Abstract
Histiocytic proliferative diseases are uncommon in cats, although recently a progressive histiocytosis of the skin with terminal involvement of internal organs has been described in cats. Here we describe 3 cats (2 males and 1 female) with pulmonary Langerhans cell histiocytosis (PLCH). The cats were euthanized due to progressive respiratory clinical symptoms and deterioration. Macroscopically, extensive, multifocal to confluent, pulmonary masses were evident. Infiltration of pancreas (2 cats), kidneys (1 cat), liver (1 cat), as well as tracheobronchial, hepatosplenic, or mesenteric lymph nodes (2 cats) was observed by gross or microscopic examination. The infiltrating cells had histiocytic morphology with cytologic atypia characterized by anisokaryosis and hyperchromasia regionally within infiltrated tissues. Lesional histiocytes expressed vimentin, CD18, and E-cadherin. Expression of E-cadherin was usually markedly reduced in extra-pulmonary lesions, which is consistent with possible down-regulation of E-cadherin associated with distant migration from the lung. Transmission electron microscopy demonstrated intracytoplasmic organelles consistent with Birbeck's granules of Langerhans cells in the lesional histiocytes in all cats, except in the pancreas of one cat. These findings were compatible with PLCH with limited organ involvement of humans. It remains unproven whether feline PLCH represents a reactive or neoplastic cell proliferation.
Keywords
Introduction
Langerhans cells (LC) are bone-marrow derived antigen presenting dendritic cells (DC) that colonize the epidermis and other epithelia, which include those of the oral cavity, vagina, and the bronchial mucosa of the lung. 27, 28, 34 After contact with antigen and subsequent antigen internalization, LC migrate to regional lymph nodes and present antigen to T-cells within the paracortex in a major histocompatibility complex (MHC) class II restricted manner. 5
In humans Langerhans cell histiocytosis (LCH) comprises a variety of rare disorders of unknown etiology, which are associated with single or multiple mass lesions composed of cells with an abnormal Langerhans cell phenotype. 18, 34 Several organs including skin, lung, bone, pituitary gland, liver, lymph nodes, and thyroid may be involved. 18, 34 The biological behavior of LCH is variable and unpredictable, which supported the belief that it was a reactive disorder. 18, 28, 34 However, LCH has been shown to represent a clonal expansion of LC and is most likely a neoplastic disease. 37 LCH is approximately three times more common in children than adults. 33 In contrast, pulmonary LCH (PLCH) is much more common in adults, in which it frequently occurs as the sole lesion and uncommonly as part of a multiorgan disease. 33, 34 The majority of PLCH cases are associated with tobacco smoke, and it is suspected that genetic and other environmental factors also play a role in the pathogenesis. 34 The exact nature of PLCH has not been entirely elucidated, but the clinical course is markedly different from systemic LCH, and is frequently associated with regression, which supports a reactive process. However, clonal emergence from a background of reactive LC hyperplasia has been documented in PLCH. 40
In veterinary medicine, proliferative disorders of LC have not been extensively documented. The best-characterized entity is canine cutaneous histiocytoma, which is a tumor of epidermal LC that express CD1 and frequently E-cadherin. Histiocytomas are usually benign, self-limiting, cutaneous epitheliotropic neoplasms, predominantly seen in young dogs. 23 Rare cases of persistent and recurrent cutaneous histiocytomas with involvement of multiple cutaneous sites, draining lymph nodes and rarely internal organs have been reported. 21, 23, 24 In cats histiocytic proliferative diseases in general are uncommon. 1, 8, 10, 14, 15, 19, 25 The most common of these diseases is feline progressive histiocytosis, which presents as a low grade histiocytic sarcoma of skin with terminal metastatic disease. 1 Despite the clinical resemblance to cutaneous LCH, lesional histiocytes were most likely of interstitial DC origin rather than LC origin based on immunophenotypic studies. 1
In the present study we describe 3 cats with histiocytic proliferative disease of LC origin, which primarily affects the lungs with limited involvement of other organs.
Material and Methods
Case selection and tissue handling
Tissue samples were sourced from 3 feline patients presented to the Veterinary Medical Teaching Hospital (VMTH), University of California, Davis between 2001 and 2007. The cats were necropsied within 2 to 16 hours of death. Tissues to be examined microscopically were preserved in neutral-buffered 10% formalin, processed in routine manner, sectioned at 5-μm thickness, and stained with HE. In addition, special stains including Giemsa, Brown and Brenn tissue Gram's stain, and Ziehl Neelsen acid-fast stains were performed on various tissues using standard protocols.
Immunohistochemistry
Immunohistochemical detection of cell differentiation antigens was performed on formalin-fixed lesional tissue from each cat as previously described. 1 Primary antibodies specific for CD3 (clone CD3-12, Leukocyte Antigen Biology Laboratory [LABL], University of California, Davis, CA), CD79a (clone HM57, Dako, Carpinteria, CA), CD18 (clone Fe3.9F2, LABL), CD20 (Rabbit polyclonal antibody, Lab Vision, Fremont, CA) CD45R (B220, clone RA3-6B2, BD Pharmingen, San Jose, CA), E-cadherin (clone 36, BD Transduction Labs, San Jose, CA), vimentin (clone 384, Dako), and pan-cytokeratin (Lu5, Biocare, Walnut Creek, CA) were employed.
Transmission electron microscopy
Selected formalin-fixed tissue samples, which included lung from all cats, kidney from cat No. 1, and pancreas from cat No. 2, were submitted to the transmission electron microscopy laboratory at the California Animal Health and Food Safety (CAHFS) Laboratory, Davis, California. The samples were transferred to Karnovsky's fixative before two washes in 0.2 M sodium cacodylate, and post fixation in 2% osmium tetroxide reduced with 2.5% potassium ferrocyanide. Following fixation, tissue was washed in 0.2 M sodium cacodylate, dehydrated through a graded ethanol series before infiltration and embedment in Spurr's formulation or Eponate 12 epoxy resin (Ted Pella, Redding, CA). Thick sections were cut, mounted on glass slides and stained with Toluidine blue O and examined by light microscopy. Thin sections were cut, mounted on 150 mesh copper grids, stained with 4% uranyl acetate in 75% ethanol and Reynold's lead citrate. The examination was performed using a Zeiss 10 C and a Zeiss 906E (Zeiss Electron Microscopy, Thornwood, NY) transmission electron microscope, respectively.
Results
Clinical features
All cats were presented to VMTH, University of California for treatment. Cat No. 1 was a 10 year old spayed female domestic shorthaired cat with a 7 month history of respiratory disease consisting of tachypnea and increased respiratory effort. Radiographs of the thorax revealed a diffuse, severe broncho-interstitial pattern characterized by severe, diffuse, miliary to nodular opacities throughout all lung lobes (Fig. 1). The cat received symptomatic treatment with glucocorticoids (dexamethasone) for 7 months. The clinical symptoms did not improve and the cat developed progressive dyspnea, exercise intolerance, sneezing with nasal discharge, decreased appetite, severe weight loss, and finally anorexia. Due to the cat's poor condition and clinical prognosis, euthanasia was elected.
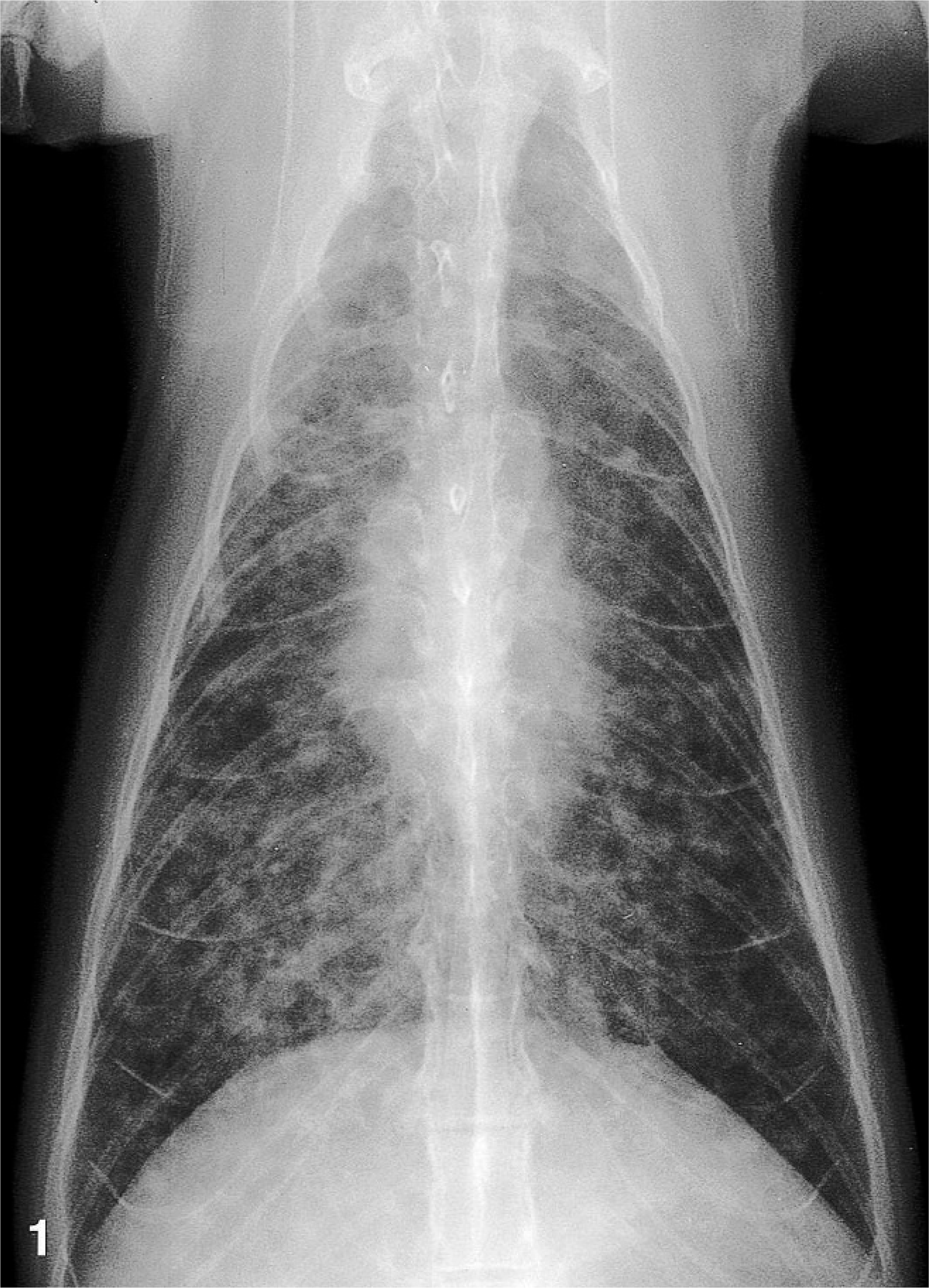
Lung; cat No. 1. Diffuse, severe broncho-interstitial pattern characterized by severe diffuse miliary to nodular pattern throughout all lung lobes. Radiograph.
Cat No. 2 was a 15 year old castrated male domestic shorthaired cat, which lived only indoors. This cat presented for severe respiratory distress characterized by tachypnea and open mouth breathing. Severe, diffuse, broncho-interstitial lung disease was observed in pulmonary radiographs. Based on the clinical evaluation, a neoplastic process was suspected. Ancillary tests to rule out infectious causes (i.e., bacterial cultures, cytology, Cryptococcus antigen test, Toxoplasma antibody titers) were negative. At various points during the 2 months of respiratory disease, the cat received treatment with theophylline, prednisolone, and furosemide. Despite the treatments the cat developed increasing lethargy and inappetance. The cat showed respiratory acidosis with a compensatory metabolic alkalosis and nonresponsive anemia. The cat was euthanized due to progression of clinical disease in the face of continuous oxygen supplementation.
Cat No. 3 was a 12 year old castrated male domestic medium haired cat, which presented with a 5 day history of increased respiratory effort and open mouthed breathing. The cat had lost weight over the last month and was inappetant for the last 2 days. Radiographic evaluation revealed a diffuse miliary pattern throughout the lung fields with an underlying broncho-interstitial pattern. The cat was euthanized due to the likelihood of underlying neoplasia and poor prognosis for recovery.
Gross pathology
The lungs from all cats were consistently and extensively involved by an infiltrative process. All lobes were diffusely firm and entirely effaced by innumerable, ill-defined, tan to pink coalescing nodular masses, which ranged from 2 to 5 mm in maximum dimension (Fig. 2). These nodules coalesced to efface larger areas of the parenchyma and extended to the visceral pleura. Masses of similar size and appearance were evident in the kidneys (cat No. 1) and pancreas (cats No. 2 and 3). Lymph nodes draining affected organs were often diffusely enlarged and tan; these included tracheobronchial (cats No. 2 and 3), mesenteric (cat No. 3) and hepatosplenic lymph nodes (cat No. 2).
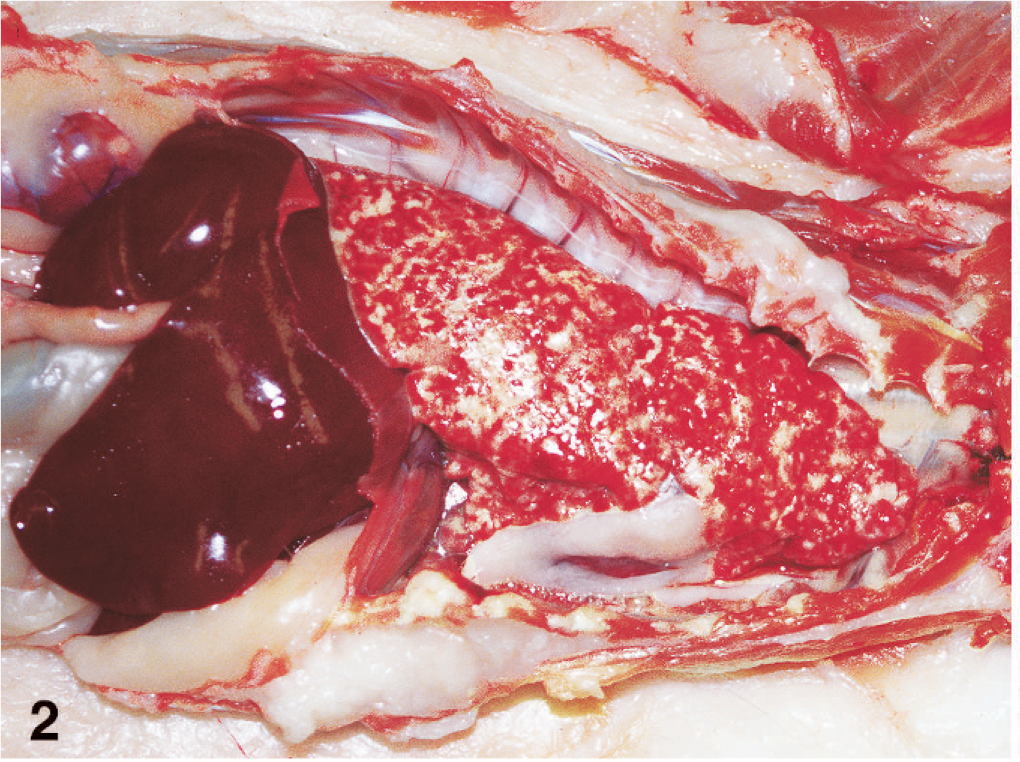
Lung, in situ; cat No. 1. All lobes of the lungs are infiltrated by myriad nodular to coalescing, cream to tan masses.
Histopathology
The pulmonary histiocytic infiltrates in all cats occurred in terminal and respiratory bronchioles. The affected terminal airway walls were partially obliterated and the lumens were filled by the histiocytic infiltrate (Fig. 3). Histiocytic infiltrates extended into contiguous alveolar ducts, and centrifugally into adjacent alveolar lumens (Fig. 4). Large tracts of pulmonary parenchyma adjacent to terminal airways and extending to the visceral pleura were thereby effaced. Affected bronchioles had smooth muscle hyperplasia, and the adjacent alveolar interstitium was thickened by fibrosis. Airways immediately proximal to the affected terminal airways were dilated (hyperinflated) in cat No. 1. Histiocytes were accompanied by aggregates of lymphocytes and fewer plasma cells, which were most numerous in perivascular, subpleural, and peribronchial interstitium. However, more diffuse lymphocytosis within the histiocyte-rich infiltrates also occurred, especially in cat No. 3.
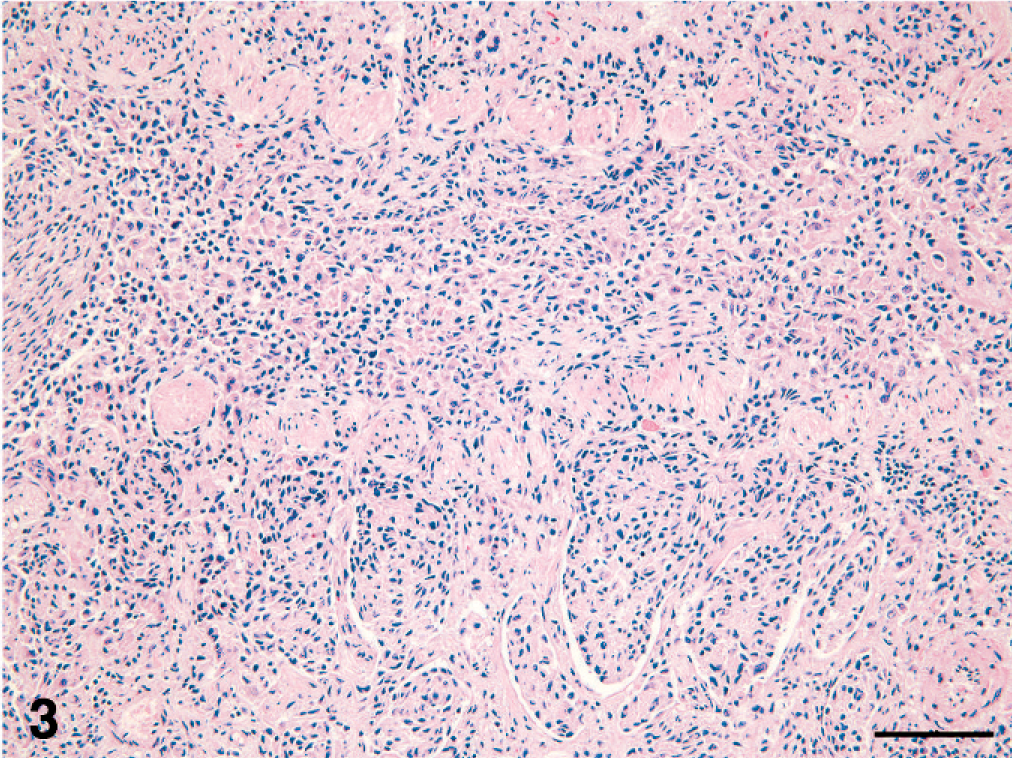
Lung; cat No. 2. Respiratory bronchiole: the lumen is infiltrated by a cohesive histiocytic infiltrate, which permeates the wall and spreads in a peribronchiolar pattern into adjacent alveoli. HE stain. Bar = 100 microns.
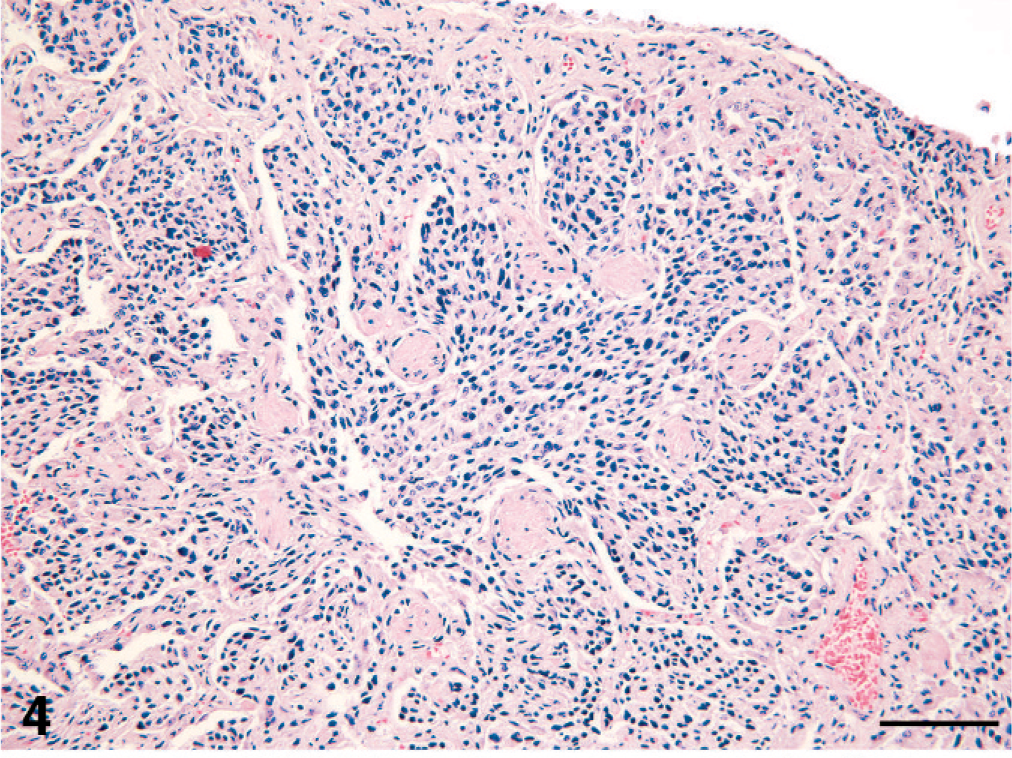
Lung; cat No. 2. Alveolar duct: the lumen of the alveolar duct is obliterated by a cohesive histiocytic infiltrate, which streams into adjacent alveoli and extends to the visceral pleura. Smooth muscle hyperplasia is present in the wall of the alveolar duct. HE stain. Bar = 100 microns.
The lesional histiocytes had indistinct cell membranes, which imparted a cohesive appearance to the infiltrate as it extended uninterrupted into adjacent airspaces. The histiocytes were pleomorphic in cell size and nuclear morphology. Cohesive histiocytes had homogeneously eosinophilic cytoplasm, while dissociated solitary histiocytes often had finely vacuolated cytoplasm (Fig. 5). The nuclear membrane profiles of these cells were oval, indented or had complex contours. The chromatin varied from euchromatic to hyperchromatic. The chromatin structure varied from finely stippled to heterochromatic and was occasionally vesicular. The mitotic rate was 0–2 per 400× microscopic field.
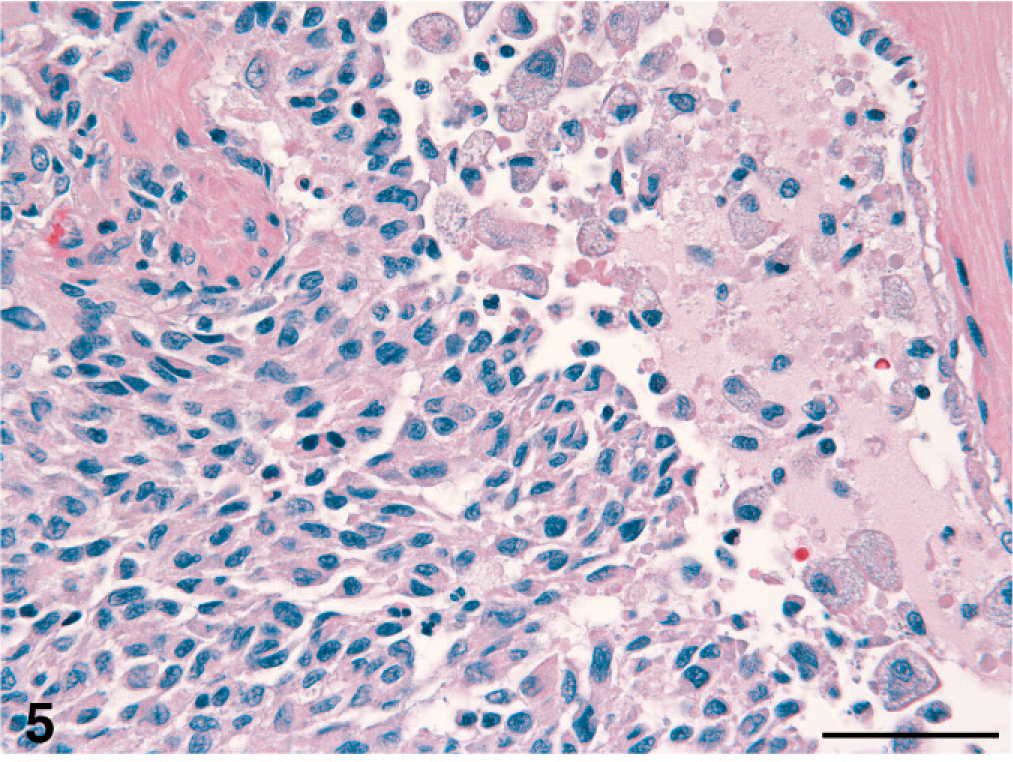
Lung; cat No. 2. A cohesive histiocytic infiltrate streams into the lumen of a respiratory bronchiole. The lesional histiocytes exhibit anisokaryosis and irregular to indented nuclear profiles. Large, foamy, individual histiocytes and cell debris fill the lumen of the bronchiole. HE stain. Bar = 50 microns.
Involved lymph nodes had subcapsular and intermediate cortical sinus infiltration, and extensive replacement of the paracortex by histiocytes with the cytological characteristics of those infiltrating the lung. In one instance (cat No. 2), the paracortical histiocytic infiltrates were noticeably more pleomorphic than the infiltrates in the lung. Histiocytes with karyomegaly and multiple nuclei were frequent; these cells also had finely vacuolated cytoplasm.
Microscopically, histiocytic infiltrates were also present in interlobular interstitium of the pancreas (cats No. 2 and 3), the cortical interstitium of the kidney (cat No. 1), and the periportal interstitium of the liver (cat No. 3). Histiocytic infiltrates were accompanied by lymphocytes and plasma cells in these organs, and were associated with adjacent parenchymal atrophy.
Pulmonary lesions were stained with Giemsa, Periodic acid-Schiff, Brown and Brenn tissue Gram's stain, and Ziehl Neelsen acid-fast stain to demonstrate microorganisms; none were detected.
Immunohistochemistry
Lesional histiocytes expressed vimentin and CD18 in all tissues (Fig. 6). They did not express cytokeratin, CD3, CD20, CD45R or CD79a. The cohesive histiocytic infiltrates in the lung intensely expressed E-cadherin, while the foamy, discrete histiocytes lacked E-cadherin expression (Fig. 7). Histiocytic infiltrates in lymph nodes, pancreas, kidney and liver usually exhibited markedly reduced E-cadherin expression by comparison to pulmonary infiltrates. Strong E-cadherin expression in these tissues was limited to subpopulations of histiocytes among histiocytes that were weak or nonexpressing. Histiocytes infiltrating tracheobronchial and mediastinal lymph nodes in cat No. 3 expressed E-cadherin with similar intensity to those invading the lung. Lymphocytes that accompanied the histiocytic infiltrates in the lung and other organs were largely CD3+ T cells, with far fewer B cells (CD20+, CD79a+).
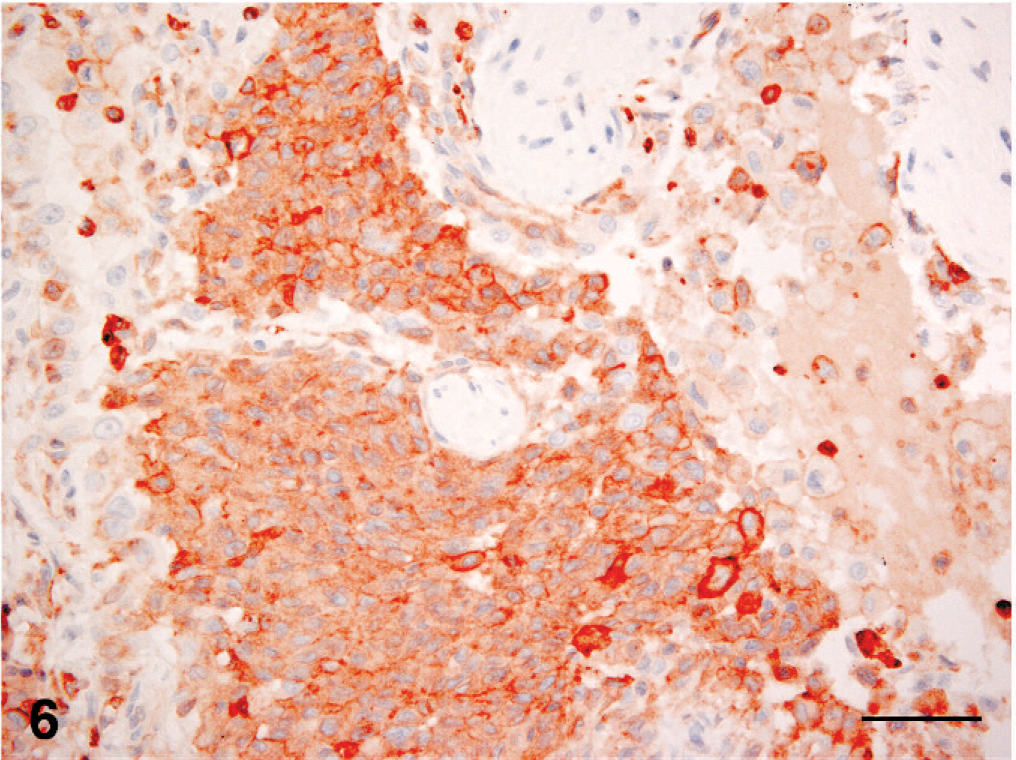
Lung; cat No. 2. Serial section to figure 5. Both the cohesive and individualized histiocytes express CD18. Immunohistochemistry; chromogen: amino-9-ethyl-carbazole, hematoxylin counterstain. Bar = 50 microns.
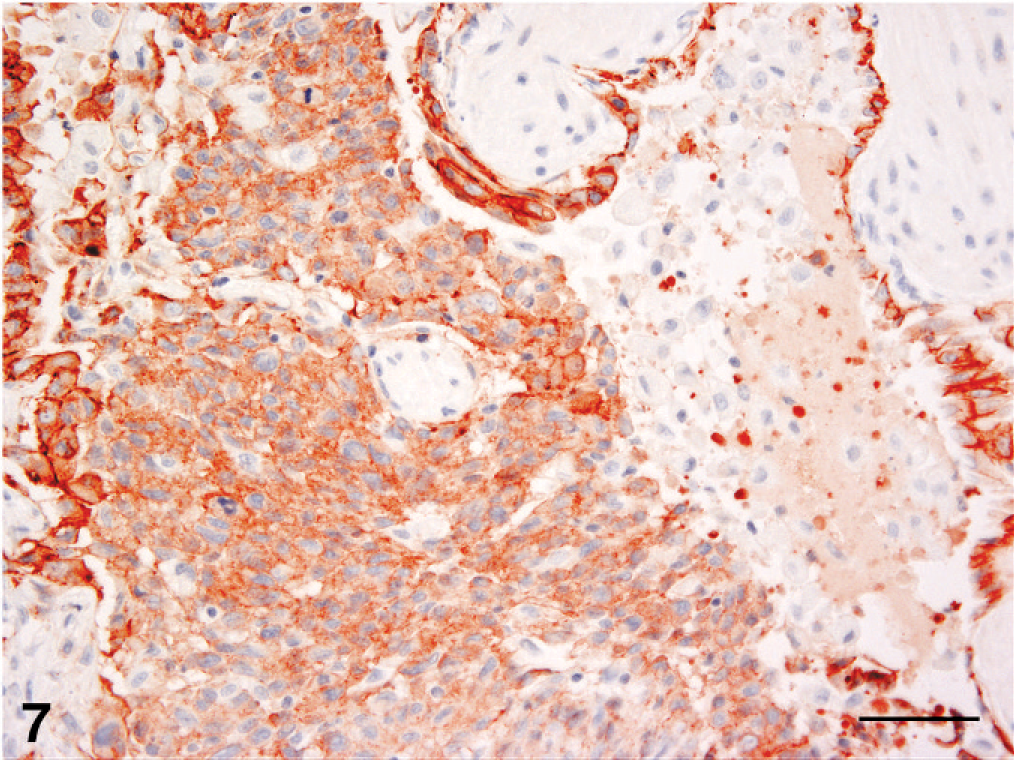
Lung; cat No. 2. Serial section to Fig. 5. Only the cohesive histiocytic infiltrate expresses E-cadherin. The bronchiolar epithelium also expresses E-cadherin. Immunohistochemistry; chromogen: amino-9-ethyl-carbazole, hematoxylin counterstain. Bar = 50 microns.
Transmission electron microscopy
Electron microscopic evaluation was performed on pulmonary lesions from all cats, the renal lesions from cat No. 1, and the pancreas from cat No. 2. Histiocytes, identified by their complex nuclear membrane profiles, contained intracytoplasmic rod-shaped organelles, which possessed a double outer membrane and a central, electron-dense, striated core (Fig. 8). In some instances, a terminal dilated vesicular structure was attached to the rod-shaped organelle. These organelles are consistent with Birbeck's granules, which are characteristically found in LC. Birbeck's granules were found in the histiocytes in all tissues examined except in the pancreas in cat No. 2.
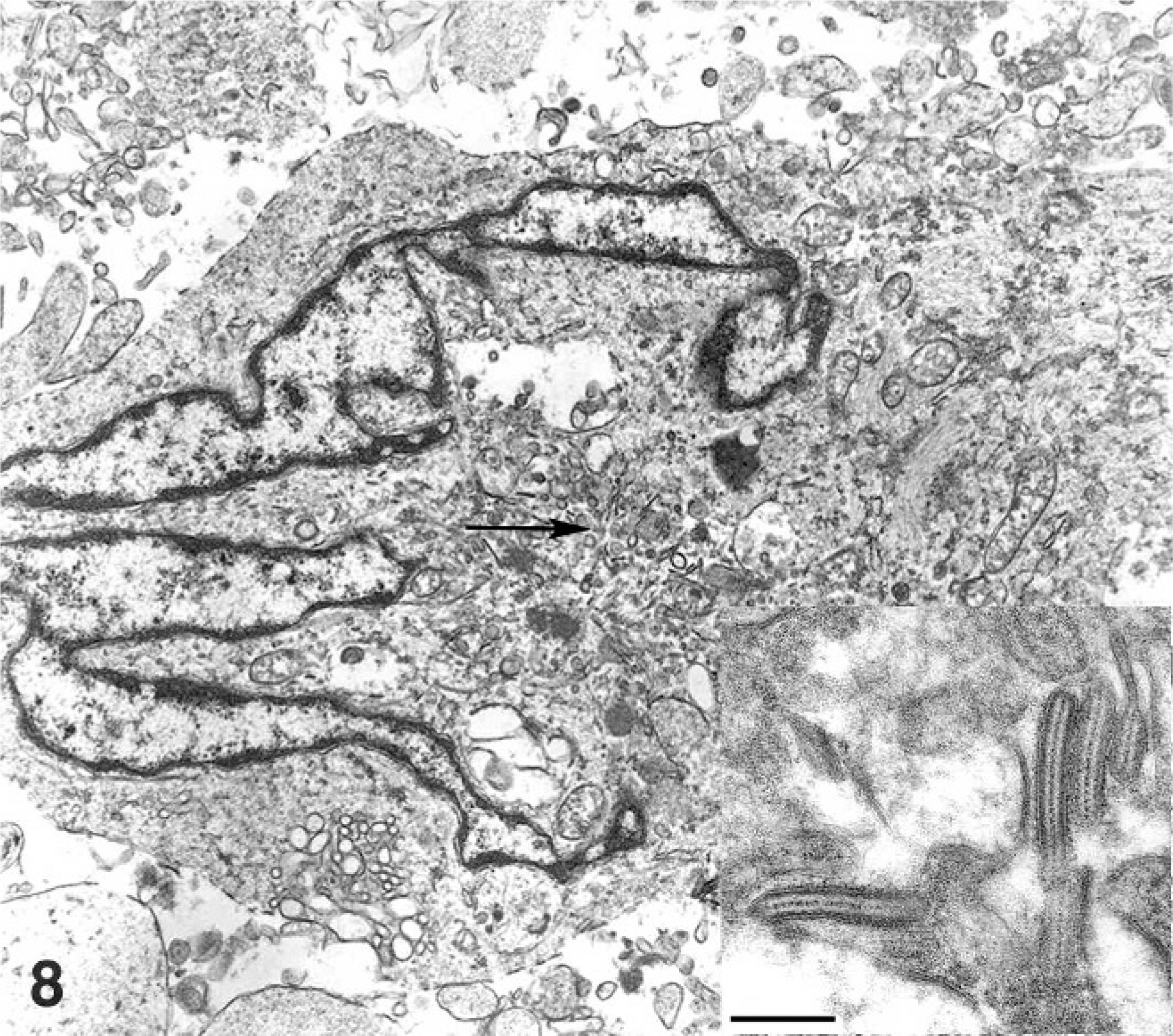
Lung; cat No. 1. Langerhans cell with a complex nuclear profile; this cell contains intra-cytoplasmic Birbeck's granules, which are rod-shaped structures composed of an outer membrane and a central electron dense striated core (inset). Arrow indicates cytoplasmic region magnified in the inset. Transmission electron micrograph; uranyl acetate and lead citrate, Bar = 100 nM.
Discussion
Feline PLCH was identified in 3 cats in which it caused respiratory failure. The disease caused obliteration of terminal airways and contiguous alveoli by cohesive sheets of histiocytes, which had the immunophenotypic and ultrastructural features of LC. Cytologically, lesional histiocytes had variable nuclear morphology, which ranged from mature to immature. In one instance, the tracheobronchial lymph node contained a higher frequency of histiocytes with immature nuclear morphology and marked anisokaryosis, which was evidence in favor of neoplastic transformation. Pulmonary LCH was accompanied by variable and limited involvement of extrapulmonary sites, which included draining lymph nodes, kidney, pancreas and liver.
Langerhans cells and interstitial DC are myeloid DC, which differentiate from a common CD34+ bone marrow precursor under the influence of various cytokines and growth factors. Important among these are GM-CSF, IL-4 and TGF-beta. 6, 7 Langerhans cells are dendritic antigen presenting cells that populate the epidermis and many other epithelial sites, which include bronchial epithelium. 11, 17, 29 Langerhans cells are demonstrable in tissues by their expression of CD1a, major histocompatibility complex class II (MHC-class II) molecules, langerin (CD207) and E-cadherin. 20 Langerhans cells also possess an ultrastructural marker, the Birbeck's granule, which is a pentalaminar rod shaped cytoplasmic organelle present exclusively within LC. 2 Birbeck's granules are found within LC in most species including humans, mice and cats, but not dogs. 2, 23, 27 The formation of Birbeck's granules is induced by langerin, a calcium dependent lectin with surface antigen capture function, which is exclusively expressed by LC. Langerin routes antigen into Birbeck's granules, which may be involved in a non-classical antigen processing pathway. 32
Histiocytes in feline PLCH expressed the leukointegrin CD18, and lacked expression of the lymphoid differentiation antigens CD3, CD20, CD45R and CD79a. Histiocytes also expressed E-cadherin, which is a major transmembrane component of the adherens junction. E-cadherin is expressed by all types of epithelial cells, and also by other cells that localize and are retained within epithelia, for example melanocytes and LC. 4, 16, 31 Hemopoietic cells other than LC mostly lack expression of E-cadherin. 26 Feline LC also express CD1a, but this molecule was not assessable in formalin fixed tissue. 39 Langerin expression was also not assessable due to the current lack of feline specific reagents. However, lesional histiocytes in the lungs of all cats were found to contain Birbeck's granules, which only form in the presence of langerin, thus confirming their designation as LC.
E-cadherin expression was most often weaker in extrapulmonary lesions in feline PLCH. Normal LC downregulate E-cadherin expression in association with migration from epithelia to lymph nodes, where they present antigens acquired in the epithelium to recirculating T cells. 9 Furthermore, reduced E-cadherin expression is also observed in several tumor types in association with invasion and metastasis. 3, 41 Hence, the reduced E-cadherin expression by lesional histiocytes was not unexpected. However, the reason for the failure to identify Birbeck's granules in lesional histiocytes in the pancreas in cat No. 2 is less clear. These histiocytes were morphologically identical to their pulmonary counterparts in other respects.
In humans, LCH comprises a large category of rare diseases with biologically variable behavior and involvement of multiple organs. 18, 28 Pulmonary LCH is an important cause of interstitial lung disease, which is predominantly diagnosed in adult cigarette smokers, whereas the other forms of LCH are most frequently observed in children. 30, 33, 34 Pulmonary involvement in LCH may occur in children as a component of multisystem disease. Multiorgan disease occurs in adult PLCH in up to 20% of cases. 28, 30, 34 The cats in this study were adult and showed clinical, radiographic, and pathological changes resembling those of PLCH with limited extrapulmonary involvement. Specifically the distribution pattern of LC throughout the lung tissue, interspersed CD3+ T lymphocytes, and interstitial fibrosis are changes described in human PLCH. In human PLCH, enlarged air spaces and tissue hyperinflation are frequently observed. 34 Similar distended terminal air spaces and alveoli of variable size suggestive of entrapped air were seen in the feline lungs; these changes were especially prominent in cat No. 1. A pivotal role for T lymphocytes in the development and progression of LCH has been proposed. 13 A rich variety of cytokines produced by T lymphocytes and LC has been detected in human LCH. 12, 13 These observations led to the “cytokine storm” hypothesis, in which it is postulated that T lymphocytes and LC participate in a cytokine amplification cascade, which influences the recruitment, maturation and proliferation of LC in LCH. 13
Despite the role proposed for T cells in amplifying lesions in human LCH, investigation of clonality of lesional histiocytes in human LCH has determined that the disease largely represents a clonal expansion of LC. 37 The clonal origin of LC was demonstrated in all forms of LCH studied; these included unifocal, intermediate (chronic) and acute disseminated disease. Assessment of clonality was possible only in female patients, and relied on the presence of appropriate methylation and polymorphism within loci on the X chromosome, and the use of methylation sensitive restriction endonucleases to distinguish maternal and paternal X-chromosomes. 37 The clonal origin of LC in human PLCH has also been investigated in a similar way. 40 Pulmonary LCH behaves more like a reactive disorder in which disease stabilization or regression often occurs. 30 Progression to respiratory failure is less common, but does occur. 35 Reactive polyclonal expansion of LC occurs frequently in human PLCH, although clonal emergence or outright clonal LC dominance is also observed. 38, 40
Currently, it is not possible to assess clonality of the LC population in feline PLCH, and the cause of this disease remains enigmatic. The presence of extrapulmonary lesions, and the enhanced anisokaryosis in the tracheobronchial lymph nodes in cat No. 2 are features that favor neoplasia. However, extrapulmonary lesions are seen in human PLCH, which is largely a reactive disease. Several potential mechanisms have been proposed in the pathogenesis of human PLCH, which occurs almost exclusively in adult smokers in contrast to other forms of LCH. 34 Langerhans cells populate the tracheobronchial epithelium and play a role in the recognition of inhaled agents. Inhalation of tobacco smoke causes tracheobronchial epithelial damage leading to release of bombesin-like peptides that stimulate alveolar macrophages to secrete cytokines such as TNF-α, GM-CSF and TGF-β which could enhance LC recruitment and activation. 34 Other genetic or environmental factors may contribute to the development of PLCH, since cigarette smoke causes an increase in Langerhans cells in affected as well as asymptomatic smokers. 34 History of tobacco smoke or other pulmonary irritant exposure was not available for the cats with PLCH. All cats were euthanized due to progressive respiratory disease leading to respiratory failure. In human PLCH, cessation of smoking often leads to resolution of symptoms. However, respiratory failure develops in a subgroup of patients and results in death. 35 Severe disseminated forms of PLCH with extensive organ involvement are also associated with high mortality in humans. 33
In cats, rare cases of histiocytosis have been described. These were either associated with internal disease 8, 15, 19, 36 or with cutaneous disease and terminal metastases to lymph nodes and various internal organs. 1, 10
In the relatively few cases of internal histiocytosis, pathological changes included bone marrow infiltration, hepatosplenomegaly with ascites, and extensive metastasis. 8, 15, 19, 36 These cats were diagnosed with malignant histiocytosis based on tumor cell morphology, marked hemophagocytosis, and variably positive immunohistochemical staining for lysozyme, α-1 antitrypsin or Mac387. 15, 19, 36 Alternatively, enzyme histochemical detection of acid phosphatase or nonspecific esterase was used for diagnosis. 8 These reports are most consistent with disseminated hemophagocytic histiocytic sarcomas of macrophage origin, which has recently been extensively characterized in dogs. 22
Feline histiocytosis initially limited to the skin was recently characterized and termed feline progressive histiocytosis (FPH). 1 FPH is a slowly progressive proliferative disease of interstitial dendritic cells that differs markedly from the feline cases presented here. The characteristic feature of FPH is the presence of solitary, or more often, multiple dermal nodules. Terminal spread to lymph nodes and other internal organs occurs. Histiocytes in FPH manifest epitheliotropism in about 40% of cats. Histiocytes express abundant CD1, CD18 and MHC class II, but largely lack expression of E-cadherin, which is consistent with an interstitial DC phenotype rather than an LC phenotype. 1
Proliferative disorders of LC are rare in veterinary medicine, but are most commonly described in dogs. 21 Canine cutaneous histiocytoma is a localized tumor of LC with a high incidence of spontaneous regression. 23 Uncommonly, histiocytomas occur as multiple lesions that persist or reoccur. Draining lymph nodes may also be obliterated by neoplastic histiocytes in a subset of these dogs. 21 In rare instances, widespread internal metastasis involving lymph nodes, lungs, and other organs occurs much like acute progressive LCH of humans. 21, 24 Pulmonary lesions in dogs with LCH are localized to peribronchial and perivascular regions. Obliteration of bronchiolar lumens and extensive effacement of alveoli characteristic of feline PLCH have not been observed (P. F. Moore, unpublished observations).
In conclusion, we have described a unique histiocytic proliferative disorder of unknown etiology in three adult cats. The disease consistently and primarily targeted the lungs with variable and limited involvement of other organs. The pathological findings were most compatible with a subset of human PLCH in which extrapulmonary spread occurs. Definitive proof regarding the reactive or neoplastic nature of this disease will require development of molecular techniques to assess clonality of the lesional LC in cats.
Footnotes
Acknowledgements
We would like to thank Bob Nordhausen and Diane Naydan for their excellent assistance in handling the electron microscopy and immunohistochemical stains.
