Abstract
Highly pathogenic avian influenza (HPAI) subtype H5N1 is an infectious systemic viral disease that results in high morbidity and mortality in poultry, and has been reported in a wide range of wild bird species during the last few years. An outbreak of HPAI H5N1 occurred in wild birds in Sweden in 2006 that affected several duck species, geese, swans, gulls, and raptors. Tufted ducks (
Introduction
Influenza A viruses (family
In 2005, an outbreak of H5N1 with mortality in several species of waterfowl, predominantly bar-headed geese (
Disease caused by experimental HPAI H5N1 infection has been studied in many avian species, including swans,
3,8
geese,
3,17
passerines, and budgerigars (
The current study describes the pathology and distribution of virus in tissues of tufted ducks naturally infected with HPAI H5N1 virus in Sweden. The purposes of the study were to obtain a better understanding of the disease in a frequently affected wild bird species and to discuss the diagnostic implications.
Materials and methods
Outbreak
Of the 83 sampled tufted ducks, 37 tested positive for HPAI. Three birds (2 of which were not included in the present study) were negative on PCR analysis of the initial tracheal swab but had similar encephalic and pancreatic histologic lesions as positive birds. Therefore, PCR analysis was conducted on brain samples from these birds by using the same method as described for the swabs.
Sample selection
For the present study, 20 tufted ducks were selected of the 37 that tested positive for the presence of HPAI viral RNA. Inclusion criteria for the study were low degree of autolysis, detection of HPAI nucleic acid by PCR in at least one sample (swab or tissue), and availability of brain and at least one other organ for histopathologic investigation. Of the 20 birds included in the study, 2 were sampled by cloacal swabs, 17 were sampled by tracheal swabs, and 1 was sampled by both cloacal and tracheal swabs for PCR analysis (Table 1). The selected ducks included 9 females, 8 males, and 3 whose sex was not recorded at necropsy. Eleven were adults, and 2 were juveniles, and, in 7, the estimated age was not recorded. Thirteen ducks were found dead, and 7 ducks were euthanized because of severe neurologic signs, including head tilt, circling, loss of balance, and drooping wings. Tissues available for histopathology from the selected ducks included brain (
Diagnostic tests for avian influenza in 20 selected tufted ducks. *
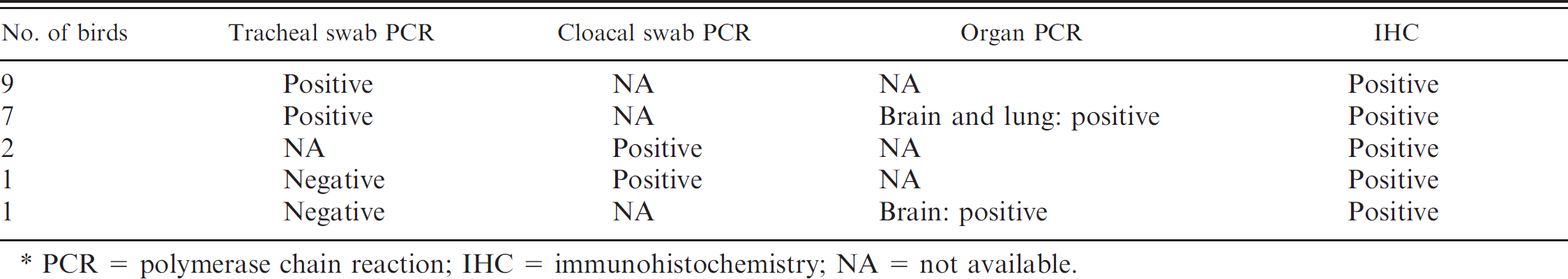
PCR = polymerase chain reaction; IHC = immunohistochemistry; NA = not available.
kidney, pancreas, and intestines were also stored at −20°C for possible additional virologic analyses.
Histopathology and IHC
After formalin fixation, the tissue samples were processed routinely, sectioned at 4–5 μm, and stained with hematoxylin and eosin. Duplicate sections for IHC were mounted onto glass slides treated with adhesive, c depar-affinized in xylene, and rehydrated. Endogenous peroxidase activity was blocked with 3% hydrogen peroxide for 7 min. The sections were treated with proteinase K d for 6 min and with 2% bovine serum albumin e for 20 min. They were then incubated at room temperature for 60 min with a commercial anti-influenza A nucleoprotein primary monoclonal antibody, f diluted 1:200. The detection was conducted with labeled streptavidin-biotin. g The color was developed with aminoethylcarbazole substrate. h Sections were counterstained with hematoxylin, coverslipped, and examined microscopically. Each immunostain included a positive reference control (pancreas from a swan that was confirmed positive for H5N1) and a negative control (pancreas of a healthy chicken). Each section was also accompanied by a primary antibody-omitted control. The presence of viral antigen was described for each tissue. The intensity and extension of the immunostaining was assessed semiquantitatively (negative, mild, moderate, or marked).
Results
Gross pathology
Most of the ducks were in good body condition (
Histopathology and IHC
The main histologic lesions associated with the presence of avian influenza antigen were found in the brain, pancreas, and upper respiratory tract sections. All 20 birds were positive by IHC in at least 1 tissue.

Pancreas of tufted duck (

Brain: influenza viral antigen (red stain) in nucleus and cytoplasm of neurons. Influenza A immunohistochemistry. Hematoxylin counterstain. Bar = 50 μm.

Nasal mucosa: influenza viral antigen in superficial epithelium and submucosa. Immunohistochemistry. Hematoxylin counterstain. Bar = 50 μm.

Liver: area of hepatic necrosis with influenza viral antigen in nucleus and cytoplasm of hepatocytes. Immunohistochemistry. Hematoxylin counterstain. Bar = 50 μm.
Fibrin and cell-rich exudates were seen in the lumen of these airways. Positive IHC staining was not observed in the secondary bronchi or in any of the air sacs that were examined. Inflammatory lesions were not observed in the air sacs.
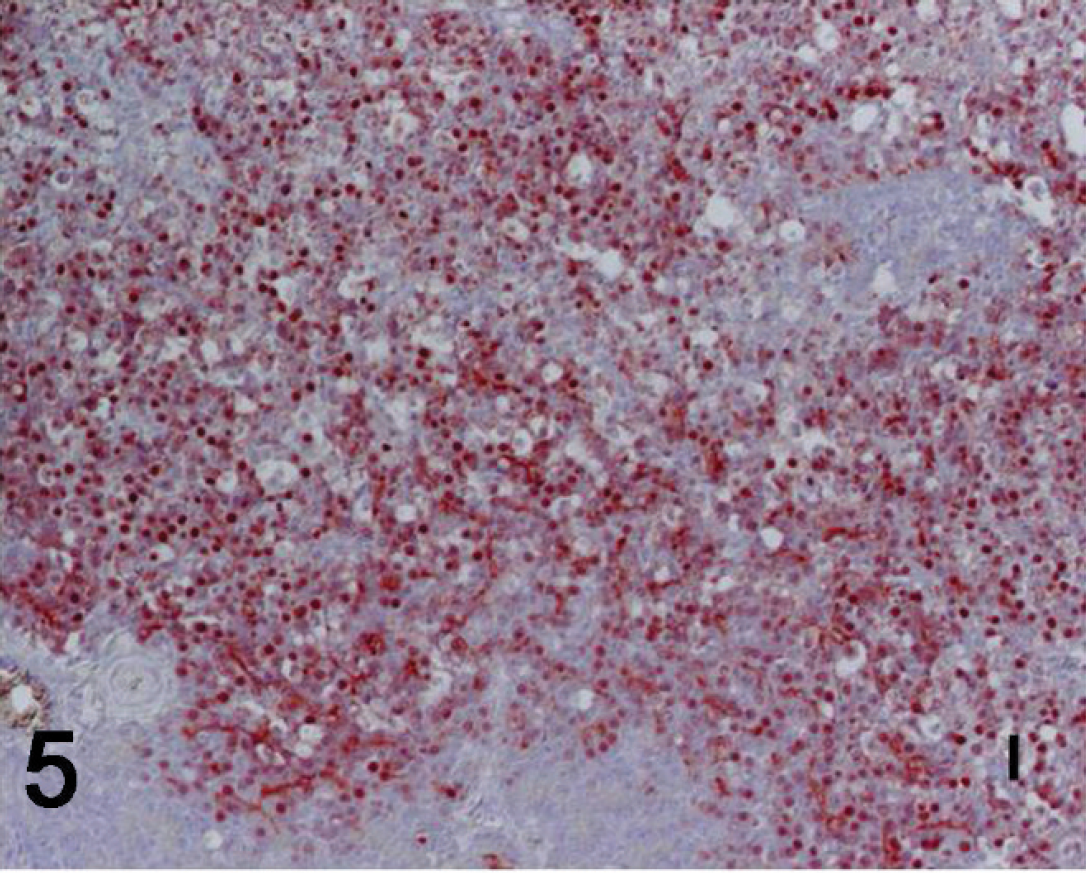
Pancreas: abundant influenza viral antigen in nucleus and cytoplasm of acinar cells bordering an area of necrosis. Immunohistochemistry. Hematoxylin counterstain. Bar = 50 μm.

Kidney: influenza viral antigen in nuclei of tubular cells. Some of the fine granular staining in the cytoplasm and the luminal portions of the tubular cells is nonspecific. Immunohistochemistry. Hematoxylin counterstain. Bar = 50 μm.

Ovary: influenza viral antigen in cytoplasm and nucleus of follicular cells. Immunohistochemistry. Hematoxylin counterstain. Bar = 50 μm.

Small intestine: marked staining of the submucosal (Meissner) plexus because of the presence of influenza viral antigen. Note the absence of antigen in the intestinal mucosa. Immunohistochemistry. Hematoxylin counterstain. Bar = 50 μm.
tubular epithelium was noted in one other bird. Viral antigen was found in these 2 ducks in renal tubular cells and in cellular debris in the tubular lumina (Fig. 6).
Discussion
In the current study, tufted ducks naturally infected with HPAI H5N1 revealed a range of mild-to-severe, acute-to-subacute inflammatory lesions that affected between 1 and 6 organs per bird, which confirmed the within-species variation of tissue tropism in H5N1 infections. These variations are likely to be more marked in natural than experimental disease. In naturally infected free-living birds, the clinical and pathologic picture of viral infection is influenced by several factors, such as the age of the bird, the amount and routes of viral exposure, the presence of concomitant infections, the levels of immunity acquired during previous exposure to influenza viruses, 8 and the time course of the infection.
The most consistent lesions in the tufted ducks were multifocal nonsuppurative encephalitis and multifocal necrotizing pancreatitis. This is in agreement with natural H5N1 infection in other wild bird species 6,11,12,14,24,25 and with experimental infection of wild birds, including tufted ducks. 9 The lesions found in the brain and pancreas were similar to those described in other natural and experimental infections. However, in the present study, histologic lesions were not always associated with the presence of viral antigen as might have been expected. In general, the most strongly IHC-positive cells were devoid of light microscopic evidence of cell injury and were located peripherally or adjacent to areas of necrosis. In contrast, necrotic cells were devoid of viral antigen. This may indicate that the preservation of virus in the tissue may be limited to a short period. Viral antigen may thus be difficult to detect in necrotic tissue and in cases with a longer time course of infection. The high number of ducks with encephalitis, in association with high levels of virus as detected by IHC, suggests that the virus is highly neurotropic, as previous studies showed. 4,9,17,25 Signs of neurologic disturbance were, in fact, the main observed clinical signs in infected birds. It is likely that the encephalitis, in combination with an inability to feed or drink, was the ultimate cause of death in most of the birds. Neurotropism of the virus was also observed in the peripheral nervous system, with detection of virus in the submucosal and myenteric plexa of the intestine and in ganglion cells. These findings in the peripheral nervous system are not well documented in wild birds with natural H5N1 infection but have been described in several experimentally infected wild bird species, such as wood ducks, 4 Canada geese, 17 and tufted ducks. 9
Rhinitis, with the presence of viral antigen in nasal epithelium, was another common finding in the tufted ducks. This has rarely been described in naturally infected birds. Rhinitis was not observed in naturally infected mute and whooper swans, 25 whereas experimentally infected mute swans 8 and Pekin ducks 15 had signs of rhinitis. Descriptions in wild birds may be underrepresented, because this tissue is not often investigated in routine postmortem examinations. The presence of mites in the nasal cavity of some of the tufted ducks may account for some of the inflammatory changes in their nasal epithelium. However, the presence of viral antigen in most of the ducks with inflammatory changes suggests that the virus is indeed related to the inflammation. It could be speculated that primary parasitic lesions in the mucosa predispose to nasal HPAI infection.
Despite the presence of congestion and inflammatory changes in the lungs of many of the tufted ducks, mild antigen staining was only found in the lungs of 3 ducks. Viral antigen was not found in the remainder of the respiratory tract (trachea and air sacs). It is probable that, at the time of death, the virus was no longer detectable in the lungs. Some of the inflammatory changes may be attributed to possible concomitant infections. The low number of antigen-positive lungs is in agreement with descriptions in naturally infected mute and whooper swans in which only vascular endothelium in the lung of 2 of 18 swans contained viral antigen, 25 as well as in magpies in which only pulmonary endothelial cells were positive. 11 In general, the lower respiratory tract seems to be less affected in naturally infected wild birds 11,24,25 than in poultry 16 and experimentally infected birds. 9 However, in experimentally infected tufted ducks inoculated intratracheally and intra-esophageally, viral antigen was found in epithelial cells of the bronchus, parabronchus, and atrium, as well as in air capillaries. 9 The route of administration (often intranasal and/or intratracheal) and the viral dose in experimental infections probably increase the likelihood of finding viral antigen in both the upper and lower respiratory tract compared with wild birds in which the route of infection and the virus dose can vary from case to case. Thus, in the naturally infected tufted ducks included in the current study, the virus seems either to be cleared from the lungs fairly rapidly or to be more rhinotropic than pneumotropic.
The heart is often described as one of the main targets for H5N1 viral replication, with the presence of viral antigen in myocytes in association with myocardial necrosis in both naturally 5,11,24,25 and experimentally 9,15 infected birds. Half of the tufted ducks in the current study had lesions in the heart, but viral antigen could not be detected, which indicated that the virus was no longer present or that the changes were not caused by viral infection. Localization of viral antigen to organs, such as liver, kidneys, and adrenal glands, was observed in only a few birds. Although the virus has pantropic potential, other factors seem to affect the distribution of virus and lesions in different organs.
Although the number of organs and the severity of lesions varied among individuals, the disseminated and multifocal distribution of lesions in the tufted ducks suggests that the virus is spread hematogenously. However, there was no evidence of actual vascular damage. Viral antigen was only found in the endothelium and tunica media of a few blood vessels in the nasal mucosa of 2 ducks. This is in agreement with experimentally infected mute swans in which antigen was found in submucosal arteries of the nasal concha in only 3 of 14 swans with signs of rhinitis and which also had large amounts of antigen in other tissues. 8 Experimentally infected Pekin ducks also had signs of rhinitis but had no lesions or antigen in the endothelium, 16 and endotheliotropism was not detected in experimentally infected tufted ducks. 9 The previous studies are in contrast to experimentally infected black swans in which the virus led to a vasculotropic infection, with the presence of viral antigen in the endothelial cells in most visceral organs and the brain. 3
The tufted ducks included in the current study were tested by tracheal and/or cloacal swabs. When considering the very small amounts of antigen visible by IHC in the lower respiratory tract and none visible in the intestinal epithelium, the question arises as to where the virus is derived in the PCR-positive tracheal and cloacal swabs. It was shown experimentally that detection of virus in pharyngeal swabs varied between species–-tufted ducks, common pochards (
It is interesting to note that in the current study, 2 birds were PCR negative in tracheal swabs but were later confirmed to be infected by HPAI by PCR of cloacal swabs or of brain tissue and thus were included in the study. Similar findings were found in other Swedish wild bird species tested with tracheal swabs. Of 45 HPAI-positive birds (including the tufted ducks in the present study) that were also analyzed histologically, 10 were PCR negative in tracheal swabs. These latter birds had histopathologic changes consistent with HPAI infection, including encephalitis and, in some cases, pancreatitis. They, therefore, were retested by PCR with brain and lung tissue and, subsequently, 5 of these were positive (unpublished data). Thus, PCR of tracheal swabs failed to detect at least 11% of positive cases. The failure to detect positive birds by using tracheal swab analysis only as well as the inter- and intraspecies variations of lesions and distribution of virus imply that sampling for HPAI should include both tracheal and cloacal swabs, and that the highest sensitivity of detection is achieved by testing brain and lung tissue. When considering the fact that nasal epithelium often contained viral antigen, oropharyngeal swabs should perhaps also be included. It should be noted that analysis of swabs alone is a screening method and not a completely reliable diagnostic method.
The acute nature of lesions in naturally infected tufted ducks and the high number of positive tufted ducks compared with other affected bird species observed during the influenza outbreak in Sweden suggest that subclinical infections with a carrier state are probably rarely encountered among tufted ducks. Therefore, tufted ducks found dead or with neurologic signs should be sampled and necropsied because they can serve as “indicator species” for HPAI. Tufted ducks are probably particularly sensitive to HPAI, as suggested in experimental infections, 9 but other factors, such as natural behavior of the ducks, population density, environmental factors, amount of viral exposure, level of viral replication in tissues, and concomitant infections, may also affect the clinical and pathologic picture of avian influenza infections in free-living birds. 15 The Swedish outbreak index cases came from a specific area during special climatic and environmental conditions (cold weather with large amounts of ice cover along the coast and a limited amount of open water) where larger than normal numbers of tufted ducks aggregated in a limited amount of space, which may have created a higher risk of efficient spreading of the virus. To assess how sensitive tufted ducks are to infection with H5N1 in a natural setting, it would have been interesting to sample asymptomatic individuals from areas where positive ducks were found. However, this was not possible during the outbreak. Ancillary diagnostic tests, such as parasitology and bacteriology, would also have been beneficial to assess possible concomitant infections, but these were not conducted because of constraints when working with a biosafety level class-3 pathogen. The description of the pathology in a natural H5N1 infection as described in the current study can serve as a complement to and in comparison with experimental studies. The current study also provides information on variations in presentation of the disease and on confounding factors that should be kept in mind when dealing with natural viral infections and samples obtained from the field.
Acknowledgements
The authors thank Jonas Malmsten and the technicians at the postmortem room for help with necropsies; Ewa Westergren, Lotta Ottander, Susanne Andersson, Gudrun Andersson, and Maria Lögdahl for technical laboratory help; and Thijs Kuiken for help with developing the IHC method. The authors are also grateful to all who helped collect the birds, especially to L. E. Carlsson, who also provided photographs and video films of affected birds. This study was funded by The Swedish Research Council for Environment, Agricultural Sciences and Spatial Planning (FORMAS 221–2007–1177).
Footnotes
a.
MagAttractr Virus Mini M48 Kit, Qiagen GmBH, Hilden, Germany.
b.
Magnatrix 8000+ 96-well extraction robot, NorDiag ASA, Oslo, Norway.
c.
VECTABONDTM, Vector Laboratories Inc., Burlingame, CA.
d.
Proteinase K Dako S3004, Dako Denmark A/S, Glostrup, Denmark.
e.
Bovine Albumin-BSA-Cohn Fraction V-A4503, Sigma-Aldrich Sweden AB, Stockholm, Sweden.
f.
mAbs AI-84, European Veterinary Laboratory, Woerden, The Netherlands.
g.
LSAB Dako 0690, Dako Denmark A/S, Glostrup, Denmark.
h.
AEC-kit Vector SK-4200, Vector Laboratories Inc., Burlin-game, CA.
