Abstract
A 10-y-old female green tree python (Morelia viridis) died of fungal pneumonia caused by Purpureocillium lilacinum, which was confirmed histologically and by PCR and subsequent DNA sequencing. The same fungal species was cultivated from a swab taken from the terrarium in which the snake was housed. Clinical and environmental P. lilacinum isolates were indistinguishable by the typing method applied, strongly suggesting clonal relatedness of both isolates. Because no other underlying predisposing respiratory infection could be detected by virus-specific PCR or histopathology, P. lilacinum was considered a primary pulmonary pathogen in this tree python.
Purpureocillium lilacinum (formerly Paecilomyces lilacinus) is an ubiquitous, saprophytic, filamentous fungus commonly occurring in soil, decaying vegetation, insects and nematodes, water, indoor air, and various viscous substances. 27 It is best known for its entomopathogenicity and has been widely used as an agricultural biocontrol agent. 9 Despite its moderate virulence, P. lilacinum may act as a primary fungal pathogen 18 causing diseases in various reptiles including chelonians,21,26 crocodilians,8,17,24 and squamates.4,23,24 Its ability to colonize synthetic materials, such as catheters or plastic implants, 16 indicates that the fungus may also have the potential to colonize inorganically furnished racks and terraria. Sampling of inorganic surfaces of the snake’s terrarium was performed as part of our study in order to validate this hypothesis. An accurate identification of the pathogen is crucial for therapeutic approaches because susceptibility to antifungal agents may differ even beyond the species level.3,10 Here we present a fatal pulmonary P. lilacinum infection in a green tree python (Morelia viridis) identified by histopathology, fungal culture, PCR, and subsequent DNA sequencing.
A 10-y-old female green tree python (Morelia viridis) was found dead in its terrarium after a history of lying stretched out on the floor of the terrarium with labored breathing for 5 d. The animal was housed individually in a large polyvinyl chloride terrarium with plastic plants as hide-out and 2 wooden branches as sole organic furnishing. The floor of the container was lined with newspaper that was changed on a regular basis. The lighting consisted of LED lights and fluorescent tubes. Aeration was guaranteed by venting slots in the walls of the terrarium. The necessary humidity was achieved by daily spraying the container with a pressure sprayer using tap water. The receptacle of the sprayer was rinsed regularly with fresh water and a detergent, but the nozzle and supply pipe had not been cleaned for over a year.
At autopsy, the snake was in good nutritional status and but had disseminated, well-circumscribed, soft, yellow, miliary nodules in the lung (Fig. 1). The other organs were macroscopically unremarkable.
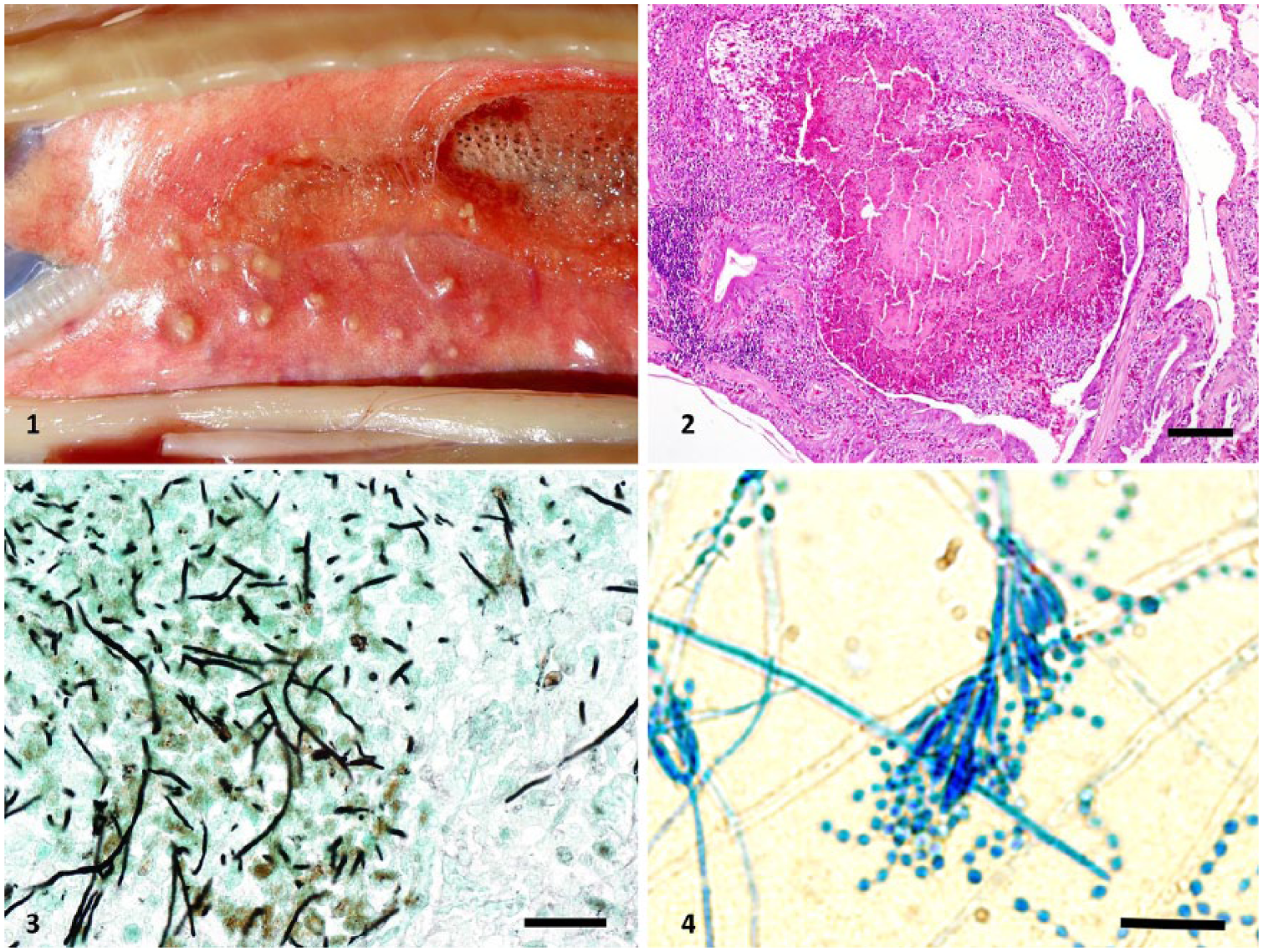
Multinodular granulomatous pneumonia in a green tree python (Morelia viridis) as the result of Purpureocillium lilacinum infection.
Microscopic examination demonstrated that the lung contained dense accumulations of heterophils, histiocytes, and necrotic debris inside the faveolar airways and that the inflammatory process infiltrated the lung trabeculae (Fig. 2). Fungal hyphae were not evident with hematoxylin and eosin (H&E) staining; using Grocott methenamine silver (GMS) and periodic acid–Schiff (PAS) stains, hyphae were visible in large numbers inside the inflammatory exudate and only sparsely infiltrating the lung tissue. The septate irregular hyphae had 45° and 90° branching (Fig. 3). Neither H&E nor special stains revealed reproductive structures, such as phialides and conidia, as signs of adventitious sporulation in tissue samples as described previously.13,26 Thus, histologically the genus of the fungi could not be determined. Lung areas not colonized by the fungus had multifocal interstitial moderate lymphoplasmacytic, histiocytic, and heterophilic infiltration, mild epithelial hyperplasia, and moderate accumulations of edematous fluid, mucus, blood, and heterophils inside the airways. The epicardium was mildly edematous, and the pancreas had moderate subacute lymphoplasmacytic interstitial inflammation with hydropic degeneration of acinar cells.
Lung, liver, kidney, spleen, and brain were tested for multiple viral pathogens including adenovirus, arenaviruses (California academy of science virus, Golden Gate virus, and Colierville virus), nidovirus, paramyxovirus, and reovirus by PCR at a commercial laboratory (Laboklin, Bad Kissingen, Germany). All tests were negative.
Lung tissue and a swab were submitted for microbiologic examination. Several pieces from the plastic plants, wooden branches, and newspaper in the terrarium were sampled, and swabs were taken from the bottom corners of the terrarium as well as the nozzle and pipe of the pressure sprayer. Isolation of bacteria and fungi from specimens was performed as described previously 25 yielding abundant growth of fungi, which were analyzed in detail by a multiphasic approach. After cultivation at 28°C for 5 d on Sabouraud agar with gentamicin and chloramphenicol (SAB, Becton Dickinson, Heidelberg, Germany), the fungal isolate from the lung tissue (isolate 2058) and an additional isolate from the bottom corner (isolate BA1) showed similar colony morphology and thus were re-grown on SAB using the same cultivation conditions. After 5 d of incubation, colonies of both isolates were 20–30 mm diameter consisting of a compact basal felt covered by a floccose aerial overgrowth, with a pale reverse. Initially white, the colonies became slightly purple after prolonged incubation. Microscopically, conidiophores with dense clusters of phialidic cells and chains of ellipsoidal conidia borne from conidiogenous cells were present (Fig. 4).
DNA was extracted from both isolates, and PCR amplification of the ITS1-5.8S-ITS2, the D1 and D2 domains of the 28S rRNA gene (D1-D2), and a fragment of 18S rDNA were performed and subsequently sequenced as described previously.14,25 BLASTn with default settings (http://www.ncbi.nlm.nih.gov/BLAST) 1 was used to compare obtained sequences to nonredundant DNA database sequences. The sequences were aligned with the most similar sequences of established species deposited in GenBank using MEGA v.6 in order to define levels of relatedness. 28 The same software was used for phylogenetic analyses. Maximum-likelihood based on Tamura 3-parameter corrected nucleotide distances obtained from DNA sequence data of 3 loci (D1-D2 of 28S rDNA, 18S rDNA, and ITS1-5.8S-ITS2) with 1,000 bootstraps was used to reconstruct phylogenetic trees. Comparisons with corresponding 18S rDNA, 28S rDNA, and ITS1-5.8S-ITS2 sequences showed that both isolates shared high similarity scores with P. lilacinum strains (Fig 5). The partial nucleotide sequences were deposited in GenBank: 18S rRNA gene, accessions KY410346–KY410347; ITS1-5.8S-ITS2, accessions KY410342–KY410343; and D1-D2, accessions KY410345–KY410346. Additionally, both P. lilacinum isolates (2058, BA1) were typed by randomly amplified polymorphic DNA (RAPD)-PCR employing 5 different primers (5’-GTGGATGCGA-3’, 5’-ACGCGCATGT-3’, 5’-ACGGCCGACC-3’, 5’-AGGCCGCTTA-3’, 5’-AGCGGGCCAA-3’, and 5’-AGCGTCACTG-3’) as described elsewhere. 15 RAPD analysis strongly suggests a clonal relatedness of the clinical and environmental P. lilacinum isolates, as both isolates were indistinguishable by the typing method applied.
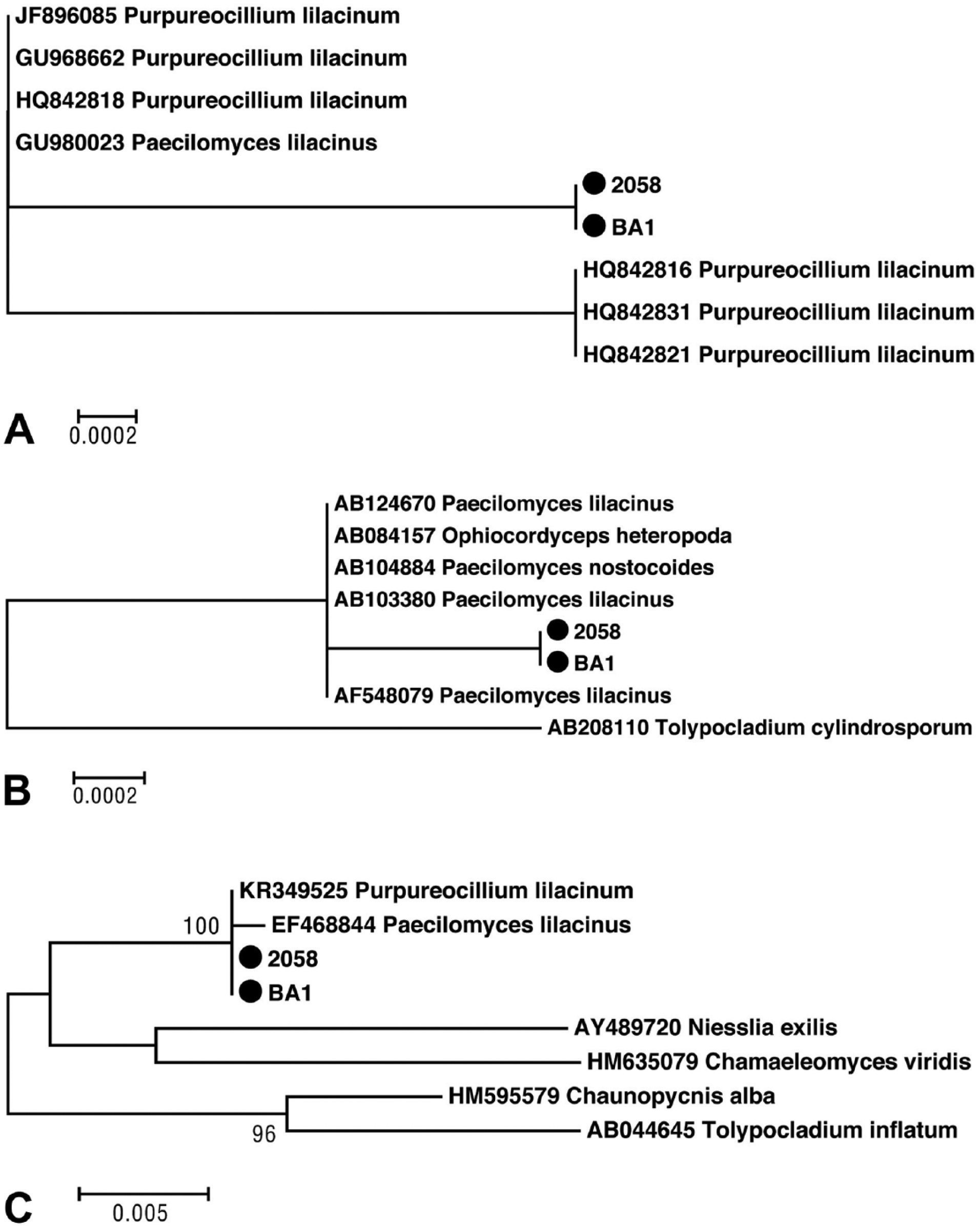
Maximum-likelihood tree for Purpureocillium lilacinum based on Tamura 3-parameter corrected nucleotide distances obtained from partial DNA sequence data of 3 loci:
The minimal inhibitory concentration (MIC) of itraconazole against both P. lilacinum isolates was determined (Etest, BioMérieux, Marcy l’Etoile, France) on casitone agar (Becton Dickinson), in accordance with the manufacturer’s instructions. Agar plates were incubated and read as described elsewhere. 20 Both isolates exhibited MIC values of 16 µg/mL.
In human medicine, hyalohyphomycosis caused by P. lilacinum is recognized as a serious threat to immunocompromised patients, although the number of reported infections in immunocompetent individuals is increasing.2,19,22 Fungal infections in reptiles involving P. lilacinum have been reported in a central bearded dragon (Pogona vitticeps) and a panther chameleon (Furcifer pardalis; Schmidt V, et al. Systemic mycosis in reptiles—an urgent disease problem. Proc 2nd Europ College Zool Med Scientific Meeting; 2013:124; Wiesbaden, Germany), an Aldabra giant tortoise (Aldabrachelys gigantea), 7 a Horsfield’s tortoise (Testudo horsfieldii), 2 Chinese soft-shelled turtles (Pelodiscus sinensis), 12 Fly River turtles (Carettochelys insculpta), 11 a loggerhead sea turtle (Caretta caretta), 26 a hawksbill sea turtle (Eretmochelys imbricata), 21 and a saltwater crocodile (Crocodylus porosus). 17 Mycotic pneumonia was only diagnosed in the panther chameleon and the loggerhead sea turtle, whereas in other cases, the skin (turtles and Horsefield’s tortoise), eye (bearded dragon), liver (Aldabra tortoise), or liver and spleen (crocodile) were affected. Wild-caught reptiles may respond to confinement with chronic stress leading to immunosuppression and subsequently to higher morbidity. Furthermore, high population densities and inappropriate climate situations put the animals at further risk. The snake in our report was captive-born, thrived well, and reproduced in captivity for 10 y. The only contact with conspecifics took place during reproductive season. Histologic examination did not reveal any evidence of immunodeficiency, although presumably unrelated inflammatory processes were present in the pancreas. All PCR tests for viruses yielded negative results. Further, no evidence of viral coinfection was observed by microscopic examination.
In breeding collections, snakes are often kept in terraria with reduced organic furnishing in order to avoid contact of occupants with environmental pathogens. The industry pays respect to these trends by offering a wide variety of naturalistic furnishings. This may lead keepers to the erroneous conclusion that the cleaning of cages is not of upmost importance. The saprophytic fungus P. lilacinum has been shown to be ubiquitous and may be recovered from decaying organic matter and soil, as well as air, water, and even paper. 5 In our case, the fungus was cultured from a swab taken from a bottom corner of the plastic terrarium where humidity may persist for a longer period of time after daily misting. Colonization of the plastic surface of the terrarium is in accord with the reported ability of the fungus to colonize plastic implants and catheters. 16
The snake was in good body condition despite severe granulomatous pneumonia. Microscopically the fungus was present solely in the exudate in the faveolar airways and sparsely infiltrating the lung trabeculae without any histologic evidence of adventitious sporulation, which could have enhanced spread to other organs within the host.13,26 No other organs were affected by the fungus, thus making the airways the most likely portal of entry of the infection. Fungal elements might have been inhaled either during misting or from mold growth in the cage corners. Cultures of swabs taken from the nozzle and the pipe of the pressure sprayer were negative for P. lilacinum. Thus, the source of the infection remains unknown, but it is very likely that environmental management played a role in the development of the infections. We believe that P. lilacinum was a primary pathogen in this tree python.
A number of different therapeutic regimens have been used to treat P. lilacinum infections in humans. Antifungal drugs have been applied as sole treatment or in combination with surgical debridement of an infection site. Cutaneous, subcutaneous, and ocular fungal infections could not be cleared in most cases by the use of older antifungal drugs such as amphotericin B, flucytosine, fluconazole, miconazole, itraconazole, or even novel triazoles.3,19 Low susceptibility was also observed in our case; the determined MIC for itraconazole was 16 µg/mL. Peak concentrations of itraconazole in plasma (2.48 µg/mL), liver (4.27 µg/mL), and muscle (0.63 µg/mL) of spiny lizards after once daily dosing with 23.5 mg/kg for 3 consecutive days 6 were far below the MIC determined for our isolates. In dermal and systemic P. lilacinum infections in Fly River turtles, a combination of malachite green and formaldehyde dip treatment and parenteral itraconazole administration led to successful elimination of the pathogen. 11 A combination of systemic and local drug application (nebulization) might be beneficial for the treatment of P. lilacinum causing pneumonia in snakes. Newer triazole antifungal drugs such as voriconazole, posaconazole, and ruvaconazole seem to be more effective in the treatment of these infections in humans based on clinical and in vitro studies. 19 However, more pharmacologic studies are necessary to elaborate effective treatment protocols in snakes.
Footnotes
Acknowledgements
The entire work was performed by the authors.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
