Abstract
A 2-year-old, male Japanese native fowl (
Keywords
Histologically, each peripheral nerve (PN) consists of axons, Schwann cells that form myelin sheaths around some axons, the endoneurium, perineurium, and epineurium. Myelinated and unmyelinated axons are individually wrapped by the endoneurium and are bound into a nerve fascicle by the perineurium. The perineurial cells that form this layer resemble fibroblasts but are 3-dimensionally flattened, polygonal, or squamous, and form many concentric layers around the axons, surrounded by a discontinuous basement membrane. Neoplasms that originate from the perineurium (perineuriomas) have rarely been described in humans 17 and are classified into 2 types, intraneural perineurioma and extraneural/soft tissue perineurioma. 8, 16 The former is grossly characterized by cylindrical enlargement of peripheral nerves and histopathologically by concentric proliferations of perineurial cells around axons, forming onion bulb–like structures. 28 In the veterinary literature, intraneural perineuriomas have only been reported in 3 dogs, 7, 12, 18 one horse, 29 and 11 chickens. 27 Because avian perineuriomas were recognized only in chickens experimentally inoculated with fowl glioma-inducing virus (FGV), a subgroup A avian leukosis virus (ALV-A), the neoplasm in this species may be associated with ALV infection. Although epithelial membrane antigen (EMA) 1, 25 and claudin1 9 are used as immunohistochemical markers of human perineurial cells, perineurial cells of domestic animals are negative for these antigens. 12, 18, 27 Recently, glucose transporter 1 (GLUT1) has also been used as a marker of human perineurium, 13 but GLUT1 expression has not been evaluated in perineuriomas of domestic animals. This paper describes naturally occurring intraneural perineuriomas in a Japanese native fowl.
A 2-year-old, male Japanese native fowl of the Kojidori breed (
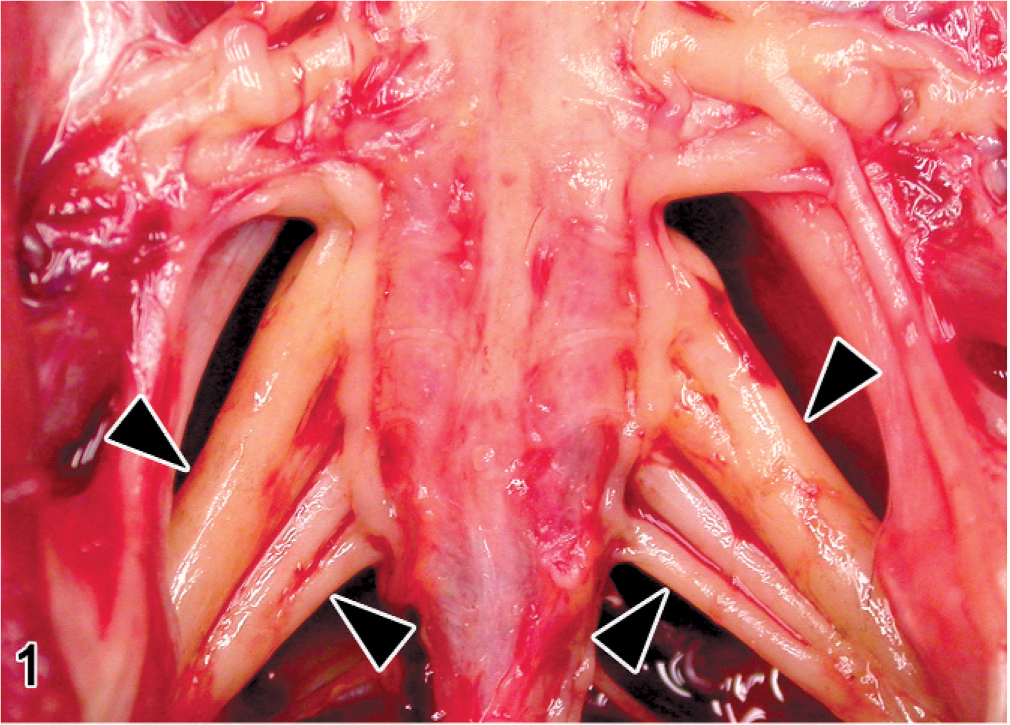
Lumbosacral plexus; Japanese native chicken. Bilateral cylindrical enlargements (arrowheads) of peripheral nerves.
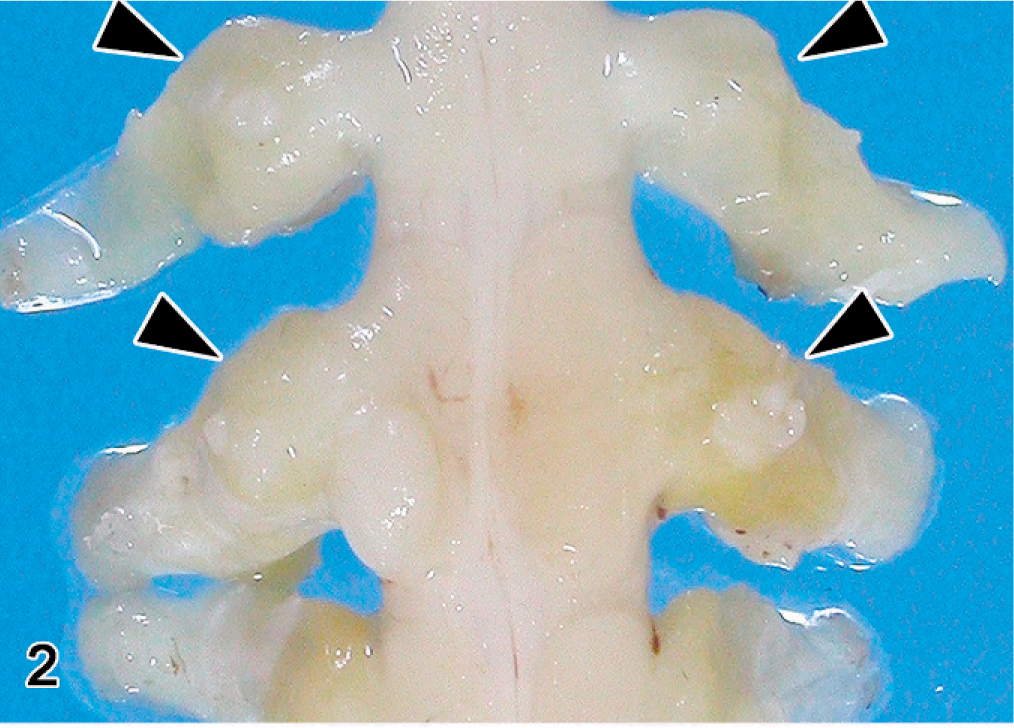
Brachial plexus and spinal ganglia; Japanese native chicken. multiple enlarged peripheral nerves (arrowheads). Formalin fixed.
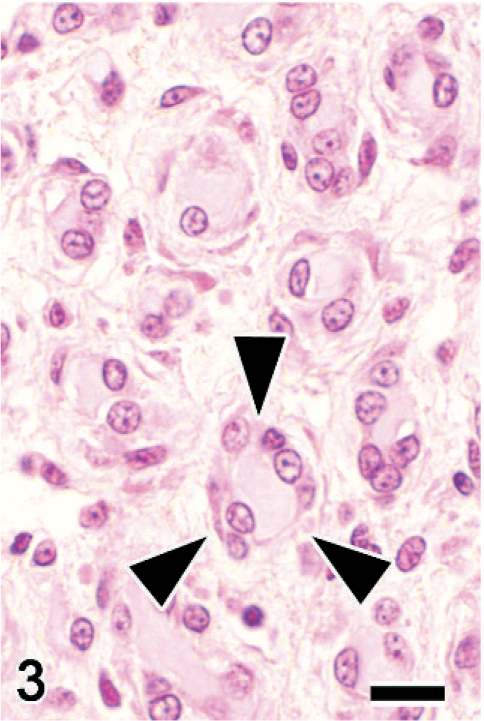
Sciatic nerve; Japanese native chicken. Cross section. Onion bulb–like structures (arrowheads) that are composed of concentric periaxonal spindle cell proliferations. HE. Bar = 10 μm.
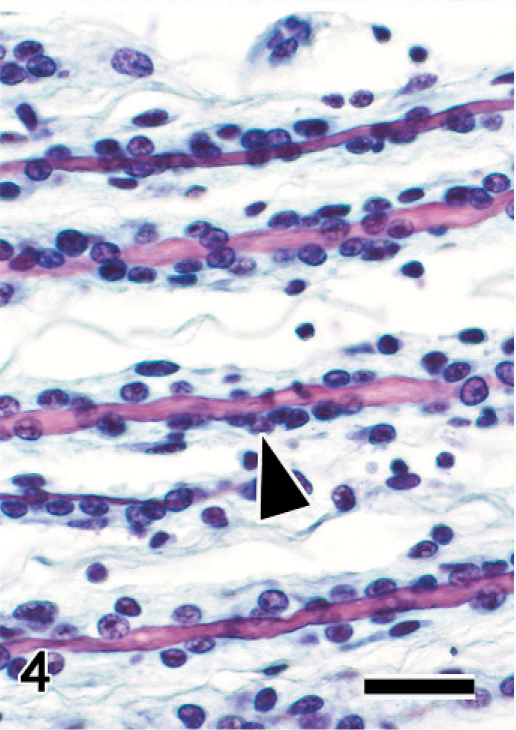
Sciatic nerve; Japanese native chicken. Longitudinal section. Spindle cells proliferate around axons (arrowhead). Bodian-Luxol fast blue double stain. Bar = 20 μm.
Immunohistochemistry was performed by the streptavidin-biotin peroxidase method, with a commercial kit (Nichirei Corp., Tokyo, Japan) as previously described. 27 The primary antibodies used in this examination were S-100 α/β (diluted 1 : 2000, DAKO, Glostrup, Denmark), neuron-specific enolase (NSE; 1 : 200, DAKO Corp., Carpinteria, CA), neurofilament protein (NFP; 1 : 100, DAKO), cytokeratin MNF116 (1 : 10, DAKO), vimentin (1 : 100, DAKO), desmin (1 : 100, DAKO Corp.), muscle actin (HHF35; 1 : 3, ENZO Diagnostics Inc., NY), claudin1 (1 : 50, Zymed Laboratories, Carlsbad, CA), and GLUT1 (1 : 400, Chemicon, International Inc., Temecula, CA). The spindle cells were positive for GLUT1 (Fig. 5) and negative for S-100 α/β, NSE, NFP, cytokeratin, vimentin, desmin, claudin1, and muscle actin (HHF35) (Table 1). Reactivity of S-100 α/β proteins was detected only in residual Schwann cells, whereas axons were positive for NSE and NFP. In cross section, a layer of S-100 α/β–positive Schwann cells covered residual axons.
Results obtained from immunohistochemical staining in normal peripheral nerves and neoplastic cells.∗

NSE = neuron specific enolase; NFP = neurofilament protein; MNF116 = cytokeratin MNF116; HHF35 = muscle actin (HHF35); GLUT1 = glucose transporter 1.
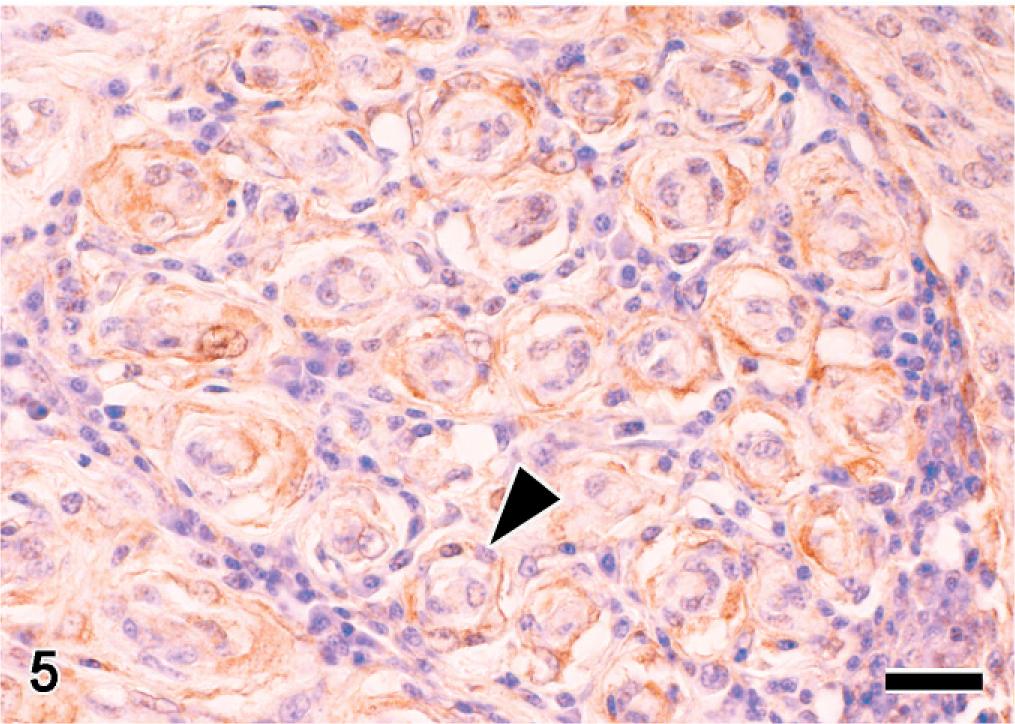
Spinal ganglion (T1); Japanese native chicken. Cross section. Immunohistochemistry for GLUT1. Spindle cells are positive for this antigen (arrowhead). Streptavidin-biotin (SAB) -DAB-H2O2, hematoxylin counter stain. Bar = 20 μm.
Nested polymerase chain reaction (PCR) for FGV was performed on the affected PNs and the brain stored at −80°C. Primer sets for FGV were designed to amplify the 135 base pair (bp) segment specific for the 3′UTR of the viral genome (GeneBank accession number AB112960).
11,
26
For positive and negative controls, total cellular DNA was extracted from FGV-inoculated and uninoculated DF-1 cells, respectively (CRL-12203; American Type Culture Collection, Manassas, VA). The PCR mixture was heated at 98°C for 1 minutes and then subjected to 25 cycles of PCR amplification (98°C for 10 seconds, 60°C for 15 seconds, and 72°C for 30 seconds) by using a Gene Amp PCR System 9700 (Applied Biosystems, Foster City, CA) in max ramp speed mode. In the second reaction, the extension time was changed to 15 seconds with 30 cycles. PCR for β-actin was performed to assure the quality of extracted DNA. The β-actin-specific fragment was detected in the examined PNs and brain, but the specific 135-bp DNA fragment of FGV was not amplified from any examined nervous tissues. To eliminate any possible relationship between the PN lesions and Marek's disease virus 1 (MDV1) infection, PCR was performed on the same materials according to the method of Chang et al.
6
The positive control was DNA extracted from MDV1-inoculated chicken embryo fibroblasts (CEF) that were obtained from the commercial SPF White Leghorn strain WL-M/O (Nippon Institute for Biological Science, Yamanashi, Japan). The MDV-1 specific
In addition, reverse transcription PCR (RT-PCR) for ALV was performed. RNA was extracted by using the PureLink Micro-to-Midi Total RNA Purification System (Invitrogen, Carlsbad, CA), according to the manufacturer's instructions. The primers were designed to target the hypervariable region in the gp85 coding region of the
The histologic findings in this fowl were consistent with those of the multiple perineuriomas previously described by Toyoda et al. 27 in FGV-infected chickens. The differential diagnosis includes Schwannomas and neurofibromas. Schwannomas consist of tumors composed of Schwann cells that are arranged in streams of elongated spindle cells. Neurofibromas are characterized by proliferation of fibroblasts or perineurial cells in addition to Schwann cells. 22 Perineurial cells in humans are immunohistochemically positive for EMA, claudin1, and GLUT1. However, perineurial cells of domestic animals, including chickens, were negative for EMA in previous reports. 12, 18, 27 In this report, immunohistochemistry of PNs for GLUT1 was investigated in a normal chicken and the affected chicken. Perineurium was positive for GLUT1 in both the normal and affected PNs. There was also positive reactivity in vascular smooth muscle, endothelial cells in small capillaries and ganglion cells. In the human, GLUT1 is occasionally detected in blood vessels in PNs and ganglion cells, as well as perineurium. 15 These findings suggest GLUT1 may be a useful marker of perineurial cells in chickens.
As mentioned in a previous report, 27 several conditions in chickens are grossly marked by swollen PNs. Swollen PNs are a common gross finding in classical Marek's disease (MD), 5, 20 but PNs in MD microscopically contain either massive accumulations of neoplastic T lymphocytes; accumulations of lymphocytes and plasma cells, with demyelination and Schwann cell proliferation; or much milder lymphocyte accumulations. 23 Histologic lesions similar to those seen in some cases of MD are rarely noted in idiopathic polyneuritis 4 and in broiler chickens experimentally infected with ALV-J. 21 Peripheral neuropathies that consist of severe edema with lymphocytes and plasma cells in nerve fascicles have also been described. 2 However, all of these PN lesions lack the characteristic onion bulb–like proliferations seen in the current case.
In some cases, FGV may be associated with avian perineurioma, because 11 chickens inoculated with FGV acquired this neoplasm.
27
In the chicken that was the subject of this report, FGV proviral DNA was not detected in the brain or PNs by nested PCR but a 2.3-kbp fragment equivalent to the
This report presents the first case of naturally occurring multiple perineuriomas in a chicken, which potentially indicates that avian perineuriomas can occur in the field as well as in the laboratory. ALVs are still a candidate for the causal agent of this rare neoplasm in chickens.
Footnotes
Acknowledgements
This work was supported in part by Grants-in-Aid for Scientific Research (B: 16405033 and B: 18380179) from the Ministry of Education, Culture, Sports, Science and Technology of Japan.
