found mainly in the dilated marginal to intermediate sinus and afferent lymphatic vessels but not in the efferent lymph vessels. No infiltration of the lymph node parenchyma was observed. Aggregated cells were encountered with individual cells, and the number of cells tended to decrease closer to efferent lymphatic vessels. Aggregated epithelioid cells had large round-to-oval nuclei with eosinophilic or clear cytoplasm and resembled histiocytes. Some were arranged in rosette-like structures that contained matrix in the center. No anisokaryosis or mitotic figures were noted (Fig. 3).
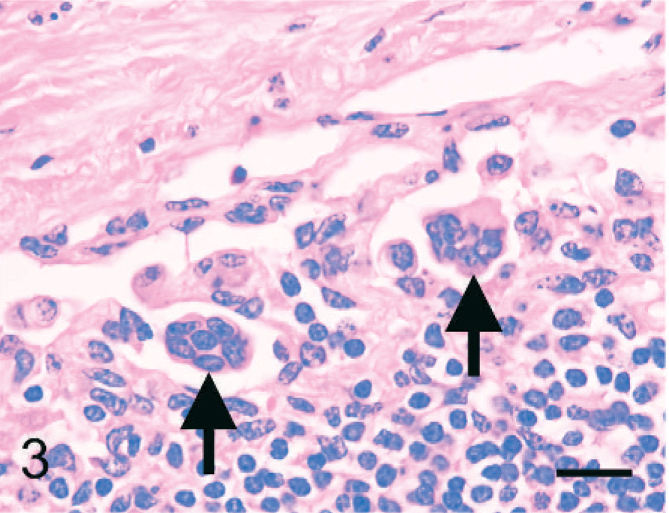
Bovine mediastinal lymph node; case No. 37. Aggregated epithelioid cells resemble the individual cells but have a syncytial appearance (arrows). HE. Bar = 20 µm.
Epithelioid cells arranged in tubular or papillary structures were located in or adjacent to nodal capsules or trabeculae. In serial sections, tubular or papillary structures were connected. Some tubular structures communicated with a sinus, and the ends of the tubular structures had a papillary appearance, whereas other tubules ended in a cystic structure. The epithelioid cells of tubular structures were mainly simple columnar, cuboidal, or squamous, and they had an oval nucleus with clear or eosinophilic cytoplasm. Almost all epithelioid cells in tubular structures were ciliated. In part of the papillary structures, pseudostratified epithelium was seen (Fig. 4). Rarely, a psammoma body was observed near the tubular structures. There were no anisokaryosis or mitotic figures (Table 2).
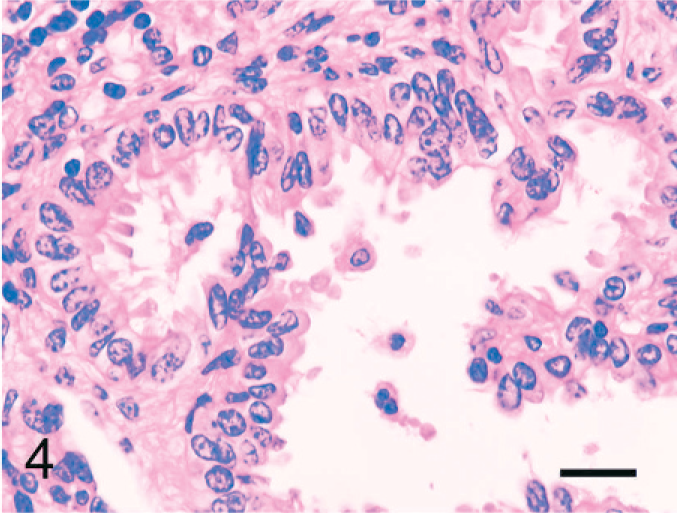
Bovine mediastinal lymph node; case No. 37. The columnar epithelioid cells lining tubulopapillary formations have an oval nucleus with eosinophilic cytoplasm. Almost all cells are ciliated. HE. Bar = 20 µm.
Immunohistochemical examination
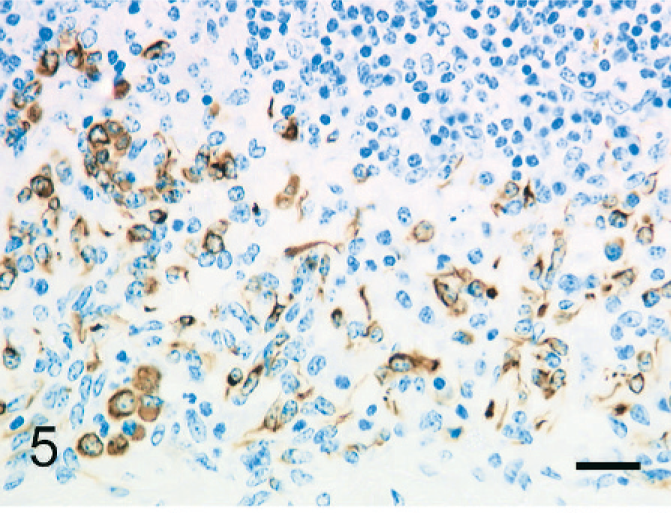
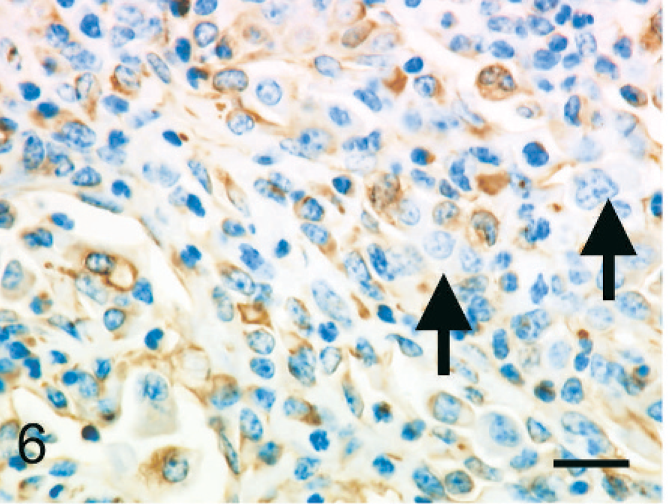
The individual epithelioid cells expressed cytokeratin. In addition, globules of cytokeratin-positive material were observed in the cytoplasm of macrophages; this was interpreted as phagocytized remnants of the cytokeratin-positive epithelioid cells (Fig. 5). Fewer than 5% of the epithelioid cells expressed vimentin (Fig. 6). Epithelioid cells did not express factor VIII–related antigen or CD68. A few cells had diffuse cell-membrane expression of E-cadherin (Fig. 7). Aggregated epithelioid cells were positive for cytokeratin (Fig. 8) and negative for vimentin (Fig. 9), factor VIII–related antigen, and CD68. The cell membrane between a few aggregated epithelioid cells was positive for E-cadherin (Fig. 10).
Bovine mediastinal lymph node; case No. 37. Individual epithelioid cells express cytokeratin. Immunohistochemistry (IHC), monoclonal antibody AE1/AE3. Mayer's hematoxylin counterstain. Bar = 30 µm.
Bovine mediastinal lymph node; case No. 37. Individual epithelioid cells are negative for vimentin (arrows), whereas macrophages and reticular cells are positive. IHC, monoclonal antibody vimentin. Mayer's hematoxylin counterstain. Bar = 20 µm.
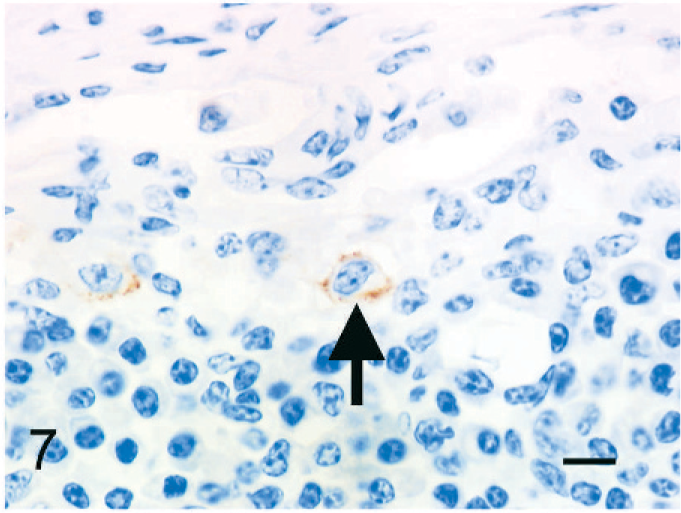
Bovine mediastinal lymph node; case No. 37. A few epithelioid cells express E-cadherin on the cell membrane (arrow). IHC, monoclonal antibody E-cadherin. Mayer's hematoxylin counterstain. Bar = 20 µm.
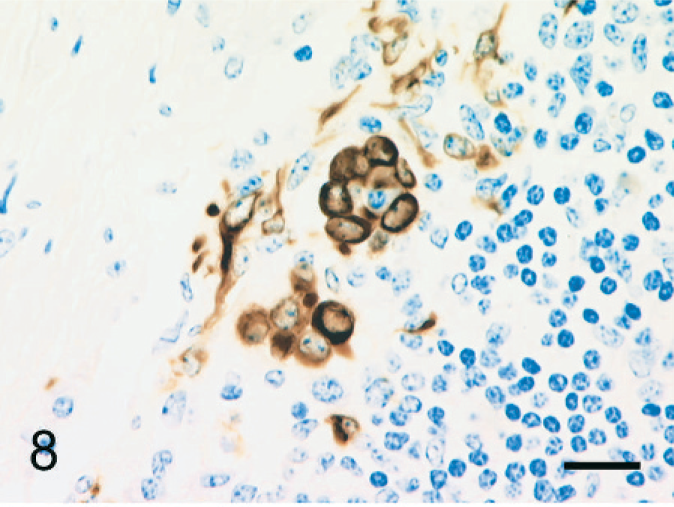
Bovine mediastinal lymph node; case No. 37. Aggregated epithelioid cells express cytokeratin. IHC, monoclonal antibody AE1/AE3. Mayer's hematoxylin counterstain. Bar = 20 µm.
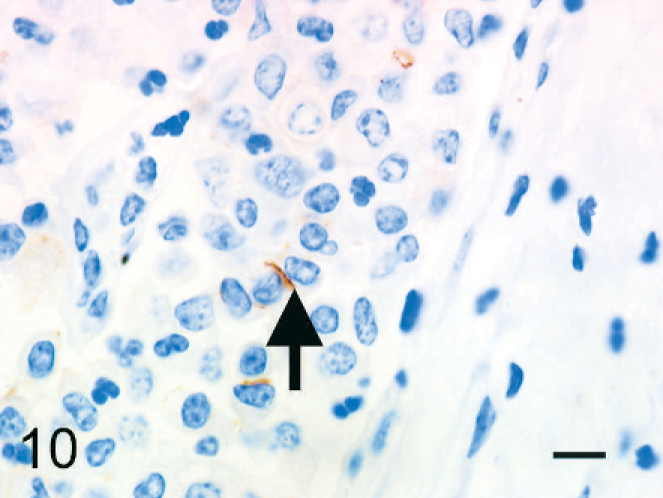
Bovine mediastinal lymph node; case No. 37. Aggregated epithelioid cells are negative for vimentin (arrows), whereas macrophages and reticular cells are positive. IHC, monoclonal antibody vimentin. Mayer's hematoxylin counterstain. Bar = 20 µm.
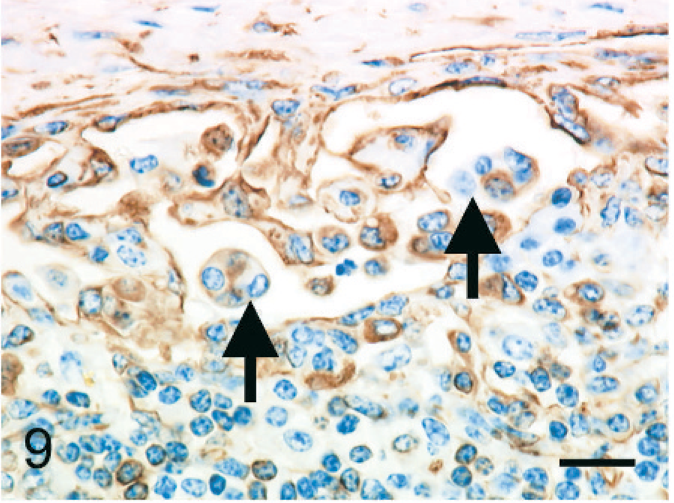
Bovine mediastinal lymph node; case No. 37. The cell membrane between two epithelioid cells expresses E-cadherin (arrow). IHC, monoclonal antibody E-cadherin. Mayer's hematoxylin counterstain. Bar = 20 µm.
Epithelioid cells arranged in tubular or papillary structures were positive for cytokeratin (Fig. 11) and negative for vimentin (Fig. 12), factor VIII–related antigen, and CD68. The cell membrane expressed E-cadherin (Fig. 13).
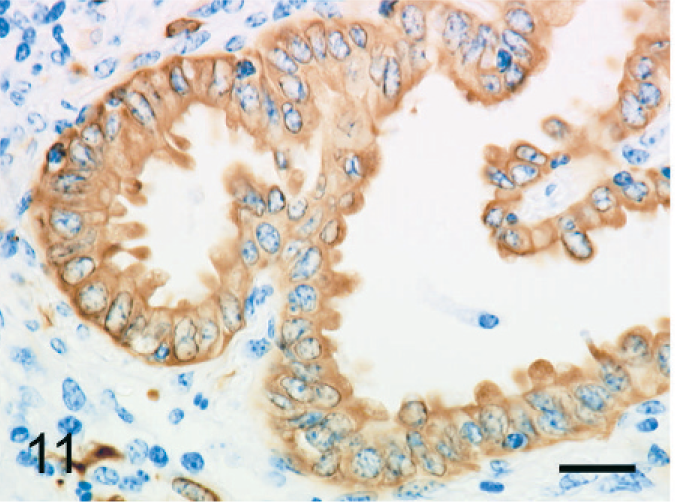
Bovine mediastinal lymph node; case No. 37. Epithelioid cells arranged in tubulopapillary formations express cytokeratin AE1/AE3. IHC, monoclonal antibody AE1/AE3. Mayer's hematoxylin counterstain. Bar = 20 µm.
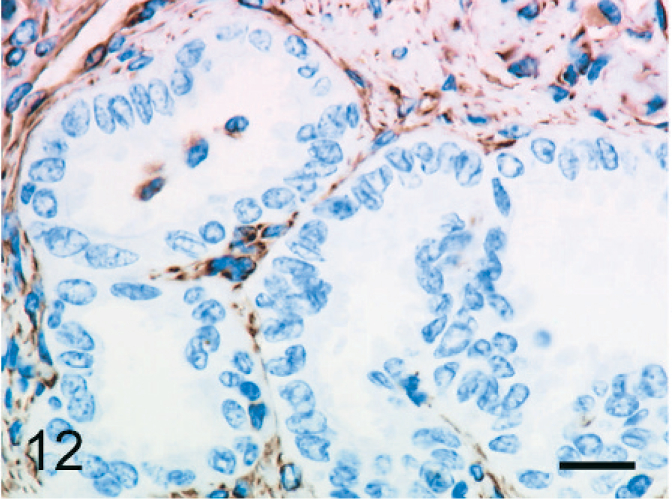
Bovine mediastinal lymph node; case No. 37. Epithelioid cells arranged in tubulopapillary formations are negative for vimentin. IHC, monoclonal antibody vimentin. Mayer's hematoxylin counterstain. Bar = 20 µm.
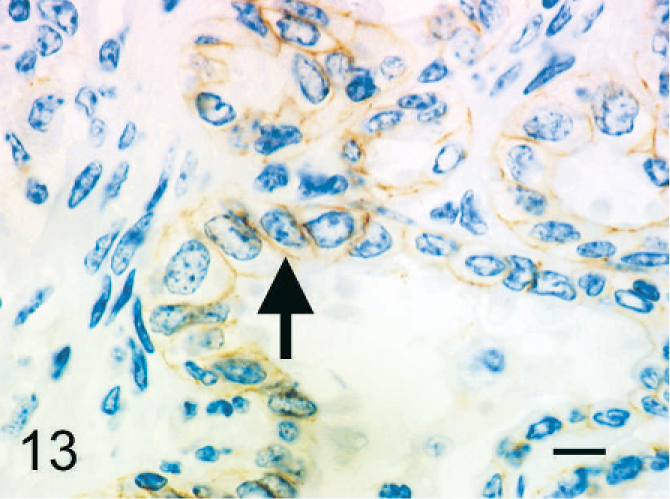
Bovine mediastinal lymph node; case No. 37. The cell membrane of epithelioid cells arranged in tubulopapillary formations expresses E-cadherin (arrow). IHC, monoclonal antibody E-cadherin. Mayer's hematoxylin counterstain. Bar = 20 µm.
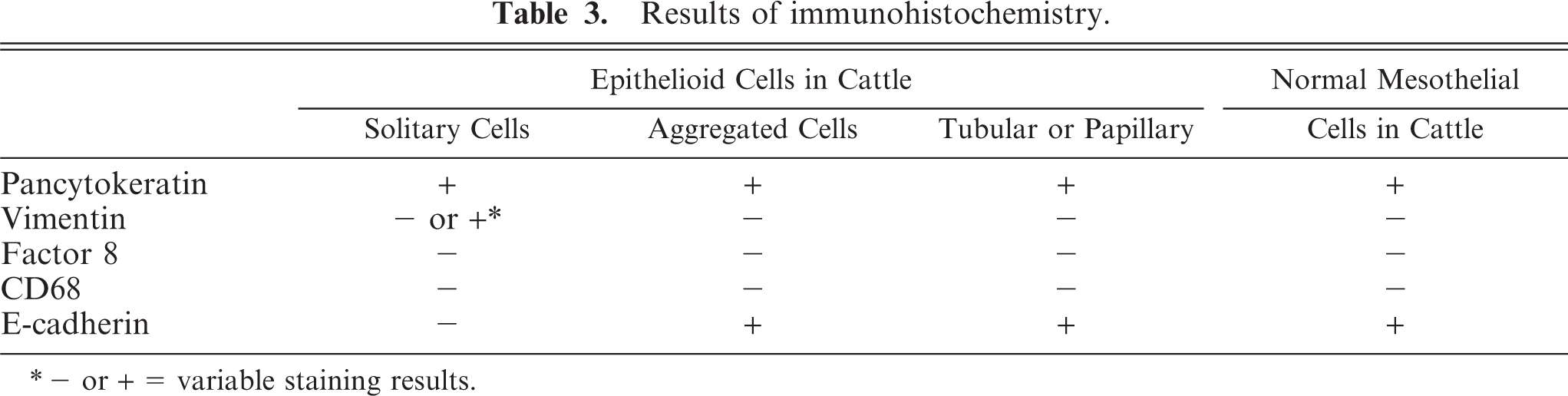
The normal bovine pleural mesothelial cells expressed cytokeratin and E-cadherin, but they were immunohistochemically negative for vimentin, factor VIII–related antigen, and CD68 (Table 3). Bovine positive control tissues (bronchial epithelium for cytokeratin and E-cadherin, vascular endothelial cells for vimentin and factor VIII–related antigen, and macrophages for CD68) were clearly positive for these markers.
Results of immunohistochemistry.
∗- or + 5 variable staining results.
Immunofluorescent examination
Although laminin was not detected, fibronectin was labeled diffusely by immunofluorescence in the cell membrane or cytoplasm of some individual or aggregated epithelioid cells. Immunofluorescence for both laminin and fibronectin was either absent or weak in the basement membrane of tubular or papillary structures.
Ultrastructural examination
Microvilli and concentric perinuclear bundles of intermediate filaments were observed in individual epithelioid cells (Figs. 14,15). Aggregated epithelioid cells had long and slender microvilli, concentric perinuclear bundles of intermediate filaments, and desmosomes (Figs. 16,17).
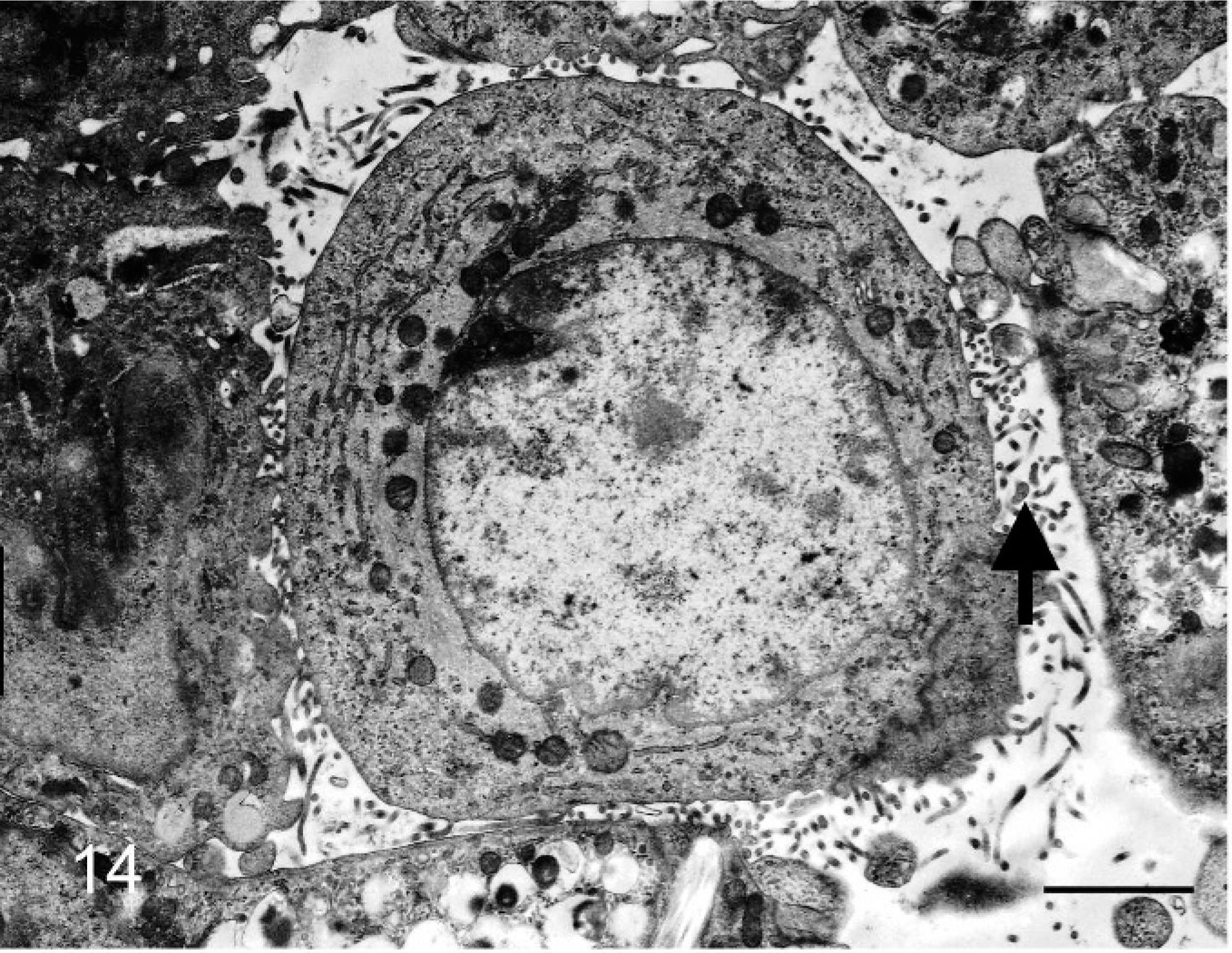
Bovine mediastinal lymph node; case No. 58. An individual epithelioid cell has microvilli. Transmission electron micrograph. Bar = 2 µm.
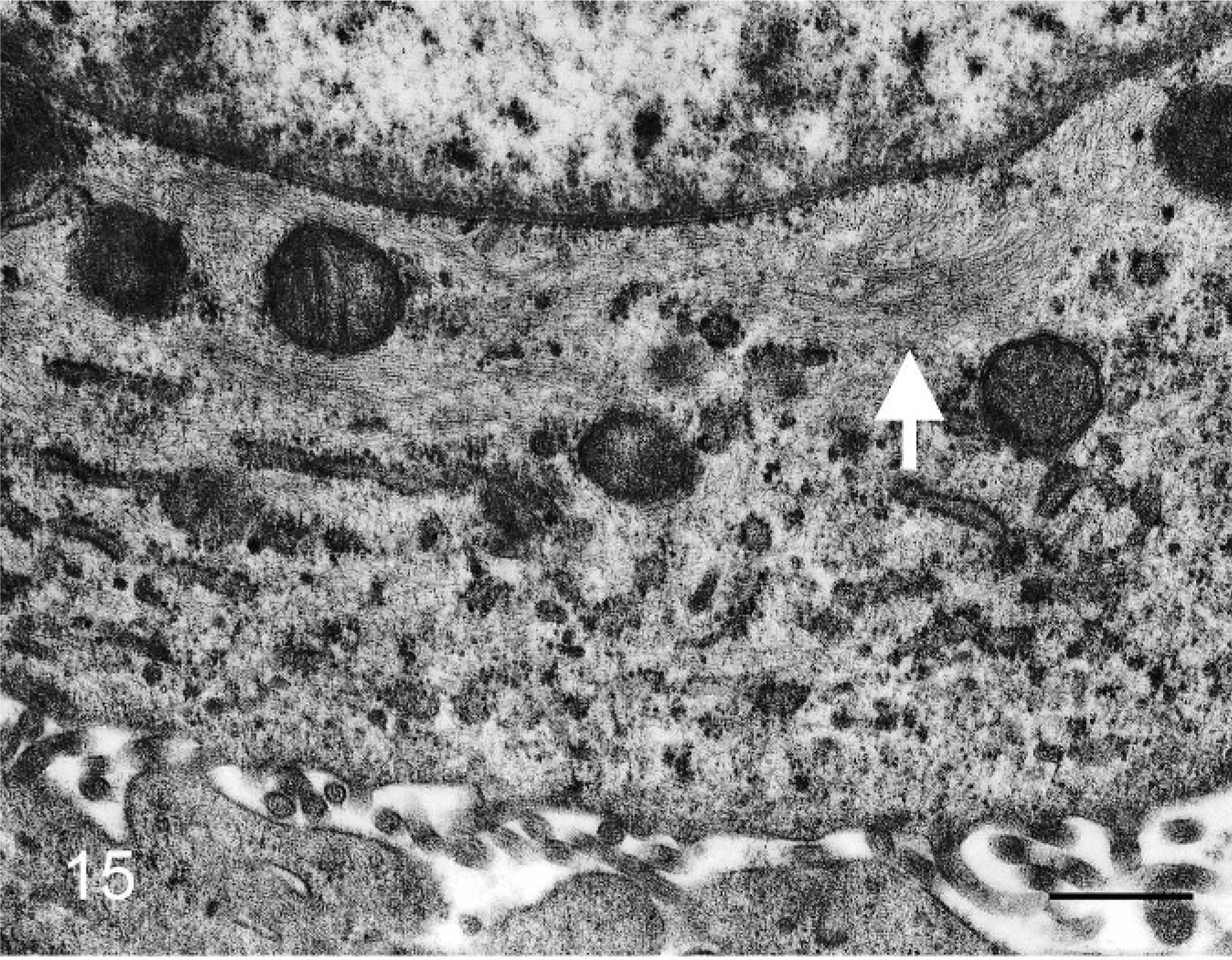
Bovine mediastinal lymph node; case No. 58. Bundles of intermediate filaments are observed in perinuclear cytoplasm of an individual epithelioid cell (arrow). Transmission electron micrograph. Bar = 500 nm.
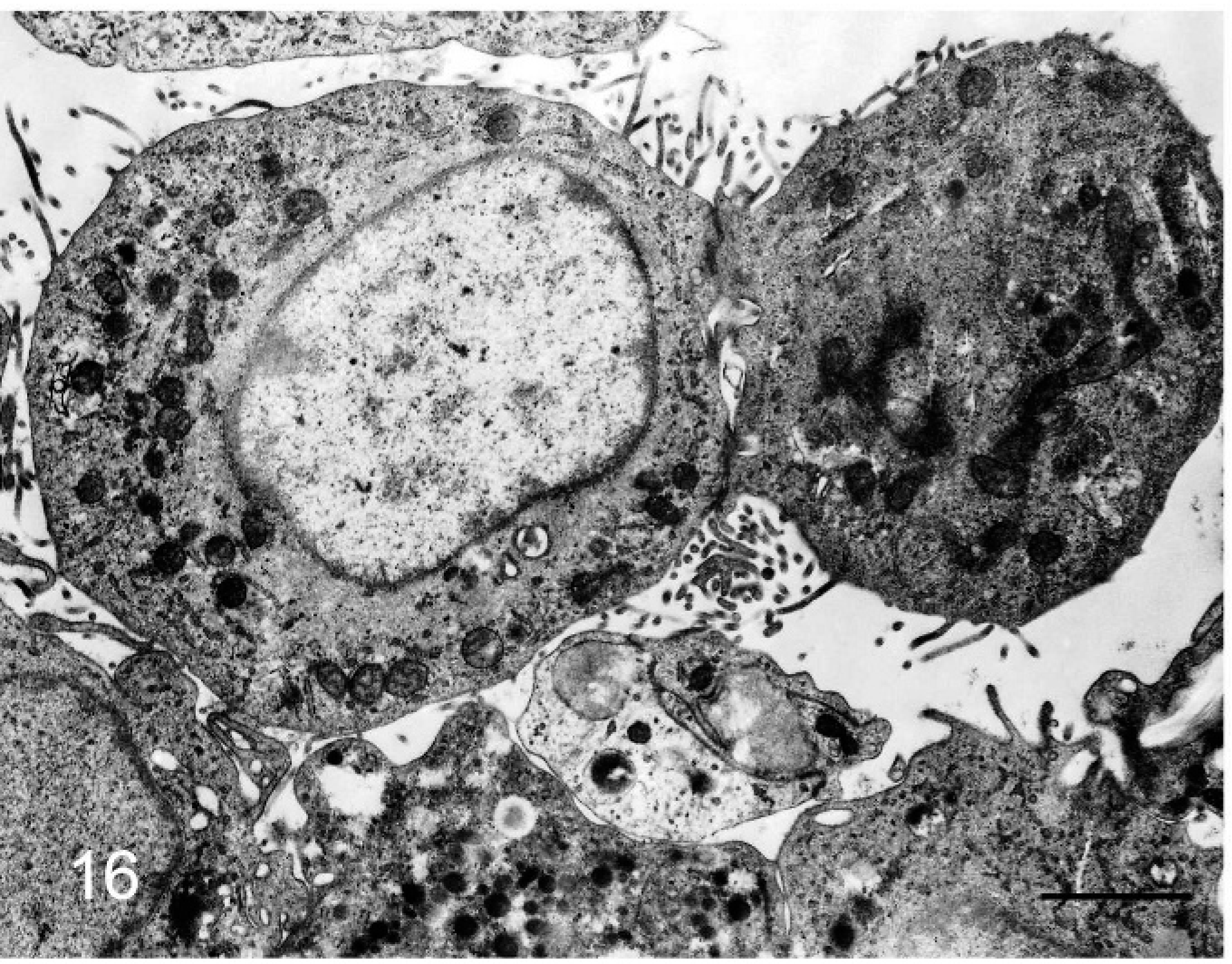
Bovine mediastinal lymph node; case No. 58. Microvilli project from aggregated epithelioid cells. Transmission electron micrograph. Bar = 2 µm.
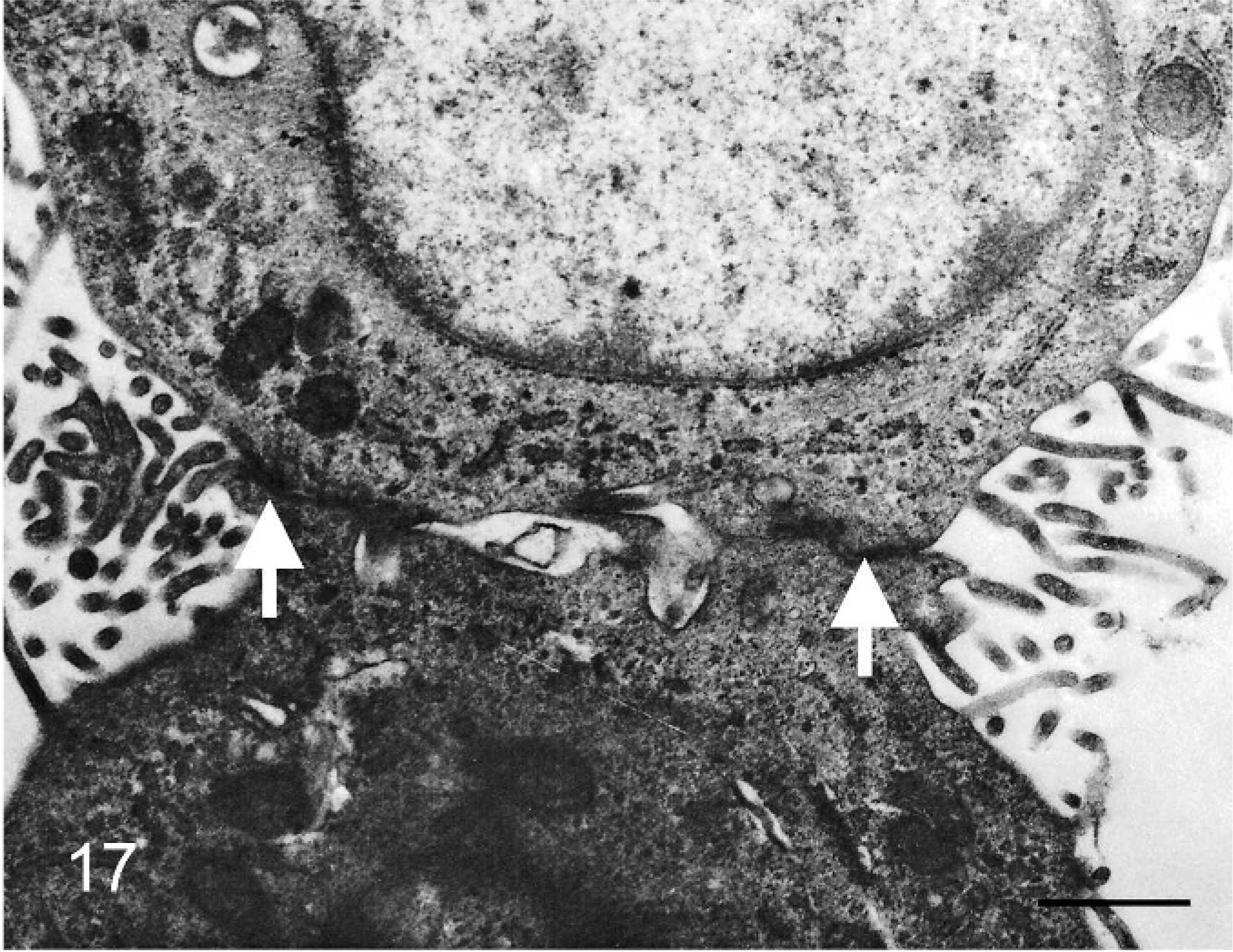
Bovine mediastinal lymph node; case No. 58. Higher magnification of the aggregated epithelioid cells in Fig. 16 showing intercellular junctions (arrows) and bundles of intermediate filaments in the paranuclear cytoplasm. Transmission electron micrograph. Bar = 1 µm.
The free border of the epithelioid cells that lined tubular structures had many cilia; the apical cytoplasm of some cells protruded into the tubular lumen (Fig. 18). Desmosomes were observed between cells (Fig. 19); cells rested on a thin basal lamina and lacked perinuclear bundles of intermediate filaments (Fig. 20). In the papillary formations, cells had cilia and apical secretory granules; some cells also had long, slender microvilli and concentric perinuclear bundles of intermediate filaments.
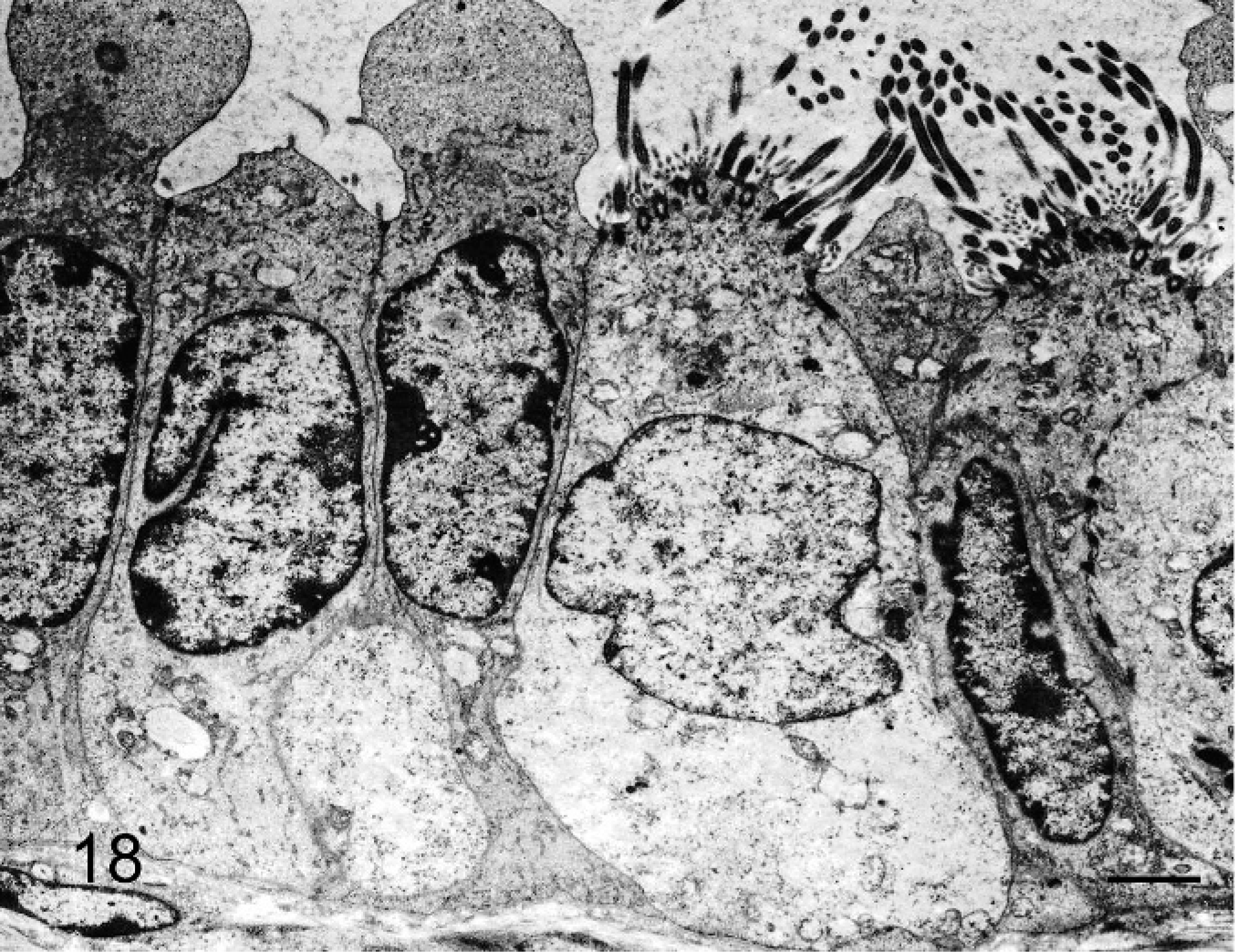
Bovine mediastinal lymph node; case No. 37. The apical border of some of the epithelioid cells that line a tubular structure have abundant cilia. Other cells are nonciliated and have a tall apical projection. Transmission electron micrograph. Bar = 2 µm.
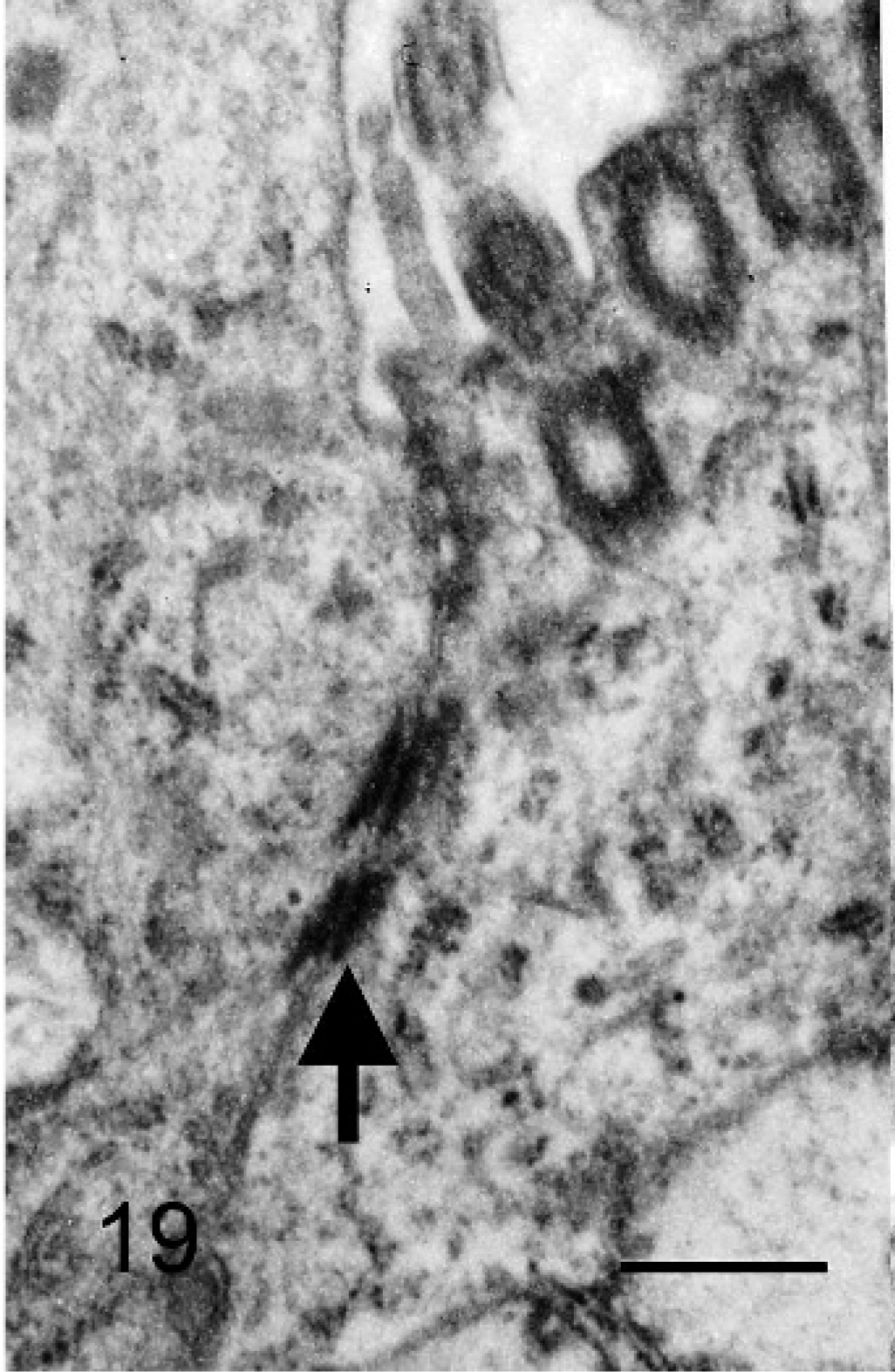
Bovine mediastinal lymph node; case No. 37. Intercellular junctions (arrow) between a ciliated and a nonciliated cell lining a tubular structure. Transmission electron micrograph. Bar = 500 nm.

Bovine mediastinal lymph node; case No. 37. Basal lamina (arrow) is evident beneath the epithelioid cells lining a tubular structure. Transmission electron micrograph. Bar = 500 nm.
Discussion
Epithelioid cells were found in 60% of mediastinal lymph nodes, although carcinoma was not evident in any of the cattle in the present study. The individual and aggregated epithelioid cells were confined to nodal sinuses; epithelioid cells in tubular or papillary structures were in the nodal capsule or trabeculae. These cells lacked atypia or mitotic figures and did not infiltrate nodal parenchyma or disrupt nodal architecture. Because there was no evidence of malignancy in the epithelioid cells, they were categorized as BEI.
Metastatic carcinoma cells tend to have not only morphologic abnormalities but also reduced E-cadherin expression and overexpression of enzymes inducing extracellular matrix proteolysis. 8, 17 Furthermore, metastatic carcinoma cells tend to alter nodal structure by proliferating and infiltrating the parenchyma. 4 However, these features of malignancy were absent in the epithelioid cells observed in bovine mediastinal lymph nodes. Human epithelioid cells with benign features and discovered accidentally in lymph nodes were first described in 1897. 13 Subsequent clinical studies demonstrated the benignity of these nodal epithelial inclusions. 3, 4, 7
The individual and aggregated epithelioid cells could have entered lymph nodes via lymphatic vessels, because they were found in both afferent lymphatic vessels and nodal sinuses; the lack of basement membrane and extracellular matrix proteins, such as laminin or fibronectin, around most epithelioid cells indicates that the epithelioid cells were free in nodal sinuses. For epithelial cells to invade lymphatic vessels and reach nodal sinuses, they would have to disrupt the extracellular matrix at the site of origin. Such invasive and metastatic abilities are characteristics of malignancy, yet none of the cattle in this study were affected by carcinoma. Furthermore, the epithelioid cells remained in nodal sinuses without infiltrating nodal parenchyma and lacked nuclear atypia or mitotic figures. Taken together, these findings indicate that the epithelioid cells were neither malignant nor of epithelial origin.
Since the first description, BEI have been reported in many regions, including pelvic, mediastinal, axillary, and cervical lymph nodes in human beings. 5, 6, 12, 16 Benign mesothelial cell inclusions have been reported, especially in mediastinal lymph nodes, in human beings 1, 3, 14, 16 and in 3 dogs, 11 in association with pleural and pericardial effusion. When mesothelial cells are injured, adjacent mesothelial cells proliferate, detach from the basement membrane into the serosal fluid, and settle on the wound surface. 9 Lymphatic spaces immediately beneath the mesothelial surface communicate directly with the pleural and peritoneal surface through so-called stomas. 3 Thus, detached mesothelial cells in serosal fluid can access lymphatic vessels via the stoma and travel to lymph nodes, even without the ability to infiltrate the extracellular matrix. Thus, it is no wonder that epithelioid cells were observed at high prevalence in bovine mediastinal lymph nodes in this study. Mesothelial cells may express both vimentin and cytokeratin. 9 In cattle, however, some normal mesothelial cells were reportedly negative for vimentin. 2, 10 In the present study, normal mesothelial cells from cattle were positive for cytokeratin and negative for vimentin. In mediastinal lymph nodes, only a few individual epithelioid cells expressed both cytokeratin and vimentin. Ultrastructurally, the epithelioid cells had long, slender microvilli with concentric perinuclear bundles of intermediate filaments; these findings are characteristic of normal or neoplastic mesothelial cells. Together, these results indicate that the individual and aggregated epithelioid cells were mesothelial cells.
Although BEI by mesothelial cells is rare in humans, BEI was common in cattle and the cells were found in lymph nodes of cattle without pleural or pericardial effusion or other thoracic lesions. This indicates that the property and the process are different between cattle and human. Thus, it may be easier for BEI to form in cattle than in humans even without pleural or pericardial effusion or inflammation. Some of the epithelioid cells were apparently phagocytized, because cytokeratin-positive cells were observed in nodal macrophages. Because the epithelioid cells were observed in 60% of the cattle, including fetuses, this may be part of the normal process of mesothelial cell turnover.
Tubular epithelioid cells were mainly simple columnar, cuboidal, or flattened ciliated cells and, obviously, differ from the endothelial cells of blood vessels or lymphatic vessels immunohistochemically and ultrastructurally. The tubular and papillary epithelioid cell formations that were observed in these cattle resemble the so-called benign glandular inclusions described in pelvic lymph nodes of women. 4, 5, 7, 12 These inclusions are lined by ciliated cells like those of the endometrium or oviduct and are considered to be a metaplastic phenomenon of peritoneal mesothelium, which is derived from coelomic epithelium from which the Mullerian duct also originates. 4, 5, 7, 12 In this study, some of the epithelioid cells that formed tubular and papillary structures had long slender microvilli, concentric perinuclear bundles of intermediate filaments, and cilia; these cells were adjacent to individual and aggregated epithelioid cells that were also considered to be mesothelial in origin. The fact that some individual and aggregated epithelioid cells expressed fibronectin along the cell membrane or in the cytoplasm could indicate that individual epithelioid cells aggregate or proliferate in lymph nodes and then settle in nodal stroma. The epithelioid cells arranged in tubular or papillary structures were only observed in adult cattle, so they may have been formed postnatally. Furthermore, the minimal or absent expression of basement membrane proteins around the tubular structures may indicate that basement membrane proteins had not been synthesized yet. These results, taken together, would indicate that the tubular or papillary structures were composed of individual and aggregated epithelioid cells, and thus are also considered to be of mesothelial origin.
In summary, epithelioid cells were observed in mediastinal lymph nodes of 60% of cattle. None of the cattle had carcinoma and many lacked pleural/pericardial effusion or inflammation. Recognition of the bland cytologic features of these cells and the retention of normal nodal architecture is useful in distinguishing BEI from metastatic carcinoma. The authors believe these cells are of mesothelial origin and they enter lymph nodes via lymphatic vessels, perhaps, in the absence of pleural disease, as part of normal mesothelial cell turnover.
Footnotes
Acknowledgements
This work was supported by a grant-in-aid to the High Technological Research Center (Rakuno Gakuen University) from the Ministry of Education, Culture, Sports, Science and Technology of Japan.
