Abstract
The H5N1 high-pathogenicity avian influenza (HPAI) viruses have caused widespread disease of poultry in Asia, Africa and the Middle East, and sporadic human infections. The guinea pig model has been used to study human H3N2 and H1N1 influenza viruses, but knowledge is lacking on H5N1 HPAI virus infections. Guinea pigs were inoculated intranasally or intragastrically with A/Vietnam/1203/04 (VN/04) or A/Muscovy duck/Vietnam/209/05 (MDk/VN/05) viruses. Mild listlessness was seen at 2 and 3 days postinoculation (DPI) in guinea pigs inoculated intranasally with VN/04 virus. At 5 DPI, the guinea pigs had bronchointerstitial pneumonia and virus was identified in bronchiolar epithelium and alveolar macrophages. Virus was isolated from the lungs but was lacking from other organs. Minimal lung lesions were seen in intranasal MDk/VN/06 group and virus was not detected, but serologic evidence of infection was observed. Intragastric exposure failed to produce infection or lesions with either virus. The localized respiratory disease in guinea pigs with H5N1 viruses was very similar to that of H3N2 and H1N1 influenza in humans and was less severe than reported for H5N1 human cases.
Avian influenza viruses of the H5N1 subtype have been responsible for the ongoing outbreak of high-pathogenicity avian influenza (HPAI) in poultry and wild birds of Asia, the Middle East, and Africa, and such viruses have sporadically caused fatal infections in humans and other mammals including Bengal tigers (Panthera tigris tigris), stone martens (Martes foina), and domestic cats (Felis catus) and dogs (Canis lupus familiaris). 10 In mammalian animal models such as ferrets, mice, and cats, infections with H5N1 isolates have produced varying syndromes ranging from respiratory disease to severe systemic infection with high mortality after intranasal exposure or when fed infected chicks. 2, 9 Guinea pigs have been proposed as a model to study infection with human influenza A viruses. 1, 3, 4 Human H3N2 and H1N1 influenza viruses were shown to replicate efficiently in the respiratory tract of guinea pigs without prior adaptation, and they produce pulmonary lesions. This study was undertaken to determine the susceptibility and pathogenesis of infection by H5N1 HPAI viruses in 8-week-old guinea pigs after upper respiratory or gastric exposure.
Female Hartley strain guinea pigs (Charles River Laboratories, Wilmington, MA) weighing 300–350 g were housed in high-efficiency particulate air–filtered isolation units maintained within a Biosafety Level 3 Agriculture animal laboratory facility. Animal experiments were conducted under approval of the Institutional Animal Care and Use Committee. Two H5N1 HPAI viruses were used: the human isolate A/Vietnam/1203/04 (VN/04) (Centers for Disease Control and Prevention, Atlanta, GA) and the domestic duck isolate A/Muscovy duck/Vietnam/209/05 (MDk/VN/05) (Dr. Nguyen Van Can, National Center for Veterinary Diagnosis, Hanoi, Vietnam). Virus stocks were propagated in 10-day-old embryonating chicken eggs, and the infected allantoic fluid was titered and reported as 50% egg infective dose (EID50), calculated using the method of Reed-Muench. 8 For each of the viruses, groups of 3 (intragastric, VN/04) to 4 (intranasal, VN/04; intranasal and intragastric, MDk/VN/05) guinea pigs were anesthetized (intramuscular, ketamine [20 mg/kg] and xylazine [2 mg/kg]) and inoculated intranasally (i.e., canula, 106 EID50 of virus in 1 ml divided between each nostril) or intragastrically (i.e., gavage catheter, 106 EID50 of virus in 2 ml). Two sham control guinea pigs were intranasally inoculated with 2 ml of sterile phosphate-buffered saline (PBS). At 5 days postinoculation (DPI), two guinea pigs from each group, except only one guinea pig from the VN/04 intragastric group, were euthanatized, and duplicate sets of the following tissues were collected for virus isolation and histologic examination: nasal turbinate, tonsils, trachea, lungs, olfactory bulbs, brain, heart, blood, spleen, liver, stomach, pancreas, small intestine, large intestine, kidney, adrenal glands, diaphragm, and skeletal muscle. For virus isolation and titration tissues were homogenized in sterile PBS to obtain 10% (w/v) homogenate tested by standard procedures in Madin-Darby canine kidney (MDCK) cells. Titers were expressed as log10 50% tissue culture infecting dose (TCID50). The lower detection limit was 101.5 TCID50/g of tissue. Similar tissue sample sets were collected at 14 DPI in 10% neutral buffered formalin and processed to 5 µm HE-stained sections for histologic examination. Duplicate sections were stained immunohistochemically using as the primary antibody a mouse-derived monoclonal antibody (P13C11) specific for type A influenza virus nucleoprotein antigen. 6 Fast red was used as the substrate chromagen, and slides were counterstained with hematoxylin. Serologic evaluation to determine infection was accomplished by the standard hemagglutination inhibition test.
Clinically, the four guinea pigs inoculated intranasally with VN/04 exhibited slight temporary lethargy between 2 and 3 DPI, and then recovered. No clinical abnormality was observed in the other guinea pig groups. VN/04 virus was isolated only from the lungs of two intranasally inoculated guinea pigs at 5 DPI (Table 1), with titers of 102.75 and 103.75 TCID50/g of tissue, respectively. No virus was isolated from any of the others tissues. In addition, virus was not isolated from any tissues of the guinea pigs that had been intranasally or intragastrically inoculated with MDk/VN/05 virus, intragastrically exposed to VN/04, or intranasally sham inoculated. However, guinea pigs intranasally inoculated with VN/04 or MDk/VN/05 and sampled at 14 DPI, seroconverted, while those that were intragastrically or sham inoculated remained negative for H5 antibodies (Table 1).
Viral isolation from lung and serology of guinea pigs inoculated with H5N1 HPAI viruses via intranasal or intragastric routes.

Nasal turbinate, trachea, tonsils, brain, olfactory bulbs, heart, blood, liver, spleen, stomach, small intestine, pancreas, large intestine, kidney, adrenal glands, diaphragm, and skeletal muscle were examined for all four groups and were HPAI virus isolation negative.
Values represent number hemagglutination inhibition–positive for the two guinea pigs euthanatized at 14DPI; individual geometric titers in parentheses.
Mean value is expressed as log10 TCID50 from two guinea pigs at 5 DPI; = no virus detected.
Mild congestion with slight edema of the lungs was observed at 5 DPI in guinea pigs inoculated intranasally with VN/04 virus. Histologically, moderate lymphocytic bronchointerstitial pneumonia with mild to moderate alveolitis characterized by peribronchiolar lymphocytic infiltration with interstitial edema around the small blood vessels (Fig. 1) as well as necrotic epithelium and debris adhering to the surface, mainly of bronchioles and less frequently in bronchi, were observed. Immunohistochemically, influenza viral antigen was commonly identified in bronchiolar epithelium (Fig. 2). The parenchyma was edematous; alveolar walls congested adjacent to bronchioles; and alveolar lumens were flooded with a mixture of fibrin, macrophages, and, rarely, neutrophils (Fig. 3a). Lymphocytes and histiocytes were seen within alveolar walls, and macrophages were identified within the lumens of terminal airways (Fig. 3b). Rarely, influenza antigen was identified in alveolar macrophages. In contrast, guinea pigs inoculated intranasally with MDk/VN/05 virus exhibited minimal bronchointerstitial pneumonia characterized by mild lymphocytic infiltration around peribronchiolar areas without demonstrating viral antigen in tissues, including lungs. Lesions and viral antigen were lacking at 14 DPI in guinea pigs intranasally inoculated with VN/04 and MDk/VN/05 viruses. Guinea pigs inoculated intragastrically with VN/04 or MDk/VN/05 viruses lacked macroscopic and microscopic lesion, and no viral antigen was demonstrated in any tissues at 5 and 14 DPI.
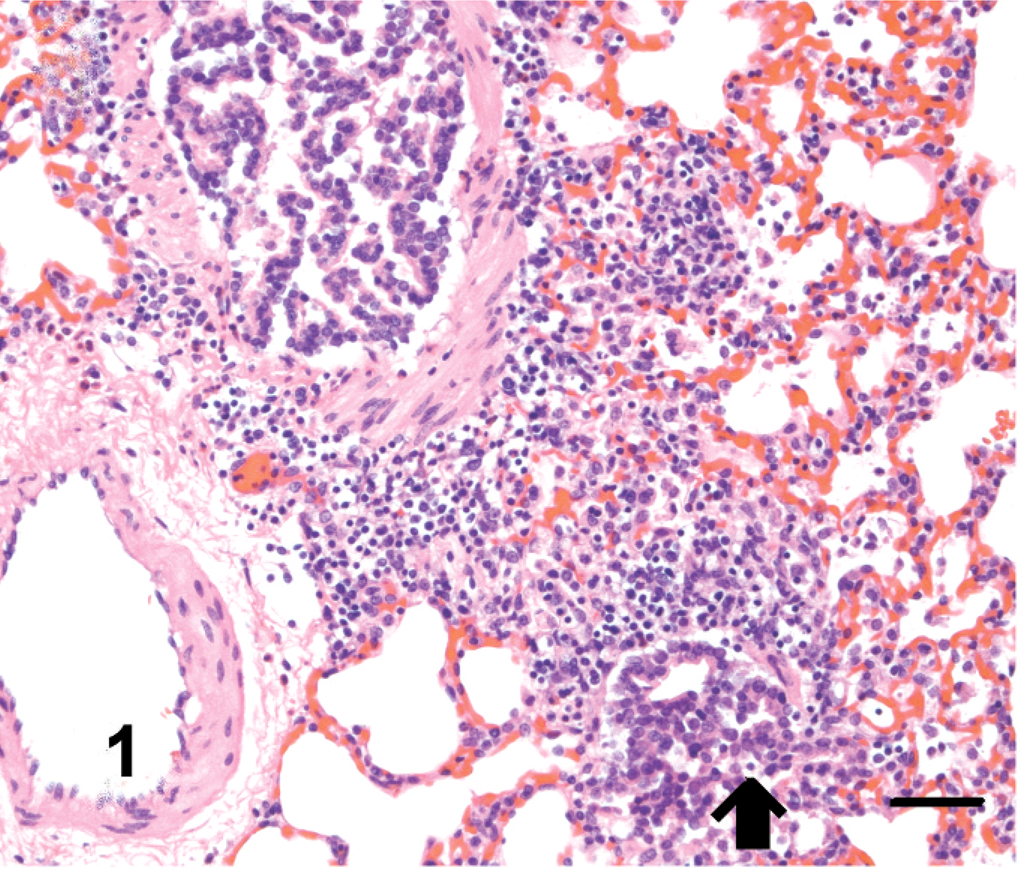
Lung; guinea pigs infected intranasally with VN/04 virus, 5 DPI. Moderate lymphocytic infiltration around bronchiolar areas with necrosis of bronchiolar mucosal epithelium (arrow). HE. Bar = 50 µm.
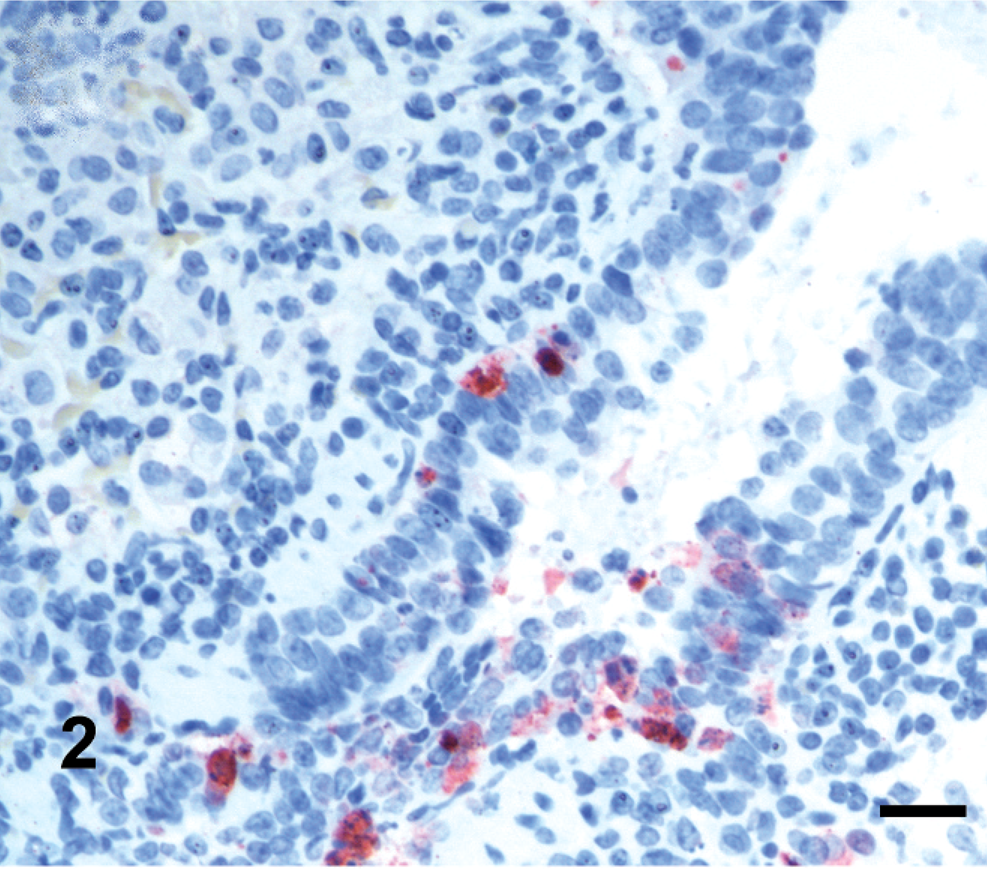
Lung; guinea pigs infected intranasally with VN/04 virus, 5 DPI, Influenza viral antigen detected to bronchiolar epithelia. Biotin-streptavidin complex with hematoxylin counterstain. Bar = 25 µm.
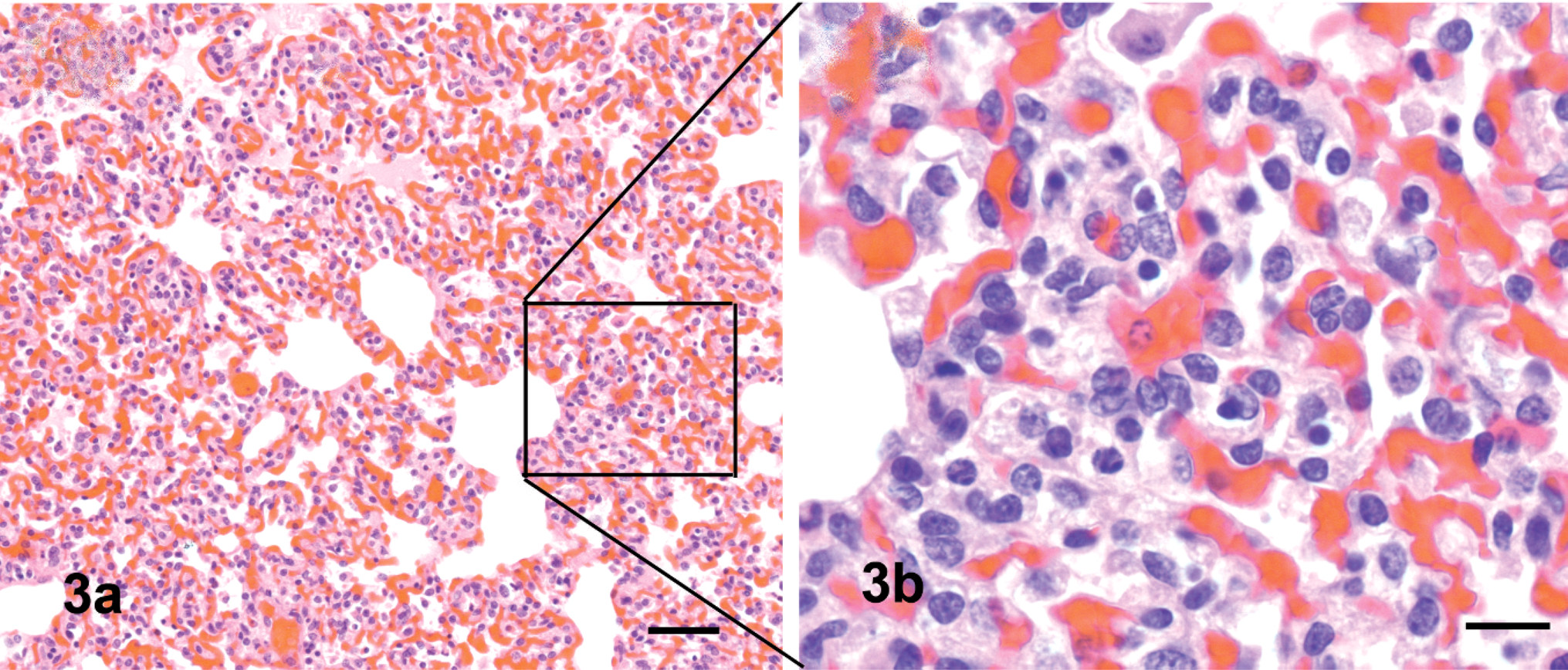
Lung; guinea pigs infected intranasally with VN/04 virus, 5 DPI.
Guinea pigs have several advantages as a mammalian animal model for studying influenza disease, including high susceptibility to infection with human influenza A viruses 4 ; in addition, their lungs contain humanlike bronchus-associated lymphoid tissue. 1 Intranasal inoculation with human influenza A viruses, A/Hong Kong/68 (H3N2) 1, 3, 11 and A/England42/72 (H3N2), 7 resulted in virus replication within the lungs and interstitial pneumonia but without overt clinical illness. Such infections produced immunity to the respective influenza A viruses. By contrast, in our H5N1 HPAI virus study, intranasal exposure to VN/04 virus produced a limited, local lower respiratory tract infection with minimal clinical signs and no mortality. Such infections were associated with bronchiolitis and adjacent lymphocytic to histiocytic interstitial pneumonia. By contrast, intranasal inoculation with MDk/VN/05 virus produced no virologic or immunohistochemical evidence of infection, but minimal peribronchiolar inflammatory lesions and anti-H5 hemagglutinin antibodies were observed, which suggests very limited virus replication. However, the lack of virus replication and lesions after gastric exposure to VN/04 or MDK/VN/05 suggests that inhalation or direct oropharyngeal mucus membrane contact is more likely to lead to infection than alimentary exposure at the same challenge dose in liquid medium, although alimentary exposure to higher doses of virus may have been responsible for H5N1 HPAI infections in domestic cats fed infected chicks. 9
Interestingly, VN/04 virus produced mild clinical disease associated with respiratory infection and lesions in the guinea pig model, but this virus strain was isolated from a fatal human case. By contrast, this virus strain was highly pathogenic in ferret and mouse models, producing systemic, fatal disease associated with a broader tissue tropism for virus replication and lesion production, including high virus titers in multiple organs ranging from 105.5 to 106.0 TCID50/g. 2, 12 However, no single animal model has exactly reproduced the human H5N1 infection and disease. Most fatal human cases have been associated with rapidly progressing fulminant primary viral pneumonia that often progressed to acute respiratory distress syndrome with virus replication in alveolar pneumocytes and macrophages. 5 Most of the available post mortem examinations of humans have not supported viral replication or viral pathology in visceral organs except lungs and intestine. In the current study, the guinea pigs became infected with H5N1 HPAI viruses without production of clinical signs, but the presence of mild specific lung pathology suggests that the guinea pig is worthy of further exploration as an animal model for studying nonlethal respiratory infections by some H5N1 HPAI viruses.
Footnotes
Acknowledgements
We thank Joan Beck for the organization of experiments and technical support. Kira Moresco, James Doster, and Roger Brock are thanked for assistance in technical aspects of this project. This study was supported by contract RSRD-06-00051 from the National Biodefense Analysis and Countermeasures Center, Department of Homeland Security. This project was part of a Research Sabbatical conducted by the senior author (Y. K. Kwon).
