Abstract
Domoic acid, produced by marine algae, can cause acute and chronic neurologic sequela in California sea lions (Zalophus californianus) from acute toxicity or sublethal exposure. Eight sea lions, representing acute and chronic cases, both sexes, and all age classes, were selected to demonstrate a concurrent degenerative cardiomyopathy. Critical aspects of characterizing the cardiomyopathy by lesion distribution and morphology were the development of a heart dissection and tissue-trimming protocol and the delineation of the cardiac conducting system by histomorphology and immunohistochemistry for neuron-specific protein gene product 9.5. Histopathologic features and progression of the cardiomyopathy are described, varying from acute to chronic active and mild to severe. The cardiomyopathy is distinguished from other heart lesions in pinnipeds. Based on histopathologic features, immunopositive staining for cleaved caspase-3, and comparison with known, similar-appearing cardiomyopathies, the proposed pathogenesis for the degenerative cardiomyopathy is the primary or at least initial direct interaction of domoic acid with receptors that are suspected to exist in the heart. L-Carnitine, measured in the heart and skeletal muscle, and troponin-I, measured in serum collected at the time of death from additional animals (n = 58), were not predictive of the domoic acid-associated cardiomyopathy. This degenerative cardiomyopathy in California sea lions represents another syndrome beyond central neurologic disease associated with exposure to domoic acid and may contribute to morbidity and mortality.
Keywords
Descriptions of heart lesions in pinnipeds are relatively limited and most are a primary or secondary consequence of infection with metazoan or protozoan parasites and bacteria. 14, 15 There is a case of toxin-induced heart pathology in a harbor seal (Phoca vitulina) due to acute lead toxicosis. 28 Heart pathology was described in California sea lions (Zalophus californianus) dying within 0 to >15 days of admittance to The Marine Mammal Center (TMMC), consequent to acute domoic acid toxicity and was hypothesized to result from an indirect, centrally mediated (brain–heart) mechanism of catecholamine surges associated with seizures or from a direct mechanism due to engagement of domoic acid receptors that may exist in the heart. 26 Gross heart pathology occurred in a few sea lions, characterized by myocardial pallor, hemorrhage, and fibrinous epicarditis progressing to fibrosis. Histopathology was characterized by mild-to-moderate myofiber degeneration, contraction band formation and necrosis with overlying fibrinous epicarditis, and multifocal hemorrhage progressing to fibrosis.
Domoic acid is a potent neurotoxic analog of the naturally occurring excitatory neurotransmitter
Investigations in several species, including humans, nonhuman primates, and rats, demonstrate that glutamate receptors are present widely in the mammalian body with similar physiologic and pharmacologic properties both in the neural and non-neural tissue and with unique distributions by anatomic site and species. 10, 11 The excitotoxicity of agonistic compounds for glutamate receptors vary with potency, dose, duration of exposure, chemical availability, rate of absorption, receptor affinity, target by anatomic site as related to variable cellular expression of receptor types, possible interaction or potentiating effects with other ligands or their analogs, and host factors, including genetic predisposition, health status, and age. 10 In the brain, domoic acid is a potent agonist of ionotropic glutamate receptors, which coincides with pathology preferentially affecting regions associated with memory function in California sea lions, humans, and laboratory models. 10, 12, 27 Glutamate receptors also were demonstrated in the heart of humans, monkeys, and rats and include both ionotropic and metabotropic classes. There is, however, interspecies variation in the receptor groups present as well as interspecies and intraspecies variation in their respective affinities and distribution in the cardiac structures but with consistent interspecies expression in the conducting system (atrioventricular node, bundle of His, and Purkinje fibers), intramural nerve fibers, ganglia cells, and cardiomyocytes. 10, 11 The role of these receptors relative to exposure to domoic acid and the manifestation of cardiac dysfunction and heart pathology remains uncertain.
A standardized, relatively rapid protocol is described here to dissect and sample the heart of California sea lions to allow for histologic examination of all regions of the heart, including the conducting system, and thus to permit more precise documentation and interpretation of heart histopathology according to lesion distribution and morphology. This protocol permitted an accurate description of the degenerative cardiomyopathy that frequently was identified in California sea lions suffering from acute or chronic effects associated with domoic acid exposure 12 and was critical to differentiating it from other causes of heart pathology.
Materials and Methods
Eight California sea lions (Nos. 1–8), which were admitted to TMMC between 2005 to 2007 and diagnosed with neurologic disease from exposure to domoic acid, 12, 25 were selected to characterize the heart lesions associated with exposure to domoic acid (Table 1). These individuals represented both sexes (male n = 3, female n = 5) and yearling through adult age classes (yearling n = 2, juvenile n = 2, subadult n = 1, adult n = 3). Sex determination was based on genital morphology, and age class was estimated using a combination of body length, tooth size, and stage of sagittal crest development. 13 Animals were selected if they had a clinical and pathologic diagnosis of acute or chronic effects from exposure to domoic acid 12 and had a necropsy performed within 1–12 hours of death using the heart dissection protocol (Figs. 1–7) and taking representative tissues from the body and brain, including both hippocampal formations with adjacent temporal regions and regions associated with cardiac function (most notably the red nucleus and mesencephalic reticular formation) for histologic examination. The domoic acid-associated brain lesions 12 were defined histologically as acute, chronic active (i.e., neuronal loss confounded by acute damage), and chronic quiescent (i.e., neuronal loss without apparent acute damage). None of the selected animals had significant concurrent disease that could affect cardiovascular function, including concurrent heart conditions, severe renal disease, evidence of infarction anywhere in the body or brain, severe lung disease, or septicemia.
Individual data provided for animals that were selected to represent and characterize the domoic acid-associated cardiomyopathy. ∗
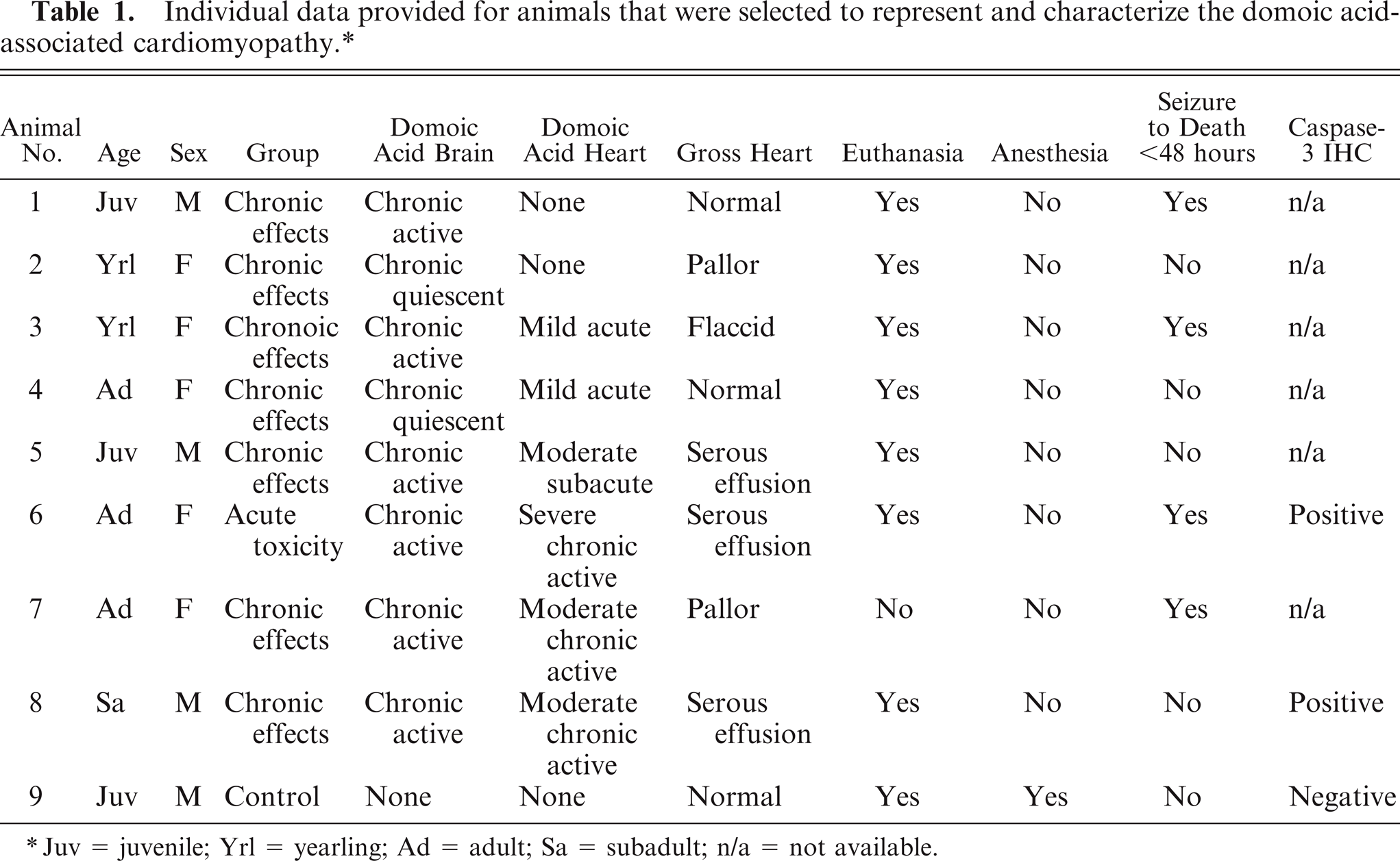
Juv = juvenile; Yrl = yearling; Ad = adult; Sa = subadult; n/a = not available.
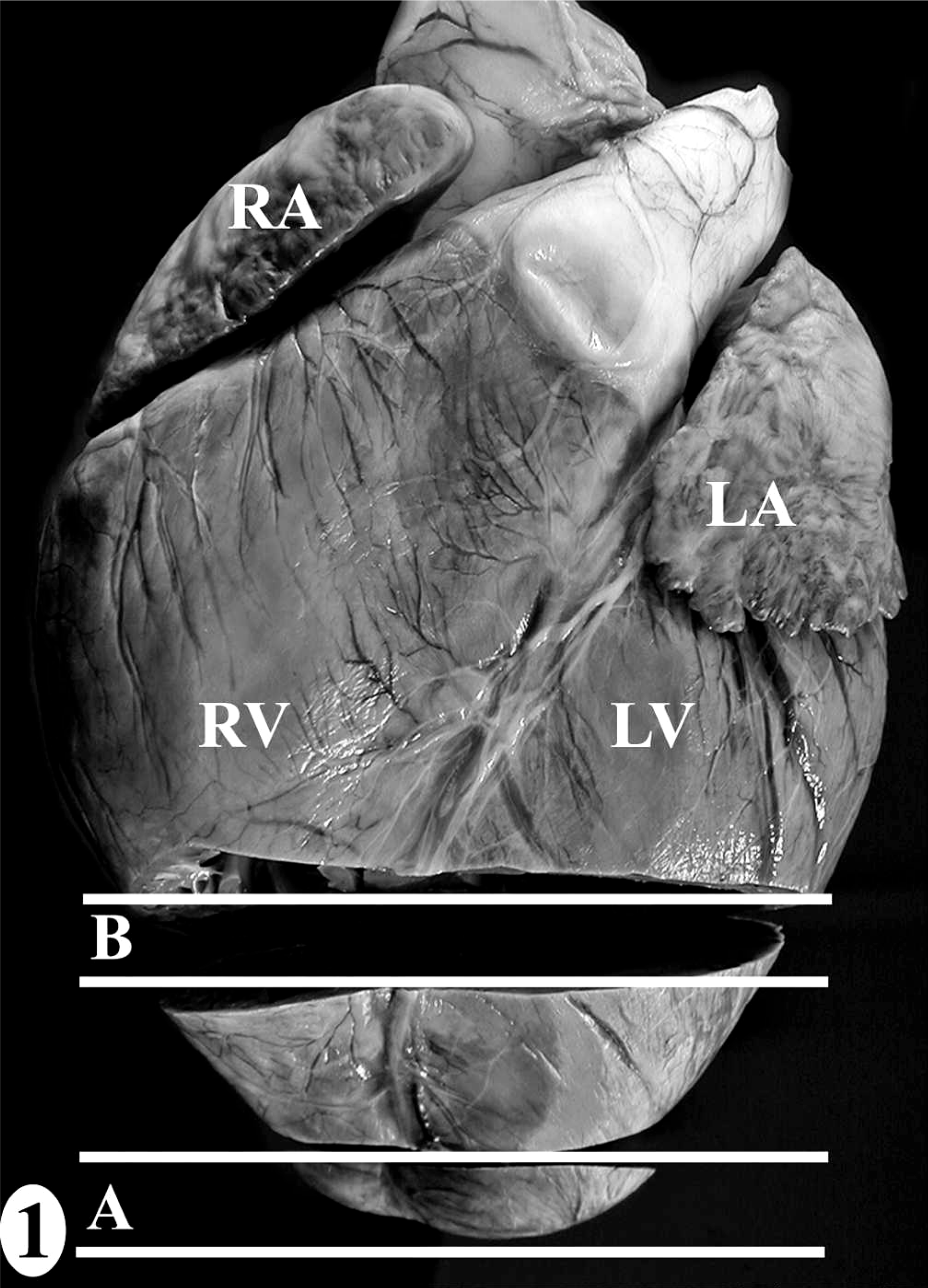
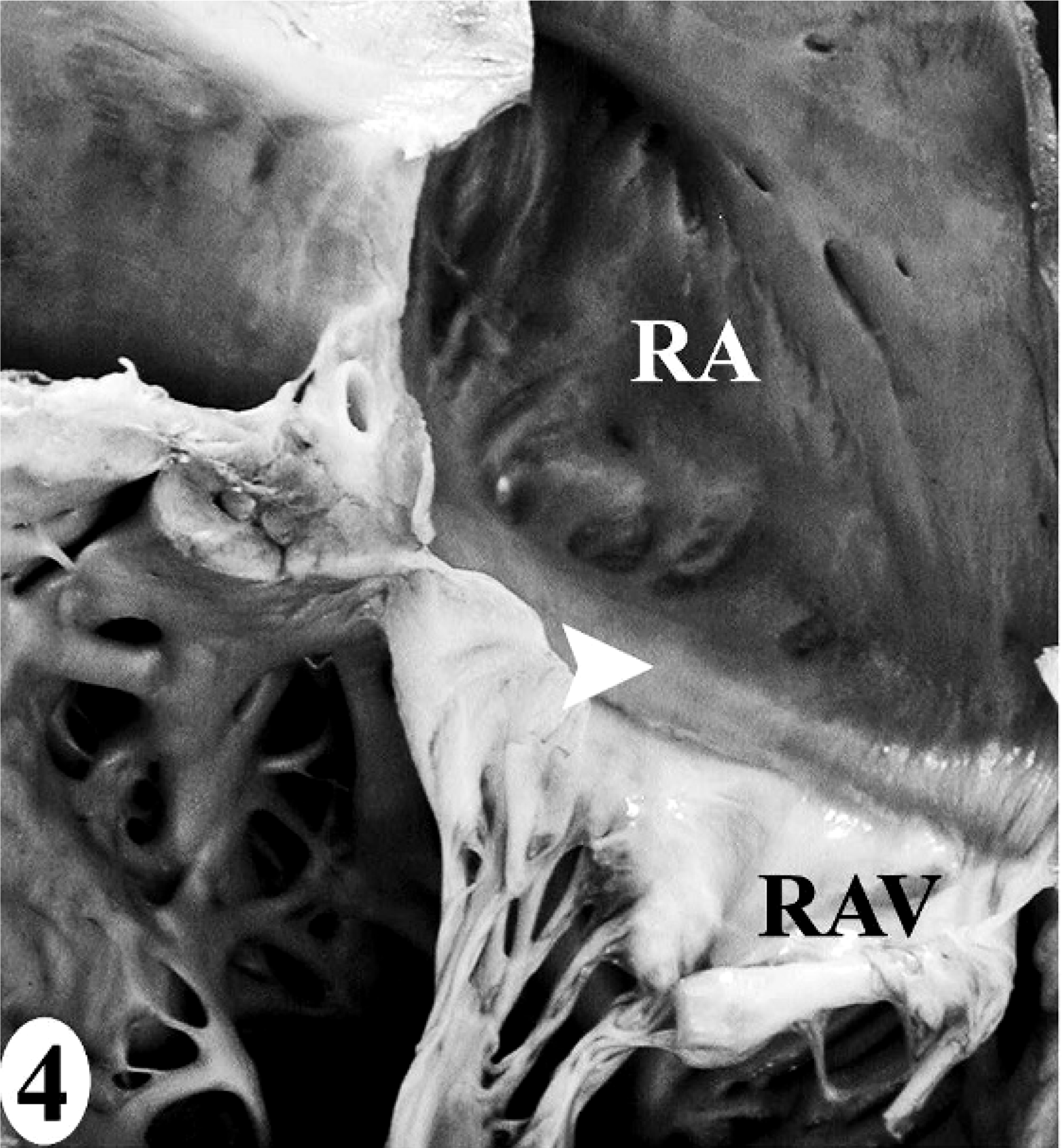
Normal heart; California sea lion No. 9. The heart dissection protocol (
For caption see above
For caption see above
For caption see above
For caption see above
For caption see abovexs
For caption see above
Animal Nos. 1–8 died (n = 2) or were euthanatized (n = 6), and tissues collected at necropsy were immersed in neutral-buffered 10% formalin, processed in a routine manner, and embedded in Tissue Prep (Fisher Scientific, Fairlawn, NJ). Five-micron-thick sections were cut, stained with hematoxylin and eosin, and examined under light microscopy by one pathologist (TSZ). Characterization of heart pathology included a review of documented findings on gross examination and evaluation of histologic sections representing all regions of the heart and most to all components of the conducting system. Gross heart pathology was considered in light of whether the animal died or was euthanatized, was anesthetized at the time of death, or had a seizure within 48 hours of death. The cardiomyopathy was evaluated histologically by region for lesion type and severity, and findings among regions were combined into an overall diagnosis of lesion type, severity, and duration. Regions were defined as the left atrium, right atrium, and the base, mid, or apical level for each the right ventricular free wall, left ventricular free wall, and interventricular septum. Lesion criteria were 1) character of morphologic change(s) in the cardiomyocyte; 2) the distribution of these changes within the myocardium relative to other structures, including the epicardium, endocardium, conducting system, and vasculature; 3) the presence and type of leukocyte infiltrate; 4) the presence of adipocyte replacement; 5) the presence of fibroplasia or fibrous replacement; and 6) the distribution of these latter three changes relative to cardiomyocyte changes. Severity was scored (in four 20× fields) as mild (a few scattered lesions not seen consistently), moderate (multifocal lesions seen consistently), and severe (coalescing lesions seen consistently).
Selected serial sections of formalin-fixed paraffin-embedded heart were stained with Masson's trichrome for collagen and with Ziehl-Neelsen acid fast for lipofuscin 1 in animal Nos. 6 and 8. Immunohistochemistry (IHC) also was performed on serial sections to detect apoptotic activity using a polyclonal rabbit anti-human cleaved caspase-3 antibody, clone Asp125 (1:125; Cell Signaling Technology, Danvers, MA), and immunoreactive cells were compared to special stained serial sections. Briefly, sections were deparaffinized, rehydrated in running water, immersed in a EDTA/Tris buffer (pH 9) and placed into an electronic pressure cooker for 60 minutes at 120°F (PASCAL, Dako Corp., Carpinteria, CA) for antigen retrieval, removed and rinsed in distilled water, placed in 3% hydrogen peroxide (10 minutes), rinsed in distilled water, placed in dual endogenous peroxidase block (Dako Corp.) (10 minutes), incubated with primary antibody (60 minutes), rinsed 3 times in Tris buffer (Dako Corp.), incubated with rabbit polymer (Dako envision, Dako Corp.) (30 minutes), rinsed again in Tris buffer, incubated with diaminobenzidine (DAB) Plus (Dako Corp.) (5 minutes), rinsed in distilled water, counterstained with Mayer's hematoxylin (30 seconds to 2 minutes), rinsed in Tris buffer, then in distilled water, dehydrated through a series of alcohols, and mounted. IHC was performed on the negative controls without primary antibody on serial heart sections from animal Nos. 6 and 8 and with the primary antibody on heart sections from animal No. 9, which did not have heart pathology. The positive control was an involuting thymus from a California sea lion.
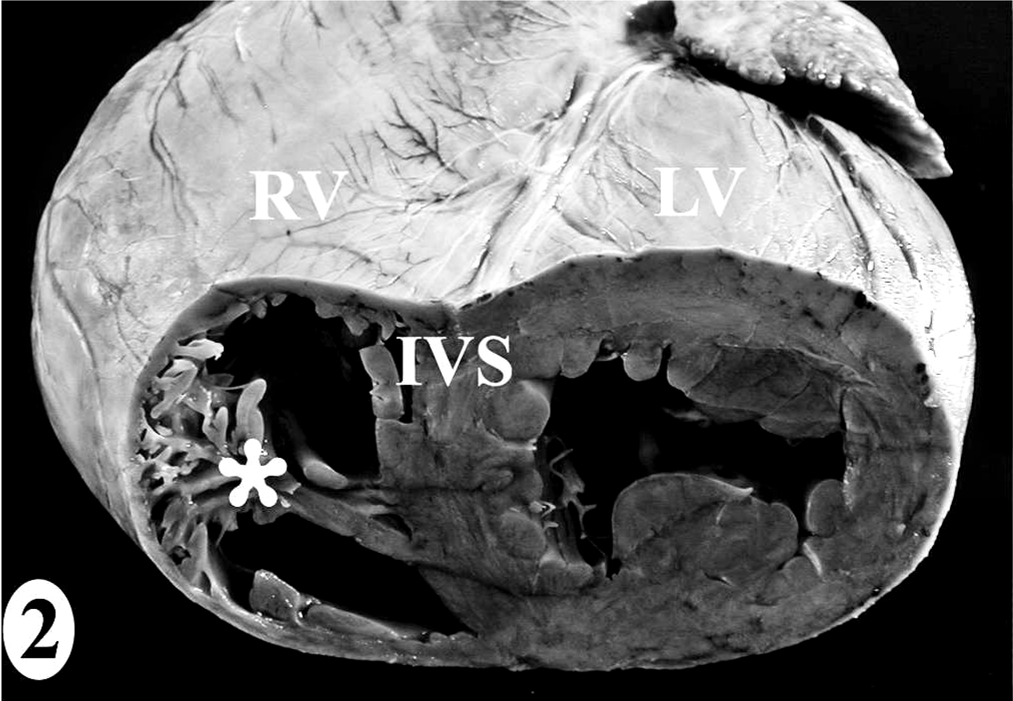
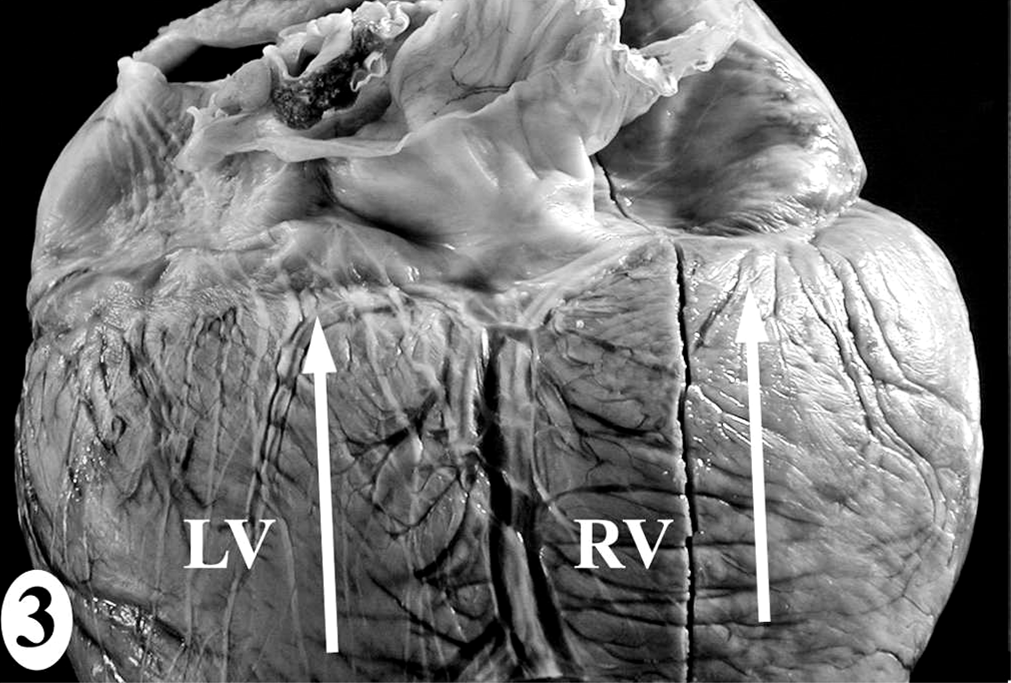
The heart dissection and trimming protocol (Figs. 1–7) was developed using the heart from one male juvenile sea lion (No. 9) that was euthanatized under anesthesia due to irreversible trauma to both eyes and did not have antemortem or postmortem lesions to suggest exposure to domoic acid or heart disease (Table 1). The gross landmarks for the sinoatrial node and atrioventricular node (Figs. 4–7) were defined by comparing photographs of serial 5-mm-thick gross sections and the corresponding histologic section. Identification of the conducting system by routine histology was confirmed by IHC using a polyclonal rabbit anti-human protein gene product 9.5 (PGP 9.5) antibody, clone UCH-L1 (1:1,500; Chemicon International Inc., Temecula, CA) that reacts with all central and peripheral neural structures and a technique with a DAB chromagen reporting system on formalin-fixed paraffin-embedded tissue, as described previously. 11 The negative control was IHC performed without the primary antibody on a serial heart section, and the positive control was peripheral nerves around the heart. Thinner sections were made when trimming for histology while maintaining the landmarks and architecture of the original sample. Half of sample 1 was submitted, maintaining the left ventricular free wall-to-interventricular septum-to-right ventricular free wall relationship. Similar representative regions from half of sample 2 were submitted, of which the interventricular septum included the septomarginale band.
Additional diagnostics tests were performed on animals Nos. 1–3, 5–8, as well as on additional animals (Nos. 10–60) admitted to TMMC that died or were euthanatized due to disease unrelated to the heart or brain effects from domoic acid, as confirmed by clinical and pathology findings. Accordingly, animals were assigned to one of three groups: 1) controls, which had trauma (n = 7, Nos. 10–12, 15–17, 21), renal failure (n = 7, Nos. 13, 14, 18, 20, 22, 23, 25), cancer (n = 1, No. 26), or thoracic bacterial infection (n = 2, Nos. 19, 24); 2) acute domoic acid neurologic toxicity; or 3) chronic neurologic effects from sublethal exposure to domoic acid (Table 2). Histopathology reports were reviewed when possible to assess whether histologic changes suggestive of the domoic acid-associated degenerative cardiomyopathy were “present” or “absent” or whether “other” heart pathology occurred. When present, other heart pathology was minimal-to-mild and scattered, except for animal Nos. 18, 21, 38, 44, 50, and 59 in which myocardial damage was moderate or regionally intense.
Individual data for levels of L-carnitine in skeletal muscle and heart and troponin-I in serum from California sea lions with and without exposure to domoic acid. ∗

The median troponin values and mean L-carnitine values are presented by group for comparison. NCP = noncollagen protein; n/a = not available.
Gross examination only.
Moderate-to-severe “other” heart pathology.
Troponin-I concentrations in serum were measured at the time of death in 34 animals (control group: n = 7, Nos. 10–16; acute toxicity: n = 13, Nos. 6, 28–35, 41–44; and chronic effects: n = 14, Nos. 1, 3, 7, 45–53, 59, 60) of both sexes (male = 8, female = 26) and all age classes (yearling = 5, juvenile = 2, subadult = 2, adult = 25). All control animals did not have any heart pathology and were included since the troponin-I assay is not validated for pinnipeds. Blood was collected from the caudal gluteal vein, placed in serum separator tubes (Beckton-Dickenson, Franklin Lakes, NJ), centrifuged, the serum was aliquoted, stored at −80°C, and submitted to the Ohio State University for testing. Troponin-I concentration was measured using an immunoassay system (Unicel DXI; Beckman Coulter, Fullerton, CA) with a detection level of 0.01 ng/ml. 2 Concentrations less than 0.11 ng/ml were considered normal. Median troponin-I values were calculated for each of the three groups using SPSS. Normality of the data was evaluated, and the data were log transformed for analysis as they were not normally distributed. Individual and median values were reported and troponin-I concentrations were compared among groups using ANOVA and Bonferroni post-hoc comparison tests.
Results
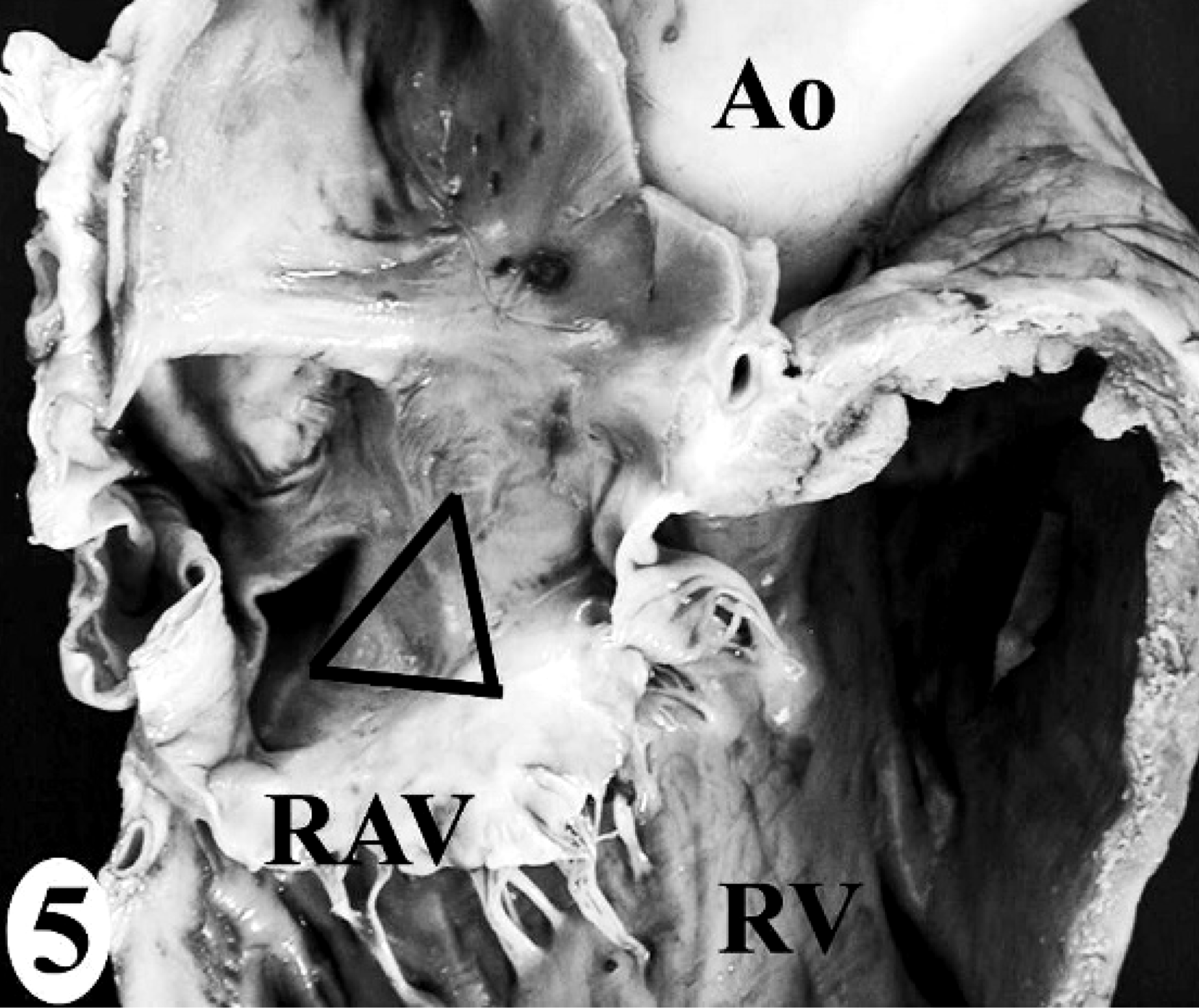
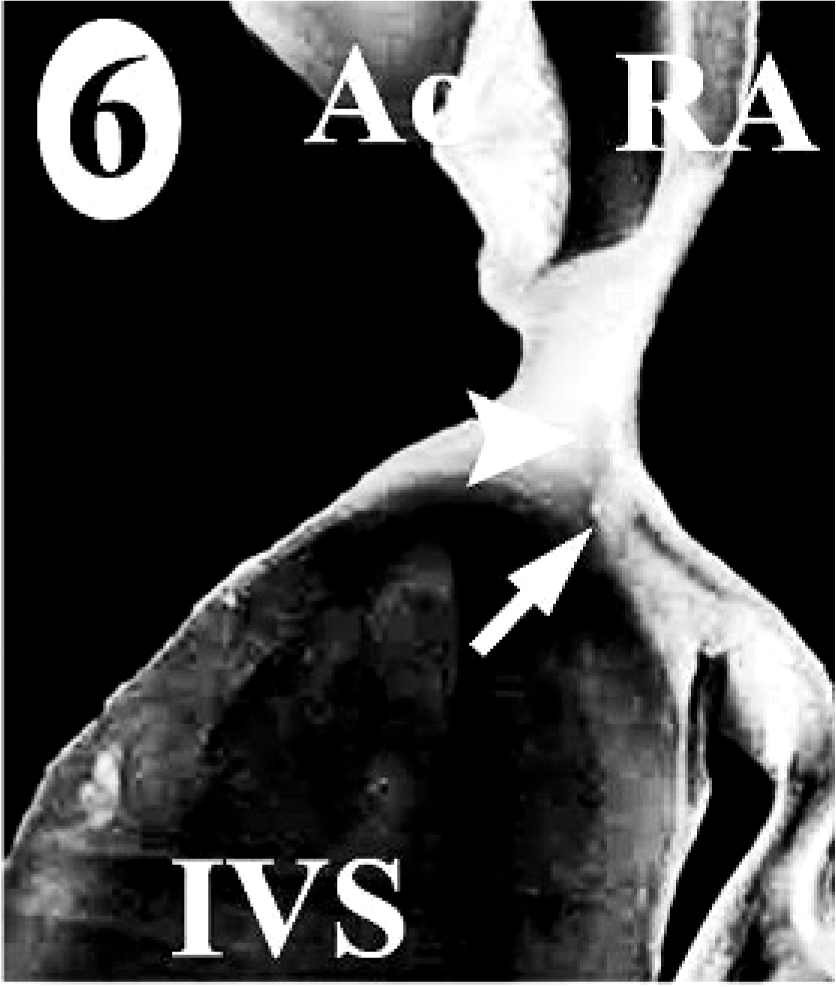
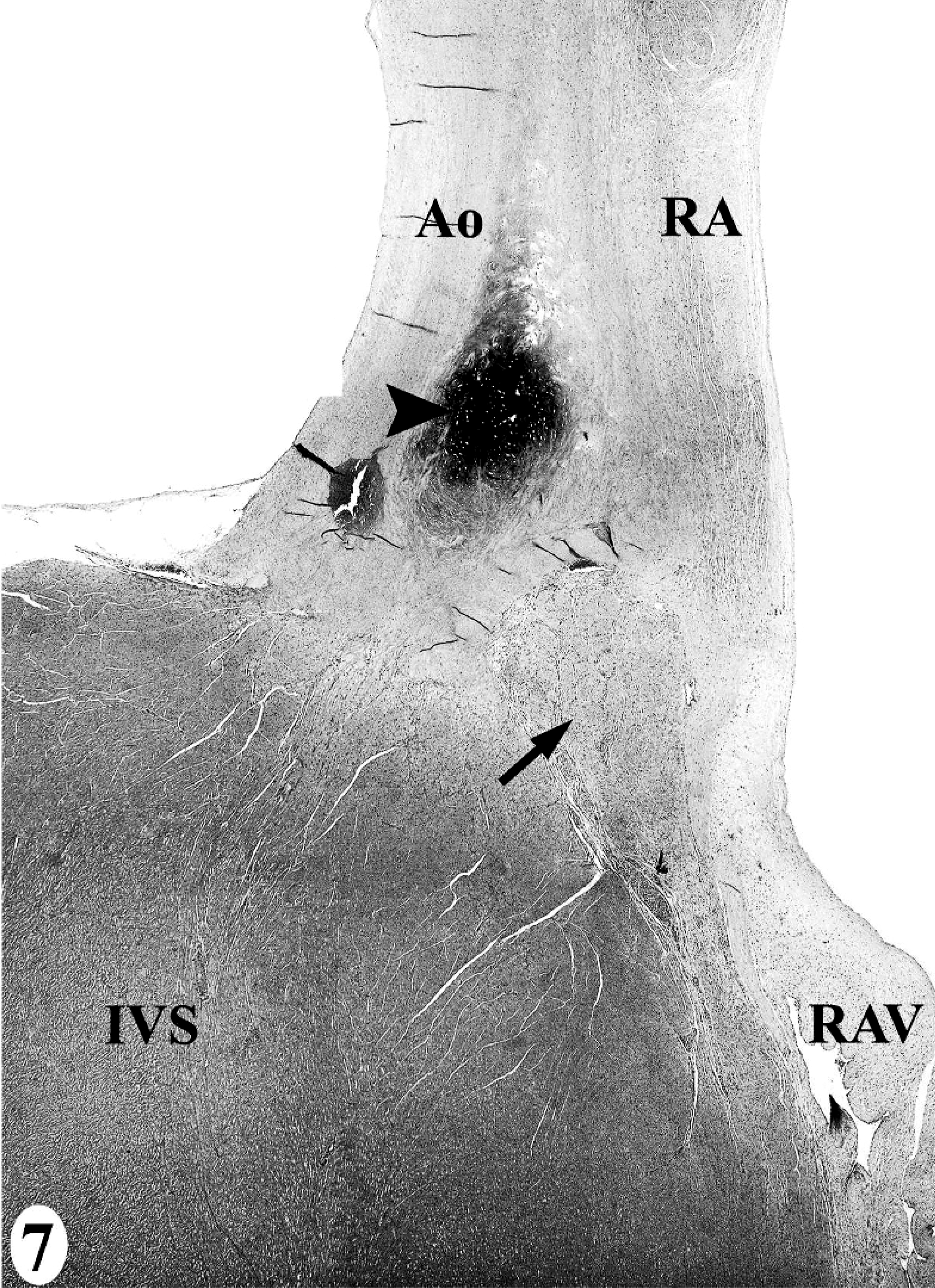
In the normal California sea lion (No. 9) heart, components of the conducting system, including the sinoatrial node, atrioventricular node, bundle of His, and Purkinje fibers, the intramural nerve fibers, and ganglia cells were strongly immunoreactive for PGP 9.5 (Fig. 8). This information was used to confirm the identification of the conducting system by routine histology and to develop a heart dissection and trimming protocol (Figs. 1–7) that allowed for relatively rapid, consistent sampling of all heart regions and the conducting system. The sinoatrial node was most difficult to sample consistently and to identify by routine histology. The atrioventricular node on gross and histologic examination was correlated tightly with the cartilaginous to osseous island in the right fibrous trigone, which can be palpated and visualized on cut-section deep to the septal cusp of the right atrioventricular valve (Figs. 5, 6). The Purkinje fibers occurred along the subendocardium, and nerve fibers and ganglia cells predominated in the subendocardium and midmyocardium in association with medium-to-small caliber blood vessels (Fig. 8 inset).
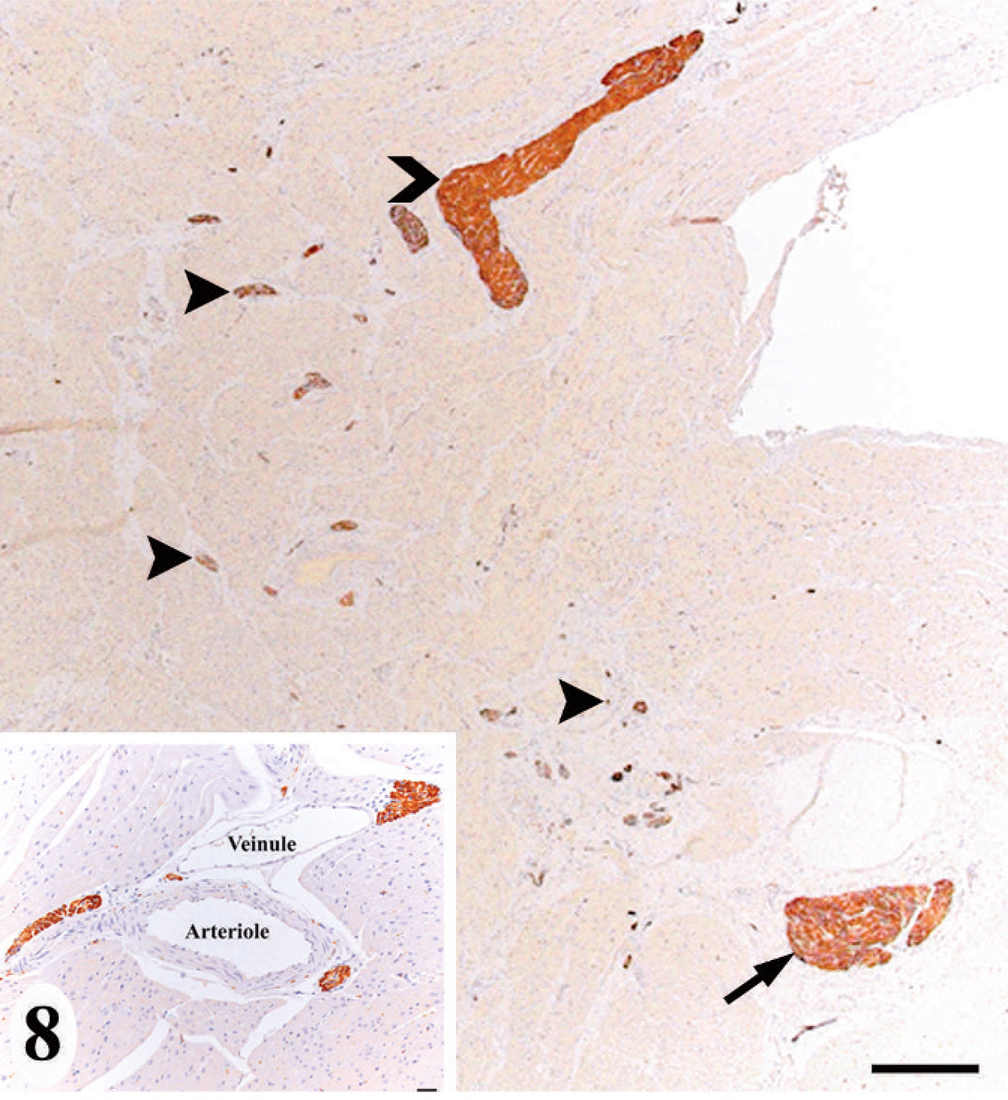
Normal heart; California sea lion No. 9. The interventricular septum at the base of the heart had immunoreactive staining (dark brown) demonstrating the atrioventricular node (arrow), bundle of His (chevron), and intramural nerve fibers (arrowheads). PGP 9.5. Bar = 500 μm. Intramural nerve fiber tended to be associated with medium-to-small caliber blood vessels. Inset: PGP 9.5. Bar = 10 μm.
Animals with the domoic acid-associated degenerative cardiomyopathy had one of three nonspecific gross findings, including a globally flaccid heart, multifocal-to-diffuse myocardial pallor, and mild serous pericardial effusion, but notably without fibrinous material (Table 1). Although these findings occurred independently of each other in these 8 animals, they sometimes occurred concurrently in the other reviewed cases of domoic acid-associated cardiomyopathy. Although the number of animals included was small, the occurrence of gross pathology did not seem related consistently to the occurrence, severity, or duration of the domoic acid-associated degenerative cardiomyopathy, nor with euthanasia, euthanasia under anesthesia, or seizure activity 48 hours before death.
Morphologically, the cardiomyopathy was characterized by vacuolization of the cardiomyocyte cytoplasm and intercardiomyocyte separation by clear space, often leaving a lacy interstitial tissue, which was consistent with interstitial edema rather than artifactual separation (Fig. 9). Involvement of the conducting system cells was difficult to discern, as these cells inherently have lightly vacuolated to feathery morphology. Affected cardiomyocytes progressed to vacuolar degeneration and less frequently, hyaline necrosis or apoptosis, as characterized by a rounded hypereosinophilic cytoplasm with loss of cross-striation and a pyknotic-to-karyolytic nucleus. Adipocytes replaced damaged and lost cardiomyocytes, sometimes with a minimal attending leukocyte infiltrate comprised of histiocytes, occasionally with phagocytized lipofuscin (as confirmed by acid fast staining), suggesting clean-up of apoptotic cardiomyocytes, as well as of unidentified mononuclear cells, and few lymphocytes and neutrophils (Fig. 10). Less frequent than adipocytes, immature and eventually mature fibrous tissue (as confirmed by trichrome staining) replaced affected cardiomyocytes.
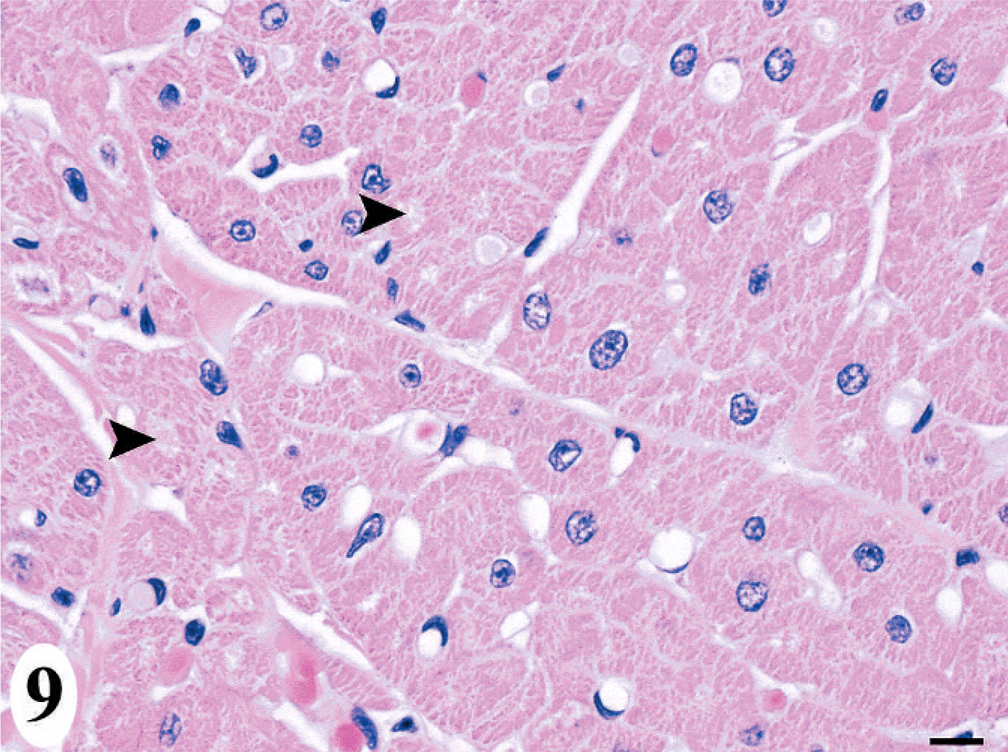
Heart; California sea lion No. 3. Acute lesions of the domoic acid-associated degenerative cardiomyopathy, which included interstitial edema and vacuolization of cardiomyocytes (arrowhead). HE. Bar = 10 μm.
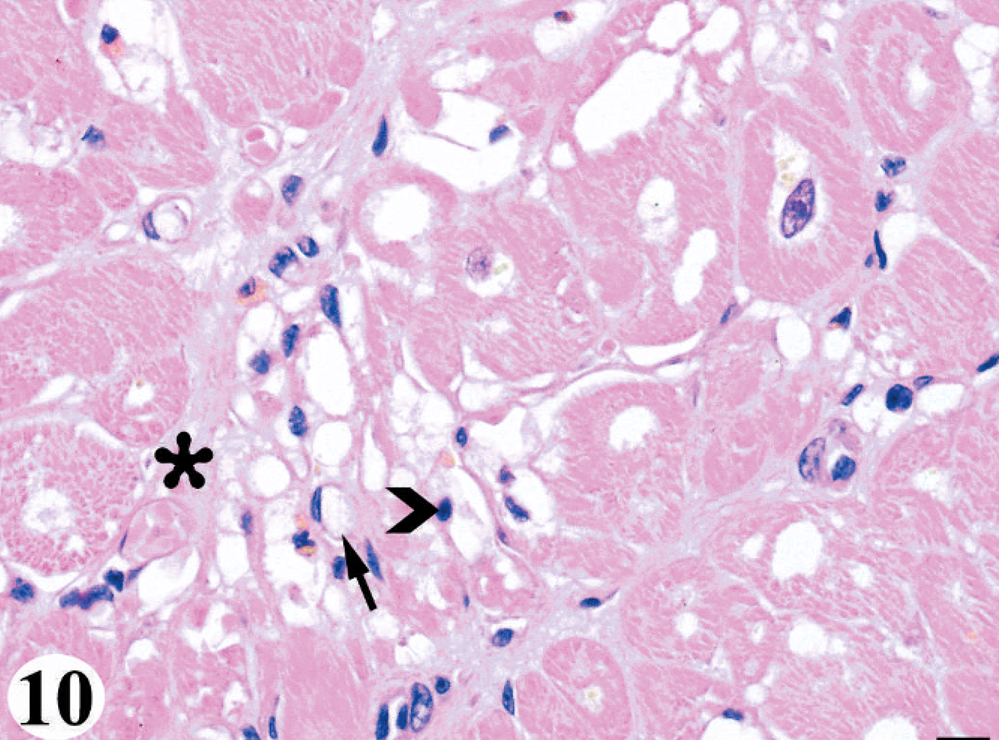
Heart; California sea lion No. 6. More advanced lesions of the domoic acid-associated degenerative cardiomyopathy, which included interstitial edema, diffuse cardiomyocyte vacuolar degeneration and loss with a minimal leukocyte infiltrate, replacement by adipocytes (arrow), and lesser fibroplasia (asterisk). The infiltrate was comprised of unidentified mononuclear cells (chevron) and histiocytes. HE. Bar = 10 μm.
Distribution of the cardiomyopathy was consistent among animals. The first region affected was the base of the interventricular septum just distal to the atrioventricular node (Fig. 11). The lesion progressed toward the apex along the interventricular septum and circumferentially first to the left ventricular free wall and then the right ventricular free wall; therefore, over time the most severe and chronic lesions would be at the base of the heart with more acute and more mild lesions occurring toward the apex. In all sea lions, the atria were not affected. Within the myocardium, the lesion seemed to follow the distribution of the conducting system distal to the atrioventricular node and other nerve tissue, tending toward the midmyocardium to subendocardium and often regional to medium-to-small caliber blood vessels.
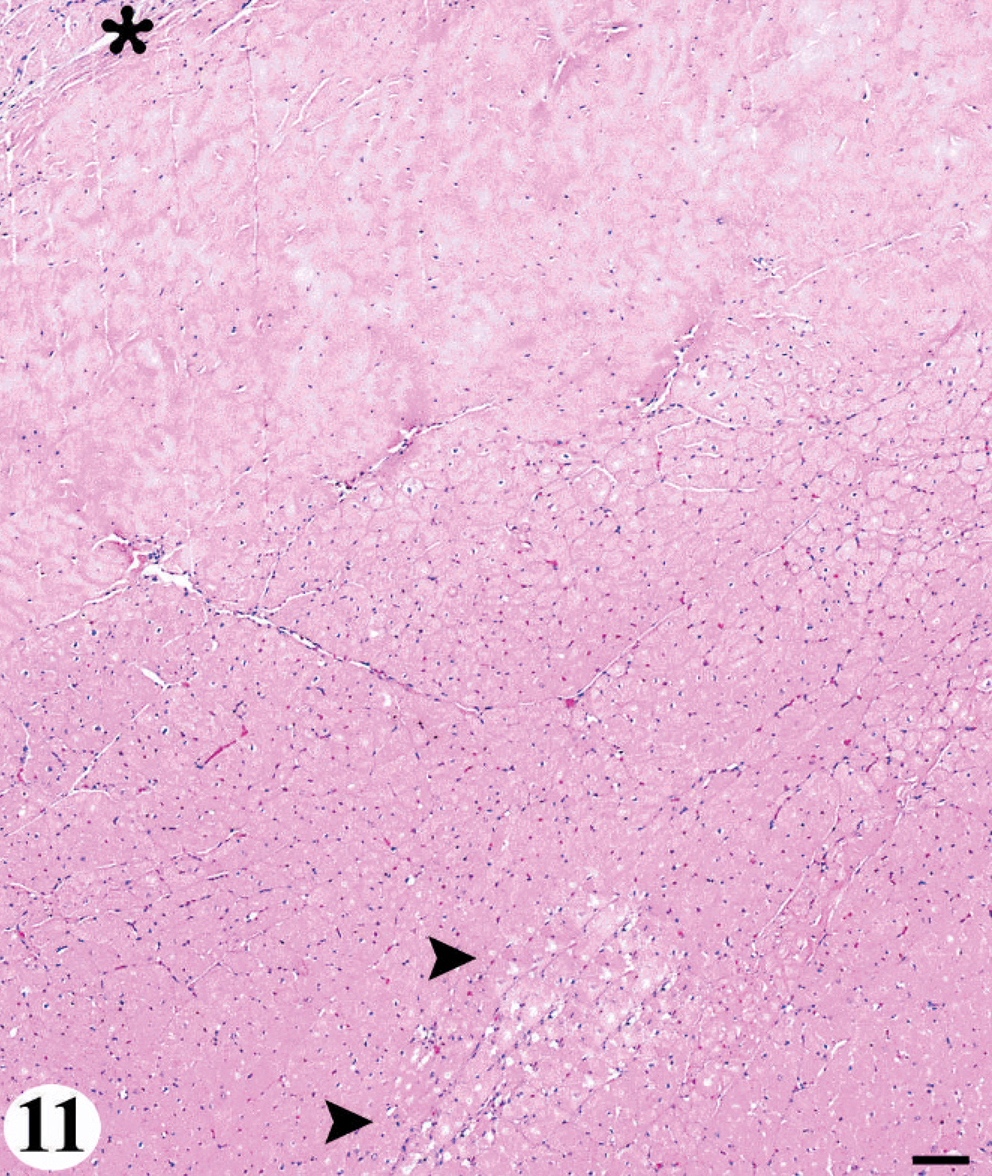
Heart; California sea lion No. 3. The consistent lesion distribution pattern of the domoic acid-associated degenerative cardiomyopathy. Acute lesions (arrow) first occurred at the base of the interventricular septum just distal to the atrioventricular node (asterisk). HE. Bar = 300 μm.
The duration of the cardiomyopathy was defined as acute having cardiomyocyte separation, vacuolization, degeneration, and necrosis with minimal-to-no leukocyte, adipocyte, or fibroblastic infiltrate (Fig. 9); subacute having the aforementioned changes with more frequent leukocyte (although still a minor component), adipocyte, and fibroblast components; and chronic active having the aforementioned changes with adipocyte replacement being a predominant feature or with mature fibrous tissue (Fig. 10). Using these morphologic, distribution, and duration criteria, study animals were diagnosed either with a mild acute, moderate subacute, moderate chronic active, or severe chronic active cardiomyopathy (Table 1). A chronic quiescent cardiomyopathy was not found in any sea lion examined.
Because the cardiomyopathy had lesion morphology suggestive of an association with the apoptotic pathway, IHC for cleaved caspase-3 activity was performed on heart sections from three animals. One animal was the juvenile male control (No. 9), animal No. 6 was an acute toxicity case with chronic active brain pathology and severe chronic active domoic acid-associated cardiomyopathy, and animal No. 8 was a chronic case with chronic active brain pathology and moderate chronic active domoic acid-associated cardiomyopathy. Animals Nos. 6 and 8 demonstrated immunopositive staining for cleaved caspase-3 (Fig. 12). Immunoreactive cells were sparse, especially in animal No. 8, and located exclusively in the ventricles within lesion areas in animal No. 6 as well as in nonlesion areas in animal No. 8. Immunoreactive cells in animal No. 6 had morphology consistent with two cell types, mature cardiomyocytes and an unidentified mononuclear cell characterized by sparse cytoplasm and a hyperchromatic nucleus. The cellular morphology and examination of serial sections stained with trichrome indicate that these latter cells were not histiocytes or fibroblasts transforming into a collagen fiber. Additionally, a few individual immunoreactive cells scattered among cardiomyocytes had microscopic features consistent with apoptosis, suggesting that they were apoptotic cardiomyocytes. The subcellular localization of immunopositive staining was both intracytoplasmic, predominantly, and intranuclear. Nonspecific immunostaining of intracytoplasmic lipofuscin was excluded by the more globular, dispersed appearance of lipofuscin versus immunoreactive material (Fig. 12), by examination of serial acid fast-stained sections for lipofuscin, and by the lack of immunostaining in the heart from animal No. 9, which had numerous lipofuscin-laden cardiomyocytes.
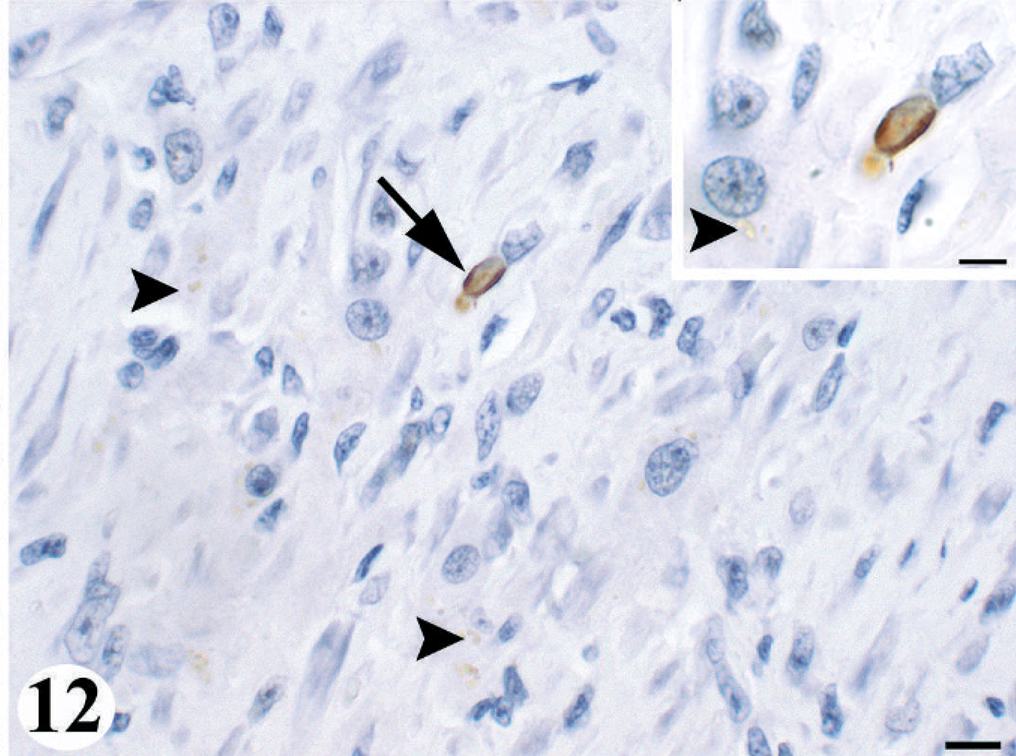
Heart; California sea lion No. 6. Chronic, active domoic acid-associated cardiomyopathy demonstrating intranuclear and intracytoplasmic immunoreactive staining (dark brown) (arrow) of a damaged cardiomyocyte, which indicated the mechanism of apoptosis. Lipofuscin (arrowheads) was the pale brown, globular intracytoplasmic material. Cleaved caspase-3. Bar = 10 μm.
The domoic acid-associated degenerative cardiomyopathy was not associated with the previously described pericarditis 26 in any sea lions. Additionally, the previously described contraction band formation with necrosis of cardiomyocytes 26 did not occur in any of the animals Nos. 1–8. When it occurred in other animals, the heart was fixed within 1–2 hours of death and concurrent contraction band necrosis was not present. Both findings suggest that the contraction band formation was the result of rapid dissection and fixation postmortem rather than a pathologic lesion. All cases had the common background changes of lipofuscin-laden cardiomyocytes, anisokaryosis (karyomegaly), multiple nuclei, and cytomegaly, which are noted in California sea lions that die from various causes, tending to increase with age. Other confounding cardiac pathology or extracardiac disease, which could affect the cardiovascular system, was not present in any animal.
The following cardiac changes are presented to distinguish them as separate from the domoic acid-associated degenerative cardiomyopathy. Such changes occurred minimally or mildly in some animals (Nos. 28–60) that had the degenerative cardiomyopathy but excluded them from the study set (Nos. 1–8). The condition of nonsuppurative polymyositis (T. S. Zabka, personal observation) began as an inflammatory lesion predominantly of lymphocytes, occasional neutrophils, and eventually histiocytes and plasma cells, with secondary segmental hyaline degeneration and necrosis of cardiomyocytes, but notably without cytoplasmic vacuolization. Within the heart, the distribution tended toward the midventricles and was random within the myocardium (i.e., without association to the conducting system). Extravascular migration of microfilaria morphologically, consistent with Acanthocheilonema odenhali, 7 sometimes was associated with small foci of eosinophilic-to-granulomatous inflammation distributed randomly in the heart, but did not cause severe disease. Bacterial septicemia or infection with protozoal tachyzoites manifested as randomly distributed foci of liquefactive necrosis and inflammation, of which the character of the inflammation was dictated mostly by the etiologic agent. In another sea lion, myocardial infarction had an apical distribution with a discrete region of necrosis, polymorphonuclear infiltrate, and fibrosis with little to no adipocyte replacement.
The additional diagnostic tests performed on animal Nos. 1–3, 5–8, and 10–60 did not facilitate identification of animals with histopathology indicative of the domoic acid-associated cardiomyopathy, as described herein. Tissues samples, which included blood, skeletal muscle, and heart, were taken perimortem and thus were reflective of the histopathology. The individual and median troponin-I concentrations in serum did not differ significantly among the three groups (P = .446) (Table 2).
Discussion
The cardiac conducting system, intramural nerve fibers, and ganglia cells were demonstrated in a California sea lion using antibody to PGP 9.5, as previously demonstrated in the human, monkey, and rat heart. 11 The conducting system had a similar distribution and landmarks to that of the dog. 8 The described heart dissection protocol for California sea lions provided a consistent, rapid means to sample the heart while still allowing for gross and histologic identification of all regions and structures of the heart. This protocol was critical to characterizing the degenerative cardiomyopathy associated with exposure to domoic acid. It allowed for the identification of lesion distribution and morphology, identification of the true occurrence, severity, and duration, as the condition did not affect the heart globally. It was useful to distinguish the lesion from other heart lesions of sea lions, including the nonsuppurative polymyositis condition, migration by microfilaria, vascular accidents (infarction), contraction band formation (either artifact from rapid fixation or pathologic in association with necrosis), infection with bacteria or protozoal tachyzoites, and age-related degenerative changes.
The domoic acid-associated degenerative cardiomyopathy affected animals of both sexes, of all age classes after the pup stage, and with either acute neurologic toxicity or chronic neurologic effects. 12 The cardiomyopathy ranged from mild to severe and acute to chronic active; a chronic quiescent cardiomyopathy was not identified. The cardiomyopathy was not associated consistently with gross lesions, and when present, the changes were nonspecific. One animal, which did not have a cardiomyopathy, had myocardial pallor, which may reflect differential contraction or congestion at the time of death. The serous pericardial effusion was mild in all animals in which it occurred and was without concurrent gross or histologic changes to suggest it was not more than an agonal event. A flaccid heart suggests that the animal did not die in left ventricular systole as expected, reasons for which may be a terminal arrhythmogenic event of cardiac or extracardiac origin or general poor contractility associated with metabolic or physiologic imbalances. Antemortem electrocardiogram is required to assess whether the domoic acid-associated degenerative cardiomyopathy correlates with a functional cardiac disturbance. Euthanasia, anesthesia, and seizures within 48 hours of death did not correlate with the occurrence or type of gross pathology. Euthanasia artifact was not appreciated microscopically in the heart, as confirmed by examination of the control animal (No. 9).
Microscopically, the primary lesion was cardiomyocyte vacuolar degeneration and loss with adipocyte replacement. The lesion started at the base of the interventricular septum distal to the atrioventricular node and progressed distally and circumferentially, especially in the midmyocardium to subendocardium. The minimal reactive infiltrate included an unidentified mononuclear cell that may be a cardiac progenitor cell; however, IHC is required for confirmation. 18 Engagement of apoptotic pathway, as at least part of the pathogenesis, was supported by immunoreactive staining for cleaved caspase-3 within heart lesions of both animals tested and by the nature of the minimal leukocyte response, including lipofuscin-laden macrophages interpreted as phagocytosis of dead cardiomyocytes. Caspase-3 was detected in the active state, as the antibody is specific for the cleaved form and immunopositive material was both intracytoplasmic and intranuclear. 23 Although immunoreactive cells also occurred in nonlesion areas, staining was limited to the most vulnerable region, the interventricular septum, suggesting it preempted morphologically apparent cell death. The relatively sparse immunoreactivity was consistent with the transient nature of cleaved caspase-3. Thus, this molecular technique may be less optimal than perhaps other more encompassing and consistent methods, such as characterizing mRNA cellular expression of apoptotic genes.
The association of the cardiomyopathy with the intracardiac nervous system, particularly distal to the atrioventricular node, suggests that it may cause arrhythmias or predispose to arrhythmias, especially under conditions of stress. The severity of domoic acid-induced brain lesions and death did not always seem to correlate in some sea lions, which may support the concept that the degenerative cardiomyopathy resulted in fatal cardiac disturbances. Findings in other species further support this possibility. In the hearts of humans, monkeys, and rodents, glutamate receptors are present on the conducting system (atrioventricular node, bundle of His, Purkinje fibers), intramural nerve fibers, ganglia cells, and cardiomyocytes, especially the intercalated discs; therefore, interference with these receptors may lead to conducting disturbances. 10, 11 An experiment on rat cardiomyocytes demonstrated that domoic acid may reduce the ATP-induced increase in intracellular calcium of electrically stimulated cardiomyocytes by inhibiting the ATP–cardiomyocyte interaction and thus disturb cardiomyocyte conduction. 21 Thus, in California sea lions exposed to domoic acid, the cardiomyopathy may be an important contributor to morbidity and mortality. Without antemortem cardiac function studies, however, this possibility remains uncharacterized.
The microscopic and molecular findings in these sea lions are suggestive of a toxic insult, nutritional imbalance, idiopathic organic heart condition, or, less likely, a neurogenic or vascular insult rather than an infectious or immune-mediated etiology, especially those known to affect pinnipeds. 14, 15 Nutritional imbalance was excluded, because animals were fed their regular diet while in rehabilitation and animals were in good-to-thin, but not emaciated, body condition. Idiopathic disease also was excluded, as the cardiomyopathy was identified only in domoic acid cases and previously was not described as a sporadic occurrence. The lesion morphology and distribution were not consistent with a vascular accident (i.e., infarct), because the latter manifests as a discrete region of polymorphonuclear leukocyte infiltrate and fibrosis and is distributed primarily in the apical to mid ventricle of the left ventricle to interventricular septum. 27 Additionally, the sea lions did not have intercurrent predisposing conditions.
Quite relevant to animals exposed to domoic acid, which manifests primarily as brain disease, is a centrally mediated (i.e., brain-heart) pathogenesis. In these sea lions, microscopic findings were not present in extratemporal regions of the brain that control cardiac function, such as the mesencephalic reticular formation and red nucleus. Thus, secondary effects on cardiac function, such as attenuation of the baroreceptor reflex demonstrated in rats after injection of domoic acid into the nucleus ambiguous 6 or the ventilatory response to hypoxia demonstrated after injection of domoic acid into the nucleus of the solitary tract 5 were not likely. Other centrally mediated mechanisms that are well established in human medicine include central nervous system-induced sudden death by intense activation of the autonomic nervous system, the release of opioids, or the release of neuroactive peptides like catecholamine and stress-related neuropeptides. 20, 27 Cortical stimulation studies suggest sympathetic predominance over the right hemisphere; therefore, similar to increased catecholamine release, stimulation of the right insular area or a vascular accident in the right middle cerebral artery can result in sympathetic-mediated tachycardia and left ventricular dysfunction. The mechanism of damage is most likely through microvascular spasm of coronary vasculature or direct catecholamine-induced free radical damage via calcium influx and overload in cardiomyocytes. Anticipated microscopic lesions are myocyte contraction band formation, with or without necrosis, and mononuclear leukocyte infiltrates comprised of lymphocytes and macrophages. The condition primarily affects the left ventricle from the apex to midaspect, sparing the base of the heart. Cortical stimulation studies in the left hemisphere show a parasympathetic predominance. Accordingly, partial complex seizures (epileptic activity), most often originating from the left temporal lobe, but occasionally the right temporal lobe or occipital lobe, can result in bradycardia, hypotension, and cardiac asystole. A similar mechanism of cortical lateralization to the left temporal lobe associated with postural changes is a proposed mechanism for neurocardiogenic syncope (or neurally mediated syncope). 19
California sea lions with neurologic effects from exposure to domoic acid have epileptic activity and demonstrate bilateral or unilateral lesions consistently in the temporal region and sometimes in the insular or occipital cortex, of which the right side is more often affected than the left in chronic cases. 12 Despite these findings, which may predispose to a centrally mediated, especially sympathetic-mediated cardiomyopathy, the morphology and distribution of such an insult is different from the degenerative cardiomyopathy described in sea lions Nos. 1–8. Further exclusion of a centrally mediated mechanism awaits investigation of glutamate receptors in the sea lion heart, antemortem cardiac function studies, and perhaps more mechanistic studies in laboratory animal models.
Doxorubicin hydrochloride cardiotoxicity has lesion morphology most reminiscent of the sea lion cardiomyopathy; however, the distribution is less similar, because it results in a diffuse cardiomyopathy, including involvement of both atria. 24 The similarity in lesion morphology may be because the pathogenesis involves apoptotic pathways. 4 Doxorubicin toxicity was dependent on repeated dosing, has an insidious onset and variable latency period of 0–2.5 year (median 3–8 weeks), and becomes rapidly progressive with cardiac failure occurring within 1 week to 2.5 months after the final dose. 24 This is interesting in light of the sea lion cardiomyopathy in which the duration of the cardiac lesions did not necessarily correspond to the duration of the brain lesion, which was evident especially in animals with the acute to subacute cardiomyopathy. This occurrence may be related to low-dose initial effects on cardiomyocytes that can be difficult to detect by routine histology or are repaired, 18 an insidious onset perhaps with a latency period as in doxorubicin cardiotoxicity, a dose or repeat dose-dependent pathogenesis as seen in doxorubicin toxicity, or the toxin preferentially engaging the metabotropic over the ionotropic glutamate receptors in the heart and thus resulting in more gradual changes in cellular control, rather than rapid changes. 9 Metabotropic receptors are distinct from ionotropic receptors because of the association with long-term aspects of cellular control rather than rapid changes such as fast synaptic neurotransmission, the bidirectional interaction with multiple second messengers in the same cell, and the more diverse and heterogenous distribution in the body with specific localization at an anatomic and subcellular level. 10 Also of note, cardiac damage was ongoing despite the animals being in rehabilitation weeks to months after exposure to domoic acid, which may suggest a mechanism of self-propagation as suspected for the domoic acid-induced brain lesions. 12
The ancillary tests may support the proposed pathogenesis of direct domoic acid cardiotoxicity in the heart of California sea lions. Cardiac troponin (I or T), which usually is assessed in conjunction with creatinine kinase-MB in serial blood samples, has a positive correlation with the syndrome of myocardial infarction in humans. 27 Conversely, human reports of myocardial stunning due to exaggerated direct sympathetic stimulation showed an absence or only mild elevation in troponin and creatinine kinase-MB, 27 and adriamycin-induced cardiomyopathy was associated with reduced mRNA expression of troponin-I and other cytoskeleton genes of alpha actin and myosin light chain. 20 Thus, should the proposed pathogenesis for the degenerative cardiomyopathy in sea lions be correct, then serum concentrations of troponin-I should not be predictive of its occurrence, as the results from these chronic active cardiomyopathy cases suggest. Caution in data interpretation, however, is warranted, as troponin analysis is not validated for sea lions and thus this study relied on comparison to the control sea lion group. Also in general, troponin analysis demonstrates variability among laboratories, assay type, and species to which it is applied.
The level of
The sea lion degenerative cardiomyopathy is dissimilar to the cardiomyopathy of sea otters, 17 as described in association with exposure to domoic acid, and of Pygmy (Kogia breviceps) and Dwarf (Kogia sima) sperm whales 3 of unknown but likely multiple and complex origin. Sea otters develop a global, multifocal (with a subepicardial and subendocardial concentration), nonsuppurative myocarditis that was hypothesized to progress to dilated cardiomyopathy when associated with exposure to domoic acid. 17 Myocardial vacuolization, congestion, and interstitial fibrosis were considered secondary changes. Thus, the sea otter cardiomyopathy was a primary inflammatory condition with a random global distribution in contrast to the sea lion cardiomyopathy being a primary vacuolar degenerative condition with a specific ventricular distribution. The cardiomyopathy described in whales was a degenerative condition of consistent but global distribution (greater detail not more defined) and hypothesized to be a continuum from myocardial degeneration to chronic cardiomyopathy. 3 The former was characterized predominantly by anisokaryosis with karyomegaly, nuclear rowing and interstitial edema, and the latter also had myofiber disarray, loss of cross-striations, homogenized sarcoplasm, and interstitial fibrosis. Thus, the whale cardiomyopathy, although a degenerative condition, was different from the sea lion cardiomyopathy, as it had a specific global distribution and had predominant morphologic features of anisokaryosis (karyomegaly), multiple nuclei, myofiber disarray, and contraction band necrosis, which were common background lesions or absent in the sea lions.
In conclusion, our data suggest that the degenerative cardiomyopathy described in California sea lions was associated with exposure to domoic acid. Based on lesion morphology and distribution, it was distinguishable from other causes of heart lesions. The degenerative cardiomyopathy may contribute to domoic acid-associated morbidity and mortality because of its association with the intracardiac nervous system and intermittent discrepancy between the severity of domoic acid-induced neurologic disease and death. A unifying proposed pathogenesis was the primary or at least initial direct interaction of domoic acid with the heart rather than a centrally mediated mechanism. This hypothesis was based on our findings in sea lions, comparison to other cardiac insults,
19,
20,
24,
27
and the demonstration of ionotropic and metabotropic glutamate receptors in the heart of nonhuman primate, rats, and humans.
10,
11
If similar to the brain, domoic acid would engage some or all of these receptors, cause a calcium-associated insult, and result in conduction disturbances, primarily in apoptotic pathways of damage, and perhaps self-propagation of damage after domoic acid was cleared from the body.
11,
12,
21
The lack of troponin-I and
Additional work to elucidate the pathogenesis and functional effects of this degenerative cardiomyopathy on the health of California sea lions exposed to domoic acid includes characterizing glutamate receptors in the sea lion heart, investigating the molecular signature in normal and affected sea lion hearts, conducting electrocardiograms, and further examining the cardiac effects on cardiomyocytes and on the conducting system in experimental models exposed to domoic acid.
Footnotes
Acknowledgements
This study was supported by funds from the US National Marine Fisheries Service, Oceans and Human Health Initiative grant number NA04OAR4600200 and through a Morris Animal Foundation Fellowship Training grant number D05ZO-401. The work was authorized under the US Marine Mammal Protection Act by Scientific Research Permit No. 932-1489-00. We appreciate the review and input on some initial cases by Dr. Terry Spraker, the caring for animals and work performed by the staff and volunteers of The Marine Mammal Center, especially the assistance of Elizabeth Wheeler and Tenaya Norris, the technical assistance in immunohistochemistry performed by Sandra Horton.
