Abstract
Histiocytic proliferative disorders are commonly observed in dogs and less often cats. Histiocytic disorders occur in most of the dendritic cell (DC) lineages. Canine cutaneous histiocytoma originates from Langerhans cells (LCs) indicated by expression of CD1a, CD11c/CD18, and E-cadherin. When histiocytomas occur as multiple lesions in skin with optional metastasis to lymph nodes and internal organs, the disease resembles cutaneous Langerhans cell histiocytosis of humans. Langerhans cell disorders do not occur in feline skin. Feline pulmonary LCH has been recognized as a cause of respiratory failure due to diffuse pulmonary infiltration by histiocytes, which express CD18 and E-cadherin and contain Birbeck’s granules. In dogs and cats, histiocytic sarcomas (HS) arise from interstitial DCs that occur in most tissues of the body. Histiocytic sarcomas begin as localized lesions, which rapidly disseminate to many organs. Primary sites include spleen, lung, skin, brain (meninges), lymph node, bone marrow, and synovial tissues of limbs. An indolent form of localized HS, progressive histiocytosis, originates in the skin of cats. Hemophagocytic HS originates in splenic red pulp and bone marrow macrophages in dogs and cats. In dogs, histiocytes in hemophagocytic HS express CD11d/CD18, which is a leuko-integrin highly expressed by macrophages in splenic red pulp and bone marrow. Canine reactive histiocytic diseases, systemic histiocytosis (SH) and cutaneous histiocytosis, are complex inflammatory diseases with underlying immune dysregulation. The lesions are dominated by activated interstitial DCs and lymphocytes, which invade vessel walls and extend as vasocentric infiltrates in skin, lymph nodes, and internal organs (SH).
Keywords
Histiocytic Lineages: Development and Identification
The cellular spectrum covered by the term histiocyte has evolved over time. Histiocyte is now considered an overarching term to describe cells of dendritic cell (DC) or macrophage lineage. 13,22
Histiocytes differentiate from CD34+ stem cell precursors into macrophages and several DC lineages, which encompass Langerhans cells (LCs) and interstitial DCs. Langerhans cells occur within epithelia of the skin and alimentary, respiratory, and reproductive tracts. 45 Interstitial DCs occur in perivascular locations in many organs except the brain, although they do occur in the meninges and choroid plexus. 19 Among the most studied DCs are those that occur in skin; these include epidermal LCs and dermal DCs, which are interstitial DCs. Dendritic cells that occur in T-cell domains in peripheral lymphoid organs (lymph node and spleen) are known as interdigitating DCs. Interdigitating DCs in lymph nodes comprise resident DCs and migratory DCs. The migratory DCs arrive in lymphatics from tissues and consist of LCs and interstitial DCs. 76 Most canine and feline histiocytic diseases involve proliferations of cells of LC or interstitial DC lineage; hemophagocytic histiocytic sarcoma (HS) is currently the only histiocytic disease of dogs and cats that has been shown to originate in macrophages. 3–5,11,24,51,54
Cytokines and growth factors that influence DC development include fms-like tyrosine kinase (FLT3) ligand, granulocyte-macrophage colony-stimulating factor (GM-CSF), stem cell factor, tumor necrosis factor alpha (TNF-α), interleukin 4 (IL-4), and transforming growth factor beta 1 (TGF-β1). 37,76 Macrophage development from CD34+ precursors is influenced by GM-CSF and macrophage colony-stimulating factor (M-CSF). Blood monocytes can differentiate into either macrophages under the influence of M-CSF or into DCs under the influence of GM-CSF and IL-4. 74–76 Langerhans cell differentiation is critically dependent on TGF-β1 stimulation. 37,45,65 Epidermal keratinocytes constitutively secrete TGF-β1, which promotes the differentiation of LCs from dermal CD14+ precursor cells. These precursor cells upregulate C-C chemokine receptor 6 (CCR6) and migrate into the epidermis to become immature epidermal-resident LCs in response to macrophage inflammatory protein 3 alpha (MIP-3α or CCL20), which is produced there. Culture of LCs in the presence of GM-CSF and IL-4 results in further differentiation of LCs into functionally mature DCs with enhanced potential to present antigen to T cells with appropriate costimulation. These latter conditions are present in states of cutaneous inflammation. 37
Dendritic cells are the most potent antigen-presenting cells (APCs) for induction of immune responses in naive T cells. The development of canine- and feline-specific monoclonal antibodies for functionally important molecules of DCs and macrophages has enabled their identification in canine and feline tissues, especially skin. 47,64,69,86 Dendritic cells occur in 2 major locations: within the epidermis (LCs) and within the dermis, especially adjacent to postcapillary venules (dermal interstitial DCs). 89 Canine and feline DCs express CD1a molecules, which, together with major histocompatibility complex (MHC) class I and class II molecules, are responsible for presentation of peptides, lipids, and glycolipids to T cells. 39,47,54,69,86 Hence, DCs are best defined by their abundant expression of molecules essential to their function as APCs. Of these, the family of CD1 proteins is largely restricted in expression to DCs in skin, while MHC classes I and II are more broadly expressed.
The β-2 integrins (CD11/CD18) are critically important adhesion molecules, which are differentially expressed by all leukocytes. β-2 Integrins are heterodimeric molecules consisting of 1 of 4 α subunits (CD11a-d) and a single β subunit (CD18 or β-2). The major hypothesis underlying the development of monoclonal antibodies specific for canine CD11/CD18 was that β-2 integrins would be differentially expressed in canine histiocytic diseases. Canine-specific monoclonal antibodies for the known human β-2 integrin α subunits CD11a (αL), CD11b (αM), and CD11c (αX) were produced. 20 Also, a monoclonal antibody specific for a novel canine β-2 integrin α subunit was discovered. This antibody was then used to characterize the canine protein, which led directly to the discovery of the human analogue designated αD and by inference CD11d. 21,81 Subsequently, CD11d expression in canine histiocytic disease was associated with macrophage differentiation in hemophagocytic histiocytic sarcoma, which differed markedly from histiocytic sarcomas of interstitial DC origin; these expressed CD11c. 5,51 These results support the original hypothesis.
CD11/CD18 expression is highly regulated in normal canine macrophages and DCs. CD11c is expressed by LCs and interstitial DCs, while macrophages predominately express CD11b (or CD11d in hematopoietic environments such as the splenic red pulp and bone marrow). A subset of dermal interstitial DCs also expresses CD11b. 20,21,64 In diseased tissues, these β-2 integrin expression patterns may be broadened. β-2 Integrins are detectable in feline tissues. However, monoclonal antibodies specific for feline CD11a and CD11c are not available. Feline CD11b and CD11d are detectable with cross-reactive anti–canine monoclonal antibodies. Canine and feline CD18 are detectable by species-specific monoclonal antibodies that were developed for use in formalin-fixed tissue. These monoclonal antibodies have greatly facilitated the investigation of histiocytic diseases in dogs and cats. 3–5,11,18,24,47,51,54
Langerhans cells and dermal interstitial DCs are also distinguishable by their differential expression of E-cadherin (LCs+) and Thy-1 (CD90) (dermal interstitial DCs+). Langerhans cells localize within epithelia via E-cadherin homotypic adhesion with E-cadherin expressed by epithelial cells. 37 Langerhans cells, in the quiescent conditions that prevail in normal skin, are subject to slow turnover and are maintained by self-renewal or renewal from a dedicated dermal precursor cell. 37,45 Dermal interstitial DCs are subject to more rapid turnover and are repopulated by a blood-borne precursor (perhaps the monocyte) and by local self-renewal. 45,89
Migration of DCs (as veiled cells) beyond the skin to the paracortex of lymph nodes, where they join forces with interdigitating DCs, occurs in the normal steady state and is enhanced in inflammatory states. 45,89 Successful interaction of DCs and T cells in response to antigenic challenge also involves the orderly appearance of costimulatory molecules (B7 family—CD80 and CD86) on DCs and their ligands—CD28 and cytotoxic T-lymphocyte antigen 4 (CTLA-4) on T cells. 9,70 In situ DCs have low expression of MHC-II and costimulatory molecules and are more receptive to antigen uptake. Migratory DCs upregulate MHC class II and B7 family members and become more adept at antigen presentation to T cells. 9
Aspects of the developmental and migratory program of DCs are recapitulated in canine and feline histiocytic diseases. Defective interaction of DCs and T cells may contribute to the development of canine reactive histiocytoses—cutaneous histiocytosis (CH) and systemic histiocytosis (SH), which are related interstitial DC disorders that likely occur in the context of disordered immune regulation. The distant migratory potential of DCs is of immense clinical significance in the adverse prognosis of histiocytic sarcomas, which largely originate in interstitial DCs and rapidly disseminate via metastasis.
An Overview of Canine and Feline Histiocytic Diseases
Several well-defined histiocytic proliferative diseases have been recognized in dogs (Table 1). Canine cutaneous histiocytoma, which exhibits evidence of LC differentiation, usually occurs as a solitary lesion in young dogs and spontaneously regresses. Cutaneous Langerhans cell histiocytosis (LCH) covers a spectrum of disease, in which multiple cutaneous lesions are consistently observed. In some cases, lymph node and systemic metastasis is also observed. Individual skin lesions otherwise resemble those in histiocytoma. Hence, these 2 diseases could be considered part of a larger spectrum of disease with diverse biological behavior. Feline counterparts of canine cutaneous histiocytoma and cutaneous LCH have not been reported. 54
Histiocytic Diseases of Dogs and Cats.
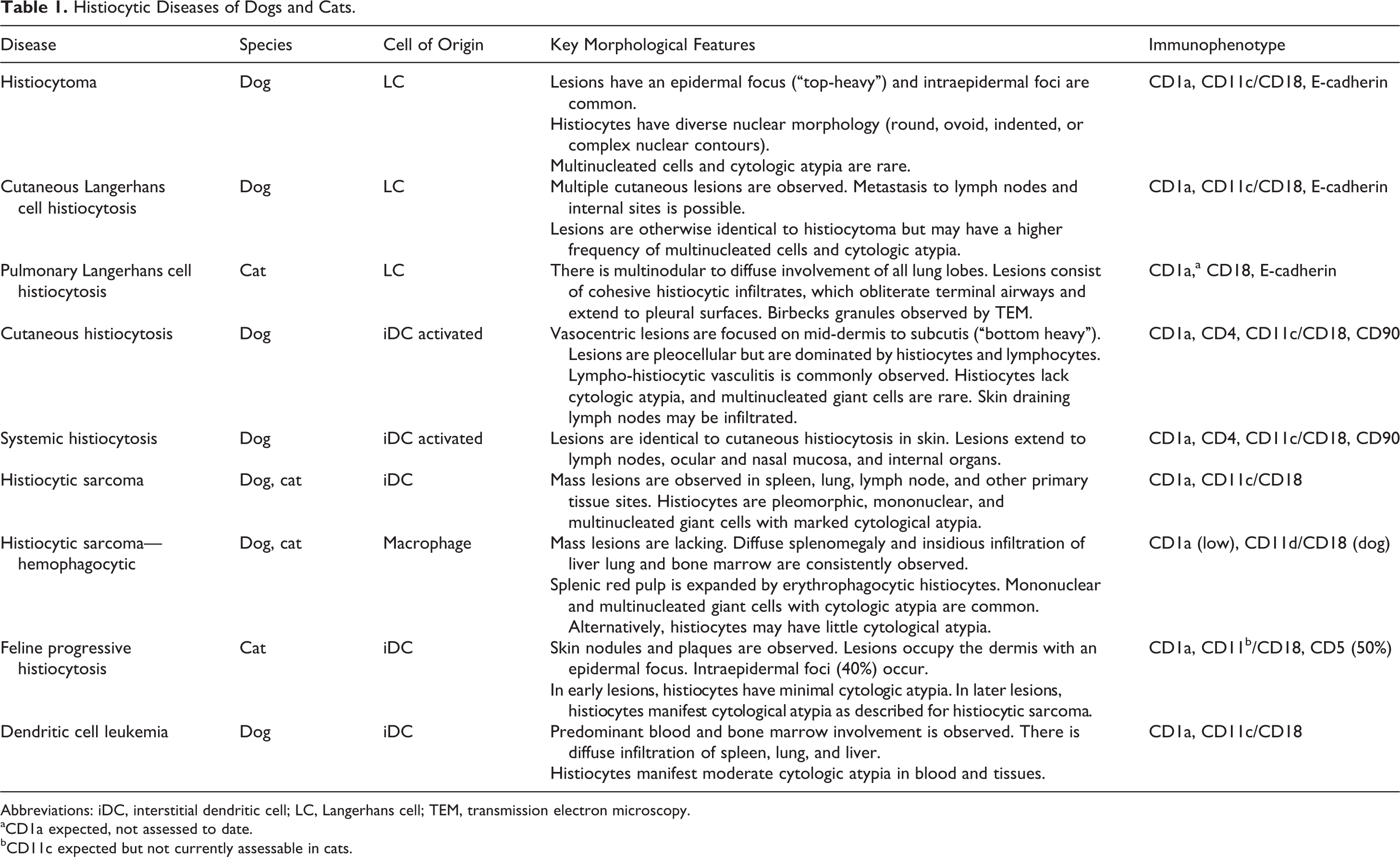
Abbreviations: iDC, interstitial dendritic cell; LC, Langerhans cell; TEM, transmission electron microscopy.
aCD1a expected, not assessed to date.
bCD11c expected but not currently assessable in cats.
The reactive histiocytoses have only been identified in dogs; there is no known feline counterpart. The reactive histiocytoses (CH and SH) are related disorders arising from activated interstitial DCs. Reactive histiocytoses are histiocyte- and lymphocyte-rich inflammatory disorders associated with a degree of immune dysregulation, which promotes lesion persistence. 3,12,49
The HS complex HS occurs in dogs and cats. The incidence of HS in cats is much lower than in dogs. Histiocytic sarcomas occur as localized lesions in spleen, lymph nodes, lung, bone marrow, skin and subcutis, brain, and articular tissue of appendicular joints. Histiocytic sarcomas may occur as multiple lesions in single organs (especially spleen and skin) and rapidly disseminate to involve multiple organs. This form of the disease was formerly called malignant histiocytosis (MH) but is now more appropriately termed disseminated HS. 22 Histiocytic sarcomas mostly arise from interstitial DCs. However, the hemophagocytic form of HS is a distinctive clinic-pathologic entity, which arises from splenic red pulp (± bone marrow) macrophages. 5,51
Two feline histiocytic diseases do not have a canine equivalent: feline progressive histiocytosis (FPH) and pulmonary LCH. Feline progressive histiocytosis is the most common histiocytic disease in cats and invariably begins as cutaneous nodules and plaques. Feline progressive histiocytosis is essentially a low-grade HS with indolent behavior initially. 4 Pulmonary LCH is a disease of aged cats (10–15 years), which causes progressive respiratory failure due to extensive obliteration of the pulmonary parenchyma by an infiltrate dominated by LCs. 11
Histiocytic Diseases of Langerhans Cell Origin
Langerhans cells are resident DCs within the epithelium of skin and mucosal sites (tongue, oropharynx, pulmonary bronchi, vagina, and ectocervix). 45 The C-type lectin, langerin (CD207), is expressed on the surface of LCs. Internalization of langerin mediates the formation of Birbeck’s granules (BGs), which are cytoplasmic structures that define LCs and distinguish them from interstitial DCs. Birbeck’s granules have been found in the LCs of mice, humans, pigs, cats, and many others. 45,58,69 All of these species express a functional langerin molecule. The lone exception to the above is the dog. BGs have not been observed in canine epidermal DCs, which are presumed to be LCs. 52 However, there is a conflicting report of BGs in canine epidermal LCs, in which the structures illustrated lack distinctive features of BGs. 42 Birbeck’s granules are present in LCs of cats and were demonstrated in the infiltrating histiocytes in feline pulmonary LCH. 11,69 The C-type carbohydrate recognition domain (CRD) of langerin has conserved amino acid residues that create the principal calcium binding site, which is necessary for mannose binding. Variations in the amino acid sequence in the CRD alter its conformation and negatively affect mannose binding. 84 In fact, a rare mutation in human langerin (W264R) abolishes mannose binding and the formation of BGs. 83,84 Canine langerin has a 4–amino acid deletion located between 2 key glutamic acid residues involved in calcium binding. The functional impact of this deletion is unknown, but it is likely to negatively affect mannose binding (Maureen Taylor, Imperial College, London, personal communication, 2008). Hence, the lack of BGs in canine LCs and in canine LC disorders may be traceable to a nonfunctional langerin molecule. This hypothesis awaits experimental verification. Another unresolved issue with canine langerin, which could have important functional ramifications, is that the predicted gene structure using Gnomon (National Center for Biotechnology Information [NCBI]) lacks exon 3 (neck region), which is required for oligomerization and mannose binding. 58,66 The exon-intron structure of canine langerin needs experimental verification using langerin transcripts from isolated canine LCs. Importantly, both predictions cast a shadow over the functionality of canine langerin; if either were true, BG formation would be abolished.
Human Langerhans Cell Histiocytosis
Langerhans cell proliferative disorders (such as LCH) have been best defined in humans. Human LCH covers a spectrum of disease ranging from localized to rapidly progressive systemic disease. 22 Localized disease is most commonly observed in bone (bone marrow), and multiple sites may be affected. Localized LCH has been described in the lungs of smokers and usually resolves with cessation of smoking. Localized LCH may also affect skin, but skin is more commonly affected as part of systemic disease. In this latter instance, LCH of skin may occur before systemic disease is present. Other sites involved in multisystem LCH include lymph nodes, spleen, liver, lung, and bone marrow. Involvement of the central nervous system in LCH, while described, is relatively uncommon. 22,31,71
Diagnosis of LCH in humans relies on demonstration of BGs, which are the ultrastructural hallmark of LCs. 22 Furthermore, expression of CD1a and langerin by atypical cells in LCH are important indicators of an LC immunophenotype. 22,31
Canine Cutaneous Histiocytoma
Histiocytomas are common, largely benign, cutaneous neoplasms of dogs. The age-specific incidence rate for histiocytomas drops precipitously after 3 years, although histiocytomas do occur in dogs of all ages. 79 Histiocytomas occur in all breeds; purebred dogs are predisposed, particularly Boxers and Dachshunds. 79 Lesions may occur in any region of the body, but the head is a frequent site, particularly the pinna. 79
Histiocytomas usually occur as solitary lesions, which usually undergo spontaneous regression. 55,79 Multiple histiocytomas (ie, cutaneous LCH) occur in <1% of cases and are more common in Shar Peis. 2,79 Recurrence rate for histiocytomas at the surgical excision site are extremely low, and development of a histiocytoma at a new site occurs at extremely low incidence. 79 Metastasis of a solitary histiocytoma to local lymph nodes has been reported. Regression of this lesion occurred within a few weeks. 54 Similarly, follow-up of an additional 4 dogs with solitary metastatic histiocytoma revealed spontaneous regression in 3 dogs within a 1-to 2-month time frame; the other dog was euthanized at the time of diagnosis (P. F. Moore, unpublished data).
Histiocytomas are progressively infiltrated by lymphocytes after a variable time course. 14 These lymphocytes likely mediate lysis of neoplastic histiocytes, since it has been shown that the tumor-infiltrating lymphocytes are highly enriched for CD8+ T cells, which are cytotoxic cells capable of mediating regression. 54 Mononuclear cells isolated from canine histiocytomas have been shown to have potent allo-stimulatory properties in a mixed leukocyte reaction (MLR). 7 These authors postulate that tumor histiocytes could potentially mediate their own destruction if they functioned as antigen-presenting cells. However, it is not possible to exclude a contribution of tumor-infiltrating/resident interstitial DCs in the allogeneic MLR. It is more likely that migration of these normal DCs (rather than tumor histiocytes) to lymph nodes would result in antigen presentation to naive T cells and subsequent recruitment of effector/memory T cells into the cutaneous lesion. 36 Regardless, the consistent spontaneous regression of cutaneous histiocytomas is a poorly understood, fascinating biological phenomenon.
Cutaneous histiocytomas originate in histiocytes that manifest LC differentiation. This is based on immunophenotypic and ultrastructural features. 8,27,33,40,41,54,62 As discussed previously, BGs have not been conclusively illustrated in normal canine LCs or in cutaneous histiocytoma. 27,33,40–42,52 Histiocytomas do exhibit ultrastructural features characteristic of DCs, and they also express CD1a and E-cadherin, which are important functional markers expressed by LCs. 8,40,41,54,62 Histiocytomas arise in the dermis and may invade the epidermis in a subset of lesions. Hence, histiocytomas do not arise directly from intraepidermal LCs. It is quite possible that they arise from dermal precursors of LCs similar to the cells described by Larregina and others. 37 These CD14+ dermal cells downregulate CD14 and express CD1a, E-cadherin, and langerin following culture with TGF-β1. They also express CCR6 and are responsive to MIP-3α (CCL20), which is a chemokine produced by keratinocytes that facilitates migration of CCR6+ LC into epidermis. CD14+ dermal cells did not possess BGs when first isolated but did develop BGs after culture with TGF-β1. CD14+ LC precursors are found in perivascular locations in superficial and deep dermis. 37 It is not known if an analogous CD14+ LC precursor exists in the dog, but the prospect that cutaneous histiocytoma might originate from a perivascular dermal LC precursor is an attractive hypothesis, which could also explain the stratified expression pattern of E-cadherin within histiocytomas—stronger expression closer to the epidermis, which is a rich source of TGF-β1. 63
Morphologic Features
Histiocytomas are most often dome-shaped, exophytic growths with complete or partial alopecia; they may attain 4 cm in diameter, but most lesions are less than 2 cm in diameter. 79 Morphological descriptions of histiocytomas emphasize the tropism of the tumor infiltrate for the superficial dermis and epidermis to create a “top-heavy” lesion. In fact, tumor histiocytes may invade the epidermis as individual cells or nests of cells (Fig. 1). 54 This can raise concern for epitheliotopic T-cell lymphoma. Immunophenotyping using antibodies specific for CD3 and CD18 is necessary to resolve this dilemma. 54
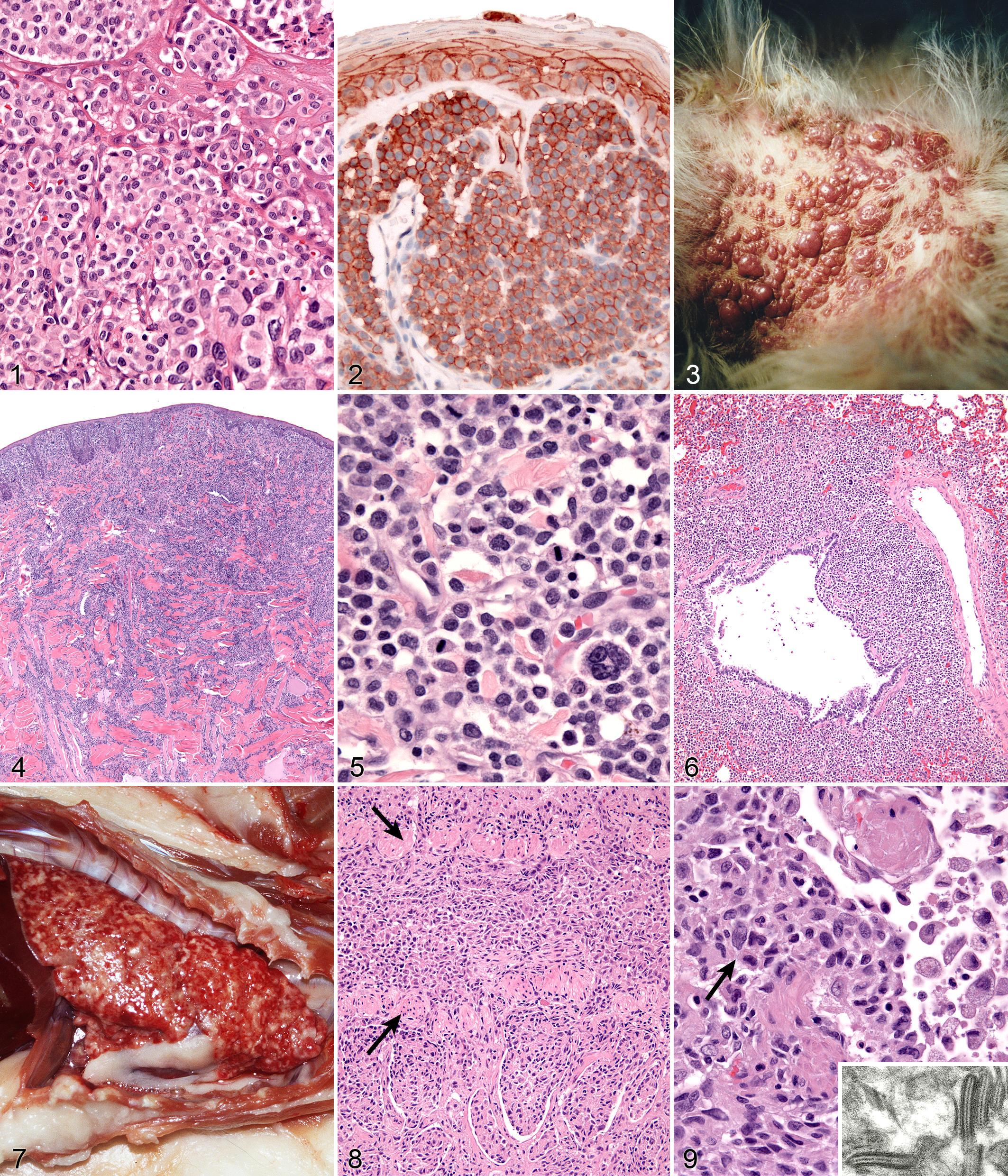
Skin; dog; canine cutaneous histiocytoma. Histiocytes possess round to ovoid, or indented to twisted and folded nuclei (inset). The cytoplasm is moderately abundant and histiocytes form intraepidermal nests. Hematoxylin and eosin (HE).
Tumor histiocytes may display a range of cytological features. Nuclei may be round to oval, indented, or complexly folded (convoluted; Fig. 1 inset). Multinucleated histiocytes may be observed at low frequency. Cytologic atypia, manifest as anisokaryosis and hyperchromatic nuclei, is unusual. The mitotic index in histiocytomas is highly variable but is often high. The cytoplasm is usually abundant and eosinophilic. Invasion of the epidermis by individual histiocytes or nests of histiocytes occurred in >60% of lesions in 1 case study. 54 In a larger context, epidermal invasion is not quite as prevalent as in that report, but it is a diagnostically useful feature and should be carefully evaluated.
Regression of cutaneous histiocytoma is accompanied by a progressive lymphocytic infiltrate, which eventually spreads throughout the lesion. 14 This infiltrate is associated with necrosis of tumor histiocytes in all but the earliest stage. 14 The heavy lymphocytic infiltrate that occurs in late-stage regression of histiocytomas can be mistaken for non-epitheliotropic T-cell lymphoma. There may be very few recognizable tumor histiocytes present amid numerous reactive histiocytes and T cells. These lesions resemble inflamed non-epitheliotropic T-cell lymphoma. 50 Molecular clonality analysis of the T-cell receptor locus (TRG) would be expected to resolve this issue. However, clonal T-cell expansions have been documented in association with the cytotoxic T-cell response to histiocytoma (about 50% incidence) (B. Gericota, V. K. Affolter, and P. F. Moore, unpublished observations).
Immunophenotypic Features
The most comprehensive immunophenotype of canine histiocytomas was established using acetone-fixed sections from snap-frozen tumor tissue. 54 The structure of the canine CD1 locus is now largely known based on the most recent canine genome assembly, and the dog has at least 8 CD1a genes, of which at least 3 are functional. The CD1 antibodies used in the original 1996 study map to various canine CD1a genes. 39 So in reconciling the results of these 2 studies, tumor histiocytes in canine histiocytoma diffusely expressed CD1a, CD11a/CD18, CD11c/CD18, CD44, CD45, and MHC class II. Tumor histiocytes variably expressed CD11b/CD18 and CD54. They did not express CD90 (Thy-1) and CD45RA. 54 Tumor histiocytes express E-cadherin, which is also expressed by normal LCs (Fig. 2). 8,45,62,63 The true incidence of E-cadherin expression in canine histiocytomas is not known, but it is certainly less than 100% observed in the published reports. 8,62,63 Histiocytomas lacking E-cadherin expression occur in our case series (P. F. Moore and V. K. Affolter, unpublished data). This could in part be due to the use of various antibodies to E-cadherin in these studies. It is important to use antibodies that stain the cell membrane where functionally relevant E-cadherin is expressed. Langerhans cells use E-cadherin to localize in the epidermis via homotypic interaction with E-cadherin expressed by keratinocytes. E-cadherin is a lineage-associated marker but not a lineage specific marker for LCs. The staining pattern of E-cadherin in many lesions is not uniform; E-cadherin expression may be limited to the histiocytic infiltrate immediately adjacent to the epidermis. Also, E-cadherin may be expressed by cutaneous round cell tumors of diverse lineage. 63 Immunophenotyping with multiple markers can clearly resolve this issue, and E-cadherin expression remains a valuable indicator of LC differentiation in cutaneous histiocytoma. Furthermore, differential expression of CD90 and E-cadherin is useful in distinguishing proliferative diseases of interstitial DCs (CD90+) and LCs (E-cadherin+). 3,54
Canine Cutaneous Langerhans Cell Histiocytosis
Histiocytomas are almost always solitary lesions. The existence of multiple histiocytomas at the same or distant sites is uncommon. 79 There have been several reports of extensive skin involvement with multiple histiocytomas. 10,25,26,29,54,57 This spectrum of disease best fits under the umbrella of cutaneous LCH, since skin is invariably involved, and even when systemic disease eventuates, skin is the initial site of origin. Cutaneous LCH is well documented in humans as a single-system disease and as a component of systemic disease. 56,71 Dogs may have literally hundreds of cutaneous lesions ranging from nodules to masses, which elevate the epidermis and may be accompanied by redness, alopecia, and ulceration (Fig. 3). Lesions at mucocutaneous junctions and in tissues of the oral cavity may also occur. Lesions may be initially limited to skin or involve skin and draining lymph nodes. Rarely, internal organ involvement also occurs.
Shar Pei dogs were overrepresented in a cohort of dogs afflicted by cutaneous LCH (about 20% of cases), but the disease occurred in many other breeds. Delayed regression of cutaneous LCH was common, and lesions persisted for up to 10 months prior to regression. About 50% of the dogs with cutaneous LCH were euthanized due to complications in management of the extensive ulcerated lesions and failure of timely regression (P. F. Moore and V. K. Affolter, unpublished observations). If cutaneous LCH involved lymph nodes, the prognosis was markedly worse. In all instances, these dogs were euthanized. The clinical course of dogs that experienced systemic spread of cutaneous LCH was even more rapid, and all dogs were euthanized or died. Necropsies were performed on some dogs. Peripheral lymph nodes and lungs were consistently affected, but metastatic disease can be widespread and involve many other organs (P. F. Moore and V. K. Affolter, unpublished observations). This spectrum of disease manifestation and diverse clinical behavior most resembles rapidly progressive multisystem LCH of humans. 56,71 Consistent treatment strategies were not employed in these cases. Cyclosporine A was used in a number of dogs because the lesions were mistaken for cutaneous histiocytosis; no treatment responses were noted. Nitrogen mustard (CCNU) treatment was effective in most instances, but the responses were not durable. Immune modulators (levamisole) were tried without success (P. F. Moore, unpublished observations). In 1 published report, a dog with cutaneous LCH was successfully treated with griseofulvin, which is an immune-modulating antibiotic. This dog died after developing systemic disease 3 months after griseofulvin was withdrawn. 57
Morphologic Features
Individual lesions of cutaneous LCH often do not differ markedly from those of cutaneous histiocytoma. The lesions are often large masses, which span the dermis and extend into the subcutis and muscle. In aggressive cases, the lesions manifest a higher degree of cytologic atypia but maintain tropism for the epidermis (Fig. 4). Histiocytes may have more anisokaryosis and multinucleated cells than is typical for solitary histiocytoma, but these are not consistent features (Fig. 5). Lymphatic invasion by neoplastic histiocytes portends lymph node involvement, which is an adverse prognostic indicator. Systemic lesions are consistently observed in lung, and the infiltrates have a peribronchial distribution (Fig. 6). Widespread infiltration of other organs is possible.
Immunophenotypic Features
Histiocytes in cutaneous LCH share the immunophenotype of histiocytes in histiocytoma.
Feline Pulmonary Langerhans Cell Histiocytosis
Pulmonary LCH is a disease of aged cats (10–15 years), which causes progressive respiratory failure leading to euthanasia. 11 The cats present with severe respiratory distress characterized by tachypnea, increased respiratory effort, or open-mouthed breathing. The symptoms can be acute or present for several months. Thoracic radiographs reveal a diffuse bronchointerstitial pattern of miliary to nodular opacities throughout all lung lobes. The original report described pulmonary LCH in 3 cats. Since then, 4 additional cases have been encountered (P. F. Moore, unpublished data). All have been diagnosed at necropsy. Pulmonary LCH occurs in humans, especially among smokers. It is believed to be a reactive disorder, which often resolves following cessation of smoking. Clonality studies have been associated with mixed results; some lesions harbor clonal expansions of LCs, but most lesions have polyclonal LC proliferation. 85,88 A neoplastic process was favored in feline pulmonary LCH based on the cytological characteristics of the LC infiltrate and the consistent extra-pulmonary lesions. However, molecular clonality studies were not conducted in support of that conclusion. 11
Morphologic Features
The macroscopic lesions are distinctive; an infiltrative process involves all lung lobes. The lungs are diffusely firm and entirely effaced by ill-defined, coalescing nodular masses (2–5 mm maximum dimension) (Fig. 7). These nodules often coalesce, thereby effacing larger areas of parenchyma, which extend from peribronchiolar locations to the pleural surface. Extra-pulmonary spread to pancreas and kidney is variably observed. The tracheobronchial lymph nodes and lymph nodes draining affected abdominal organs are also enlarged and diffusely effaced.
Pulmonary lesions are unique and consist of histiocytic infiltrates within terminal and respiratory bronchioles. The infiltrates partially obliterate the airway walls and fill the lumens. Extension of the infiltrates into adjacent alveolar ducts and alveoli occurs (Fig. 8). Histiocytes form cohesive infiltrates with indistinct cell borders. They are moderately pleomorphic in cell size and nuclear morphology, which is often complex (convoluted, twisted, or folded) (Fig. 9). Transmission electron microscopy was used to demonstrate characteristic BGs in the cytoplasm of lesional histiocytes; these structures are only present in LCs, and their formation is dependent on the C-type lectin, langerin (Fig. 9 inset).
Immunophenotypic Features
The lesional hisiocytes express CD18 and E-cadherin. CD1 expression has not been evaluated in pulmonary LCH, since only formalin-fixed tissues have been available. E-cadherin expression is most often markedly reduced in extra-pulmonary lesions. This may be indicative of active downregulation of E-cadherin expression associated with LC migration; this occurs in normal LCs when they migrate from epithelia to lymph nodes.
Histiocytic Diseases of Interstitial DC and Macrophage Origin
Histiocytic sarcomas are most often derived from cells with the phenotypic profile of interstitial DCs. The reactive histiocytoses (CH and SH) are complex inflammatory disorders composed of lymphocytes and activated interstitial DCs. Interstitial DCs occur in almost all tissues with the exception of the brain. Interstitial DCs do occur in the meninges and choroid plexi. 19 Interstitial DCs are identifiable in tissues by immunophenotyping. This requires the use of sections derived from snap-frozen tissue or unfixed cell smears. Interstitial DCs express CD1a, CD11c, CD11b (subset), and MHC class II. In skin, dermal DCs are of interstitial type and they also express CD90 (Thy-1). Resident unstimulated interstitial DCs express low levels of costimulatory molecules, some of which are members of the B7 family—CD80 and CD86. Interstitial DCs are potent APCs, and they possess all of the molecules involved in presentation of peptides, lipids, and glycolipids to T cells (CD1a, MHC classes I and II).
Macrophages are an important component of all tissues. The identification of macrophages in tissues relies on immunophenotyping. Macrophages differentially express β-2 integrins in normal tissues. For instance, in most tissues, macrophages express CD11b/CD18 (Kupffer cells, microglia, synovial type A cells). Macrophages in hematopoietic sites, such as the splenic red pulp, bone marrow, and medullary sinuses of lymph nodes, express CD11d/CD18. 20,21 Other markers used for the identification of macrophages include Mac387 (calprotectin), MHC class II, CD163, and CD204—the latter two are scavenger receptors. 30 The scavenger receptors, CD163 and CD204, are also expressed by DC subsets in some species. 43,87 Expression patterns of CD163 and CD204 in normal canine DCs have not been reported. Of the markers mentioned, only CD11b is not assessable in formalin-fixed tissues.
Histiocytic Sarcoma Complex
Given the ubiquitous distribution of interstitial DCs, HS can arise in almost any tissue. The neoplastic cells in most canine HS lesions have been shown to express an immunophenotype consistent with an interstitial DC origin. 5 Histiocytic sarcomas may be localized—that is, they originate at a single tissue site or in a single organ (with solitary or multiple foci). Once the lesions spread beyond the local draining lymph node to involve distant sites, the disease is then considered disseminated HS. Disseminated HS is the currently accepted terminology for what was originally reported as malignant histiocytosis. 22,53 Localized and disseminated HS have been observed in cats at a far lower incidence than in dogs. 16,23,24,35,61 In essence, feline HS complex resembles the canine counterpart in terms of location of lesions and disease progression.
The HS complex of diseases was first recognized in Bernese Mountain Dogs, in which a familial association was apparent. 53,59 Pedigree analyses support a polygenic mode of inheritance in Bernese Mountain Dogs. 1,53,59 Other breeds are predisposed to HS complex diseases, and these include Rottweilers, Golden Retrievers, and Flat-Coated Retrievers. 5,15 HS complex is not limited to just these breeds and can occur sporadically in any breed. Recently, studies of the genomic loci involved in HS in Bernese Mountain Dogs and Flat-Coated Retrievers have pointed to abnormalities in tumor suppressor gene loci (CDKN2A/B, RB1, and PTEN) that are broadly similar to those described in human HS. 28,73 Other genomic loci are probably involved as well. 28
Clinical signs of HS are vague and include anorexia, weight loss, and lethargy. Other signs depend on the organs involved and are a consequence of destructive mass formation. Accordingly, pulmonary symptoms such as cough and dyspnea have been seen. Central nervous system (CNS) involvement (primary or secondary) can lead to seizures, incoordination, and paralysis. Lameness is often observed in articular HS. Dogs with HS of interstitial DC origin often have mild, nonregenerative anemia. 1,51,53 More severe regenerative anemia, thrombocytopenia, hypoalbuminemia, and hypocholesterolemia have been consistently documented in hemophagocytic HS. 51 Hypercalcemia may also be encountered with HS, although the incidence is low (3%). 77
Morphologic Features
Primary lesions of HS occur in spleen, lymph node, lung, bone marrow, CNS, skin and subcutis, and periarticular and articular tissues of the limbs. Secondary sites are widespread but consistently include liver and lungs (with splenic primary) and hilar lymph nodes (with lung primary). In the original report of HS in Bernese Mountain Dogs, a high incidence of primary lung lesions was apparent. 53 Pulmonary HS may have been mistaken for anaplastic large cell carcinoma of the lung before the advent of cell markers for lineage determination. 53 Similarly, about 50% of synovial tumors of dogs were shown to be localized HS by using an anti–canine CD18 monoclonal antibody, which retained reactivity in formalin-fixed tissue sections. 5,18
Lesions of HS that originate from interstitial DCs (see below) are typically destructive mass lesions with a uniform, smooth cut surface and are white/cream to tan in color (Fig. 10). Lesions may be solitary or multiple within an organ (especially spleen). Articular HS has a distinctive appearance: it occurs as multiple tan nodules located beneath the synovial lining; these are further described below.
Spleen; dog; canine histiocytic sarcoma (HS). Top: Discrete mass formation is characteristic of HS of interstitial dendritic cell origin. Bottom: Diffuse splenomegaly with ill-defined mass formation is characteristic of hemophagocytic HS of macrophage origin.
Hemophagocytic HS, which originates from macrophages (see below), does not initially form mass lesions in the primary sites (spleen and bone marrow). Typically, diffuse splenomegaly is consistently observed. Within the grossly enlarged spleen, ill-defined mass lesions may be perceptible as well (Fig. 10). Hemophagocytic HS may coincide with HS of interstitial DC origin. In these instances, both diffuse splenomegaly and discrete mass formation are observed. 51
Lesions of HS are composed of sheets of large, pleomorphic, mononuclear, and multinucleated giant cells, which usually have marked cytological atypia and numerous bizarre mitotic figures (Fig. 11). Some lesions may consist of spindle cells, either alone or mixed with the mononuclear and multinucleated giant cells. Pure spindle cell lesions often lack discrete cell borders and resemble spindle cell sarcomas of diverse cell lineage (fibroblast, myofibroblast, and smooth muscle origin). Confirmation of histiocytic lineage is best achieved with IHC in these instances. 5 Phagocytosis of red cells, leukocytes, and tumor cells occurs in HS, but it is not usually pervasive except in hemophagocytic HS.
Immunophenotypic Features
Immunostaining of most HS lesions reveals that tumor histiocytes express leukocyte surface molecules characteristic of interstitial DCs, which include CD1a, MHC class II, and CD11c/CD18. 5 Expression of CD4 by neoplastic histiocytes has not been observed in HS. In comparison, histiocytes in canine reactive histiocytoses regularly express CD4, which indicates an activation phenotype. The β-2 subunit of the leukointegrins (CD18) is readily demonstrable in formalin-fixed tissue sections of HS lesions (Fig. 11 inset). Since lymphomas may express abundant CD18, it is important to exclude a lymphoid origin (eg, large cell lymphoma) by staining for lymphocyte antigen receptor–associated molecules (CD3 and CD79a) and CD20. The exact sublineages of DCs involved in HS have not been determined in most instances. The most likely candidates include interdigitating DCs in lymphoid tissues and perivascular interstitial DCs in other involved tissues. In hemophagocytic HS, histiocytes express markers most consistent with macrophage differentiation (CD11d/CD18) rather than DC differentiation, in which abundant expression of CD1a and CD11c/CD18 is expected. 51
Distinctive Histiocytic Sarcoma Syndromes
Several presentations of HS deserve separate additional description because of particular clinical or clinicopathological associations that are often crucial to successful diagnosis.
Hemophagocytic HS
Dogs with hemophagocytic HS present with a distinctive clinicopathologic syndrome. These dogs usually have a moderate to marked, regenerative, hemolytic anemia and thrombocytopenia. They have mild hyperbilirubinemia without jaundice at initial presentation. Most dogs have hypoalbuminemia and often hypocholesterolemia. 51 This clinical picture is often confused with immune-mediated hemolytic anemia and thrombocytopenia (Evans syndrome) despite the lack of erythrocyte-bound IgG (Coombs negative) in tested dogs. Hemophagocytic HS carries the worst prognosis of all forms of HS with a median survival of only 4 weeks (mean, 7 weeks) after diagnosis. 51 Factors contributing to poor outcome include development of severe anemia and coagulopathy with disease progression. In another study, in which the efficacy of CCNU was assessed as a treatment for HS, a cohort of dogs that presented with anemia, hypoalbuminemia, and thrombocytopenia had a median survival of 28 days or less (vs 106 days for all dogs). The form of HS was not further classified in this subset of dogs, but the hematological and biochemical values are shared by hemophagocytic HS. 77
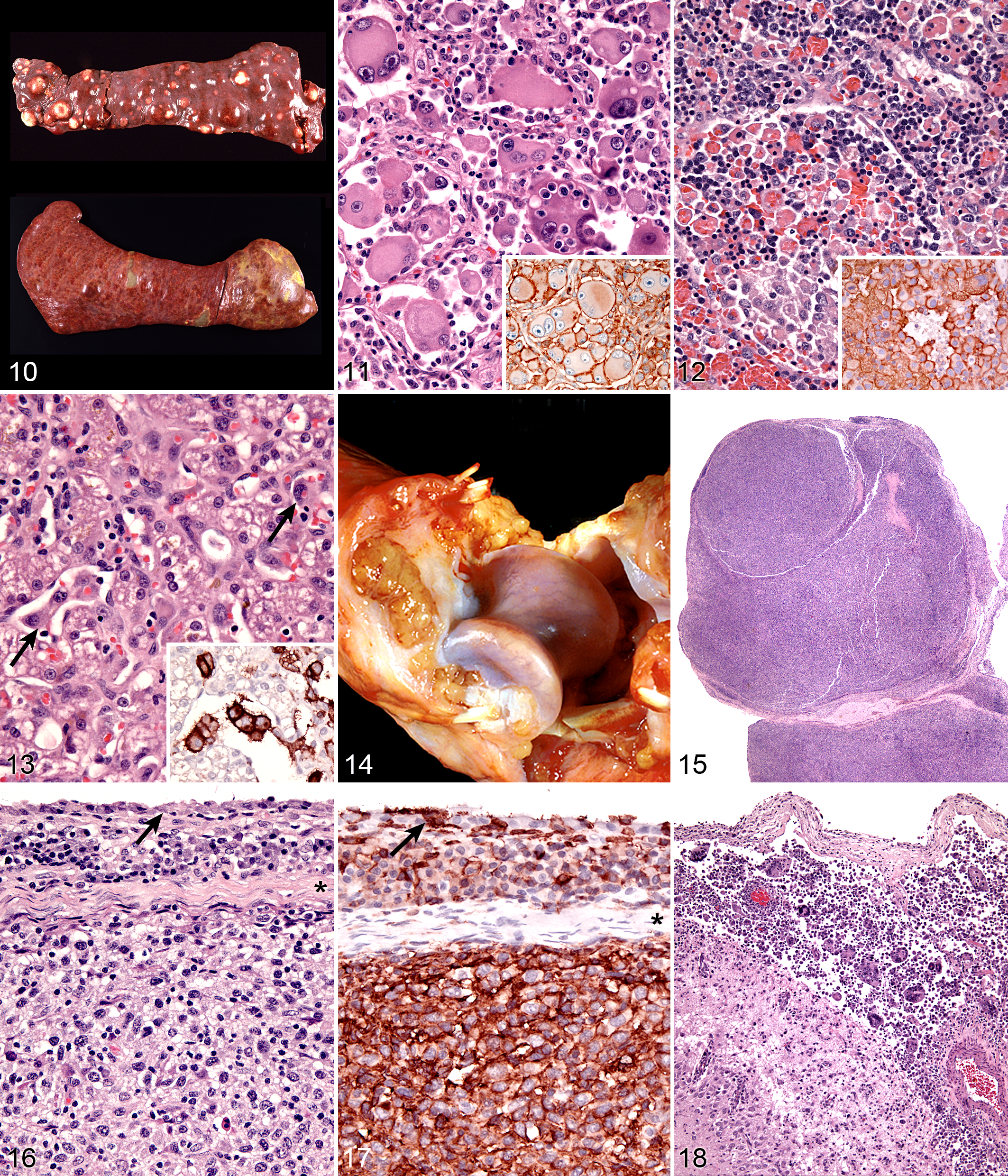
Dogs with hemophagocytic HS have diffuse splenomegaly as already described (Fig. 10), and they lack grossly visible masses (<1–2 mm) in metastatic sites such as liver and lung. This is enforced by the growth pattern of the neoplastic histiocytes. If diffuse splenomegaly and discrete masses are present, immunophenotypic investigation will reveal both types of histiocytic differentiation (DC and macrophage); this is an uncommon event. 51 In hemophagocytic HS, histiocytes expand the splenic red pulp more or less diffusely. They exhibit marked erythrophagocytosis and are accompanied by interspersed foci of extra-medullary hematopoiesis (Fig. 12). They invade red pulp sinuses and travel to the liver, where they insidiously invade the sinusoids (Fig. 13). Examination of sections of lung will reveal erythrophagocytic histiocytes within the pulmonary vasculature. The bone marrow is simultaneously infiltrated and erythrophagia is observed there as well. 51 There are 2 reports of HS in cats, in which the lesions in 3 cats closely resembled the pattern and distribution of hemophagocytic HS of dogs. 24,35
Cytologically, the histiocytes in hemophagocytic HS may appear well differentiated (especially in bone marrow); in fact, there is often asynchrony between spleen and bone marrow; usually there is more atypia in the spleen compared with bone marrow. 51 However, even the well-differentiated cells are aggressive in their behavior, as evidenced by invasion of the liver and lung vasculature.
Neoplastic histiocytes in hemophagocytic HS express a distinctive surface antigen profile much like that expressed by macrophages in splenic red pulp and bone marrow. They express CD11d/CD18 (Fig. 12 inset), lack expression of CD11c, and have variable CD1a expression—usually weak to negative. 51 Both CD11d and CD18 are assessable in formalin-fixed tissue sections, and this greatly facilitates the diagnosis of hemophagocytic HS and reveals the often inconspicuous metastatic, intravascular lesions in liver and lung (Fig. 13 inset). 51 Feline hemophagocytic HS is also thought to arise in macrophages, based on lack of CD1 expression. An origin from splenic red pulp macrophages could not be confirmed due to lack of expression of CD11d. 24
Articular/Periarticular HS
Articular HS is a localized form of HS, which occurs on the limbs. The lesions originate either within the joints (intra-articular) and/or adjacent to the joints (periarticular) of the appendicular skeleton. 18 Articular HS is the most common tumor affecting the joints of dogs and also occurs in cats at much lower incidence. 18,61 The stifle and elbow joint are most commonly affected, and lesions may occur in the coxofemoral joint and carpus. More than half of the dogs were Rottweilers. 18 Invasion of bone can occur with advanced disease. Since skeletal lesions are observed in disseminated HS in the same sites as articular HS, it is important to determine if the skeletal involvement is subsequent to a soft tissue mass originating near the synovial tissues. 72 Early detection and treatment of articular HS is potentially curative, while disseminated HS has a worse prognosis. 5,34 However, articular HS is capable of widespread dissemination since histiocytes, especially those of the various DC lineages, are actively migratory cells. This is reflected in the median survival of dogs with articular HS and evidence of metastatic disease (about 5–8 months). 18,34
Articular HS has a distinctive appearance: it occurs as multiple tan nodules located beneath the synovial lining. Hence, these lesions begin outside of the synovial space either within the joint and/or in a periarticular location. The lesions may encircle the affected joint (Fig. 14). Microscopically, the lesions display a micronodular growth pattern (Fig. 15). These nodules may coalesce into masses in advanced lesions. The lesions most often originate beneath the synovium, and the synovial lining is intact above the tumor (Fig. 16). In advanced lesions, these topographical relationships may be obscured by effacing mass formation. Cytologically, articular HS resemble HS in other sites—cytologically atypical, pleomorphic histiocytes predominate. However, a marked inflammatory infiltrate is usually recognizable within the lesions. Lymphocytes (mostly T cells), neutrophils, and well-differentiated histiocytes are prevalent. Inflammation of the adjacent synovium with lymphocyte, plasma cells, and histiocytes is also observed. 18
A history of anterior cruciate rupture or other traumatic injury to joints is associated with the development of articular HS. 18,82 Synovitis has been reported in association with rupture of the cranial cruciate ligament. T cells and dendritic cells are major components of the inflammatory response. 38 While no causal relationship necessarily exists between synovial inflammation and the development of HS, there is ample evidence of progression of inflammation to neoplasia in many inflammatory disease states. 17
The immunophenotype of histiocytes in articular HS is identical to that of perivascular interstitial DCs (CD1a+ CD11c/CD18+) rather than synovial type A cells (CD1a– CD11b/CD18+), which are intercalated between synovial-type B cells (synovial fibroblasts) in the synovial lining. Synovial type A cells do not express CD11c (R. A. Fairley, L. LeFranco, and P. F. Moore, unpublished data). Synovial cell sarcoma (SCS) is a fibroblastic proliferation, which is likely derived from synovial-type B cells (specialized synovial fibroblasts). 18 Synovial fibroblasts are highly synthetic cells and can produce a diverse array of inflammatory mediators, including chemokines, which attract macrophages. 67 Hence, it is not surprising that synovial cell sarcomas are infiltrated by large numbers of CD18+ histiocytes in most instances (P. F. Moore, unpublished data). This may lead to confusion of SCS with articular HS. However, a dominant CD18– neoplastic population (interpreted as synoviocytes) is present among the many CD18+ histiocytes. This contrasts with the uninterrupted sheets of CD18+ cytologically atypical histiocytes observed in articular HS (Fig. 17).
Central Nervous System HS
The central nervous system may be infiltrated by HS as a primary location or as a consequence of metastatic spread of disseminated HS. Extracranial metastasis of primary CNS HS must be extremely rare, since it has yet to be reported. Histiocytic sarcoma of the CNS most often originates in the leptomeninges (Fig. 18), and the lesions are composed of large numbers of mixed inflammatory cells (lymphocytes, histiocytes, and plasma cells) and cytologically atypical histiocytes (Fig. 19). The frequency of the latter cells is quite variable between cases. In the only case series of CNS HS published, Pembroke Welsh Corgis were overrepresented (7 of 15 dogs). This is not a breed that is otherwise commonly affected by HS. In reviewing the UC Davis data (>70 dogs with primary CNS HS), Pembroke Welsh Corgis were not represented (P. J. Dickinson, University of California, Davis, personal communication, 2013). The CNS lesions most commonly presented as a focal, solitary subdural mass and less commonly as a diffuse meningeal infiltrate. The lesions appeared to arise in the leptomeninges and subsequently involved the brain. Metastasis beyond the CNS did not occur. Neoplastic histiocytes in solitary mass lesions expressed MHC class II molecules, Iba1 (ionized calcium binding adaptor molecule 1), and scavenger receptors expressed by macrophages (CD163 and CD204). 30 The macrophage scavenger receptors were not expressed in the diffuse meningeal HS. The cell and tissue distribution of the macrophage scavenger receptors (CD163 and CD204) have not been studied in canine tissues, so it is difficult to reconcile these data with our data, which indicate interstitial DC markers (CD1a, CD11c, and MHC class II) are expressed by neoplastic histiocytes in CNS HS in our published report and subsequent unpublished data. 5 Macrophage scavenger receptors are present on subsets of interstitial DCs, so this could be one potential point of reconciliation of the divergent data. 43,87 Also, dendritic cells are located in the meninges and choroid plexi but not in the brain. 19 Hence, the apparent site of initiation of CNS HS does have a resident population of interstitial DCs from which HS could develop.
Dendritic Cell Leukemia
Peripheral blood involvement in HS is rare. There have been 2 reports of DC leukemia in dogs; in both instances, large numbers of atypical histiocytes were observed in peripheral blood (30,000 - 60,000 per μl). 6,68 In both dogs, the DC lineage (CD1+ CD11c+ CD11d– MHCII+) was confirmed by extensive immunophenotyping of cell smears or by flow cytometry. Diffuse infiltration of affected organs (bone marrow, spleen, lung, and liver) without mass formation was characteristic of a leukemic infiltrate. 6 This is also the pattern of infiltration exhibited by hemophagocytic HS. However, this latter disease does not involve peripheral blood and is associated with a responsive extravascular hemolytic anemia and marked erythrophagocytosis by neoplastic histiocytes, which express CD11d. 51 CD11d was not expressed by neoplastic histiocytes in either dog. 6,68
Feline Progressive Histiocytosis
Feline progressive histiocytosis (FPH) is a disease of middle-aged to older cats (age range, 7–17 years). The initial presentation of FPH many be a solitary skin nodule, although usually multiple papules, nodules, or plaques develop, measuring up to 1.5 cm in diameter. The nodules are firm, nonpruritic, and nonpainful. The surface is often alopecic and may be ulcerated (Fig. 20). The lesions are mostly located on the head, lower extremities, or trunk. Occasionally, the lesions are limited to one extremity. The lesions may wax and wane, but spontaneous regression does not occur. In FPH, a proportion of cats develop invasive, expansile masses in lymph nodes and internal organs, including the lungs, kidneys, spleen, and liver.
Feline progressive histiocytosis behaves as a low-grade HS, which originates in skin from resident interstitial DCs. 4 The initial clinical course is indolent and the histiocytes are cytologically well differentiated, and it is important to rule out infectious agents with special stains. However, later in the course, histiocytes manifest a higher frequency of cytological atypia more consistent with HS (based on serial biopsies from the same cat). Feline progressive histiocytosis has a poor long-term prognosis, since the wide variety of treatments used have lacked efficacy.
Microscopic lesions consist of diffuse dermal histiocytic infiltrates, which may extend into the subcutis. The overlying epidermis is either intact or ulcerated. Some lesions manifest epitheliotropism (about 40% incidence), characterized by intraepidermal single cells or cell aggregates; the remaining cases lack epithelial involvement (Fig. 21). Histiocytes have irregular, vesicular nuclei and finely dispersed chromatin (Fig. 22). Electron microscopic investigation of FPH in 2 cats has not demonstrated BGs (indicative of LC differentiation) in the neoplastic histiocytes. Cytological atypia is present in a minority of lesions early in the course. Biopsies of later lesions (from the same cat) reveal a higher incidence of multinucleated tumor cells. The mitotic activity varies, and atypical mitoses are seen (Fig. 22). Late-stage lesions are essentially morphologically identical to HS. The extent of reactive infiltrates, composed of dispersed lymphocytes and fewer neutrophils, varies between cases.
Immunophenotyping of the histiocytes in FPH revealed expression of CD1a, CD18, and MHC class II. The histiocytes most often lacked E-cadherin expression and expressed CD5 in about half of the cases. This immunophenotype is most consistent with an interstitial DC origin. The reactive lymphocytes that infiltrate the lesions mostly express CD3 and CD8. 4
Canine Fibrohistiocytic Nodule
The term splenic fibrohistiocytic nodule was introduced in 1998 with the intent of providing useful prognostic information for clinicians and freeing pathologists the burden of deciphering complex splenic lesions of uncertain type. 78 Unfortunately, fibrohistiocytic nodule is just a wrapper within which there are several specific entities, which include marginal zone B-cell lymphoma, high-grade B-cell lymphoma, HS of DC and macrophage origin (ie, hemophagocytic HS), nodular hyperplasia (lymphoid and complex), and a variety of stromal cell sarcomas. 46 Some of these entities (marginal zone B-cell lymphoma and hemophagoctyic histiocytic sarcoma) were described after a fibrohistiocytic nodule was introduced; the other entities were well known at the time. 51,80 Hence the diagnosis of splenic fibrohistiocytic nodule is lacking in precision and should no longer be used. 46
Canine Reactive Histiocytoses
Reactive histiocytoses have been described in dogs; a feline counterpart has not been recognized to date. Reactive histiocytoses are either confined to skin and draining lymph nodes (CH) or involve skin and extracutaneous sites (SH). Systemic histiocytosis was first described in Bernese Mountain Dogs, and a familial association was apparent. 49 Histiocytic sarcomas occurred in the same families of Bernese Mountain Dogs as SH, but progression of SH to HS was not observed. 53 Systemic histiocytosis occurs in many other large breeds of dogs. 3 Overall, SH is a rare disease, and CH is far more common. Cutaneous histiocytosis was first described in dogs as a waxing and waning dermatitis and panniculitis of unknown etiology, in which the lesions were dominated by histiocytes. 44
The concept that CH and SH are related diseases emerged from an study in which an identical lesion topography and histiocyte immunophenotype was described in CH and SH. 3 Cutaneous and systemic histiocytosis are related histiocytic inflammatory diseases that are believed to have an element of immune dysregulation in their pathogenesis. 3 The lesions are thought to be antigen driven, but no etiologic agent/antigen has been discovered. The lesions are dominated by activated dermal interstitial DCs and T cells, which frequently infiltrate the walls of dermal vessels, creating a lymphohistiocytic vasculitis. The lesions radiate from affected vessels and coalesce to form masses, especially in the deep dermis and panniculus. Therefore, the lesions have a “bottom-heavy” topography and should never be confused with histiocytomas, which have a “top-heavy” topography. The lymphoid component of the lesions is enriched for T cells, which are most often CD8+. T-cell numbers vary throughout the lesion and can vary markedly between lesions. 3 The role played by T cells is unknown. Regression of the lesions by CD8+ T cells does not occur as it does in cutaneous histiocytomas. Instead, T cells as an antigen responsive element may be involved in the proliferation and activation of DC via T-cell–derived cytokines such as GM-CSF and TNF-α, which are known to influence the proliferation and differentiation of DCs. 37,76
The hypothesis that an immunoregulatory deficit underlies the development and progression of CH and SH is bolstered by the clinical behavior and response to therapy. The clinical course in both diseases is marked by spontaneous remissions and relapses, particularly early in the course. Also, the only effective therapies used to date involve the use of immunosuppressive or immunomodulatory drugs—prednisolone, cyclosporine A, leflunomide, and tetracycline/niacinimide. 48,60 The lesions of CH and SH may wax and wane, and spontaneous resolution without therapy occurs. This makes it difficult to assess the effects of therapy.
Cutaneous Histiocytosis
Cutaneous histiocytosis is an inflammatory lymphohistiocytic proliferative disorder that primarily involves skin and subcutis. 44 CH occurs in a number of breeds; there is no clear breed predisposition. 3,44,60 Lesions may extend to involve the local lymph nodes, which are part of the skin immune system. The incidence of lymph node involvement is unknown, since lymph nodes are not commonly sampled when skin biopsies are taken unless there is marked lymphadenomegaly. The lesions most often occur as multiple cutaneous and subcutaneous nodules up to 4 cm in diameter; solitary lesions are uncommon. Overlying skin ulceration is common. Lesions may disappear spontaneously or regress and appear at new sites simultaneously. Topographically, lesions may be found on the face, nose, neck, trunk, extremities (including foot pads), perineum, and scrotum. 3,44,60 Morphological features of CH are similar to those of SH in skin, so they will be described together.
Systemic Histiocytosis
Systemic histiocytosis was originally described in related Bernese Mountain Dogs. 49 SH has been observed in other breeds less commonly (Rottweiler, Labrador Retriever, Basset Hound, Irish Wolfhound, and others). Familial occurrence of SH is evident in Irish Wolfhounds in the Pacific Northwest (P. F. Moore, unpublished observations).
SH is a generalized histiocytic proliferative disease with a marked tendency to involve skin, ocular and nasal mucosae, and peripheral lymph nodes (Fig. 23). The disease predominately affects young to middle-aged dogs (2–8 years). Clinical signs vary with the severity and extent of the disease and may include anorexia, marked weight loss, stertorous respiration, and conjunctivitis with marked chemosis. Multiple cutaneous nodules may be distributed over the entire body but are especially prevalent in the scrotum, nasal apex, nasal planum, and eyelids. These are similar sites in which CH occurs. Ulceration of the skin overlying the nodules is common. Peripheral lymph nodes may be palpably enlarged by the histiocytic infiltrates. The disease course may be punctuated by remissions and relapses, which may occur spontaneously, especially early in the disease course.
Morphologic Features—Reactive Histiocytoses
Microscopic features of SH and CH are essentially identical in skin. The lesions usually extensively involve the deep dermis and subcutis. Discrete perivascular lesions affecting the mid-dermis often coalesce in the deep dermis and subcutis. Involvement of the superficial dermis is inconsistent, and epidermotropism by histiocytes is unusual. Overall, the lesions have a bottom-heavy topography (Fig. 24). Histiocytes and lymphocytes frequently invade vessel walls (ie, lymphohistiocytic vasculitis), and this may lead to vascular compromise and infarction of surrounding tissues, which contributes to ulceration of the cutaneous lesions in CH and SH (Fig. 25). The infiltrate is pleo-cellular, but histiocytes and lymphocytes are more numerous than neutrophils, plasma cells, and eosinophils. Histiocytes have large round to oval, indented, folded, or twisted nuclei. Histiocytes have abundant pale eosinophilic cytoplasm with occasional vacuoles. Multinucleated histiocytes and histiocytes with bizarre, misshapen nuclei and mitotic figures, which are characteristic of HS, are lacking in CH and SH (Figs. 25, 26). Lesions may occur in skin draining lymph nodes in CH and SH. Histiocytes selectively invade the paracortex and sinuses. In severe lesions, the cortex is obliterated by histiocytic infiltration, which also effaces lymph node trabeculi and the capsule. Significant perinodal infiltration is possible (Fig. 27).
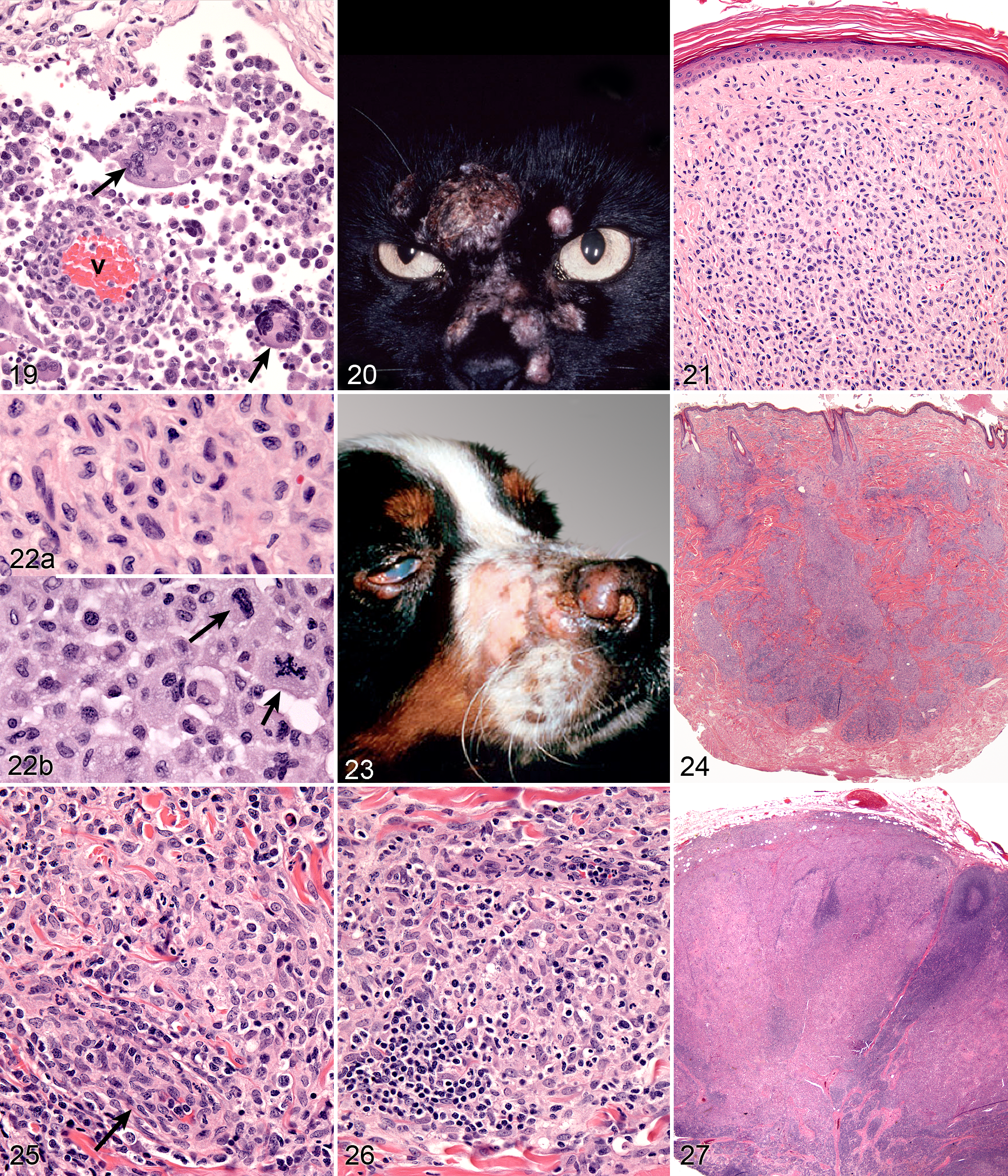
Cerebral cortex; dog; canine central nervous system (CNS) histiocytic sarcoma (HS). The histiocytic infiltrate in the leptomeninges manifests variable cytological atypia, which is especially evident in the multinucleated cells (arrows). The vessel wall (v) is infiltrated by lymphocytes and histiocytes. Hematoxylin and eosin (HE).
Inflamed cutaneous non-epitheliotropic T-cell lymphoma is readily confused with CH. 50 The key discriminatory features are identification of clusters of cytologically atypical T cells and variable expression of CD3 by neoplastic T cells—so-called CD3 antigen loss. Definitive diagnosis is reliant on T-cell receptor γ gene rearrangement analysis, which will detect clonal T-cell expansion of the lymphoma cells within the reactive/inflammatory T-cell and histiocyte infiltration. 32,50
Beyond the skin, the widespread distribution of the lesions of SH is only fully appreciated at necropsy. Histiocytic lesions have been observed in lung, liver, bone marrow, spleen, peripheral and visceral lymph nodes, kidneys, testes, orbital tissues, nasal mucosa, and other sites. The lesions in these sites are vasocentric, and the infiltrates radiate to obliterate surrounding tissue. The lesions may only be microscopic, but mass formation has also been observed.
Immunophenotypic Features—Reactive Histiocytoses
The immunophenotypic expression pattern of SH and CH is virtually identical. 3 Histiocytes in SH and CH express markers expected of DCs such as CD1a, C11c/CD18, and MHC class II. Histiocytes express CD4 (a marker of DC activation) and CD90 (Thy-1), which is expressed by normal dermal interstitial DCs. Histiocytes lack expression of E-cadherin. Hence, the immunophenotype of histiocytes in CH and SH is the inverse of that seen in cutaneous histiocytoma and cutaneous LCH with respect to the expression of CD4, CD90, and E-cadherin. 54 The activated interstitial DC immunophenotype of CH and SH is not observed in HS—CD4 expression has not been encountered to date. 5 Unfortunately, CD1a, CD4, and CD11c are not assessable in formalin-fixed tissue sections. CD90, CD18, MHC class II, and E-cadherin are assessable in formalin-fixed tissue sections.
Conclusion
Histiocytic proliferative diseases occur as neoplastic processes and inflammatory processes with immune dysregulation in dogs. The latter do not occur in cats. Most histiocytic proliferative diseases originate in LCs or interstitial DCs in both dogs and cats. Macrophage differentiation has only been demonstrated in hemophagocytic HS in dogs and cats. The finer definition of histiocytic diseases going forward will rely on the development of a greater array of markers of histiocytic lineage and the application of gene expression profiling on lesional histiocytes. Studies of this nature will hopefully lead to more effective targeted therapies for individual histiocytic diseases.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
