Abstract
A metastatic intestinal papillary adenocarcinoma was diagnosed histologically in an emerald tree boa (Corallus caninus). Metastasis was detected in the liver, both kidneys, lung, and coelomic wall. Ultrastructural examination of the metastatic intestinal epithelial cells in the liver revealed the presence of a moderate number of viral particles that most closely resembled type A retroviral particles and were mainly associated with granular endoplasmic reticulum membranes. This case is the first description of type A-like retroviral particles in a neoplasm of a snake. The role of the virions in the etiology of the intestinal adenocarcinoma is uncertain. In addition, this is the first confirmed report of a metastatic intestinal adenocarcinoma in a snake.
Neoplasms of the gastrointestinal tract in snakes are considered uncommon. 7,14 Neoplasms of unspecified intestinal origin have been reported in a canebrake rattlesnake (Crotalus horridus atricaudatus), a timber rattlesnake (Crotalus horridus horridus), and a black rat snake (Elaphe obsoleta). 19,20 Several neoplasms of colonic origin have also been reported: a carcinoma in a reticulated python (Python reticulatus), 10 and four colonic adenocarcinomas in a rat snake (E. obsoleta), 18 a bull snake (Pituophis sayi), 5 a corn snake (Elaphe guttata guttata), 11 and in a Burmese python (Python molurus bivittatus). 2 To date, occurrence of metastasis from carcinomas of the reptilian lower alimentary tract has not been confirmed.
Several retroviruses have been reported in snakes with neoplasms. Morphologically, almost all these retroviruses were type C virions. In snakes, intracytoplasmic type A retroviral particles have only been described in a spleen cell line from a Russell's viper (Vipera russelli) bearing a myxofibroma, 12 and in the venom glands of apparently healthy Brazilian Jararacussu vipers (Bothrops jararacussu). 1 Here, we describe a metastatic intestinal adenocarcinoma in an emerald tree boa (Corallus caninus) in which type A–like retroviral particles were detected.
A free-ranging, 1.2-kg adult female emerald tree boa (C. caninus) was imported in 1999. Since then it was one of a reptile collection exhibited at the Zoo Aquarium, Madrid. The first sign of overt clinical disease in this snake was a reduced appetite, refusing the last two meals. Several weeks later, the snake was discovered on the floor of the terrarium, showing a cloacal prolapse. On physical examination, the snake was severely dehydrated and depressed. The prolapsed tissue was firm and partially necrotic. The prolapse was reduced by external pulsion, and the snake was treated with enrofloxacin (5 mg/kg, intramuscularly) and rehydrated with lactated Ringer's solution (25 ml/kg, intracutaneously). Palpation revealed a firm mass, 7–10 cm cranial to the cloaca. The snake was prepared for an exploratory laparotomy, but it died 2 hours later.
At necropsy, a severe intestinal intussusception was observed 10 cm cranially to the cloaca. The intestinal wall immediately caudal to the intussusception was very thick, and palpation of the intestinal wall revealed a firm mass (4 mm in diameter) (Fig. 1). At the same level, a firm, round (5 mm in diameter) mass was observed adhered to the intestinal wall. Several firm, round (1–4 mm in diameter), white nodules were observed in the liver (Fig. 2) and both kidneys. No gross lesions were observed in other organs.
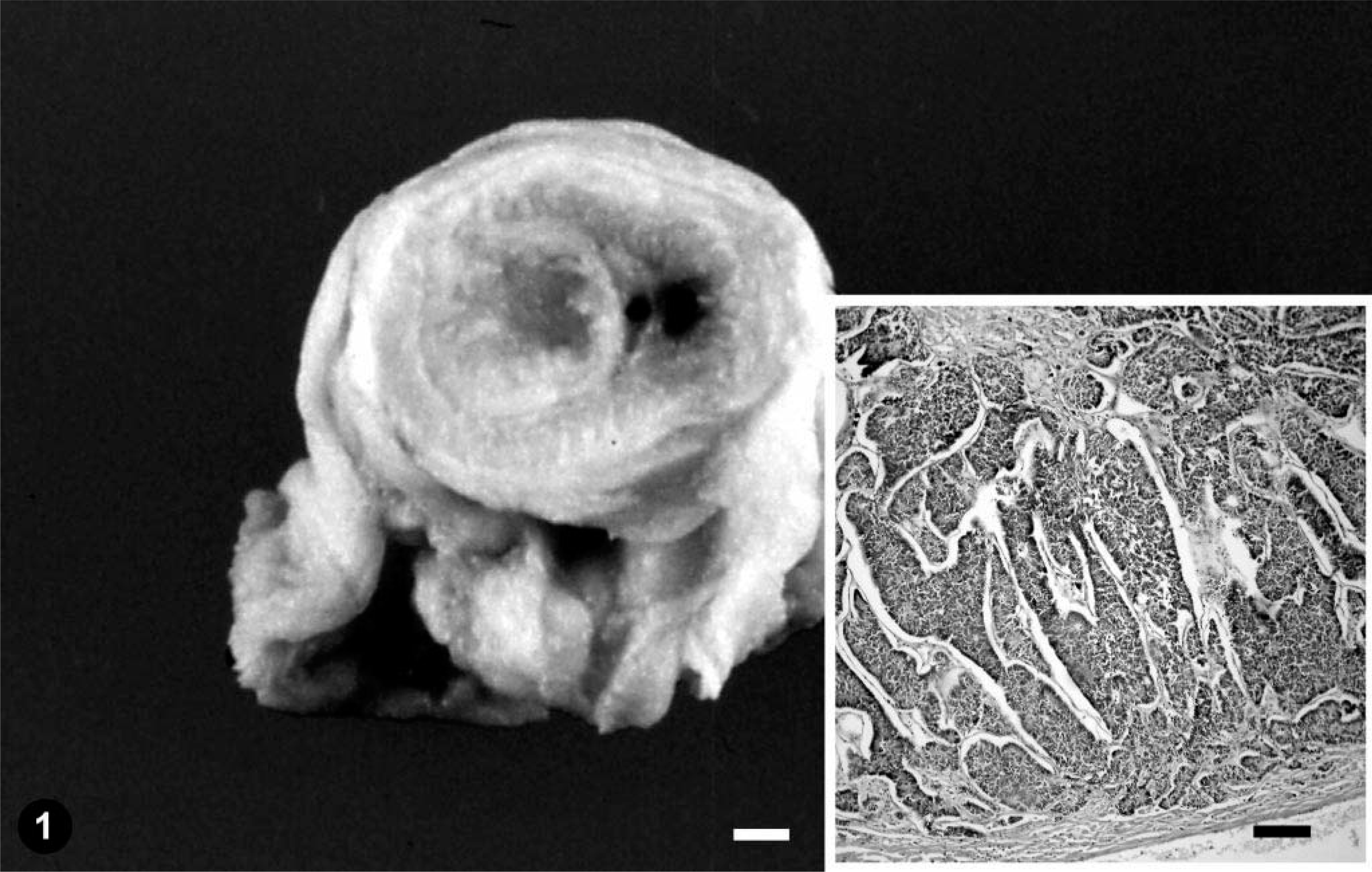
Intestine; emerald tree boa. Gross section of the primary neoplasm. Bar = 0.15 mm. Inset: proliferation of pleomorphic epithelial cells densely arranged in irregular papillae and solid cords. HE. Bar = 300 µm.
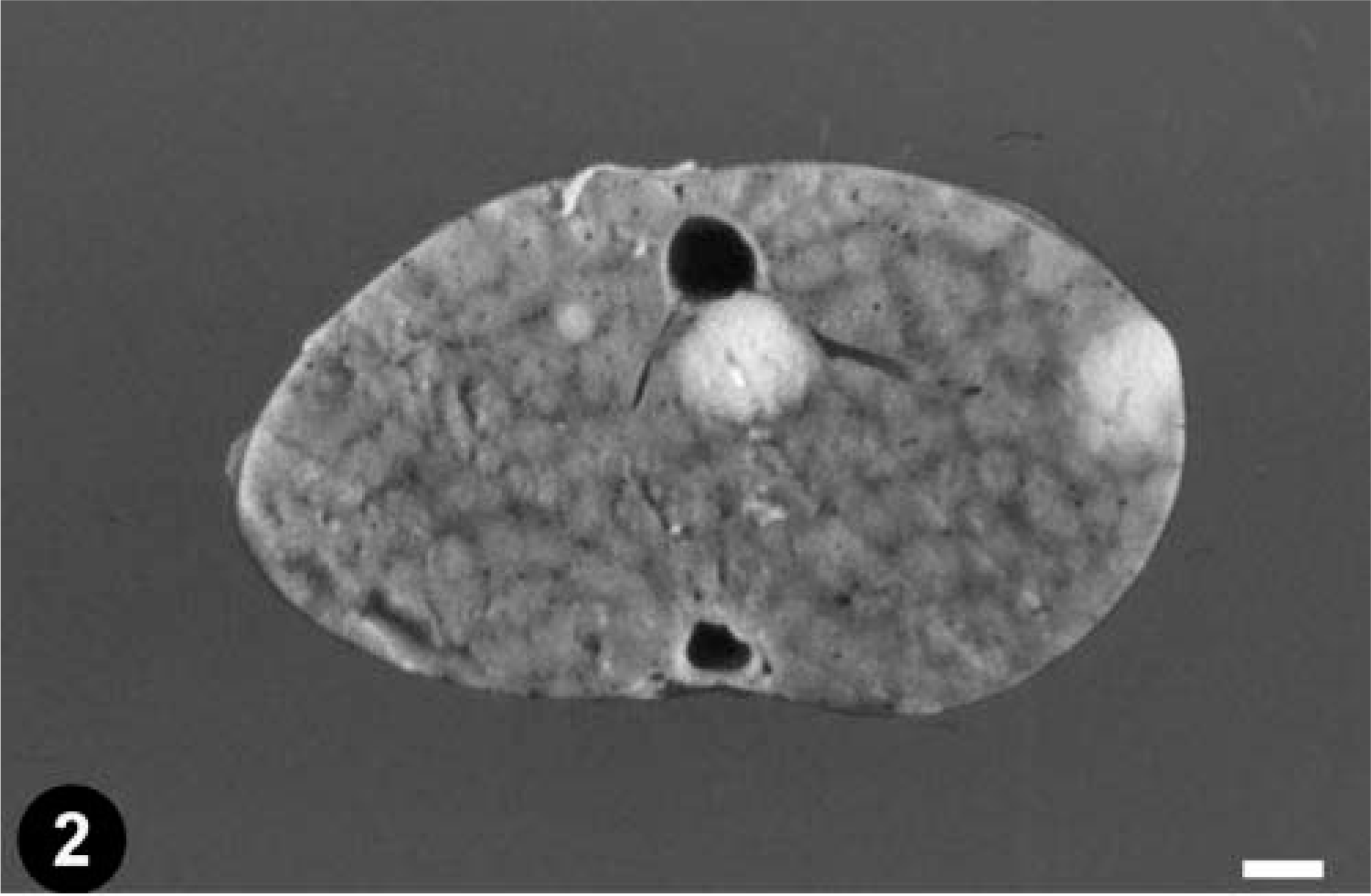
Liver; emerald tree boa. Gross section with two metastases. Bar = 2.5 mm.
Samples from small intestine, large intestine, coelomic wall, heart, liver, kidneys, lung, pancreas, and spleen were collected, fixed in neutral buffered 10% formalin, and sent to the Veterinary Faculty, University of Las Palmas de Gran Canaria, for histopathologic studies. Samples were routinely processed, embedded in paraffin, sectioned at 4 µm, and stained with hematoxylin and eosin (HE). Selected formalin-fixed tissues from the liver were cut into 1-mm3 blocks, fixed in 2.5% buffered glutaraldehyde, routinely processed for transmission electron microscopy, and embedded in epoxy resin. Ultrathin sections were mounted on grids, stained with uranyl acetate and lead citrate, and examined on a Zeiss 910 transmission electron microscope. Retrospective ultrastructural examination was not conducted on samples from the intestine and kidneys because both tissues were not fixed adequately.
Histologically, a severe necrotizing enteritis was observed in the intussuscepted intestinal tract. The mucosa at the end of the small intestine immediately caudal to the intussuscepted intestinal tract had fused crypt glands with proliferation of pleomorphic epithelial cells densely arranged in irregular papillae and solid cords that extended from the mucosa to the submucosa (Fig. 1, inset). Necrosis was observed in some of the solid cords of pleomorphic epithelial cells. The nuclei of the anaplastic cells were oval, and numerous mitotic figures were observed. Individual cells had a moderate amount of eosinophilic cytoplasm. No intracellular mucus production was observed. The neoplasm was consistent with intestinal papillary adenocarcinoma. Microscopic examination of the liver revealed a diffuse vacuolar degeneration of hepatocytes and several scattered nests of pleomorphic epithelial cells arranged in irregular papillae (Fig. 3). Individual epithelial cells had characteristics similar to those described for the intestinal adenocarcinoma. Numerous mitotic figures were detected (Fig. 3, inset). No inflammatory reaction was observed around the tumor nests. Similar nests of tumoral cells were observed in kidneys, lung, and coelomic wall. A diagnosis of intestinal papillary adenocarcinoma with metastasis to the liver, kidneys, lung, and coelomic wall was made.
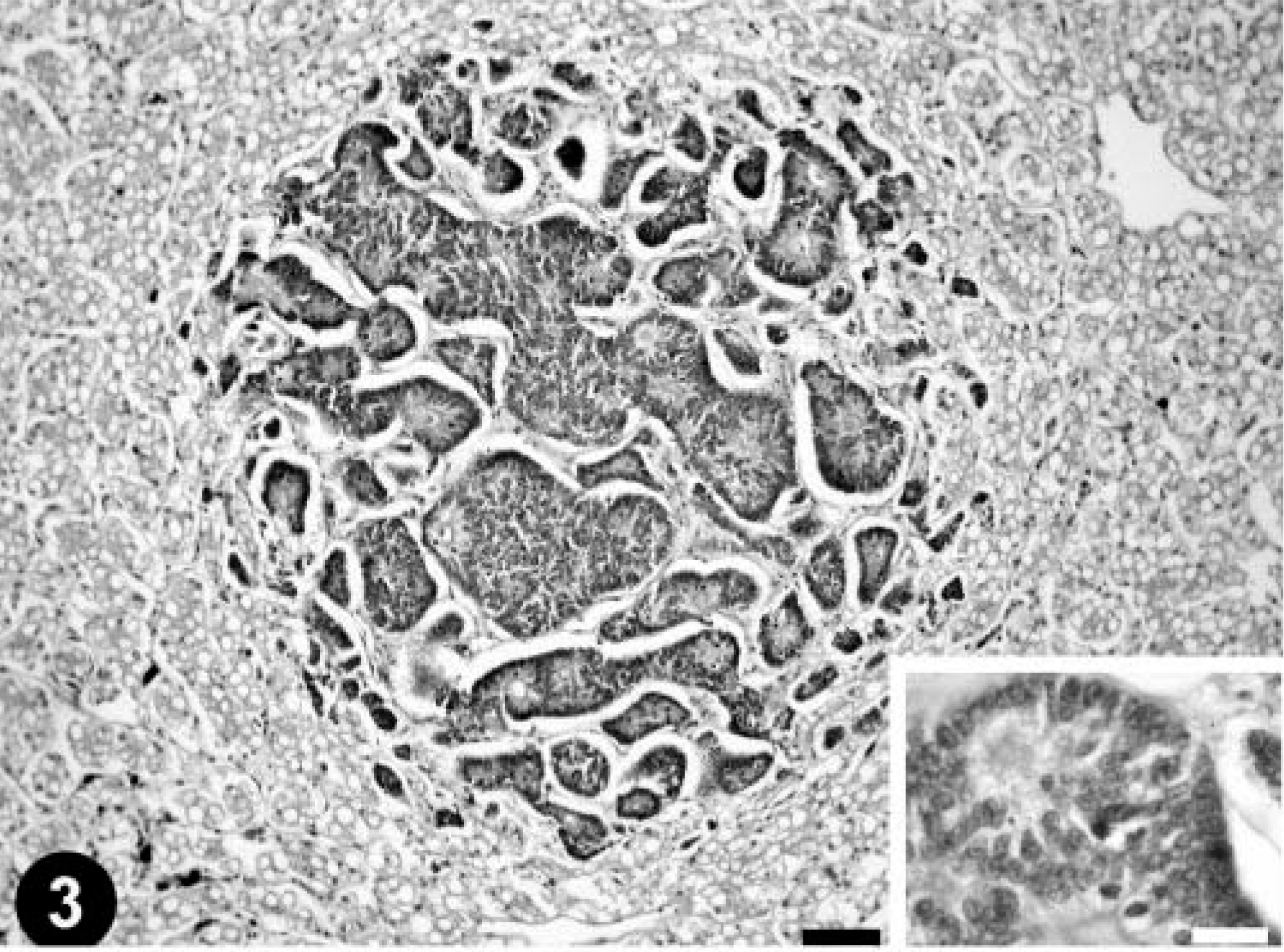
Liver; emerald tree boa. Metastatic intestinal papillary adenocarcinoma. HE. Bar = 250 µm. Inset: Three mitotic figures can be observed in this field. HE. Bar = 15 µm.
Ultrastructural examination of the metastatic intestinal epithelial cells in the liver revealed the presence of a moderate number of viral particles mainly associated with granular endoplasmic reticulum membranes (Fig. 4). Individual particle profiles measured 70 to 80 nm in total diameter and consisted of a central moderately electron-dense core 27 to 30 nm in diameter surrounded by a markedly electron-dense ring. Electron-dense granules apposed on the surface of the particles were identified as ribosomes. Morphologically, the virions were consistent with those belonging to the family Retroviridae. Ultrastructural examination of nonneoplastic hepatic tissue from this snake did not reveal viral particles.
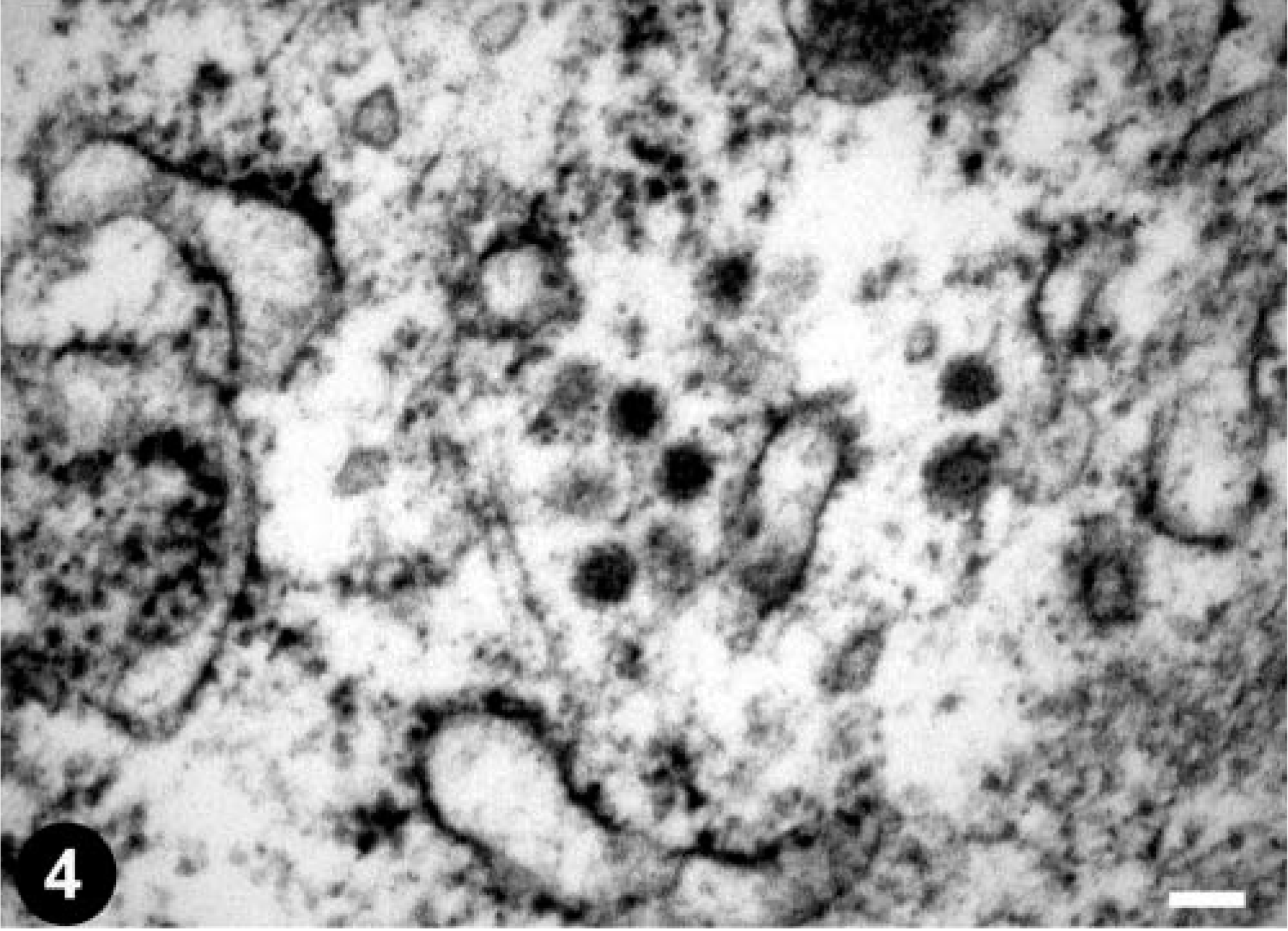
Electron micrograph. Liver; emerald tree boa. Type A–like retroviral particles in a neoplastic epithelial cell from the central hepatic metastasis shown in Fig. 2. Uranyl acetate and lead citrate. Bar = 0.1 µm.
The particles observed in the metastatic intestinal epithelial cells in the liver most closely resemble type A retroviral particles. Type A particles were first viewed as the immature intracellular forms of mouse mammary tumor virus, and the term is still reserved for strictly intracellular structures, comprising fully formed immature cores of B-type viruses, D-type viruses, and spumaviruses. 4 In the snake used in this study, they were never seen budding from plasmalemma or endomembranes, but they were closely associated with endoplasmic reticulum membranes. Similar type A retroviral particles were described in the venom glands of healthy Brazilian Jararacussu vipers 1 and in a spleen cell line from a Russell's viper (V. russelli) bearing a myxofibroma, 12 but no viruses were found within the neoplasm of the tumor-bearing viper. 23,24 Our case is the first description of type A–like retroviral particles in a tumor of a snake. The role of the virions in the etiology of the intestinal adenocarcinoma is uncertain. Similar structures are sometimes seen in human tumors and cell lines and are the products of one to more endogenous proviruses. 22
Neoplasms associated with retroviruses have been well documented in some fish populations. 6,15,16,21 Retroviruses were first detected in ectothermic species during ultrastructural examination of a spleen cell line from a Russell's viper (V. russelli) bearing a myxofibroma. 23,24 Isolation and characterization of type C particles from a rhabdomyosarcoma of a corn snake (E. guttata) have been described. 3,13 Type C retroviral particles have been detected in neoplasms from several snakes, including a boa constrictor (Boa constrictor) with erythroleukosis, 9 Brazilian lancehead vipers (Bothrops moojeni) with renal tumors, 8 a four-lined chicken snake (Elaphe obsoleta quadrivittata) with lymphoid leukemia, 25 and recently, four Burmese pythons (P. m. bivittatus) from a single collection that had a lymphosarcoma in the oral cavity, a colonic adenocarcinoma, a renal carcinoma, and an inter-mandibular fibrosarcoma. 2
However, although the identification of retroviral particles in several species of snakes suggests an etiologic relationship between the retroviral particles and neoplasms, the role of the retroviral particles in the etiology of the tumors is uncertain. Neither in vivo transmission experiments nor in vitro assessment of the transforming capacity of these retroviruses have been performed. 17 In addition, retrovirus-like particles have also been described in the venom glands of vipers without tumors. 1
The differential diagnosis for palpable enlargement of the distal intestine in snakes includes fecal impaction, foreign body, bacterial, fungal or parasitic granulomata, intussusception, neoplasia, urolithiasis, and egg impaction in females. 11 Prolapse of the colon is generally secondary to tenesmus. Intestinal intussusception was detected in this case; it occurred secondary to stricture of the intestinal lumen from thickening of the bowel wall by neoplastic cell infiltration.
This is the first report of a metastatic intestinal adenocarcinoma in a snake. To date, carcinomas of the reptilian lower alimentary tract had not been reported to metastasize, and consequently, surgical resection has been the treatment of choice. However, the snake used in this study had numerous metastasis to the liver, both kidneys, lung, and the coelomic wall. Before the surgical resection of intestinal carcinomas in snakes, evaluation of the hepatic and renal function, ultrasonography, or laparotomy may be advisable to detect visceral metastasis.
Footnotes
Acknowledgements
We would like to thank P. Castro, Department of Morphology, University of Las Palmas de Gran Canaria, for technical assistance, and S. Tucker and Dr. E. R. Jacobson, College of Veterinary Medicine, University of Florida, for their help with the bibliographic research.
