Abstract
Tissues of stranded California sea lions (Zalophus californianus) naturally infected with a hyperviruluent strain of Klebsiella pneumoniae were examined by histopathology and immunohistochemistry against the K. pneumoniae K2 capsular antigen. In 7 of 8 animals, there was severe purulent bronchopneumonia, sometimes complicated by fibrinonecrotizing pleuritis with pyothorax. In affected areas of lung, large numbers of degenerate neutrophils and macrophages were admixed with rare large extracellular and intracellular gram-negative bacilli surrounded by a prominent capsule. Through serotyping, polymerase chain reaction, sequencing, and immunohistochemistry, these bacteria were confirmed to be a K2 serotype of K. pneumoniae. The same bacteria were identified through double immunolabeling within macrophages in blood vessels, lymph nodes, spleen, and liver. Intact K. pneumoniae were identified on epithelial surfaces of the nasopharyngeal, tracheal, and small intestine mucosae and within distal renal tubules. Our findings indicate that hypervirulent K. pneumoniae causes severe respiratory disease and intrahistiocytic bacteremia in California sea lions.
Hypervirulent strains of the gram-negative bacterium, Klebsiella pneumoniae, can cause localized or disseminated infections in healthy hosts. 6,7,10 In human patients, these infections affect primarily the lungs and liver and are usually caused by 1 K. pneumoniae genotype of the capsular serogroup K1 or unrelated genotypes of the capsular serogroup K2. 3,7,9 The main phenotypic feature of some hypervirulent strains is their ability to produce large amounts of capsular mucopolysaccarides when cultured on blood agar, resulting in a mucoid string of more than 5 mm when an inoculating loop is lifted from the colonies (string test). 7 The gene responsible for this hypermucoid phenotype has been identified in human and animal isolates as the regulator of mucoid phenotype gene (rmpA), and its presence is highly suggestive of a hypervirulent strain. 3,7,9
In the past decade, rmpA-positive K. pneumoniae emerged as an important pathogen of nonhuman primates 8,9 and otariid seals. 5,6 In New Zealand sea lions (Phocarctos hookeri), K. pneumoniae infection is the most important cause of pup mortality, 6 and in California sea lions (CSLs) (Zalophus californianus), localized and disseminated hypervirulent K. pneumoniae infections have been a minor yet constant cause of strandings. 5
Detailed descriptions of K. pneumoniae pathology in CSLs and the distribution of the bacteria are not available in the literature yet are important to understanding its pathogenesis. The objective of this study was to describe pathologic features and distribution of hypervirulent K. pneumonia in CSL tissues to better understand the pathogenesis of this disease in pinnipeds.
Animals
The medical record database of rescued animals at The Marine Mammal Center, Sausalito, California, was searched for cases of CSLs stranded from 2005 to 2014 and from which K. pneumoniae was cultured. A total of 52 cases had K. pneumoniae isolates with a positive string test. Out of this group, 8 animals were selected for the study because they met the following inclusion criteria: (1) a necropsy was performed on a fresh carcass, and (2) complete histology sets were available.
All animals were found stranded alive along the central California coast and transported to The Marine Mammal Center, where they died between 2 and 24 hours following admission. Complete necropsies were performed within 12 hours postmortem.
Sections from major organs and tissues were placed in 10% buffered formalin and processed routinely for histopathologic examination. Selected sections of lung, brain, liver, and spleen were stained with acid-fast and gram stains. In all animals, sections of nasopharyngeal mucosa, tonsil, trachea, lung, tracheobronchial lymph node, diaphragm, heart, brain, small intestine, mesenteric lymph node, liver, pancreas, kidney, and spleen were further examined by immunohistochemistry for K. pneumoniae K2 capsular antigen using a rabbit polyclonal antibody (Statens Serum Institute, Copenhagen, Denmark). Double immunolabeling was performed in selected sections of lung, brain, spleen, and liver using anti–Iba-1 (macrophages) and the anti-K2 K. pneumoniae antibodies. The detail of the 2 protocols is provided as supplemental file (immunohistochemistry methods). The histopathology and immunohistochemical analyses were interpreted by reaching a consensus diagnosis by at least 2 pathologists.
To determine the cross-reactivity of the K. pneumoniae K2 antiserum with other bacterial species, a K2 K. pneumoniae imunohistochemical protocol was performed on formalin-fixed sections of agar containing pure colonies of hypermucoviscous K. pneumoniae belonging to the K2 serotype (positive controls), non-hypermucoviscous K. pneumoniae isolated from dogs (unknown serotype), Escherichia coli, and Proteus mirabilis. In addition, lung sections of sea lions and fur seals with marked bronchopneumonia where Staphylococcus aureus or β-hemolytic Streptococcus sp. were isolated in pure cultures were used as negative controls and to assess cross-reactivity with these bacteria. Using the mentioned immunohistochemistry protocol, there was no labeling of any of the other bacteria. Only K2 K. pneumoniae in agar had marked diffuse positive immunolabeling.
Bacteria Identification
Sterile swabs were collected from any tissue displaying purulent lesions (e.g., pyothorax, purulent bronchopneumonia, subcutaneous abscesses) in the 52 animals with a K. pneumoniae positive string test, including the 8 CSLs of the present study. Swabs were placed in transport media, stored for up to 24 hours at 4°C, directly plated onto 5% sheep blood agar (Hardy Diagnostics, Springboro, OH, USA) and MacConkey agar (Hardy Diagnostics), and incubated at 37°C in the presence of 5% CO2 in air. Bacteria genus and species were determined by using commercially available biochemical tests. Hypermucoid K. pneumoniae isolates were further characterized through polymerase chain reaction (PCR) and sequencing of the rmpA gene as previously described. 5 The capsular serotype of the isolates was determined by wzi sequencing as previously described. 1
All 8 animals included in this study were infected with hypermucoviscous (string test positive), rmpA positive, capsular serotype 2 (K2), K. pneumoniae. Table 1 shows the principal gross, histologic, and immunohistochemical findings in each animal. The most common gross finding was severe purulent bronchopneumonia, sometimes complicated by fibrinonecrotizing pleuritis and pyothorax (Fig. 1). Microscopically, large numbers of degenerate and viable neutrophils and macrophages, admixed with cellular debris, fibrin, edema, and scant hemorrhage, effaced up to 80% of lung architecture and filled airways and alveoli (Fig. 2). In adjacent areas, occasional macrophages expanded alveolar septa. In some foci within the lungs, inflammatory infiltrates were admixed with small to moderate numbers of extracellular and intrahistiocytic, 2.0 × 0.7-μm, gram-negative bacilli sometimes surrounded by a clear prominent halo. The bacilli were not acid fast, but with this stain, a clear 1.0- to 3.0-μm capsule (halo) was more evident (Fig. 3).
Gross, Histologic, and Immunohistochemical Findings in 8 California Sea Lions Naturally Infected With Hypervirulent K2 Klebsiella pneumoniae.
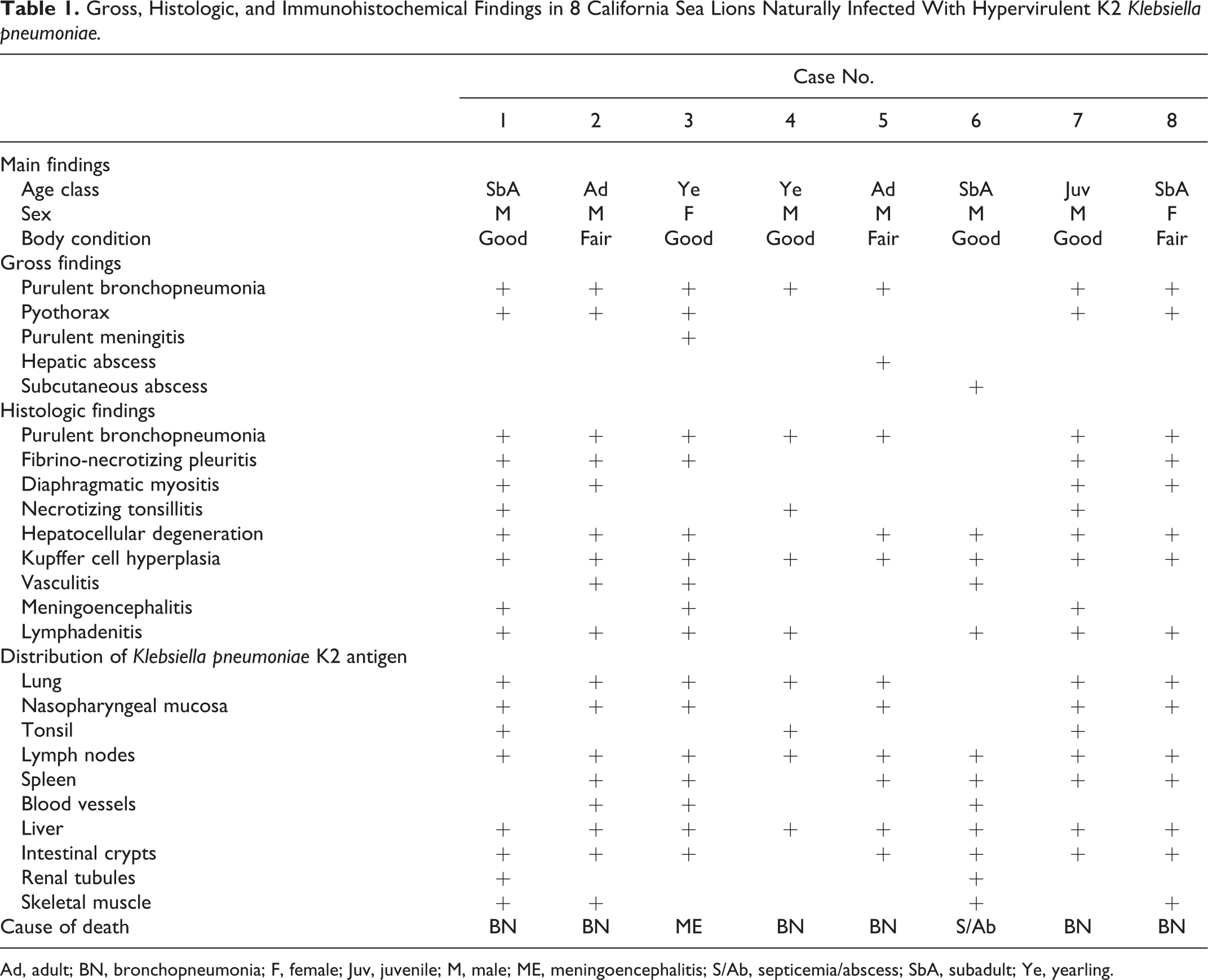
Ad, adult; BN, bronchopneumonia; F, female; Juv, juvenile; M, male; ME, meningoencephalitis; S/Ab, septicemia/abscess; SbA, subadult; Ye, yearling.
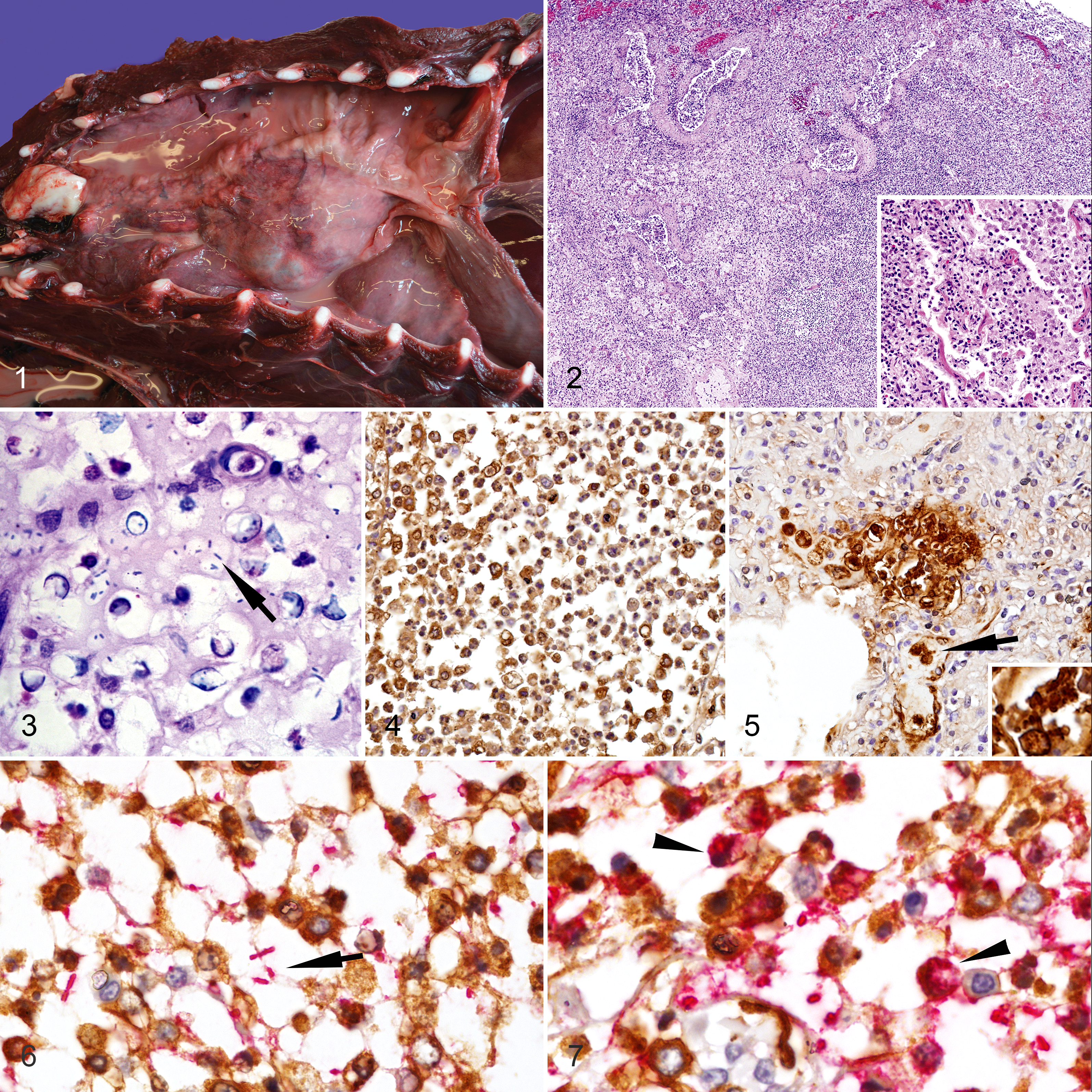
The distribution of K. pneumoniae K2 capsular antigen differed among lung sections (Figs. 4, 5). In areas with marked inflammation, intact bacteria were rare, but most leukocytes and cellular debris had mild to marked, positive, K2 capsular antigen staining. Double immunolabeling using Iba1 and anti-K2 antibodies showed that the leukocyte population in areas with intact lung architecture were primarily histiocytes. In addition, the staining pattern differed among locations within the same sections. In areas with intact bacteria surrounded by a prominent clear halo, macrophages rarely contained K2 K. pneumoniae antigen (Fig. 6). However, in areas where bacteria were partially disrupted and lacked prominent peripheral halos, macrophages contained a larger amount of cytoplasmic K. pneumoniae antigen (Fig. 7).
In all animals with bronchopneumonia, there was mild to marked histiocytic and neutrophilic tracheobronchial lymphadenitis, with moderate to marked necrotizing tonsillitis in 3 CSLs. Pyothorax and fibrino-necrotizing pleuritis was found in 5 CSLs, which was complicated by a marked histiocytic diaphragmatic myositis in 4 cases. All animals had Kupffer cell hyperplasia and mild, randomly distributed vacuolar degeneration, as well as hepatocyte individualization and necrosis. In the brain of 1 CSL, there was fibrinoid necrosis of small-caliber blood vessels associated with numerous macrophages and bacteria within vessels and the surrounding parenchyma (Suppl. Figs. S1, S2). This animal had less severe involvement of the meninges, and another 2 animals had mild lymphohistiocytic meningoencephalitis. In all described inflammatory processes, the macrophages in the parenchyma, blood vessels or sinusoids, the cellular debris, and the extracellular bacteria had moderate to marked, positive, staining with anti-K2 antibody.
In most animals (7/8), small numbers of intact K. pneumoniae bacteria were observed without major signs of inflammation on the jejunal crypt epithelium and along the nasopharyngeal mucosal surface. In 2 animals, intact colonies of K. pneumonia occupied distal renal tubules and occasionally compressed the tubular epithelium.
Hypervirulent K. pneumoniae infection in CSLs is primarily an acute to subacute respiratory infection. The pattern of lung involvement and the more acute lesions in the pleura suggest airborne bronchopneumonia complicated by pleuritis and pyothorax. The point of entry could be the upper respiratory tract, as suggested in African green monkeys 8 and humans. 7,11
In our study, there were intrahistiocytic and intracapillary bacteria in the alveolar septa adjacent to highly inflamed areas. The pattern of lung involvement could indicate seeding of intrahistiocytic and/or free bacteria from the lung to other tissues such as liver and spleen. Furthermore, moderate amounts of K. pneumoniae antigen within hepatic and splenic macrophages, as well as the more marked and chronic processes in the lung and subcutaneous tissue, reinforce this hypothesis for nearly all cases.
The thick capsular or extracapsular polysaccharide produced by hypervirulent strains of K. pneumonia results in a hypermucoviscous phenotype on agar plates and one of the most significant factors contributing to in vivo virulence. 4,7 The polysaccharide capsule allows bacteria to survive and spread within the host by protecting against leukocyte oxidative killing and through avoiding opsonization and macrophage uptake. 4 In the present study, areas of inflammation with prominent bacterial capsules contained mostly intact microbes, and adjacent macrophages had little or no cytoplasmic bacterial antigen. This could indicate that the hypervirulent K. pneumoniae capsule probably impairs macrophage uptake of bacteria in CSLs; however, additional studies are necessary to confirm this observation. In CSL tissues, intravascular macrophages sometimes contained intact bacteria. Even though the presence of intact bacteria indicates intrahistiocytic bacteremia, it is unclear if these intrahistiocytic bacteria were viable. If that is the case, this could be an important means of dissemination to different tissues.
The pathology of hypervirulent K. pneumoniae in CSLs differs from that of New Zealand sea lions, where K. pneumoniae primarily has caused meningoencephalitis with meningeal hemorrhage. 5 However, an important difference between our study and New Zealand sea lions, beside the bacterial strains, is the fact that animals were juvenile, subadult, or adult males in the present study, while in the case of New Zealand sea lions, K. pneumoniae epidemics primarily affected 1- to 3-month-old pups. 5,6 Therefore, some of the observed differences could be due to the higher susceptibility of neonates to bacterial meningitis. 2 Although in the described case of severe meningoencephalitis, we considered this lesion the most likely cause of the animal’s death, the yearling CSL also had severe bronchopneumonia and pyothorax. In addition, the pattern of bacterial distribution within the brain resembled classical bacteremic spread of gram-negative bacteria to the brain, with necrosis of small white matter blood vessels and spread of bacteria into the brain parenchyma. 2 In the case of New Zealand sea lions, bacterial invasion of white matter blood vessels and parenchyma was rare. 6
The presence of K2 K. pneumoniae on the oropharyngeal and intestinal mucosa could indicate that this bacterium inhabits these mucosae as normal flora, representing potential portals of entry to the lung and liver, but we cannot rule out that some of these bacteria represent postmortem overgrowth or expectorated and ingested microorganisms. In a similar manner, the presence of intact K2 K. pneumoniae in the distal renal tubules could indicate late bacteremic spread of this bacterium to the kidney with potential elimination through urine 7 or postmortem overgrowth.
To conclude, hypervirulent K. pneumoniae causes severe respiratory disease and intrahistiocytic bacteremia in CSLs. Further understanding of the epidemiology and pathogenesis of this strain in CSLs is warranted.
Footnotes
Acknowledgements
We appreciate the necropsy and laboratory assistance of Lauren Rust, Carlos Rios, and the suggestions and comments of 2 anonymous reviewers.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
