Abstract
This article reports 5 cases of oviduct adenocarcinoma in adult captive snakes from Smithsonian's National Zoological Park. This neoplasm was found in 1 of each of the following species: emerald tree boa (
Neoplasia is commonly encountered in the practice of reptile medicine and the prevalence is higher in snakes, followed by lizards, chelonians, and crocodilians. The prevalence of neoplasia in snakes ranges from 12.4% 2 to 15%, 5 and tumors are predominantly malignant and mesenchymal neoplasms. 2 Among snake groups, colubrids have the highest incidence of neoplasia, followed by crotalids, vipers, and boids. 5 Soft tissue sarcoma, lymphoma, renal adenocarcinoma, fibrosarcoma, and melanoma are the most prevalent tumors in snakes. 5 The possible role played by oncogenic viruses in neoplastic disorders in reptiles has been discussed. 3, 10 In many cases, type C retrovirus particles are found in snakes with tumors, but they are also seen in normal tissues from the same snake. Neither in vivo transmission experiments nor in vitro assessments of transforming capacity of these retroviral isolates have been performed, and the relationship between retroviruses and those tumors is uncertain. 3, 10
Reproductive tract tumors are more common in lizards than snakes, with the exception of granulosa cell tumors. 5 Oviduct adenocarcinoma is uncommon in both lizards and snakes and has been described as exophytic tubulopapillary tumors that invade the submucosa and muscular tunics with discrete desmoplasia and without metastasis or carcinomatosis. 5
Reptile oviducts are paired structures derived from embryonic Mullerian ducts and have multiple functions. They act as a conduit for the eggs between ovulation and oviposition or parturition. They act as a site of fertilization and, in some species, sperm storage. They provide an oocyte with albumen and multilayered eggshell. They function with extraembryonic membranes to form placental interactions providing gas and water exchange, and in some cases nutrient transfer, between the mother and embryos. 6
Five adult female captive snakes, 3 boids and 2 colubrids, from the Smithsonian's National Zoological Park are involved in this report and are detailed in Table 1. The boids include 1 emerald tree boa (
Species, age, gross, and microscopic findings of 5 cases of oviduct adenocarcinoma in captive snakes.
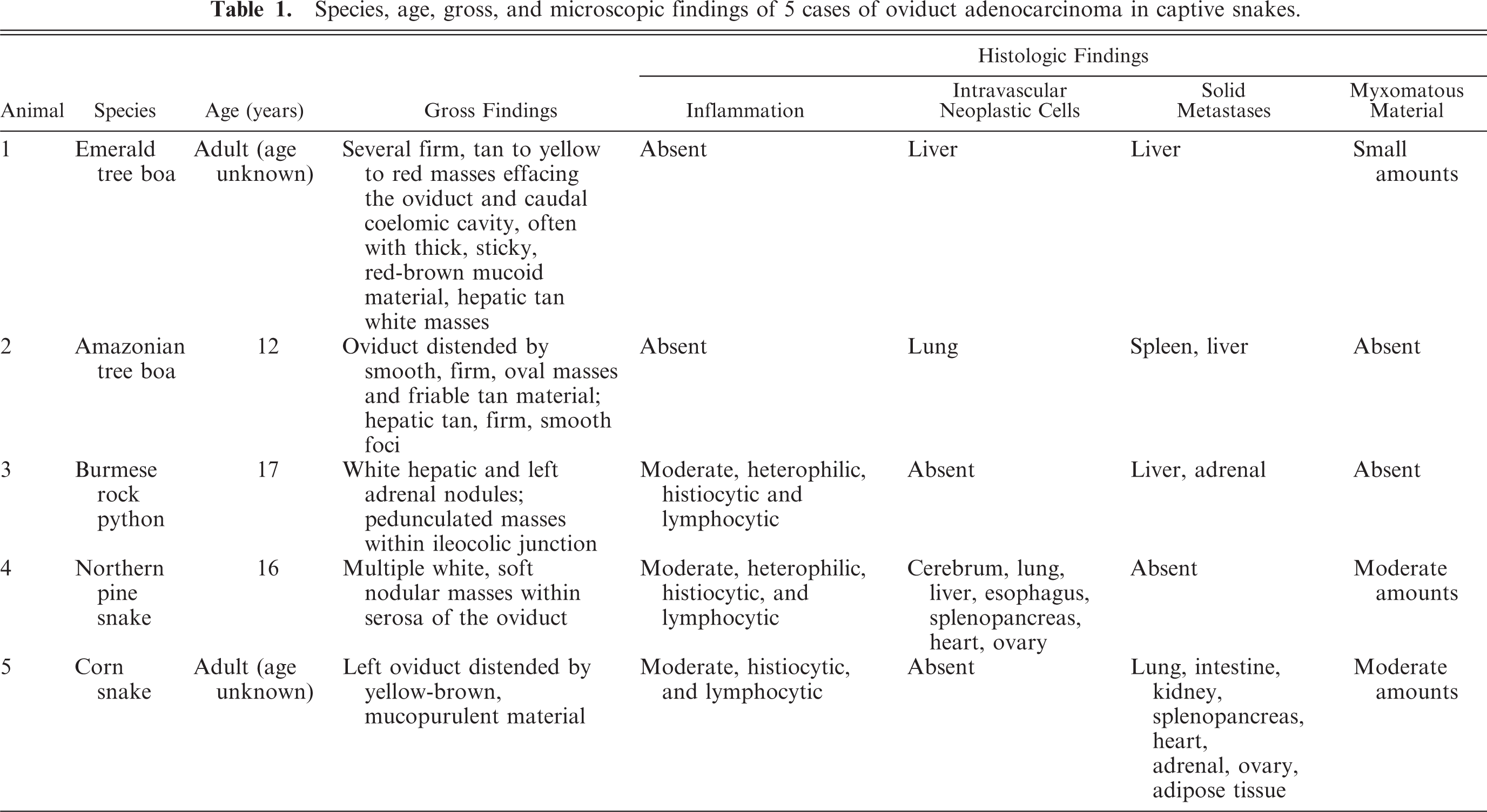
The clinical signs ranged from refusing food, weight loss, lethargy, chronic loss of muscle tone, cloacal and ocular edema to a 16-month history head tilt.
Grossly, multiple 0.2 to 7 cm in diameter, white or tan, soft to firm, nodular masses were expanding the wall of the oviducts in 3 snakes including snakes Nos. 1, 2, and 4 (Fig. 1). In addition, similar masses were within the hepatic parenchyma of snakes Nos. 1 and 2 (Fig. 2). No mass was grossly identifiable in the oviduct of 2 snakes (snakes Nos. 3 and 5), despite the presence of hepatic, adrenal, and intestinal nodules in 1 case (snake No. 3) and a distended oviduct by mucopurulent material in the other (snake No. 5).
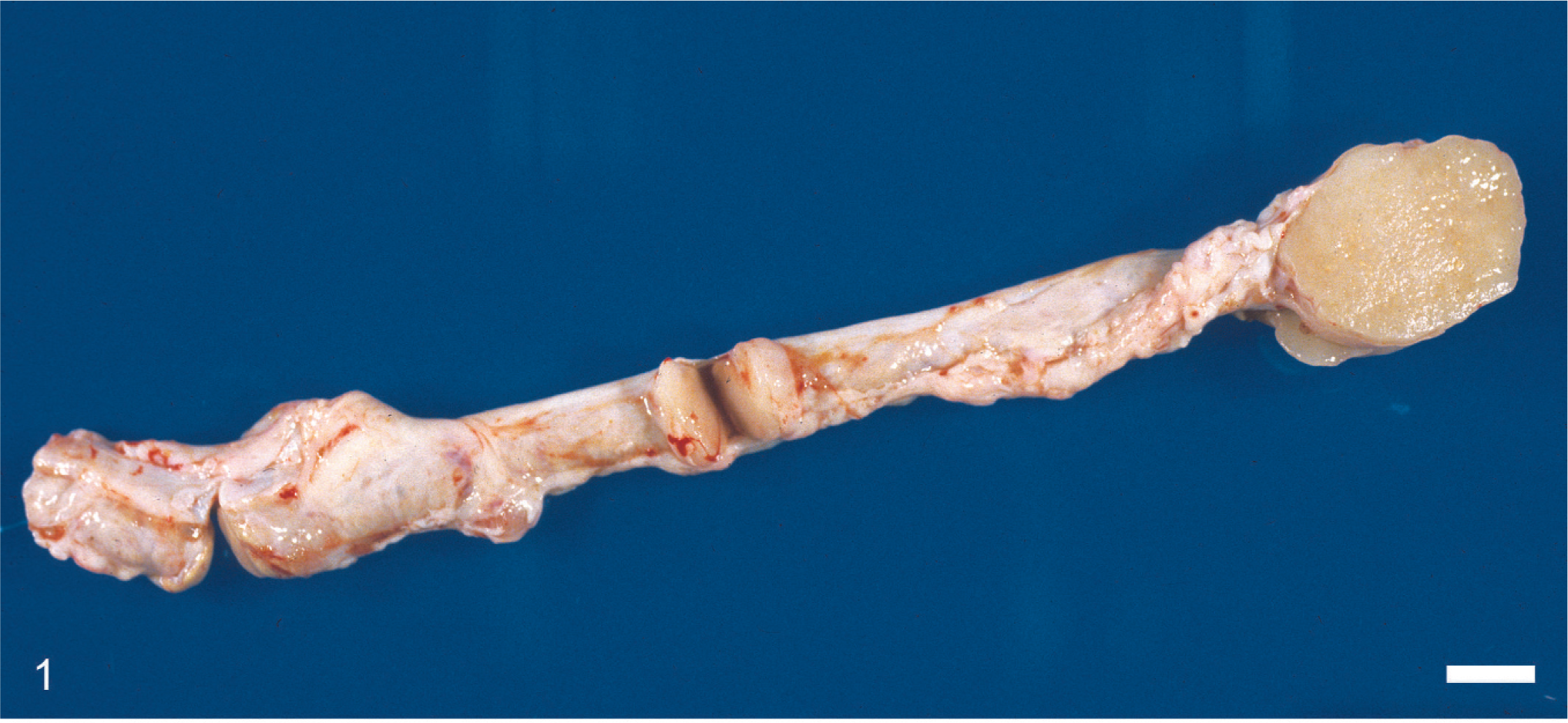
Oviduct; Burmese rock python (
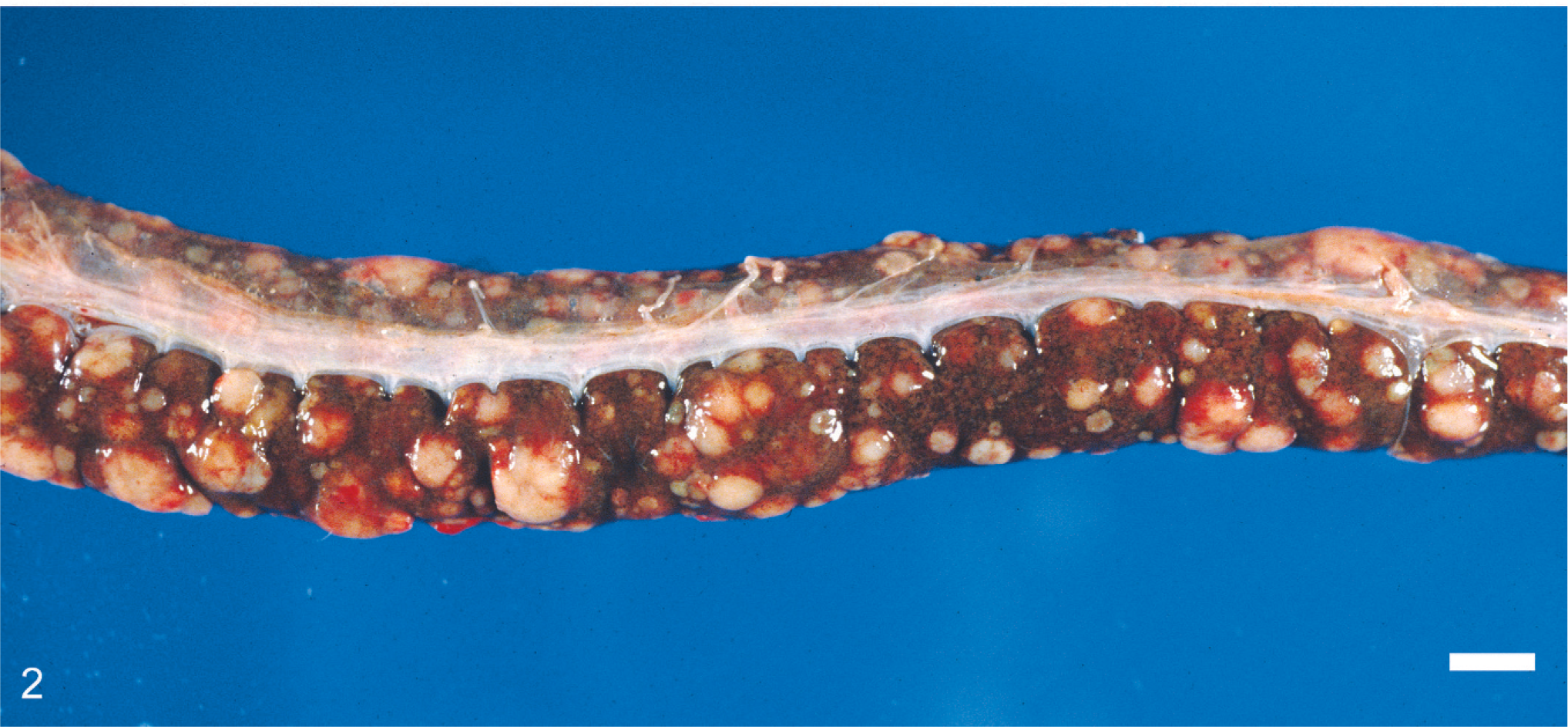
Liver; Amazonian tree boa (
Histologic changes are outlined in Table 1 and were similar in all 5 cases. Microscopically, neoplasms were papillary, infiltrating, and effacing the serosa in the Northern pine snake (snake No. 4; Fig. 3) and transmurally infiltrating all layers of the oviduct, and extending into the cut borders in all other snakes (Nos. 1, 2, 3, and 5). All neoplasms were poorly circumscribed, unencapsulated, and infiltrative. Tumor cells were organized in acini, islands, and nests or cords and ducts of polygonal cells surrounded by a moderate to abundant fibrovascular stroma. The neoplastic cells had variable cell borders, a moderate amount of eosinophilic cytoplasm, irregularly round to elongated nuclei with vesiculate chromatin and 1 distinct nucleolus (Fig. 4). The mitotic rate was 1 to 10 per high power fields. Within all the neoplasms, there was necrosis within the stroma and within the center of the neoplastic nests, and hemorrhage. Intratumor inflammation consisted mostly of heterophils, macrophages, and lymphocytes, mostly within the stroma (snakes Nos. 3–5) and intravascular neoplastic cells (snakes Nos. 1, 2, and 4) were seen. Three carcinomas contained myxomatous material (snakes Nos. 1, 4, and 5). Desmoplasia and intravascular bacterial colonies were seen in 1 case (snake No. 5). Similar cell populations were seen within the metastases in the liver (snakes Nos. 1–3; Fig. 5); spleen (snakes Nos. 2 and 5) and adrenal (snakes Nos. 3 and 5); and kidney, ovary, heart, intestine, and lungs (snake No. 5). Neoplastic cells were seen intravascularly within the lung (snakes Nos. 2 and 4); esophagus, liver, splenopancreas, heart, ovary, and cerebrum (snake No. 4). There were multifocal areas of neuronal necrosis and gliosis in areas associated with emboli of neoplastic cells in the Northern pine snake (snake No. 4; Fig. 6). The Burmese rock python (snake No. 3) had an additional intestinal lymphoma without metastases. No inclusions were evident in any of these snakes.
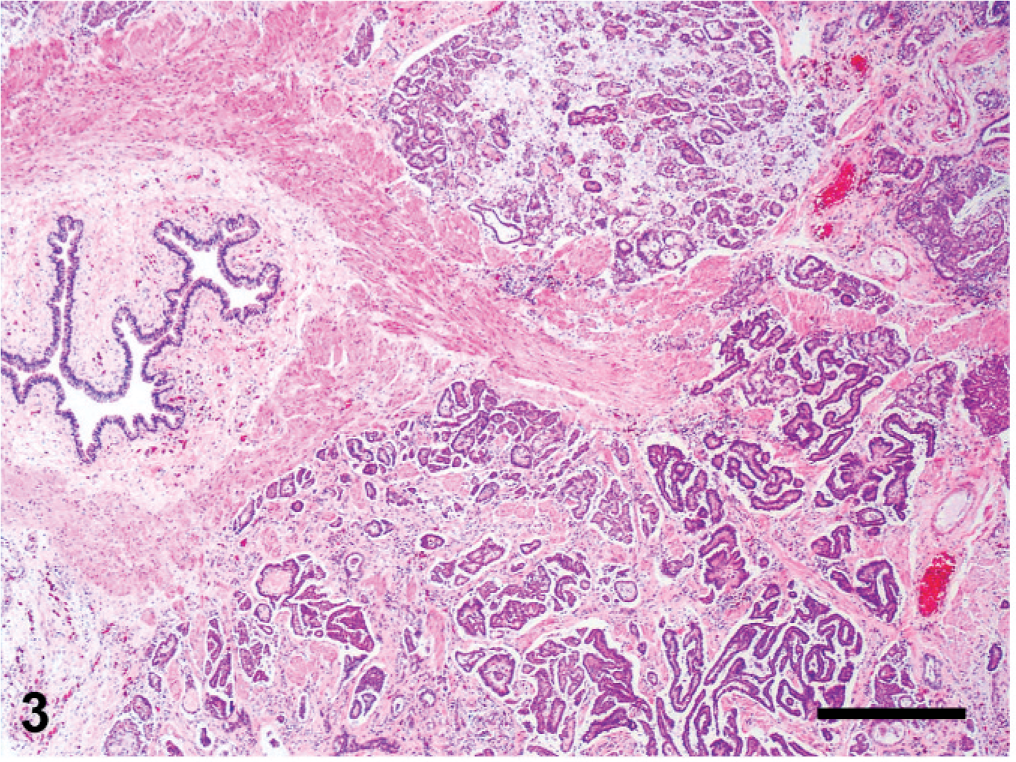
Fig. 3. Oviduct; Northern pine snake (
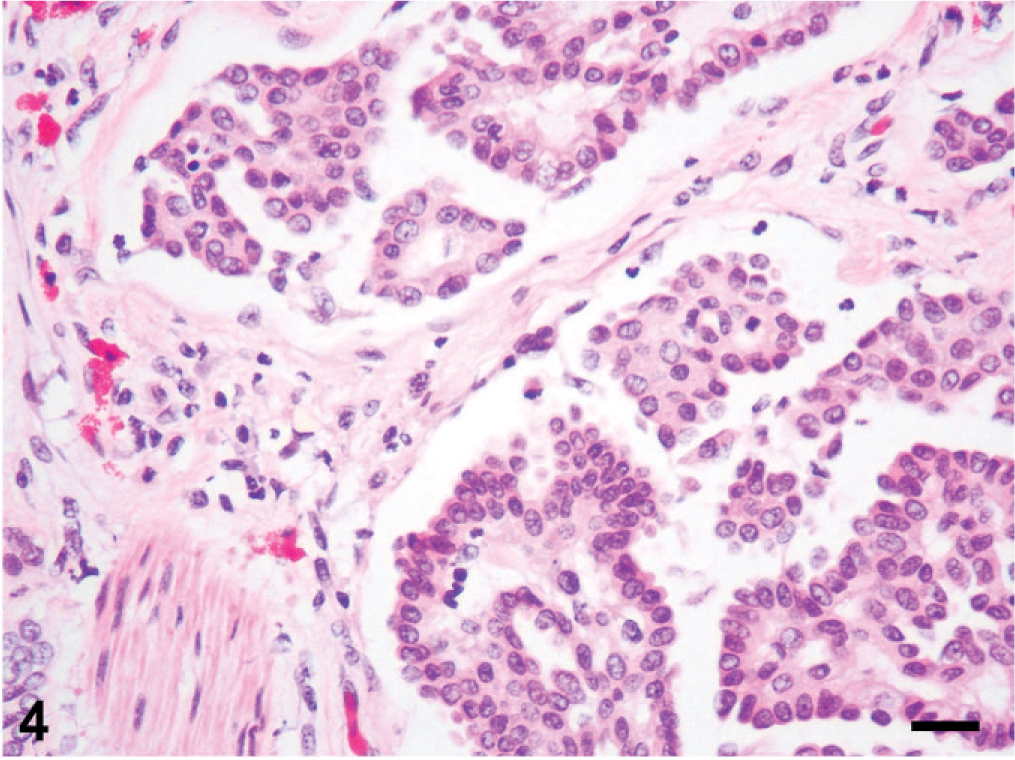
Oviduct; Northern pine snake (
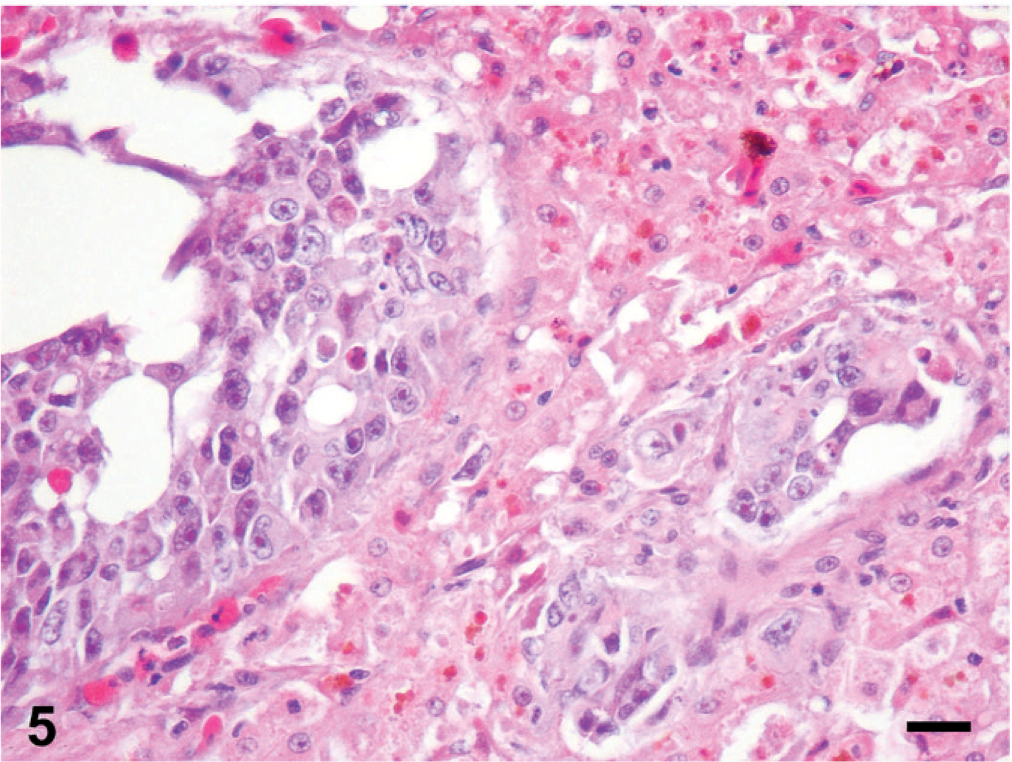
Liver; Emerald tree boa (
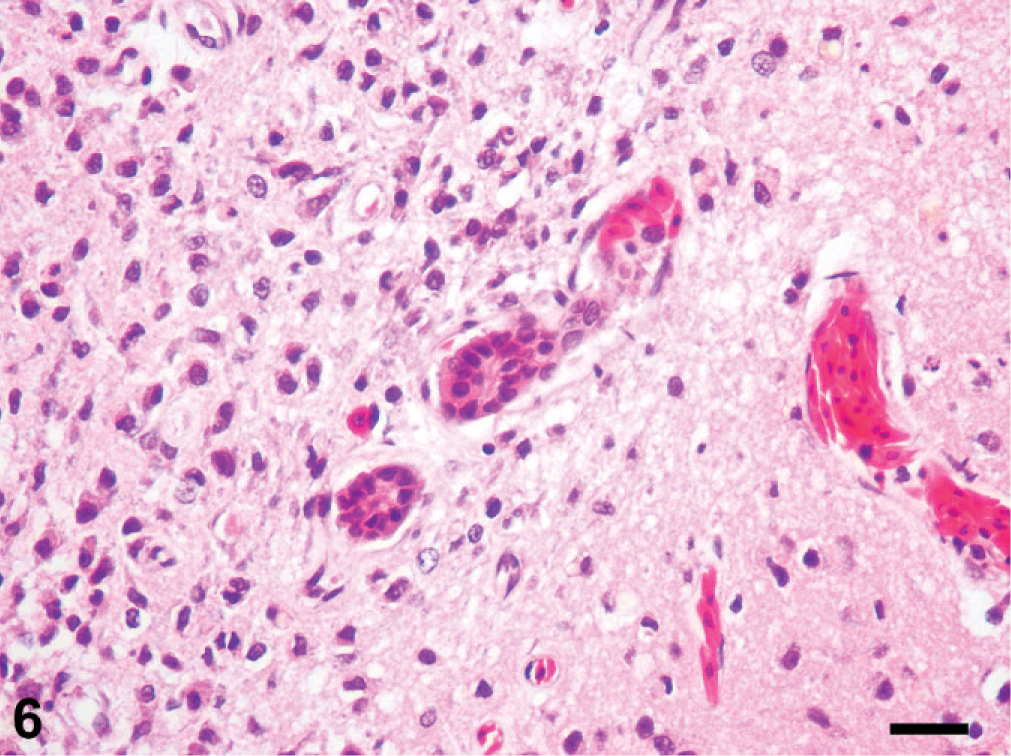
Cerebrum; Northern pine snake (
Grossly, these masses were similar to those described in uterine adenocarcinoma in cows 8 and rabbits. 12 The microscopic papillary feature is comparable with that of oviductal adenocarcinoma in the bitch, mare, 7 and rabbit doe. 12 Although no mass was grossly evident within the oviduct in 2 of these snake cases, the histologic findings were consistent with adenocarcinomas arising from the oviductal wall.
All tumors described here had solid and/or emboli of neoplastic cells to several organs. Previous studies did not find metastases arising from any oviduct adenocarcinoma in snakes. 5 With the exception of rabbit does 11, 12 and hens, tumors of the oviduct are rare in domestic mammals, with few cases reported in the dog and mare. 7, 8 Only 1 study reported venous and lymphatic metastases. 8 In the cases of the present study, several organs had intravascular neoplastic cells and metastatic clusters that occasionally surrounded lymphatic vessels. Hematogenous metastases to the lungs, liver, and sometimes brain and bones may occur within 2 years in rabbits. 11 Hematogenous spread to other viscera is not uncommon in oviductal adenocarcinoma of hens. 13
Uterine mesenchymal tumors are uncommon in domestic animals but are more frequent than uterine epithelial tumors, which are very rare. In marked contrast, uterine carcinomas are frequent in women, most often arising from the body of the uterus. 8 Uterine adenocarcinoma is the most common neoplasm in female rabbits, with an incidence of 80% in does aged 4 to 6 years. 11 In these cases, the adenocarcinoma often involves both uterine horns. 11, 12
Relatively little is known about the cause of oviductal adenocarcinoma in general. In women, chronic salpingitis has been suggested in the literature as a cause of tubal carcinoma. 14 In does, the carcinogenic effect of the estrogens has been implicated but is still conflicting. 12 It is established that higher endogenous beta-oestradiol could naturally increase the risk of oviduct tumors in hens. 1, 13 There is no reference in the literature to the possibility of a retrovirus as the direct cause of reptile oviduct adenocarcinoma, although viruses are associated with intestinal adenocarcinoma, lymphoma, and fibrosarcoma in several species of snakes. 3, 10 Retroviruses are more common in boids, as this group is most susceptible to inclusion body disease. Though no inclusion bodies were seen in any snake in this study, it is possible that retroviruses may be involved in the initiation or growth of oviductal adenocarcinoma. These possibilities warrant further studies into the development of oviductal neoplasia in boids.
Although rare, neurologic complications of tubal adenocarcinoma can develop in humans due to metastases to the brain. 9 Metastasis to the brain is not uncommon in rabbits. 12 The Northern pine snake in this study had a long history of head tilt, and microscopic findings of cerebral emboli of neoplastic cells with neuronal necrosis and gliosis point to ischemia as a likely event. The vascular endothelium of the reptile brain, based on studies on lizards, is similar to that of mammals with respect to very tight intercellular junctions and impermeability to horseradish peroxidase and microperoxidase, 4 which may have prevented a solid tumor in the brain of this animal.
In general, the histologic patterns of the adenocarcinomas in these snakes are very similar to each other and similar to those previously described, 5 but with more extensive necrosis, inflammation, hemorrhage, and desmoplasia. This is the first report in snakes, to our knowledge, of emboli of metastatic cells from oviduct adenocarcinoma to the brain.
