Abstract
Histiocytic proliferative diseases are rare in cats, and their pathogenesis is poorly understood. In the present study, 25 cases of histiocytic sarcoma (HS) and 6 of feline progressive histiocytosis (FPH) were examined, and survival times were recorded in 19 cases. The immunophenotypes of tumor cells in these cases as well as of nonneoplastic feline histiocytes were characterized using formalin-fixed, paraffin-embedded tissues. An FPH cell line (AS-FPH01) and xenotransplant mouse model of FPH were also established. The median survival time of HS (150 days) was significantly shorter than that of FPH (470 days). Immunohistochemically, nonneoplastic histiocytes were immunopositive for various combinations of Iba-1, HLA-DR, E-cadherin, CD204, CD163, CD208, and MAC387. By immunohistochemistry, dermal interstitial dendritic cells (iDCs) and macrophages were CD204+/E-cadherin−, while epidermal Langerhans cells (LCs) were CD204−/E-cadherin+. Neoplastic cells of 4 FPH and 18 HS were CD204+/E-cadherin− (iDC/macrophage immunophenotype), while 2 FPH and 2 HS were CD204−/E-cadherin+ (LC immunophenotype), and 5 HS were CD204+/E-cadherin+ (LC-like cell immunophenotype). Furthermore, immunohistochemical and western blot analyses of AS-FPH01 cells derived from E-cadherin-negative FPH revealed that cultured cells were immunopositive for both CD204 and E-cadherin in vitro and in vivo. These results indicate that the neoplastic cells of feline HS and FPH were variably positive for iDC/macrophage and LC markers, and their immunophenotype changed in different microenvironments. The novel cell line established in the present study may serve as an experimental model of FPH that will enable further molecular and therapeutic studies on this disease.
Keywords
Histiocytic proliferative diseases are less common in cats than in dogs. Four types of histiocytic proliferative diseases have been clinically and histopathologically characterized in cats: feline progressive histiocytosis (FPH), histiocytic sarcoma (HS), hemophagocytic HS, and feline pulmonary Langerhans cell histiocytosis (LCH). 28,29 The clinical course of FPH tends to be indolent, while the other feline histiocytic proliferative diseases have a poor clinical outcome due to aggressive behavior. 1,4,16 Among these 4 types, HS and FPH are the most common, while pulmonary LCH and hemophagocytic HS are extremely rare. 28,29 The lesions of HS develop in various organs, such as subcutaneous tissue, spleen, liver, esophagus, nose, eyes, and brain, and metastatic lesions are often detected in regional lymph nodes. 17,33,35,36,46 The lesions of FPH frequently develop in the dermis of the head, limbs, and trunk. As the disease progresses, lesions extend to the lymph nodes and internal organs, similar to HS. 1,34
The origin of neoplastic cells is considered to be interstitial dendritic cells (iDCs) in FPH and HS, Langerhans cells (LCs) in pulmonary LCH, and macrophages in hemophagocytic HS. 2,4,23,28,29 However, only a few immunohistochemical markers have the capacity to distinguish different types of histiocytes in feline tissue. Therefore, the immunophenotypes of neoplastic as well as nonneoplastic histiocytes in cats have not yet been examined in detail. In some FPH cases, neoplastic cells were positive for E-cadherin, an LC marker; therefore, the origin of neoplastic cells remains controversial. 5,34
In order to elucidate the pathological and immunophenotypic characteristics of feline histiocytic proliferative diseases, histopathological, immunohistochemical, ultrastructural, and western blot analyses were performed on formalin-fixed tissues of 31 cats with histiocytic proliferative diseases and the cultured cells of a case of FPH. Furthermore, nonneoplastic histiocytes in feline tissues were examined to assess the immunoreactivity of the primary antibodies. In this study, histiocytes were defined as differentiated and undifferentiated cells of myeloid-derived monocytes/dendritic cells (DCs) including iDCs, Langerhans cells, and macrophages.
Materials and Methods
Samples and Clinical Information
Tissue samples from 31 cases of feline histiocytic proliferative diseases, including 24 biopsy and 7 necropsy cases, were examined (Table 1). Information on treatments and survival after lesion detection was available for 19 cases. Survival of FPH (n = 4) and HS (n = 15) were compared by the log-rank test. P value of <.05 was considered statistically significant. Statistical analysis was performed using the Statcel4 software (OMS publishing Inc). Feline tissues with nonhistiocytic proliferative diseases, including bone marrow, skin, lymph nodes, spleen, liver, intestines, lungs, and brain, were collected from 5 necropsied cats (age range: 2–15 years) and examined to assess the immunoreactivity of the primary antibodies for nonneoplastic histiocytes. All tissue samples were fixed in 10% neutral-buffered formalin and routinely embedded in paraffin. A fresh tissue sample was collected from the skin lesion of case 2 and used in cell culture.
Anatomical Locations of Lesions and Clinical Information on 31 Cats With Histiocytic Tumors.
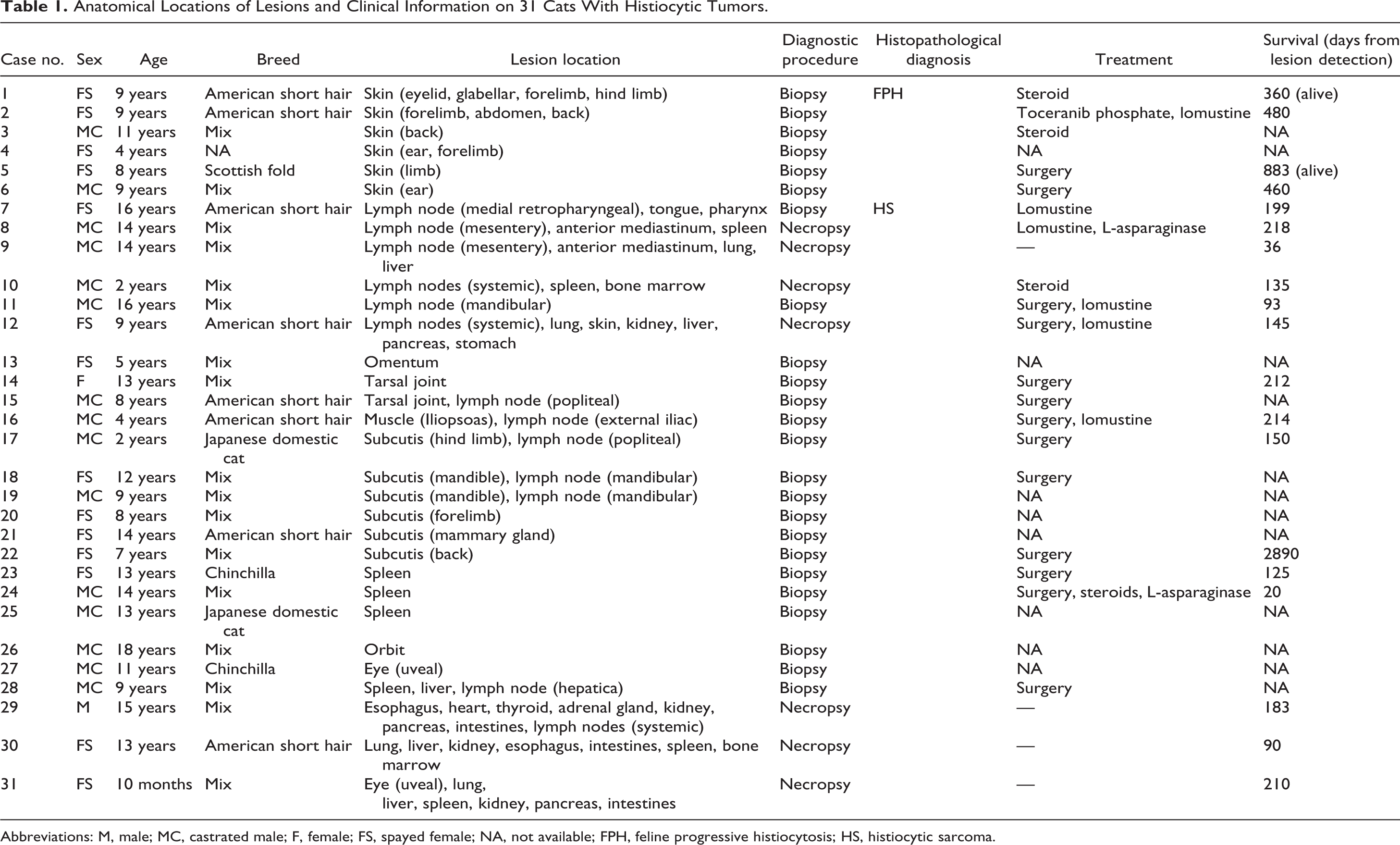
Abbreviations: M, male; MC, castrated male; F, female; FS, spayed female; NA, not available; FPH, feline progressive histiocytosis; HS, histiocytic sarcoma.
Histopathology
Four-micrometer-thick paraffin sections were deparaffinized, rehydrated through graded alcohols, and stained with hematoxylin and eosin. Each slide was evaluated and diagnoses were reached based on the consensus of 2 Japanese College of Veterinary Pathologists board-certified pathologists (JKC and KU). According to the current classification of feline histiocytic proliferative diseases, FPH was diagnosed by the presence of the main lesion in the dermis as well as weak to moderate anisokaryosis and nuclear membrane irregularities in neoplastic histiocytes. 28,29 HS was diagnosed based on neoplastic histiocytes making nodular lesions in a single or multiple organs. Furthermore, skin lesions composed of neoplastic histiocytes with severe nuclear atypia were also diagnosed as HS. Neoplastic cells were morphologically divided into 2 types: the round/polygonal cell type, >50% of neoplastic cells were round- and/or polygonal-shaped; the spindle cell type, >50% of neoplastic cells were spindle-shaped. Anisokaryosis was graded as − (negative), difference in nuclear size of less than 1.5-fold; + (mild), difference in nuclear size of 1.5- to 2-fold; ++ (moderate), difference in nuclear size of 2- to 3-fold; +++ (severe), difference in nuclear size of more than 3-fold. Nuclear membrane irregularity was graded as − (negative), almost no neoplastic cells showed irregularity; + (mild), 1% to 25% of neoplastic cells showed irregularity; ++ (moderate), 26% to 50% of neoplastic cells showed irregularity; +++ (severe), >50% of neoplastic cells showed irregularity. Hemophagocytosis was graded as − (negative), less than 20% of neoplastic cells showed erythrophagia; + (positive), more than 20% of neoplastic cells showed erythrophagia.
Immunohistochemistry
Details of the primary antibodies and antigen retrieval procedures used are summarized in Table 2. Following antigen retrieval, endogenous peroxidase was inactivated with 3% hydrogen peroxide in methanol at room temperature for 5 minutes. Sections were blocked with 8% skim milk in Tris-buffered saline (TBS) at 37 °C for 30 minutes and then incubated with each primary antibody at 4 °C overnight. After washing with TBS, sections were incubated with the anti-rabbit or anti-mouse Dako EnVision+ system hydrogen peroxidase-labeled secondary antibody polymer (Dako) at 37 °C for 30 minutes. To detect CD208, sections were incubated with biotin-conjugated anti-rat immunoglobulin G (1:400; KPL) at 37 °C for 30 minutes, and then incubated with horseradish peroxidase-conjugated streptavidin (1:400; Dako) at 37 °C for 30 minutes. Labeled complexes were visualized with 3,3′-diaminobenzidine (DAB) chromogen and 0.03% hydrogen peroxidase in Tris-HCl buffer, and counterstained with hematoxylin. Nonneoplastic histiocyte type was defined by the location and the morphology. The immunoreactivity of histiocytes in each tissue was evaluated as follows: −, negative; ±, some cells positive; +, positive. The immunohistochemical scoring of feline histiocytic proliferative diseases was graded as − (negative), negative in neoplastic cells; + (weakly positive), positive in 1% to 25% neoplastic cells; ++ (moderately positive), positive in 26% to 50% neoplastic cells; +++ (strongly positive), positive in >50% neoplastic cells.
Primary Antibodies Used for Immunohistochemistry.
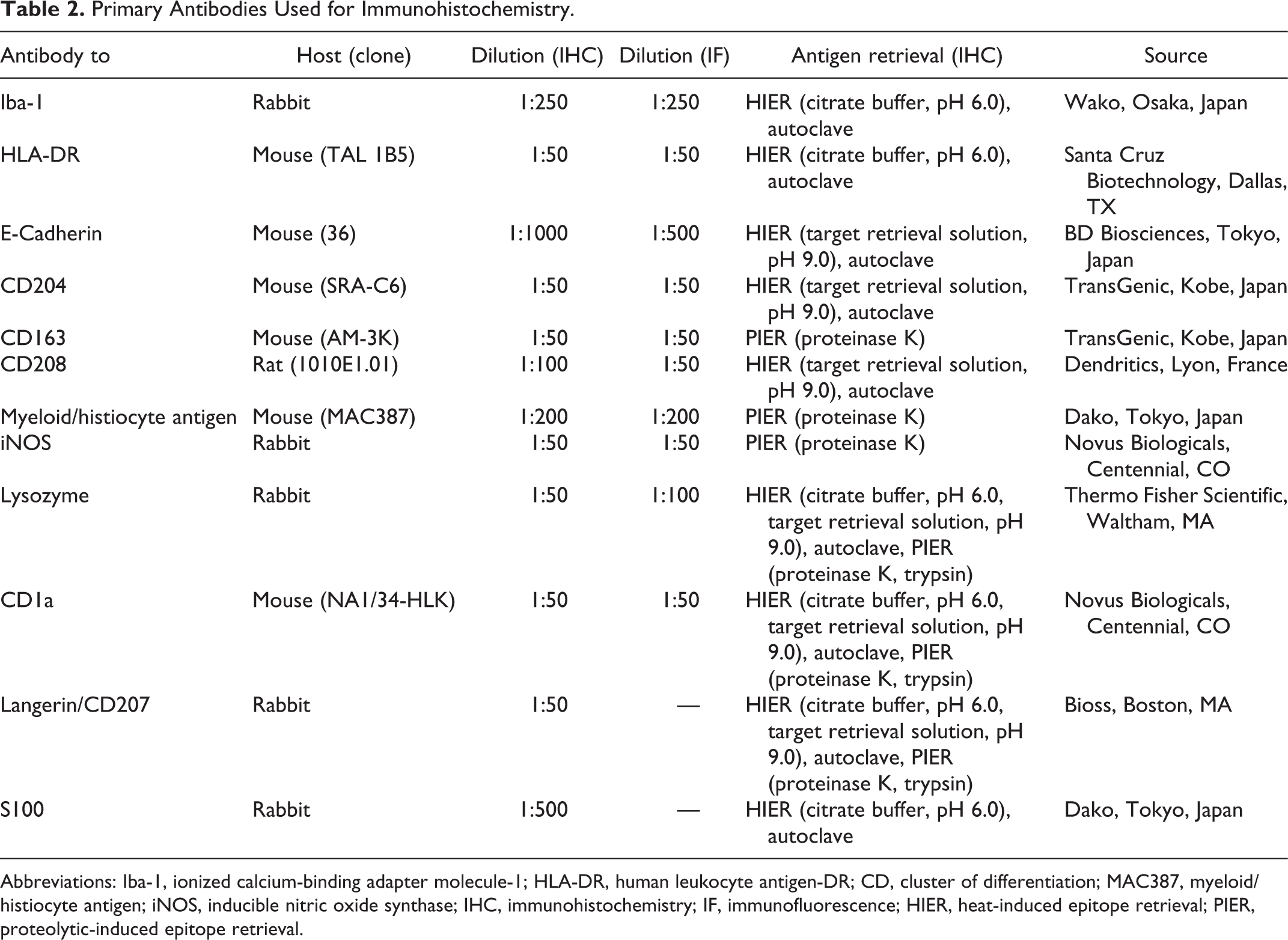
Abbreviations: Iba-1, ionized calcium-binding adapter molecule-1; HLA-DR, human leukocyte antigen-DR; CD, cluster of differentiation; MAC387, myeloid/histiocyte antigen; iNOS, inducible nitric oxide synthase; IHC, immunohistochemistry; IF, immunofluorescence; HIER, heat-induced epitope retrieval; PIER, proteolytic-induced epitope retrieval.
Double-labeling immunohistochemistry was performed on feline lymph node sections to detect CD204- and E-cadherin-double-positive cells. After heat-induced epitope retrieval (pH 9.0), endogenous peroxidase was inactivated with 3% hydrogen peroxide in methanol at room temperature for 5 minutes. Sections were blocked with 8% skim milk in TBS at 37 °C for 30 minutes and then initially incubated with the anti-E-cadherin antibody at 37 °C for 90 minutes. After washing with TBS, sections were incubated with the anti-mouse Dako EnVision+ system hydrogen peroxidase–labeled secondary antibody polymer (Dako) at 37 °C for 30 minutes. Labeled complexes were visualized with DAB chromogen and 0.03% hydrogen peroxidase in Tris-HCl buffer. Sections were then heated at 121 °C for 1 minute in the citrate buffer (pH 6.0) to inactivate the anti-E-cadherin antibody and the anti-mouse secondary antibody. Then sections were blocked with 8% skim milk in TBS at 37 °C for 30 minutes and incubated with the anti-CD204 antibody at 4 °C overnight. After the treatment with N-Histofine Simple Stain AP (M) (Nichirei Biosciences), which is an alkaline phosphatase-conjugated anti-mouse secondary antibody, the immunoreaction was visualized using the N-Histofine New Fuschin substrate (Nichirei Biosciences) and counterstained with hematoxylin.
Immunofluorescence
Double-labeling immunofluorescence was performed on feline skin and lymph node sections to detect Iba-1 and E-cadherin-double-positive cells and Iba-1 and CD204-double-positive cells. After antigen retrieval, a mixture of 2 primary antibodies was applied to each section and incubated at 4 °C overnight. After washing with TBS, sections were incubated with an Alexa 488-conjugated donkey anti-mouse IgG antibody (1:200; Invitrogen) and Alexa 594-conjugated donkey anti-rabbit IgG antibody (1:200; Invitrogen) at room temperature for 1 hour and then counterstained with DAPI (Vector Laboratories). Slides were examined with a Zeiss LSM 700 laser scanning confocal microscope (Carl Zeiss Meditec).
Transmission Electron Microscopy
Portions of formalin-fixed lymph node tissue from an E-cadherin-positive HS (case 8) were washed with 0.1 M phosphate buffer (pH 7.4), fixed in 1% osmium tetroxide, and then dehydrated through a graded ethanol series. Specimens were then embedded in Luveak-812 resin (Nacalai Tesque). Ultrathin sections were counterstained with uranyl acetate and lead solution. Prepared sections were examined with a transmission electron microscope (JEM-1400Plus; JEOL).
Cell Culture of FPH
Primary neoplastic cells were isolated from an E-cadherin-negative FPH (case 2), washed, and then centrifuged with sterile phosphate-buffered saline (PBS; pH 7.4) containing 1% penicillin-streptomycin (FUJIFILM Wako Pure Chemical). Neoplastic cells were suspended in Dulbecco’s modified Eagle’s medium (DMEM; Gibco, Thermo Scientific) containing 0.25% crude trypsin (BD Difco) and incubated at 4 °C for 16 hours. Thereafter, cells were centrifuged at 1000 rpm at 4 °C for 5 minutes and pelleted cells were resuspended in DMEM supplemented with 10% heat-inactivated fetal bovine serum (FBS; GE Health Life Sciences) and 1% penicillin-streptomycin, and then filtered through a cell strainer (70 μm; Falcon). The cell suspension was seeded on collagen-coated 60-mm Petri dishes (CORNING) and maintained in a humidified atmosphere of 5% carbon dioxide (CO2) and 95% air at 37 °C. Cells were cloned by a serial dilution method. Once cultured cells reached 90% confluence, they were removed and seeded on collagen-coated 60-mm petri dishes with 10% FBS in DMEM. Cell passages were performed weekly. A cell line named AS-FPH01 was established and characterized as described later.
Morphological Characterization of FPH Cells
AS-FPH01 cells (third passage) were seeded at 1 × 105 cells in 2-well chamber slides (Thermo Scientific) and incubated at 37 °C in an atmosphere of 5% CO2 and 95% air. At approximately 80% confluence, cells were fixed in methanol and stained with Giemsa solution (Merck Millipore).
Growth Curve
AS-FPH01 cells (12th passage) were seeded at 1 × 105 cells on 16-well multiplates and maintained in growth medium at 37 °C for 14 days. The number of cells in each well was counted 1, 2, 4, 6, 8, 10, 12, and 14 days after the initiation of the cell culture. After the growth curve had been obtained, the cell population doubling time was calculated using the following equation: doubling time (hours) = T × log (2)/log (Ne/Nb), where T is the duration (hours) of the culture, Nb is the cell number at the beginning of the incubation time, and Ne is the cell number at the stationary phase.
Chromosome Analysis
Confluent cultured AS-FPH01 cells (third passage) were incubated with 0.02 μg/mL of colcemid (KaryoMAX; Thermo Scientific) at 37 °C for 2 hours in a humidified atmosphere of 5% CO2 and 95% air and then treated with prewarmed hypotonic 0.075 M potassium chloride (FUJIFILM Wako Pure Chemical). After centrifugation, cell pellets were resuspended in fixative solution (methanol/acetic acid; 1:1), dropped on a slide, and subsequently stained with Giemsa solution. Two hundred metaphases were counted under a microscope with an oil immersion objective lens (100×) to estimate the modal chromosome number.
Immunofluorescence of FPH Cells
Primary cultured cells and AS-FPH cells (18th passage) were each seeded and grown on chamber slides 48 hours prior to staining. After fixation with 4% paraformaldehyde, the cell membrane was permeabilized by incubating cells with 0.25% Triton X-100 for the detection of intracellular antigens. After being rinsed with PBS, cells were treated with 1% bovine serum albumin in PBS-Tween 20 to block nonspecific reactions. Primary antibodies (Table 2) were applied at 4 °C overnight. Cells were further incubated with the Alexa 488-conjugated donkey anti-mouse or anti-rabbit IgG antibody (1:400; Invitrogen) at room temperature for 1 hour. To detect CD208, cells were incubated with biotin-conjugated anti-rat immunoglobulin G (1:400; KPL) at room temperature for 30 minutes, and then incubated with fluorescein-conjugated streptavidin (1:400; Vector Laboratories) at room temperature for 1 hour. Cells were counterstained with DAPI (Vector Laboratories). Negative controls were performed by omitting the primary antibodies. Slides were examined with a Zeiss LSM 700 laser scanning confocal microscope (Carl Zeiss Meditec).
Tumorigenicity Assay and Metastasis Assay of AS-FPH01 Cells
Five 5-week-old female severe combined immunodeficient (SCID) mice (FOX CHASE SCID C.B-17/Icr-scid/scidJcl; CLEA Japan) were subcutaneously (SC) injected with 200 µl of the AS-FPH01 cell (fourth passage) suspension (5 × 106 cells) at the lower back. These mice were housed in a cage with free access to water and a standard purified rodent growth diet (AIN-93G, Oriental Yeast). When the masses were palpable at the injection site, tumor volumes were evaluated every 3 days using the following equation: tumor volume (mm3) = (length × width 2 )/2. 19 All mice were euthanized with isoflurane 2 months after the injection or when the tumor volume exceeded 1000 mm3. To assess metastatic ability, 100 µl of the AS-FPH01 cell (fourth passage) suspension (5 × 105 cells) was injected into the lateral tail vein of 6 SCID mice. All mice were euthanized with isoflurane 2 months after the injection. Tumor masses and organs were collected at necropsy and fixed in 10% neutral-buffered formalin. IHC was performed as described above. All animal experiments were approved by the Committee of Animal Experiments, Graduate School of Agriculture and Life Sciences, the University of Tokyo (Approval Number P17-017).
Western Blot Analysis
Based on the results of immunohistochemistry and immunofluorescence on feline tissues, CD204 and E-cadherin were considered to be reliable markers for detecting iDCs/macrophages and LCs, respectively. Thus, western blot analysis using the anti-CD204 antibody and anti-E-cadherin antibody was performed. Furthermore, western blot analysis was performed to confirm the reactivity with an appropriate molecular weight antigen of anti-HLA-DR and anti-CD208 antibodies in the nonneoplastic lymph nodes of cat and dog. The canine lymph node was used as control because cross-reactivity to this anti-HLA-DR and anti-CD208 antibodies had been indicated. 42 Protein extraction from original tumor tissue (case 2), AS-FPH01 cells (22nd passage), AS-FPH01 cell line-derived xenograft (CDX) tissues, feline skin, and feline lymph nodes was performed. RIPA lysis buffer (Santa Cruz Biotechnology) was used to extract the CD204 protein, HLA-DR protein and CD208 protein, and Triton X-100 lysis buffer (20 mM Tris-HCl, pH 7.5, 150 mM NaCl and 1% Triton X-100) was used to extract the E-cadherin protein. A proteinase inhibitor cocktail (Santa Cruz Biotechnology) was mixed with each lysis buffer. Thirty micrograms of protein samples diluted (4:1) with a loading buffer containing dithiothreitol were electrophoresed on a 5% to 20% polyacrylamide gel (e-PAGEL; ATTO) at 200 V, 300 mA for 50 minutes. Protein bands were transferred to a polyvinylidene fluoride membrane (Immobilon-P transfer membrane; Millipore) at 25 V, 500 mA for 25 minutes. After nonspecific binding had been blocked, the following primary antibodies were applied: a mouse anti-E-cadherin antibody (1:500; BD Transduction Laboratories) as a LC marker, a mouse anti-CD204 antibody (1:500; TransGenic) as an iDC/macrophage marker, a mouse anti-HLA-DR antibody (1:300; Santa Cruz Biotechnology) as a histiocytic marker, a rat anti-CD208 antibody (1:200; Dendritics) as a interdigitating cell marker, and a mouse anti-β-actin antibody (1:10000; Cell Signaling Technology) as a loading control. The enhanced chemiluminescence detection system (GE Healthcare) and chemiDoc XRS+ system (Bio-Rad Laboratories) were used for visualization.
Results
Clinical Information on Cats With Histiocytic Proliferative Disease
Information on each case is summarized in Table 1. Fifteen cats were females (14 neutered) and 16 were males (15 neutered). Mean age at the time of diagnosis was 9 years (range: 10 months to 18 years). Breeds were mixed breed (n = 17), American short hair (n = 8), Chinchilla (n = 2), Japanese domestic cat (n = 2), Scottish fold (n = 1), and unknown (n = 1).
Six cats (cases 1–6) had single or multiple skin nodules with ulcers. In 6 cats (cases 7–12), swollen lymph nodes were the main lesion. In case 12, multiple nodular lesions were also observed in the lungs. In 15 cats (cases 13–27), mass lesions were detected in a single organ, including the omentum, tarsal joint, muscle, subcutis, spleen, orbit, and eye; however, 5 cats (cases 15–19) had metastatic lesions in the regional lymph nodes. In 4 cats (cases 28–31), a large mass or nodular lesions were detected in multiple organs indicating systemic involvement.
Information on treatments and survival days after lesion detection was available for 19 of 31 cats. Seven of the 19 cats had received chemotherapy, 6 of which were treated with lomustine. Median survival from lesion detection was 191 days (range: 20–2890 days). The survival time of HS (median, 150 days; range, 360–883 days) was significantly shorter than that of FPH (median, 470 days; range, 20–2890 days; P = .025).
Histopathology
Among the 31 cats that were histopathologically examined, 6 (cases 1–6) were diagnosed with FPH and 25 (cases 7–31) with HS. The morphological characteristics of neoplastic cells in each case are summarized in Supplemental Table S1. In both FPH and HS cases, neoplastic cells had a moderate to large amount of amphophilic cytoplasm and an oval nucleus with a thick and irregular nuclear membrane, coarse nuclear chromatin, and a distinct nucleolus. Hemophagocytosis score was negative in all cases. In FPH cases, round or polygonal neoplastic cells infiltrated the dermis close to the epidermis (Fig. 1). In these cases, anisokaryosis and nuclear membrane irregularities were mild to moderate. In HS cases, neoplastic cells infiltrated several organs. In these cases, neoplastic cells were round, polygonal (Fig. 2), or spindle-shaped (Fig. 3), and different forms of cells were often mixed (Fig. 4). Also, neoplastic cells were often multinucleated. In most cases, anisokaryosis and nuclear membrane irregularities were moderate to severe.
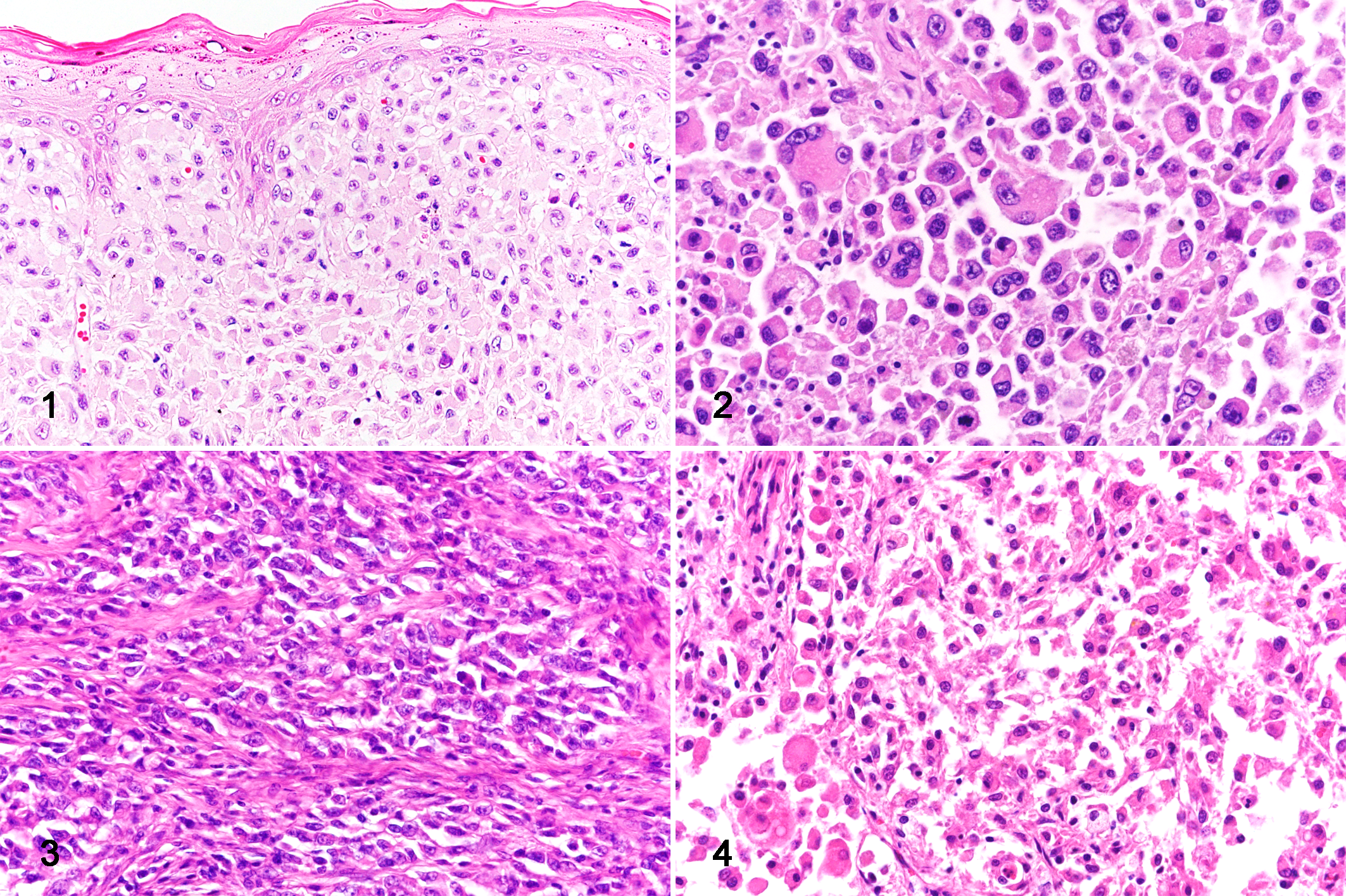
Histiocytic proliferative diseases, cat.
Immunohistochemistry and Immunofluorescence of Nonneoplastic Histiocytes
The immunohistochemical findings of feline histiocytes in each organ are listed in Table 3. Most histiocytes were immunolabeled with antibody to ionized calcium-binding adapter molecule-1 (Iba-1; Figs. 5a, 6a) and human leukocyte antigen-DR (HLA-DR; Fig. 5b, and Suppl. Figs. S1 and S2), while follicular DCs in lymph nodes were negative for Iba-1 and microglia were negative for HLA-DR. Some macrophages and follicular DCs in lymph nodes and lymphoid follicles of the intestinal mucosa were positive for E-cadherin (Fig. 5c). Monocytes, the majority of macrophages, iDCs, and Kupffer cells were positive for CD204 (Figs. 5d, 6b). Although the staining pattern of CD163 was similar to that of CD204, alveolar macrophages were negative for CD163. LCs in the epidermis, follicular DCs, and microglia were negative for CD204 (Fig. 6b) and CD163. Interdigitating cells in the lymph nodes (Suppl. Fig. S3) and spleen (Suppl. Fig. S4) were positive for CD208. Bone marrow monocytes, intravascular blood monocytes, and some macrophages were positive for the myeloid/histiocyte antigen (MAC387). Double-labeling immunohistochemistry of the lymph nodes revealed CD204-positive cells and E-cadherin-positive cells in the sinus (Fig. 7a), and some cells were CD204 and E-cadherin double-positive (Fig. 7b). Double-labeling immunofluorescence of the skin revealed Iba-1 and E-cadherin double-positive LCs in the epidermis (Fig. 8a, b, c). In the sinus of the lymph nodes, there were macrophages that were double-positive for Iba-1 and E-cadherin (Fig. 9a, b, c) and double-positive for Iba-1 and CD204 (Fig. 10a, b, c).
Immunohistochemical Findings of Nonneoplastic Histiocytes in Feline Organsa.
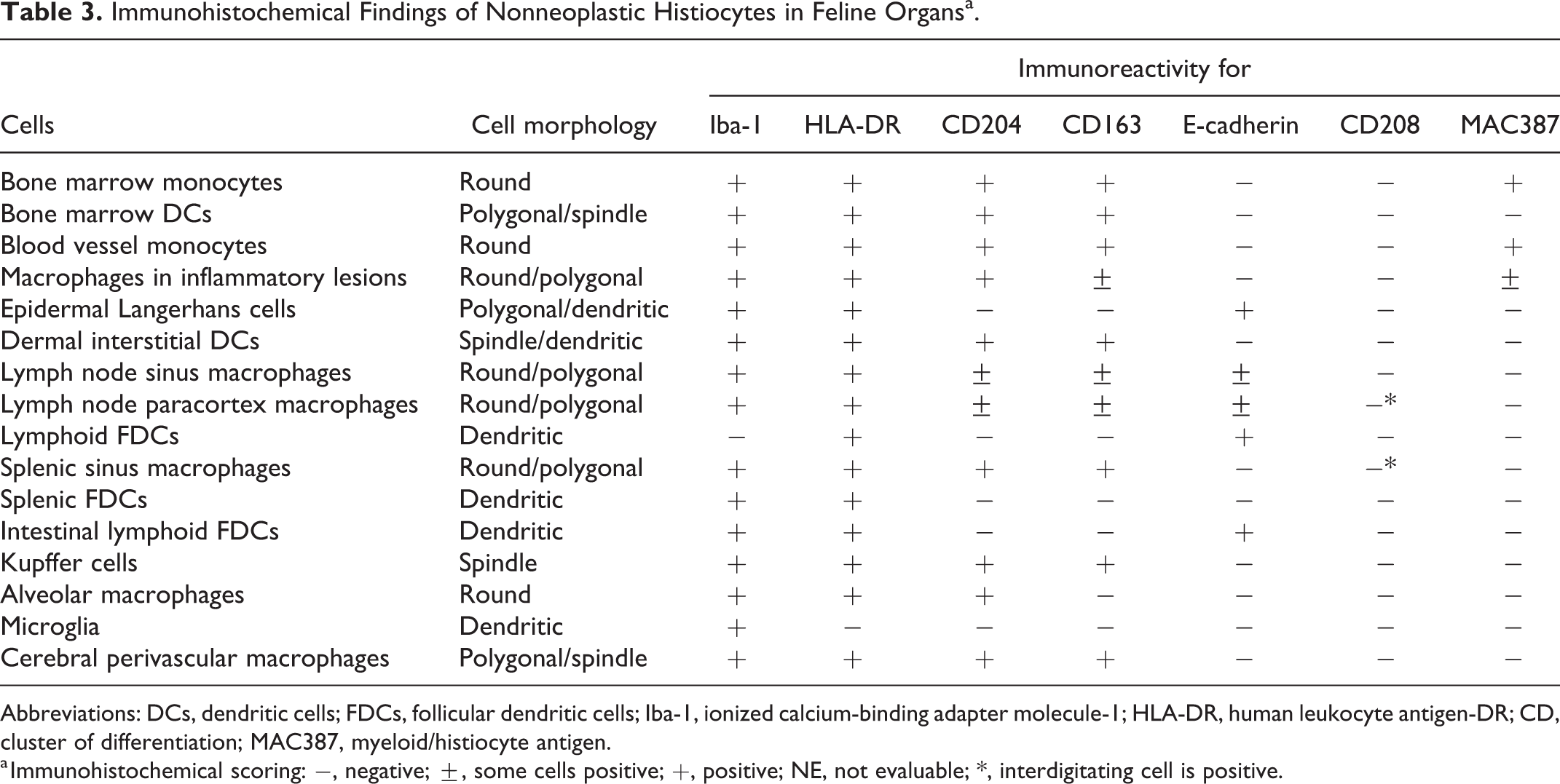
Abbreviations: DCs, dendritic cells; FDCs, follicular dendritic cells; Iba-1, ionized calcium-binding adapter molecule-1; HLA-DR, human leukocyte antigen-DR; CD, cluster of differentiation; MAC387, myeloid/histiocyte antigen.
a Immunohistochemical scoring: −, negative; ±, some cells positive; +, positive; NE, not evaluable; *, interdigitating cell is positive.
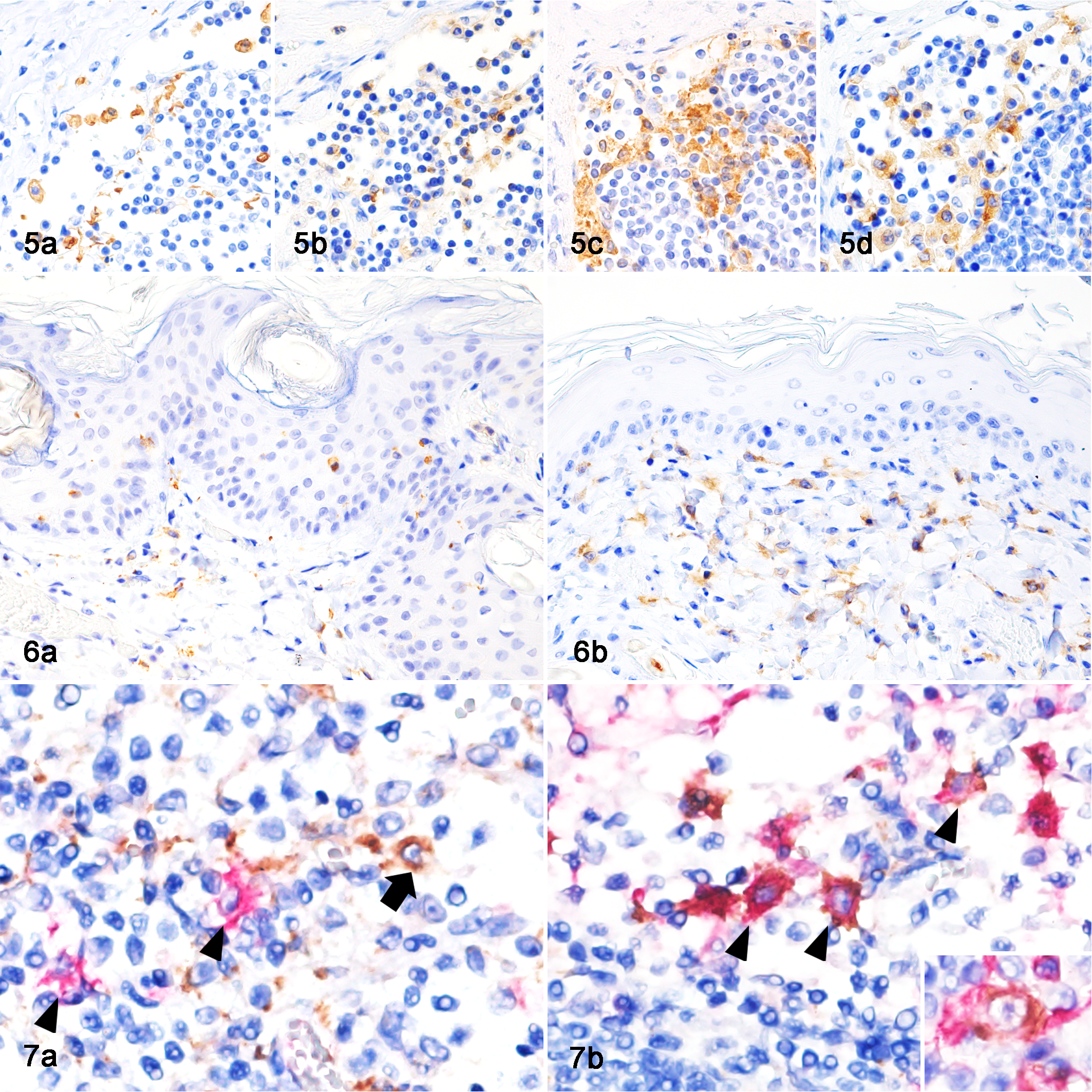
Lymph node, sinus, cat. Histiocytes are positively immunolabeled for (a) Iba-1, (b) HLA-DR, (c) E-cadherin, and (d) CD204. Immunohistochemistry (IHC).
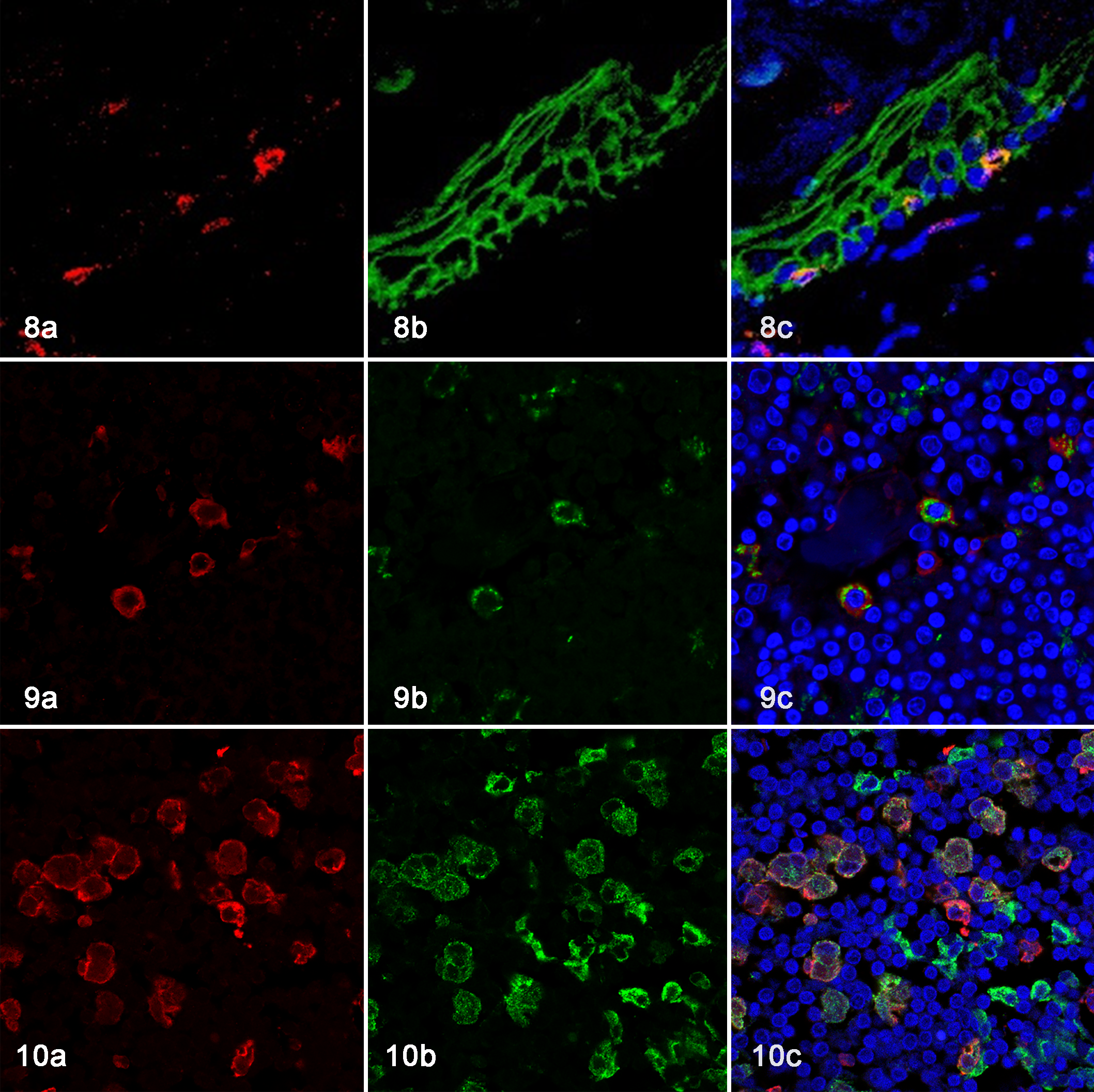
Skin, cat. Double-labeling immunofluorescence (IF). (a) Iba-1-positive cells (red), (b) E-cadherin-positive cells (green). (c) A merged image shows that Langerhans cells are double-positive for Iba-1 and E-cadherin. DAPI nuclear counter staining (blue).
Immunohistochemistry and Immunofluorescence of Feline Histiocytic Proliferative Disease Tissues
The immunohistochemical results of cats with histiocytic proliferative disease are listed in Table 4. The neoplastic cells of all FPH and HS cases were positive for Iba-1 (Figs. 11a, 12a, 13a, 14a), and HLA-DR (Figs. 11b, 12b, 13b, 14b), and negative for CD208 and MAC387. The neoplastic cells of 4 FPH and 23 HS were positive for CD204 (Figs. 11c, 13c, 14c), and the neoplastic cells of 1 FPH and 9 HS were positive for CD163. In 2 FPH (cases 5 and 6) and 7 HS (cases 7–13), neoplastic cells were positive for E-cadherin (Figs. 12d, 13d). Moreover, neoplastic cells were positive for both CD204 and E-cadherin in 5 HS (cases 9–13). In E-cadherin and CD204 double-positive HS (case 12), the immunoreactivity of E-cadherin was strong in skin lesions and weak to moderate in lymph node and lung lesions.
Immunohistochemical Results of Feline Progressive Histiocytosis and Histiocytic Sarcoma.
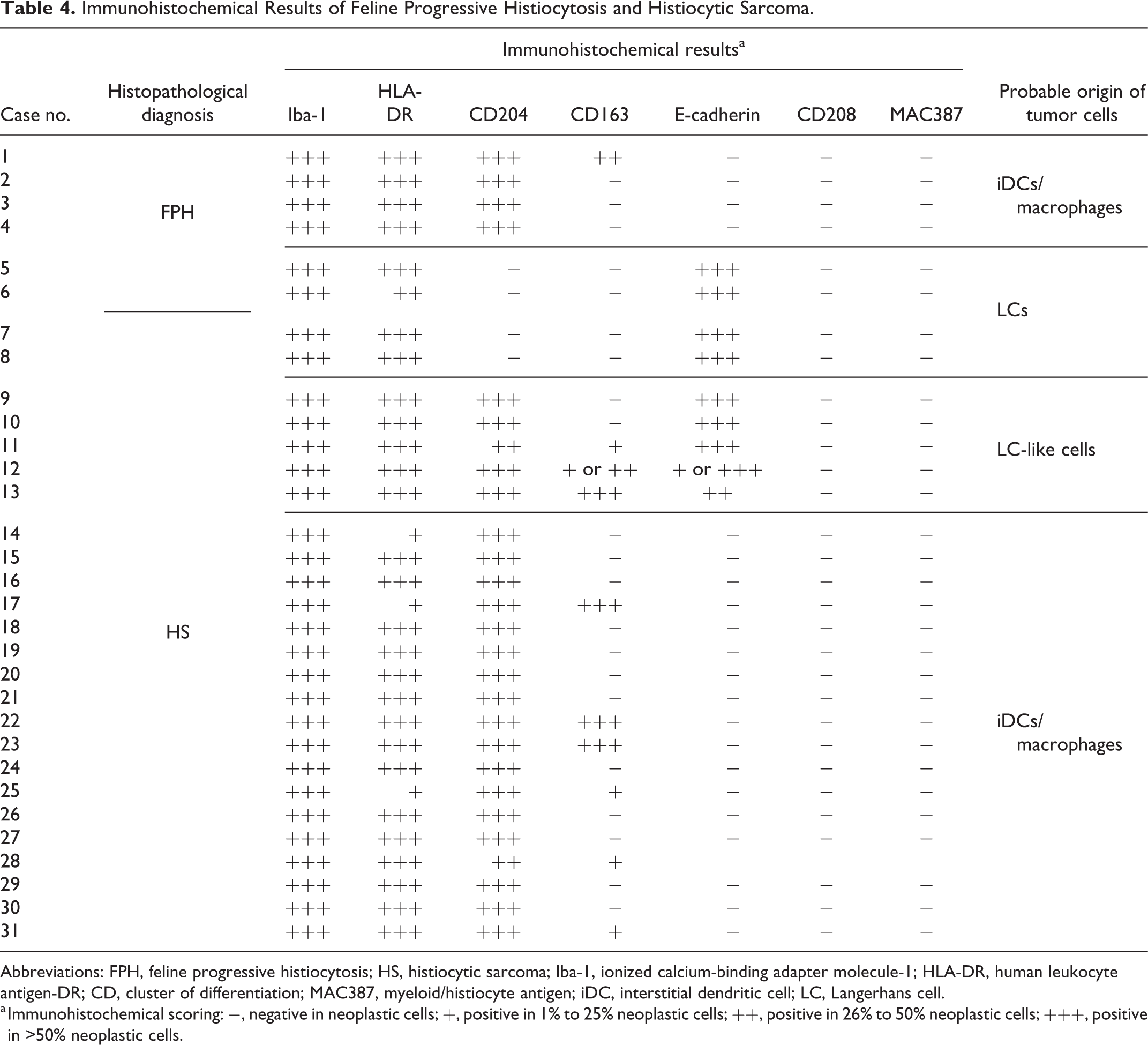
Abbreviations: FPH, feline progressive histiocytosis; HS, histiocytic sarcoma; Iba-1, ionized calcium-binding adapter molecule-1; HLA-DR, human leukocyte antigen-DR; CD, cluster of differentiation; MAC387, myeloid/histiocyte antigen; iDC, interstitial dendritic cell; LC, Langerhans cell.
a Immunohistochemical scoring: −, negative in neoplastic cells; +, positive in 1% to 25% neoplastic cells; ++, positive in 26% to 50% neoplastic cells; +++, positive in >50% neoplastic cells.
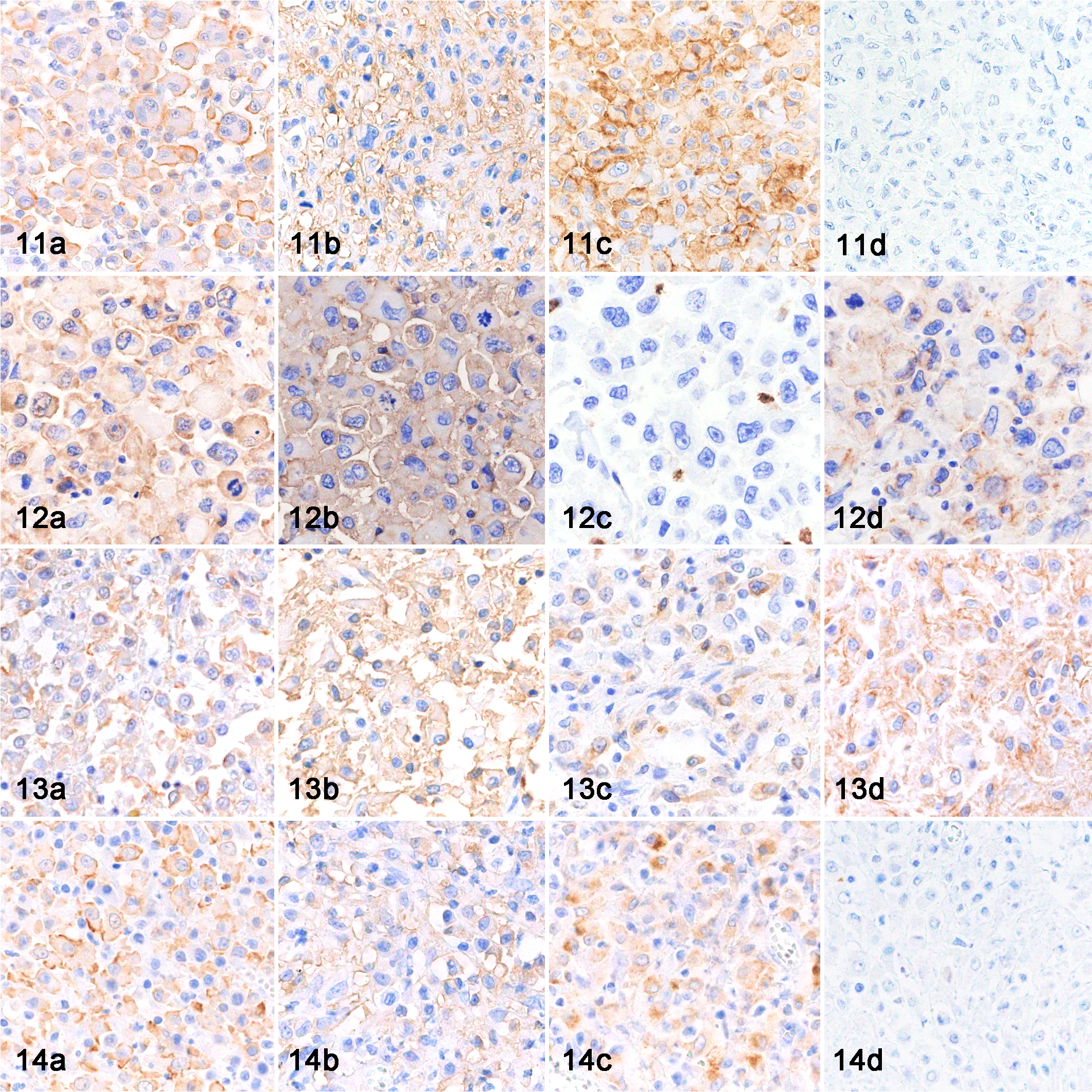
Histiocytic proliferative diseases, cat. Immunohistochemistry (IHC).
Transmission Electron Microscopy
Neoplastic cells in E-cadherin-positive HS (case 8) had relatively smooth cell membranes. Neoplastic cells had an oval to oblong nucleus, and the nuclear membrane was mildly irregular and partially depressed. The nucleus contained coarse nuclear chromatin and a distinct nucleolus (Fig. 15a). Neoplastic cells contained many vesicular structures in the cytoplasm, and tennis racket-shaped structures were observed in some cells. However, a central straight line, similar to Birbeck’s granules of LCs, was not observed in the rod-shaped part (Fig. 15b).
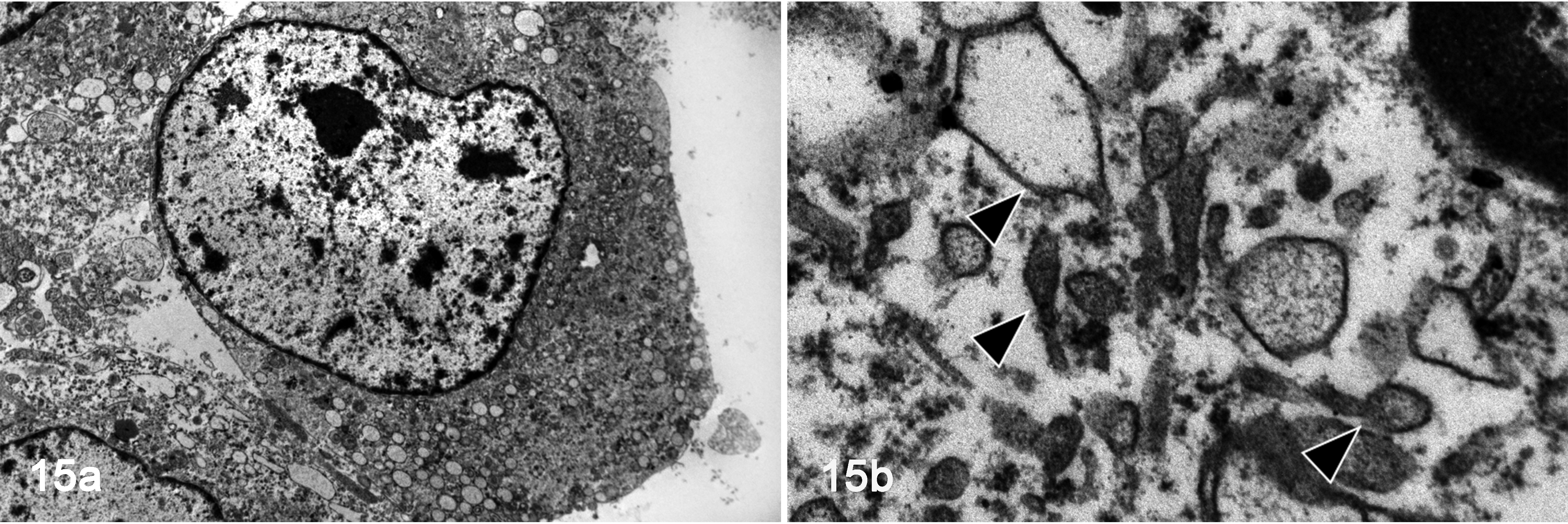
E-cadherin-positive histiocytic sarcoma, lymph node, cat, case 8. Transmission electron microscopy. (a) The neoplastic cell has an oval nucleus with coarse nuclear chromatin and a distinct nucleolus. (b) The cell contains many vesicles in the cytoplasm. There are tennis racket-shaped structures in the cytoplasm.
Characterization of the Established FPH Cell Line (AS-FPH01)
AS-FPH01 cells had adhesive properties to the collagen-coated cell culture vessel and were round with an abundant cytoplasm (Fig. 16). Their nuclei were round with prominent single or multiple nucleoli. Anisocytosis and anisokaryosis were moderate. Bizarre binucleated and multinucleated giant cells were occasionally present.
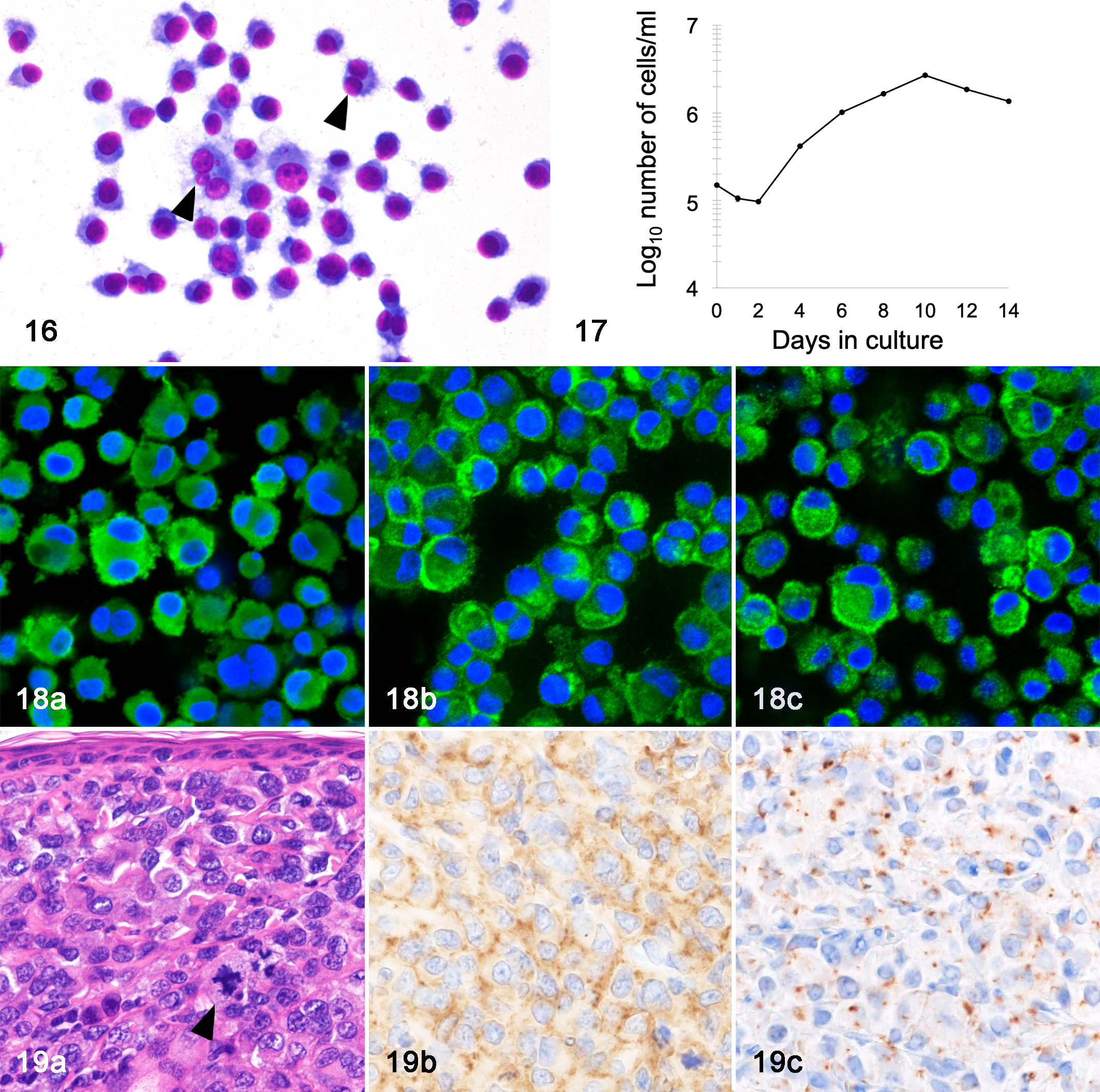
AS-FPH01 cell line, cat, case 2. Cultured cells are round with abundant cytoplasm. Multinucleated cells are occasionally found (arrowheads). Giemsa stain.
Karyotyping revealed that the number of chromosomes of AS-FPH01 ranged between 19 and 142 (mean 62.4, median 67). The population-doubling time of AS-FPH01 was 57.6 hours (Fig. 17). AS-FPH01 cells were maintained for more than 60 passages.
AS-FPH01 cells were immunopositive for Iba-1 (Fig. 18a), HLA-DR, CD204 (Fig. 18b), CD163, E-cadherin (Fig. 18c), CD1a, iNOS, and lysozyme, and were immunonegative for MAC387 and CD208. E-cadherin positivity was detected not only in the cell membrane, but also in the cytoplasm. Primary cultured cells and AS-FPH01 cells showed the same immunophenotype.
Xenotransplantation to SCID Mice
Six weeks after the cell inoculation, a dome-shaped subcutaneous mass was observed in 4 out of 5 SCID mice SC injected with AS-FPH01 cells. These masses were located in the subcutis and dermis, and composed of polygonal neoplastic cells. Neoplastic cells had a moderate amount of amphophilic cytoplasm and an oval to oblong nucleus with a thick, irregular nuclear membrane, coarse nuclear chromatin, and single or multiple nucleoli. Multinucleated cells and mitotic figures were often observed. In 2 out of 4 SCID mice, neoplastic cells exhibited epitheliotropism (Fig. 19a). In 2 out of 4 SCID mice, metastatic lesions were detected in intraperitoneal adipose tissue, pancreas, and ovary. Transplanted AS-FPH01 cells were positive for Iba-1, HLA-DR, CD204 (Fig. 19b), CD163, and E-cadherin (Fig. 19c), and negative for CD208 and MAC387 by immunohistochemistry. E-cadherin positivity was detected focally in the cytoplasm and weakly in the membranes of neoplastic cells.
Neoplastic lesions were grossly and histopathologically detected in the skeletal muscle and adrenal glands of 1 out of 6 mice intravenously injected with AS-FPH01 cells. The morphological and immunohistochemical features of neoplastic cells in these lesions were consistent with those in the transplanted tumors of SCID mice with the SC injection.
Western Blot Analysis
In the western blot analysis using the anti-CD204 antibody, a band at approximately 72 to 77 kDa was detected in protein extracts from feline skin, feline lymph node, original tumor tissue (case 2), AS-FPH01 cells, and AS-FPH01 cell line-derived xenograft (CDX) tissue (Fig. 20a). The expression levels of CD204 were higher in the lymph node, original tumor tissue (case 2), and AS-FPH01 CDX tissue than in skin and AS-FPH01 cells. Regarding the anti-E-cadherin antibody, a band was detected at approximately 120 kDa in protein extracts from feline skin and lymph node, but not from original tumor tissue, AS-FPH01 cells, or AS-FPH01 CDX tissue. A band at approximately 45 to 50 kDa was instead detected from AS-FPH01 cells and AS-FPH01 CDX tissue (Fig. 20b). In the western blot analysis using the anti-HLA-DR antibody, a band at approximately 34 kDa was detected in protein extracts from feline and canine lymph nodes, and the band was consistent with the molecular weights of HLA-DR (HLA-DRα: 36 kDa; HLA-DRβ: 27 kDa in human; Suppl. Fig. 5). In the western blot analysis using the anti-CD208 antibody, bands at approximately 40 kDa and 50 kDa were detected in protein extracts from feline lymph node, and bands at approximately 40 kDa and 60 kDa were detected in protein extracts from canine lymph node. These bands were comparable to the molecular weights of CD208 (44 kDa, glycosylation: 90 kDa in human; Suppl. Fig. S5).
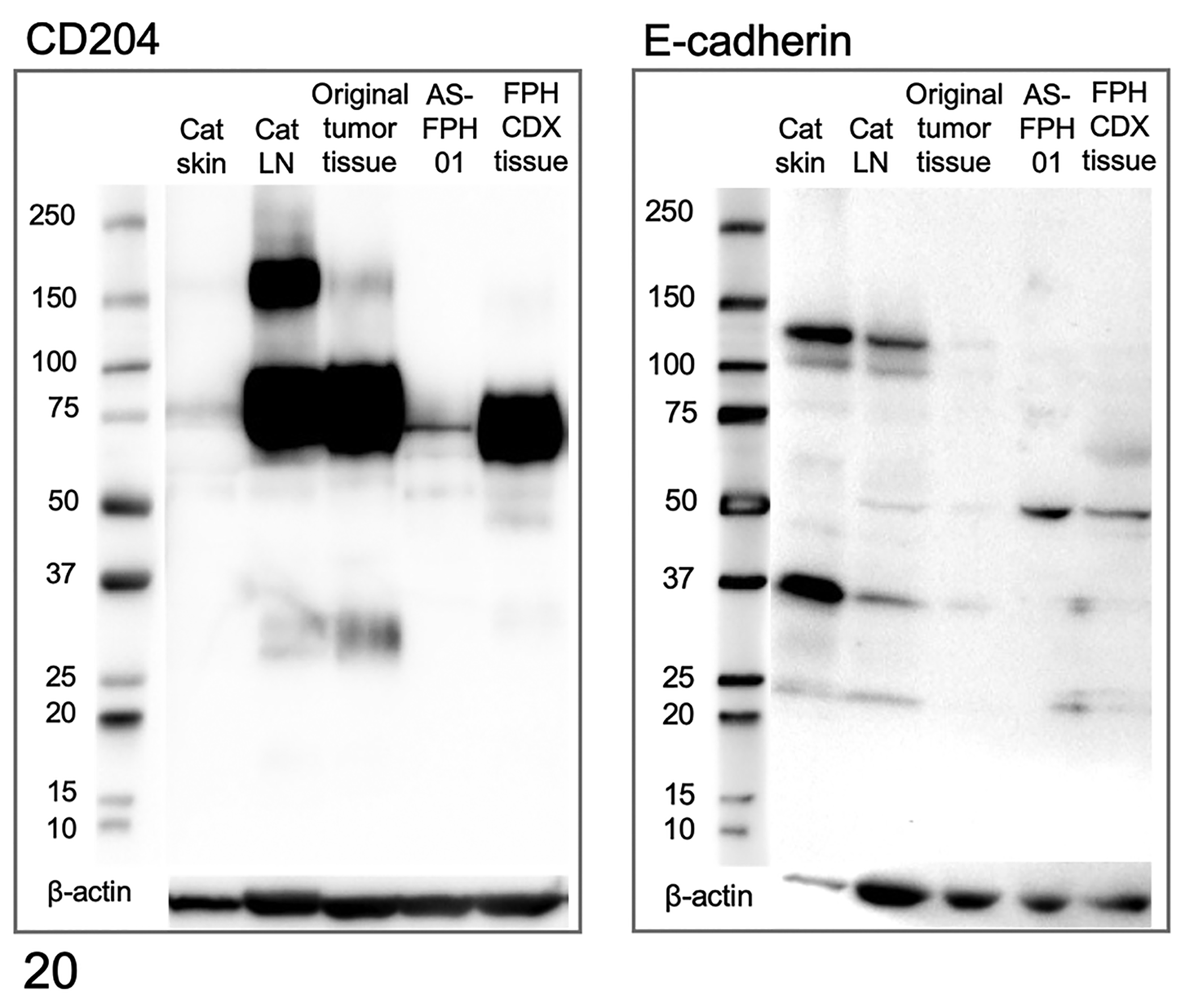
Western blots for CD204 and E-cadherin. In the Western blot for CD204 (left), a band is detected at approximately 72 to 77 kDa from feline skin, feline lymph node, original tumor tissue (case 2), AS-FPH01 cells, and AS-FPH01 cell line-derived xenograft (CDX) tissue. In the Western blot for E-cadherin (right), a band is detected at approximately 120 kDa from feline skin and lymph node, but not in original tumor tissue (case 2), AS-FPH01 cells, or AS-FPH01 CDX tissue. A band is detected at approximately 45 to 50 kDa from the feline lymph node, AS-FPH01 cells, and AS-FPH01 CDX tissue. Beta-actin is used as a loading control.
Discussion
Feline HS and FPH were diagnosed according to previously described histological features. 28,29 Neoplastic cells of HS were round or spindle-shaped, exhibiting significant cellular pleomorphism, whereas neoplastic cells of FPH were uniformly round. Nuclear atypia of neoplastic cells, such as anisokaryosis and nuclear membrane irregularities, was more severe in HS than in FPH. In FPH cases, multiple lesions developed in the limb, trunk, and/or the head of middle-aged to elderly cats, which is consistent with previous findings. 1,5,34 Regarding feline HS, there have only been a few case reports. 17,33,35,36,46 The present study is the first to describe the clinical and pathological features of feline HS in a series of cases. The age of cats with HS ranged widely between 10 months to 18 years. The anatomical locations of feline HS lesions varied, most commonly affecting the lymph nodes, subcutis, and spleen, similar to canine HS. Of note, the median survival time of HS (150 days) was shorter than that of FPH (470 days). These results indicate that the prognosis of HS is poor in cats, dogs, and humans. 37,38,41
Nonneoplastic histiocytes in cats were immunopositive for Iba-1, HLA-DR, E-cadherin, CD204, CD163, CD208, and MAC387 with various combinations (Table 4). Although these antibodies have been used to study feline histiocytic diseases, the immunohistochemical characterization of nonneoplastic histiocytes had not been performed, except for CD163 and CD204. 43,47 Nonneoplastic histiocytes were mostly positive for Iba-1 and HLA-DR, while the immunoreactivity of other antibodies varied among histiocytes that resided in different organs. Dermal iDCs and macrophages were positive for CD204 and negative for E-cadherin. Epidermal LCs were negative for CD204 and positive for E-cadherin. However, antibodies for CD1a, langerin/CD207, and S100, which are regarded as specific markers of human LCs, were not available for detecting LCs on formalin-fixed, paraffin-embedded tissue sections of the cat, as well as those of the dog. 29,39 Double-labeling immunohistochemistry of the lymph nodes revealed E-cadherin-positive CD204-negative histiocytes and E-cadherin and CD204-double positive histiocytes in the sinus, which may be veiled cells and LC-like cells, respectively. Veiled cells are LCs that have migrated to the lymph nodes for antigen presentation. 7,18,40 LC-like cells that co-express LC markers and iDC/macrophage markers have been reported in human and mouse lymph nodes and skin, and are considered to be derived from bone marrow DC precursors or monocytes. 11,14,21,24,26,27,31
Neoplastic histiocytic cells were immunohistochemically positive for Iba-1 and HLA-DR in all cases of FPH and HS (Table 4). Immunoreactivity for CD204, CD163, and E-cadherin varied among cases. Of the 25 HS cases, the neoplastic cells of 18 cases presented with the iDC/macrophage immunophenotype (CD204+/E-cadherin−), 2 with the LC immunophenotype (CD204−/E-cadherin+), and 5 with the LC-like cell immunophenotype (CD204+/E-cadherin+). Feline HS with the LC immunophenotype may correspond to LCH or LC sarcoma (LCS) in humans. 9,30,44 The lesions of LCH and LCS commonly develop in the skin, lymph nodes, bones, and lungs, whereas those in cats (cases 7 and 8) were located in the lymph nodes, tongue, and spleen. Moreover, Birbeck’s granules, an intracytoplasmic structure that is characteristic of LC, was not observed in HS with the LC immunophenotype by electron microscopy (case 8). Further studies with a larger number of cases and additional markers for LC are needed to clarify whether E-cadherin-positive HS in cats are actually LC tumors.
Regarding HS with the LC-like cell immunophenotype (CD204+/E-cadherin+), most lesions were located in the lymph nodes, in accordance with the distribution of LC-like cells in normal tissue. Histiocytic tumors co-expressing LC markers and iDC/macrophage markers have been reported in humans, in whom lesions were mostly located in bone and the lymph nodes. 8,12,13 Cases of HS with the LC-like cell immunophenotype (CD204+/E-cadherin+) had a shorter median survival time (114 days) than those of iDC/macrophage immunophenotype of HS (CD204+/E-cadherin−, 183 days) and LC immunophenotype of HS (CD204−/E-cadherin+, 208 days). However, the different treatments performed may have influenced the survival time of the cats examined in this study.
Among 6 FPH cases, the neoplastic cells of 4 cases presented with the iDC/macrophage immunophenotype (CD204+/E-cadherin−) and 2 with the LC immunophenotype (CD204−/E-cadherin+). In previous studies on FPH, 3/25, 4/5, and 24/26 cases were immunohistochemically positive for E-cadherin; however, CD204 was not examined. 1,5,34 Moreover, the epitheliotropism of neoplastic cells was reported in some E-cadherin-positive cases. Therefore, LC has been suggested as the origin of FPH. 5
In the present study, the E-cadherin expression of FPH was further examined in vitro and in vivo using a novel cell line (AS-FPH01) and its xenotransplant mouse model of FPH. AS-FPH01 was established from an E-cadherin-negative FPH tissue (case 2). Immunocytofluorescence on AS-FPH01 cells revealed the expression of various histiocytic markers (Iba-1, HLA-DR, CD204, and CD163), LC markers (E-cadherin and CD1a), and macrophage markers (iNOS and lysozyme). The xenotransplantation of AS-FPH01 cells into immunodeficient mice resulted in the successful development of a tumor mass in the injection site and metastatic lesions in visceral organs, replicating the pathology of FPH.
Since the expression pattern of E-cadherin differed between the original tumor tissue (E-cadherin-negative) and AS-FPH01 cells (E-cadherin-positive, in vitro and in vivo), this phenomenon was further confirmed by the western blot analysis. Consistent with the results of immunohistochemistry, the western blot analysis revealed that the original tumor had no immunoreactive band for E-cadherin (120 kDa). Protein extracts from AS-FPH01 cells and xenograft tumor had an immunopositive band at approximately 45 to 50 kDa, instead of the 120-kDa band of the full-length E-cadherin protein. E-cadherin is a transmembrane protein that is composed of an extracellular domain and cytoplasmic domain. The extracellular domain mediates cell-cell adhesion, whereas the cytoplasmic domain plays a role in signal transduction together with p120-catenin and β-catenin. 32 The E-cadherin protein is cleaved by various proteases, such as extracellular metalloproteases, γ-secretase, and kallikrein, producing fragmented E-cadherin peptides in the cytoplasm. 10,15,20,22,32 The loss of membrane E-cadherin expression and production of fragmented E-cadherin have been reported in human tumors, such as prostate cancer, pancreatic cancer, and squamous cell carcinoma. 3,6,20,22,45 Therefore, the E-cadherin-positive 45 to 50 kDa band may correspond to fragmented E-cadherin. Furthermore, the cytoplasmic immunostaining of E-cadherin in AS-FPH01 may represent fragmented E-cadherin. Further molecular studies are needed to verify whether the 45 to 50 kDa band is actually fragmented E-cadherin protein.
The absence of membrane E-cadherin expression is associated with the loss of cell-cell adhesion, and also with the invasive and metastatic abilities of tumor cells. 22,25,26,45 In a previously reported case of feline pulmonary LCH, E-cadherin expression by neoplastic cells was decreased in extrapulmonary metastatic lesions. 4 In one of the HS cases in the present study (case 12), the E-cadherin immunoreactivity of neoplastic cells varied in different organs. These results suggest that the expression of E-cadherin by feline neoplastic histiocytes was altered by their microenvironments.
In conclusion, immunohistochemical markers for different types of feline histiocytes were demonstrated in the present study. The neoplastic cells of HS and FPH in the cat were variably positive for iDC/macrophage and LC markers, which may indicate different phenotypes of these neoplasms. The novel cell line AS-FPH01 established in the present study may serve as an in vitro and in vivo model of FPH that will enable further studies of the disease.
Supplemental Material
Combined_supplemental_materials-Hirabayashi_et_al - Immunophenotyping of Nonneoplastic and Neoplastic Histiocytes in Cats and Characterization of a Novel Cell Line Derived From Feline Progressive Histiocytosis
Combined_supplemental_materials-Hirabayashi_et_al for Immunophenotyping of Nonneoplastic and Neoplastic Histiocytes in Cats and Characterization of a Novel Cell Line Derived From Feline Progressive Histiocytosis by Miyuki Hirabayashi, James K. Chambers, Ayumi Sumi, Kei Harada, Makoto Haritani, Tetsuo Omachi, Tetsuya Kobayashi, Hiroyuki Nakayama and Kazuyuki Uchida in Veterinary Pathology
Footnotes
Acknowledgements
We would like to thank the Japan Animal Referral Medical Center, NORTH LaB, and Small Animal Pathology Service for contributing samples.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
