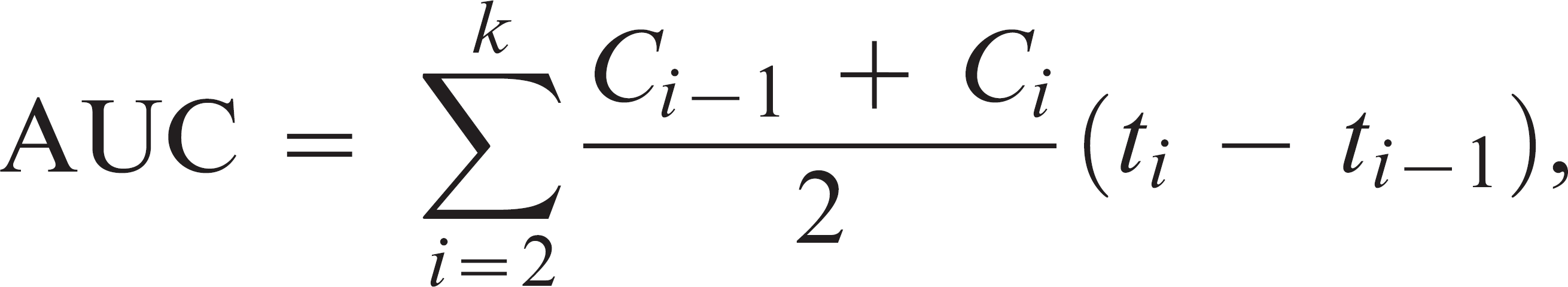Abstract
Terrestrial wild birds commonly associated with poultry farms have the potential to contribute to the spread of H5N1 highly pathogenic avian influenza (HPAI) virus within or between poultry facilities or between domesticated and wild bird populations. This potential, however, varies between species and is dependent on several virus and host factors, including habitat utilization, susceptibility, and viral shedding patterns. To provide data on susceptibility and shedding patterns of house sparrows (Passer domesticus) and rock pigeons (Columba livia), 20 birds from each species were inoculated with decreasing concentrations of A/whooper swan/Mongolia/244/05 (H5N1) HPAI virus, and the birds were evaluated for morbidity, mortality, viral shedding, and seroconversion over a 14-day trial. The house sparrows were highly susceptible to the H5N1 HPAI virus as evidenced by low infectious and lethal viral doses. In addition, house sparrows excreted virus via the oropharynx and cloaca for several days prior to the onset of clinical signs. Based on these results, house sparrows could play a role in the dissemination of H5N1 HPAI virus in poultry. In contrast, pigeons were resistant to the HPAI virus, requiring a high concentration of virus to produce infection or death. When infection did occur, the duration of viral shedding was brief, and viral titers were low. The data suggests that pigeons would contribute little to the transmission and spread of H5N1 HPAI virus in poultry.
Keywords
Introduction
Over the last decade, H5N1 highly pathogenic avian influenza (HPAI) viruses have exhibited an unprecedented ability to infect and cause disease in an extremely diverse group of hosts, including wild and domestic birds and a variety of mammalian species. 21,25,27 The frequency of interspecies transmission of H5N1 HPAI virus in Southeast Asia is the result of a wide host range and an extensive interface between humans, domestic animals, and wildlife. This large interface serves as a constant and significant risk for virus spillover into nonreservoir hosts, such as humans or wildlife, and makes eradication difficult, if not impossible, in endemic areas.
Though domesticated fowl and ducks represent a significant reservoir for H5N1 HPAI viruses in Southeast Asia, there is field and experimental evidence to support that some species of migratory waterfowl can potentially contribute to the spread of these viruses over long distances. 11,12 Viral transmission between migratory waterfowl and domestic bird populations, in either direction, can occur through several mechanisms, including direct contact in areas where the 2 groups share environments, scavenging on H5N1 HPAI virus-infected carcasses, and through “bridge” species that can transmit the virus between domestic poultry and migratory waterfowl populations. Small terrestrial birds commonly found around poultry environments have a strong potential to serve as important “bridge” species owing to their peridomestic behavior and extensive interaction with both free-living and domesticated avian populations. In addition, because of their behavior and habitat utilization, small terrestrial birds may also pose a risk for transmitting H5N1 HPAI virus to humans.
Since 2002, H5N1 HPAI viruses have occasionally been isolated from wild terrestrial birds, including Eurasian tree sparrows (Passer montanus) and rock pigeons (Columba livia), and mortality associated with natural H5N1 HPAI virus infection in these species has been reported. 9 Experimentally, an early H5N1 HPAI isolate (A/chicken/Hong Kong/220/97 [H5N1]) was only capable of low-level replication in house sparrows (Passer domesticus), a species closely related to tree sparrows, and caused mild clinical signs but no mortality. 22 An infectivity trial performed with 4 different 2005–2006 H5N1 HPAI viruses, however, indicated that more recent isolates were highly virulent for house sparrows causing 66–100% mortality in intranasally inoculated birds. 2 House sparrows infected with the 4 viruses in the latter study shed moderate concentrations of virus via the oropharynx (maximum titers 102.6–4.7 median egg infectious dose [EID50]/ml) and cloaca (101.3–4.1 EID50/ml) prior to death. Of interest, however, despite their high susceptibility, house sparrows in this study did not transmit virus to contact sparrows that were in the same enclosures. The data on H5N1 HPAI in species of the order Columbiformes is variable, with columbids exhibiting a range of susceptibility under both natural and experimental settings. Pigeons have been resistant to H5N1 HPAI virus infection in the majority of experimental trials 2,18,20 ; however, 2 recent studies have reported that inoculation of pigeons with high concentrations (10 6–8 EID50) of H5N1 HPAI virus resulted in infrequent morbidity and mortality. 13,27 Similarly, mortality in columbids associated with natural and confirmed H5N1 HPAI virus infections have been reported but generally are rare events involving a few individual birds. 9
Pigeons and sparrows likely represent 2 extremes of susceptibility to H5N1 HPAI virus among wild terrestrial birds commonly found around poultry environments. However, discrepancies and knowledge gaps in the existing body of data on these 2 species make it difficult to understand the potential role that either species may play in the epidemiology of H5N1 HPAI virus. The overall goals of the current study were to evaluate the potential for house sparrows and pigeons to become infected with and shed H5N1 HPAI virus and to serve as a “bridge species” between wild and domestic birds or between geographically separated poultry flocks. Specifically, the objectives of the study were 1) to determine the concentration of H5N1 HPAI virus required to cause infection or death in rock pigeons and house sparrows; 2) to evaluate the concentration and duration of viral shedding in relation to clinical signs for each of these species; and 3) to examine gross and microscopic lesions and distribution of viral antigen, using immunohistochemistry (IHC), associated with infection.
Materials and methods
Virus
The H5N1 HPAI virus strain, A/whooper swan/Mongolia/244/05 (H5N1), used in the current study was obtained from the Southeast Poultry Research Laboratory (SEPRL; Athens, GA). This H5N1 HPAI virus strain is in the clade 2.2 and was originally isolated from a dead whooper swan (Cygnus cygnus) in Mongolia during a large mortality event involving over 80 migratory wild birds (World Animal Health Organization: 2005. Highly pathogenic avian influenza in Mongolia in migratory birds. Disease Information. Accessed on Aug. 12, 2005. Available at: http://www.oie.int/eng/info/hebdo/ais_58.htm#Sec4). Viral stock was propagated by second passage in 9- to 11-day-old specific pathogen-free (SPF) embryonating chicken eggs. Infective allantoic fluid from the inoculated eggs was diluted in brain-heart infusion (BHI) medium to yield calculated titers of 102.0 (low dose), 104.0 (medium dose), and 106.0 (high dose) EID50 per 0.1 ml (single-bird inoculum) for the rock pigeons trial and per 0.05 ml for the house sparrow trial. Actual back-titers determined in embryonating chicken eggs using standard techniques28 were 103.1, 104.9, and 106.1 EID50/0.1 ml for the rock pigeon trial and 102.4, 104.6, and 106.6 EID50/0.05 ml for the house sparrow trial. A sham-inoculum was prepared by diluting sterile allantoic fluid 1:30 in BHI.
Animals
Twenty wild-caught adult house sparrows and 20 wild-caught adult rock pigeons were collected in Oconee County, Georgia and Macon County, Georgia, respectively, by personnel of the Southeastern Cooperative Wildlife Disease Study (SCWDS; University of Georgia [UGA], Athens, GA). Both males and females were included for both species in approximately equal numbers. All birds, for both species, were housed individually in self-contained isolation units, which were ventilated under negative pressure with high efficiency particulate air (HEPA)-filtered air. All birds were maintained under continuous lighting, and food and water were provided ad libitum. General care was provided in accordance with the guidelines of the Institutional Animal Care and Use Committee, as outlined in the Guide for the Care and Use of Agricultural Animals in Agricultural Research and Teaching 7 and under an animal use protocol approved by the Institutional Animal Care and Use Committee at both SEPRL and UGA. All experiments were performed in a BSL 3 enhanced facility at SEPRL. 1
Experimental design
House sparrows and rock pigeons were evenly divided into 4 treatment groups, per species, with 5 birds in each group. Each group was intranasally inoculated with either a sham-inoculum or 1 of 3 different viral doses of the A/whooper swan/Mongolia/244/05 virus. Prior to inoculation, blood was collected from all birds for serologic testing via the agar gel precipitin (AGP) and blocking enzyme-linked immunosorbent assay (bELISA) tests to determine whether any of the birds possessed antibodies to avian influenza (AI) virus. In addition, oropharyngeal and cloacal swabs were collected from each bird for virus isolation prior to inoculation to verify that no birds were actively shedding AI virus at the start of the study. After inoculation, birds were observed daily for morbidity and mortality. Morbidity was defined as any clinical abnormality detectable upon physical examination after inoculation with the A/whooper swan/Mongolia/244/05 virus, including weakness, shivering, ruffled feathers, respiratory dysfunction, and neurologic signs. Birds exhibiting severe neurologic signs were euthanized as described below. Oropharyngeal and cloacal swabs were collected from all birds on 0, 2, 4, 5, 7, and 10 days postinoculation (DPI), in addition to all birds that were found dead or were euthanized due to the severity of their clinical condition. At 14 DPI, blood was collected from the surviving birds for serologic testing, and all remaining birds were euthanized by intravenous administration of sodium pentobarbital (100 mg/kg). Full pathologic examinations, including gross necropsy, routine microscopic evaluation, and IHC, were performed on all 5 birds, for each species, in the high-dose group and also for all sham-inoculated control birds. The median bird infectious dose (BID50) and median bird lethal dose (BLD50) were calculated for each of the 3 viral-inoculated groups using the Spearman and Karber method 10 and expressed as EID50. Infection for BID50 calculation was determined based on positive virus isolation (in birds that died) or seroconversion (in surviving birds). Lethality for the BLD50 calculation was determined based on mortality.
Virus isolation and serologic testing
Oropharyngeal and cloacal swabs were stored at −70°C after collection until virus isolations and titrations were performed. Virus isolations from oropharyngeal and cloacal swabs were performed in SPF embryonating chicken eggs using standard procedures. 28 Viral concentration in positive samples was determined by microtiter endpoint titration using primary cultures of chicken embryo fibroblasts (CEFs). 24 The titrations were performed using a previously reported protocol. 6 Viral titers were expressed as median tissue culture infectious dose (TCID50)/ml. The minimal detectable limit of this assay was 101.87 TCID50/ml. When virus was isolated in eggs at a concentration below the tissue culture detection limit, a value of 10 1 TCID50/ml was used for statistical analysis. Serologic testing was performed via the AGP test using standard procedures 28 and a commercially available AI virus antibody test bELISA kit a following the manufacturer's instructions.
Histopathology and immunohistochemistry
Tissues samples were collected at necropsy and preserved in 10% neutral buffered formalin. After fixation, the tissues were routinely processed and embedded in paraffin. Duplicate sections were cut at 5 μm and routinely stained with hematoxylin and eosin and immunohistochemically stained using a mouse-derived monoclonal antibody (P13C11) specific for type AI virus nucleoprotein antigen as the primary antibody (SEPRL). Procedures used to perform and interpret the IHC examination followed those previously described. 23
Statistical analysis
Survival functions for inoculation dosage groups were calculated using the Kaplan-Meier method, and the equivalence of survival functions was evaluated using the log rank test for trend. Viral shedding was quantified over time by calculating the area under the curves (AUCs) formed by plotting viral titers (log10TCID50/ml) versus the number of DPI at the time of sample collection. AUCs were calculated using the trapezoidal method:
where Ci is the TCID50/ml at time ti . The AUCs of different dosage groups were compared using the Kruskal-Wallis test, and comparisons between different sample types collected within the same birds were made using the Wilcoxon signed rank test for paired data. Maximum titers observed for birds on any DPI were also compared between groups using the Kruskal-Wallis test and between sample types using the Wilcoxon signed rank test. All testing was performed assuming a 2-sided alternative hypothesis and P values < 0.05 were considered statistically significant. Analyses were performed using commercially available statistical software. b
Results
None of the pigeons or sparrows used in the current study had preexisting antibodies to AI virus or were excreting virus at the start of the experiment. Sham-inoculated birds, in both species, did not exhibit any evidence of infection during the course of the study, including viral shedding, clinical signs of disease, expression of viral antigen in tissues, or seroconversion. All 5 sparrows in the medium- and high-dose groups died, and 3 of the 5 sparrows in the low-dose group died. Survival functions were estimated for sparrows in the different inoculation dosage groups as shown in figure 1. Survival times were longest for the low-dose group and decreased significantly with increasing inoculation doses (log rank test for trend, P = 0.002). The 2 surviving sparrows in the low-dose group did not exhibit clinical signs during the trial or shed virus that was detected, but both birds developed post-exposure antibodies to AI virus as determined by the AGP and bELISA. The endpoints for BID50 and BLD50 calculations were not determined but were less than the lowest inoculation dose (i.e., the calculated BID50 and BLD50 were <102.4 EID50). The mean survival times for the sparrows in the low-, medium-, and high-dose groups were 8.2 DPI, 6.2 DPI, and 5.0 DPI, respectively. Most sparrows, in all 3 groups, either exhibited no clinical signs or had a brief duration of sickness prior to death (average duration 1 day; range: 0–2 days). Clinical signs, when detected, varied in severity and were nonspecific, including weakness, lethargy, puffed up feathers, imbalance, and severe neurologic signs such as head tremors and seizures.
Kaplan-Meier survival curves for sparrows in the low- (10 2 ), medium- (104), and high-dose (106) inoculation groups (n = 5 per group). Log rank test for trend, P = 0.002.
The quantity of virus detected in oropharyngeal and cloacal swabs was plotted over DPI for each sparrow (fig. 2), and information on AUCs and maximum titers was summarized by group (table 1). There was no significant difference between inoculation dose groups with respect to the AUCs for either the oropharyngeal swabs (Kruskal-Wallis, P = 0.594) or the cloacal swabs (P = 0.532). However the AUCs were significantly lower for the cloacal swabs than for the oropharyngeal swabs (Wilcoxon signed rank test, P = 0.002), suggesting that a lower quantity of virus was shed from the cloaca. Comparison of maximum viral titers observed on any DPI for birds in the different groups yielded results that were similar to the AUC analysis. There was no significant difference between groups with respect to the maximum viral titer for either the oropharyngeal (P = 0.857) or cloacal swabs (P = 0.411), but the maximum titers of cloacal swabs were significantly lower than those of oropharyngeal swabs (P = 0.043).
Gross lesions were variably detected in the 5 highdose group house sparrows that were pathologically examined, but the most consistent lesion was red mottling of the pancreatic parenchyma. Other gross lesions that were observed in these sparrows included splenomegaly and pulmonary edema and congestion. Viral antigen and associated microscopic lesions were detected in multiple organs (table 2). The distribution of viral antigen varied between individual house sparrows, but the H5N1 HPAI virus had the most consistent and strong tropism for the adrenal gland, pancreas, and testis (fig. 3). The most frequent and severe microscopic lesions were multifocal to coalescing necrosis of acinar cells in the pancreas and multifocal to confluent areas of necrosis in the adrenal glands admixed with variable infiltrates of heterophils. Other variably detected lesions included myocardial necrosis, lymphoplasmacytic myocarditis, degeneration and necrosis of Sertoli cells, lymphocytic orchitis, heterophilic oophoritis, severe neuronal degeneration and necrosis in the cerebrum and cerebellum, lymphoplasmacytic perivascular encephalitis, lymphocytic ganglioneuritis in peripheral nerves innervating the heart and adrenal gland, lymphocytic air sacculitis, degeneration and necrosis of myocytes in the skeletal muscle, and lymphocytic to heterophilic nephritis.
Two of the 5 pigeons inoculated with the high dose of H5N1 HPAI virus died, with a mean survival time of 7.5 days (range: 7–8 days). One of these birds was found dead without exhibiting clinical signs, but the other exhibited moderate weakness and lethargy for 2 days prior to being euthanized due to severe neurologic signs. One of the 3 remaining pigeons in this viral dose group became mildly weak and lethargic for 2 days (6–8 DPI) prior to making a full recovery. The remaining 2 pigeons inoculated with the high dose of A/whooper swan/Mongolia/ 244/05 virus remained clinically normal for the duration of the trial. All 3 surviving pigeons at this dose developed post-exposure antibodies to AI virus as detected by AGP and bELISA. None of the pigeons inoculated with low or medium doses of A/whooper swan/Mongolia/244/05 virus became ill, shed virus, or developed post-exposure antibodies. The BID50 titer was determined to be 105.5 EID50, but the maximum titer of the A/whooper swan/Mongolia/244/05 virus did not produce death in 50% or more of the pigeons and thus the calculated BLD50 was >106.1 EID50.
All 5 of the pigeons in the high-dose group excreted H5N1 HPAI virus as detected by isolation in embryonating chicken eggs. However, viral shedding in all 5 pigeons was for a brief duration and at low titers that were below the detectable limit of the tissue culture titration assay (101.87 TCID50/ml). The routes of viral shedding varied between individual pigeons; two pigeons only excreted virus via the oropharynx (1 pigeon was positive on 4 and 6 DPI and the other was positive only on 7 DPI), 2 pigeons only excreted virus via the cloaca (both only positive on 2 DPI), and 1 pigeon excreted virus via the oropharynx and cloaca (positive oropharyngeal swabs on 2 and 4 DPI and positive cloacal swab only on 2 DPI). Red mottling of the pancreatic parenchyma in the 2 pigeons that died was the only gross lesion observed in the 5 pigeons from the high-dose group. Viral antigen and microscopic lesions associated with H5N1 HPAI virus infection were only detected in the 3 pigeons that exhibited clinical signs of disease; the distribution of antigen and lesions is summarized in table 2. The A/whooper swan/Mongolia/244/05 virus had a tropism for the brain in pigeons with viral antigen demonstrated in neurons and microglial cells. In the 2 pigeons that died, numerous foci of viral staining were associated with lymphoplasmacytic encephalitis or meningoencephalitis and marked neuronal necrosis. Viral antigen expression was greatly reduced in the pigeon that survived and was restricted to infrequent neurons in a limited number of foci in the cerebrum and brainstem. Microscopic lesions in the brain of this surviving bird consisted of lymphoplasmacytic encephalitis.
Individual oropharyngeal (OP) and cloacal viral shedding for house sparrows (Passer domesticus) intranasally inoculated with varying doses of A/whooper swan/Mongolia/244/05 (H5N1) highly pathogenic Avian influenza virus: 1A, low dose/OP; 1B, low dose/cloacal; 2A, medium dose/OP; 2B, medium dose/cloacal; 3A, high dose/OP; and 3B, high dose/cloacal. Enlarged data points for each viral shedding curve represent sampling points when morbidity or mortality was detected. TCID50 = median tissue culture infectious dose.
Discussion
The results of the study presented herein provide data that are consistent with existing literature and further support the observations that house sparrows are highly susceptible to H5N1 HPAI virus and pigeons are resistant. In addition, the data on viral shedding, clinical signs, and pathogenesis of infection provide new insights into the potential for these species to contribute to viral transmission and spread of H5N1 HPAI and an explanation for some of the variation in published data on the susceptibility of pigeons.
The risk of a small terrestrial bird species contributing to viral transmission during an outbreak of HPAI in domestic poultry is dependent on a variety of conditions, including the extent of the interface between the 2 bird populations and other factors relating to susceptibility, clinical disease, and viral shedding. House sparrows are widespread and ubiquitous birds that are commonly found around human-modified habitats (Lowther PE, Cink CL: 2006, House sparrow [Passer domesticus]. In: The birds of North America online, ed. Poole A. Cornell Lab of Ornithology, Ithaca, NY. Available at: http://bna.birds.cornell.edu/bna/species/012/articles/introduction. Accessed Dec. 2, 2008), including farms, and therefore share a large interface with poultry across which an HPAI virus could be transmitted. The results of the present study indicate that house sparrows are highly susceptible to the A/whooper swan/Mongolia/244/05 virus as evidenced by low BID50 and BLD50 titers. The infectious and lethal doses for house sparrows were similar to wood ducks (Aix sponsa), a highly susceptible wild bird species, and white leghorn chickens (Gallus gallus domesticus) using the same H5N1 HPAI virus strain and route of inoculation. 6 Despite their high susceptibility to the A/whooper swan/Mongolia/244/05 virus, however, all 3 dose groups of house sparrows excreted virus for several days via the oropharynx and cloaca prior to becoming clinically ill or dying (fig. 2). Thus, the habitat utilization, susceptibility, and viral shedding suggest that house sparrows could contribute to the dissemination of H5N1 HPAI virus within a poultry farm or between farms that are geographically close. The idea that small terrestrial birds may play a role in the transmission of HPAI during an outbreak in domestic poultry, including viral introduction or dissemination, is not novel for the recent Eurasian lineage of H5N1 HPAI viruses. In particular, field and experimental data from an outbreak of H7N7 HPAI in chickens in Victoria, Australia, indicated that sparrows and starlings on the affected premises were infected with the H7N7 HPAI virus and, potentially, could have been involved in the introduction or spread of virus to poultry. 8,19
Median area under the curve (AUC) and maximum titer by swab location for sparrows challenged with 1 of 3 viral inoculation doses (n = 5 per group).*
IQR = interquartile range; TCID = tissue culture infectious dose. Within a row, different superscripts indicate medians that are significantly different (P < 0.05) using the Wilcoxon signed rank test for paired data.
AUC calculated by using the trapezoidal rule to estimate the area under the TCID50/ml × days postinfection curves illustrated in figure 2.
Distribution and severity of microscopic lesions and viral antigen in house sparrows and rock pigeons inoculated intranasally with 106 EID50 of A/whooper swan/Mongolia/244/05 (H5N1) highly pathogenic Avian influenza virus.*
EID = egg infectious dose; HE = hematoxylin and eosin; IHC = immunohistochemistry; NE = not examined. Scores were based on birds in which lesions and/or viral antigen were detected (5 sparrows and 3 pigeons). Examined house sparrows consisted of 4 males and 1 female; examined rock pigeons consisted of 3 females.
HE score: - = no lesions detected; + = mild; ++ = moderate; +++ = severe.
IHC score: - = no viral antigen detected; + = sporadic; ++ = common; +++ = widespread.
Viral antigen was detected in peripheral ganglia innervating the heart, adrenal gland, and gastrointestinal tract.
Distribution of avian influenza (AI) virus antigen in tissues of house sparrows (Passer domesticus) infected with A/whooper swan/Mongolia/244/05 (H5N1). Tissue sections were immunohistochemically stained for the nucleoprotein of AI virus using a biotin streptavidin detection system and hematoxylin counterstain. A, testis: viral antigen in epithelial cells of seminiferous tubules. Bar = 160 μm; B, adrenal gland: viral antigen in corticotrophic cells (arrows) and chromaffin cells (arrowheads). Bar = 100 μm; C, pancreas: viral antigen in the pancreatic acinar epithelial cells. Bar = 100 μm; D, heart: viral antigen in neurons in a peripheral ganglion. Bar = 100 mm.
Although viral excretion in house sparrows was predominately via the oropharynx, most small terrestrial birds found around poultry facilities tend to live above the ground level and it may be unlikely that adequate contact with poultry occurs to allow for respiratory transmission of H5N1 HPAI virus. Other potentially viable mechanisms in which house sparrows could contribute to the transmission and spread of H5N1 HPAI virus between poultry include viral contamination of the poultry environment or feed with infective feces or sparrow carcasses from birds that died of H5N1 HPAI virus infection. The house sparrows in the current study excreted virus in their feces for several days (fig. 2), suggesting that the former could potentially occur. In addition, previous studies have shown that ingesting H5N1 HPAI virus infected tissues is a viable mechanism of transmission in susceptible avian species, including gulls and chickens. 5,26 Thus, carcasses of sparrows that died of H5N1 HPAI virus infection could serve as a risk for H5N1 HPAI transmission to poultry if carcasses are scavenged. House sparrows that die of H5N1 HPAI virus infection could also transmit virus to susceptible mammals (such as pigs or domestic cats) that ingest the carcass found on or around poultry farms. 15,17
The A/whooper swan/Mongolia/244/05 virus replicated systemically in house sparrows and had a strong tropism for the pancreas, adrenal glands, and testis. Based on the microscopic lesions and distribution of viral antigen, the cause of death was likely due to failure and/or dysfunction of multiple critical organs, including the pancreas, adrenal gland, heart, or liver. House sparrows that were examined histopathologically had viral antigen in neurons of the central and peripheral nervous system, but generally the extent was greatly reduced compared with other wild avian species that have been experimentally infected with H5N1 HPAI viruses. 3,4,12
Based on the very low BID50 and BLD50 of the A/whooper swan/Mongolia/244/05 virus for house sparrows, it would seem likely that bird-to-bird transmission within this species could occur, and it is unknown why transmission of H5N1 HPAI virus between co-housed sparrows in previous experimental trials was unsuccessful. 2 Although this suggests a low probability of intraspecific transmission, additional studies are warranted to further investigate potential H5N1 HPAI virus transmission between sparrows, particularly as multiple genotypes of H5N1 HPAI virus have been isolated from terrestrial birds. 14,16
The results of this study are consistent with other experimental infection trials and suggest that pigeons generally are resistant to infection with H5N1 HPAI viruses. 2,18,20 The high BID50 and BLD50 titers for the A/whooper swan/Mongolia/244/05 virus in pigeons provide evidence that the dose of virus is an important factor in determining susceptibility to H5N1 HPAI viruses in this species and may be responsible for the low mortality in pigeons observed in previous field and experimental studies. The BID50 and BLD50, however, are dependent upon route of exposure, virus strain, and host characteristics, so variation in susceptibility could be expected under different experimental or natural conditions.
The A/whooper swan/Mongolia/244/05 virus preferentially replicated in the brains of the pigeons that exhibited morbidity or mortality, and this is consistent with previous experimental studies. 13 Viral excretion was at a very low concentration and for a brief duration. This is also consistent with previous experimental infection studies on pigeons, in which viral shedding was minimal even in birds that became sick or died. 29 Taken together, the data from the current study suggest that pigeons can become infected with H5N1 HPAI virus, and mortality associated with infection is possible, but this species is unlikely to contribute to the transmission or maintenance of this virus.
Acknowledgements
The authors thank Joan Beck, James Doster, and Kira Moresco for technical assistance; Jay Cumbee and Ben Wilcox for assistance in acquiring house sparrows and pigeons; and Kevin Keel for assistance in preparation of the microscopic images. Funding for this work was provided by the Morris Animal Foundation (D05ZO-107), the ARS CRIS project No. 6612-32000-048-00D, the Specific Cooperative Agreement No. 58-6612-2-0220 between the Southeast Poultry Research Laboratory and the Southeastern Cooperative Wildlife Disease Study, and through the continued sponsorship of Southeastern Cooperative Wildlife Disease Study member state and federal agencies.
Footnotes
a.
FlockChek® AI MultiS-Screen Antibody Test Kit, IDEXX Laboratories, Westbrook, ME.
b.
Stata 10.1, StataCorp LP, College Station, TX.