Abstract
Intraneural perineurioma is an extremely rare condition characterized by perineurial cell proliferation within peripheral nerve (PN) sheaths. In the veterinary field, this entity has been reported only in a dog. We examined multiple enlargements of PNs in 11 chickens (Gallus gallus domesticus) (9 Japanese bantams and 2 specific pathogen-free White Leghorn), which were inoculated with an avian leukosis virus (ALV) causing so-called fowl glioma. All chickens clinically exhibited progressive leg paralysis. Lumbosacral plexus, brachial plexus, and/or spinal ganglion were commonly affected, and these nerves contained a diffuse proliferation of spindle cells arranged concentrically in characteristic onion bulb-like structures surrounded by residual axons and myelin sheaths. The spindle cells were immunohistochemically negative for S-100α/β protein. Electron microscopy revealed that these cells were characterized by short bipolar cytoplasmic processes, occasional cytoplasmic pinocytotic vesicles, and discontinuous basal laminae. These features are consistent with those of intraneural perineurioma. Furthermore, the specific sequence of the ALV was detected in the PN lesions of 8/11 (73%) birds by polymerase chain reaction. These results indicate that the multiple intraneural perineuriomas of chicken may be associated with the ALV-A causing fowl glioma.
Keywords
Perineurium is one of the basic components of peripheral nerves (PNs). 5 Each nerve fascicle consists of myelinated or unmyelinated axons, or both, wrapped by endoneurium, with each fascicle contained by perineurium, and multiple fascicles bound into a PN encased by epineurium. The perineurium is formed by concentric layers of perineurial cells, which histologically resemble fibroblasts but are three-dimensionally flattened, polygonal, and surrounded by a discontinuous basement membrane. 33
Perineuriomas have been described in humans as rare neoplasms derived from perineurial cells 14, 15 and classified into two entities: 1) an extraneural soft tissue type 13, 24, 25 and 2) an intraneural type grossly characterized by segmental cylindrical PN enlargement. 36 Several authors consider the latter to be a benign intraneural neoplasm, 3, 10, 17, 21 whereas others reported the lesion as a localized hypertrophic neuropathy regarded as a reactive change to nerve injury. 22, 37 Thus, the designation as a true neoplasm is still controversial. 23 In the veterinary field, this entity has previously been described only in one canine. 8
We have previously reported that an avian leukosis virus (ALV), which belongs to subgroup A, causes so-called fowl glioma in Japanese bantam and specific pathogen–free (SPF) chicken. 19, 26, 34, 35 Japanese bantam (Gallus gallus domesticus) is one of ornamental chicken breeds. In these experiments, we encountered an unusual PN lesion in nine of 203 (4.4%) bantams and two of 27 (7.4%) SPF chickens inoculated with a homogenate of brains affected with fowl glioma or viral suspension. This report describes the characteristic morphology of the proliferative lesions of PNs, which was consistent with intraneural perineurioma.
Materials and Methods
The proliferative lesions of PNs were obtained from nine Japanese bantams and two SPF chickens used for the transmission experiment of so-called fowl glioma (Table 1). 19
Clinical summary and degree of proliferation of spindle cells in lumbosacral plexus and brachial plexus of 11 chickens inoculated with an avian leukosis virus causing fowl glioma.
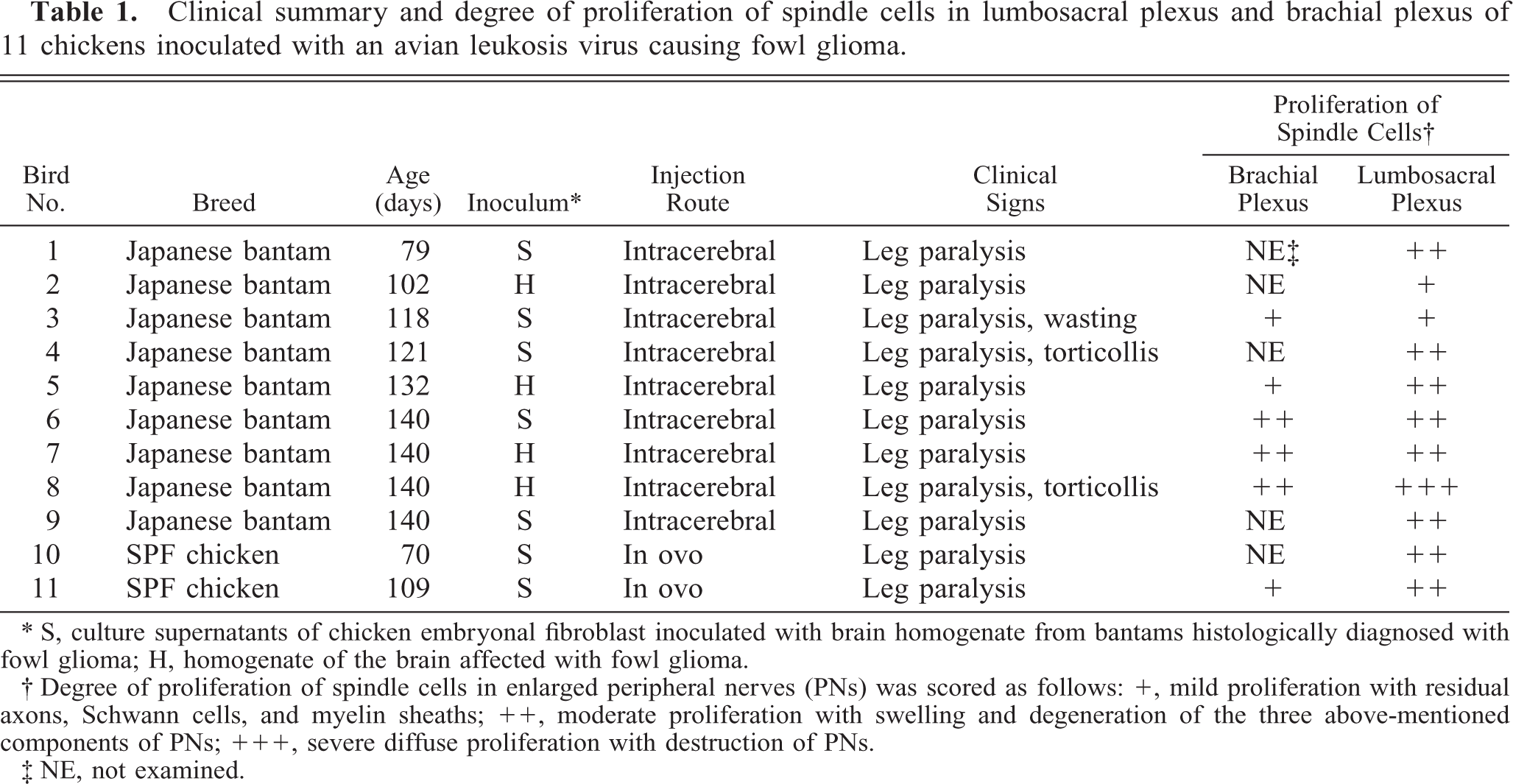
∗ S, culture supernatants of chicken embryonal fibroblast inoculated with brain homogenate from bantams histologically diagnosed with fowl glioma; H, homogenate of the brain affected with fowl glioma.
† Degree of proliferation of spindle cells in enlarged peripheral nerves (PNs) was scored as follows: +, mild proliferation with residual axons, Schwann cells, and myelin sheaths; ++, moderate proliferation with swelling and degeneration of the three above-mentioned components of PNs; +++, severe diffuse proliferation with destruction of PNs.
‡ NE, not examined.
Virus
Fertile eggs of the commercial SPF White Leghorn strain WL-M/O were purchased from Nippon Institute for Biological Science (Yamanashi, Japan). Chicken embryo fibroblasts (CEF) were prepared from 10-day-old SPF chicken embryos of the WL-M/O (C/O) strain. This strain lacks both chicken helper factor and group-specific antigen and is susceptible to ALV subgroups A to E (International registry of poultry genetic stocks, Bulletin 476, March 1988, Ralph G. Somes Jr, PhD, University of Connecticut). The presence of endogenous virus genes other than chicken helper factor and group-specific antigen in this strain is not clear. Cell culture and preparation of a brain homogenate and viral suspension containing the fowl glioma causing ALV were performed using methods described previously. 19 For inoculation, 100-fold–concentrated culture supernatant was prepared according to the method described previously. 4 A resistance-inducing factor test indicated that the ALV belonged to subgroup A. 34
Animals and experimental design
Fertile eggs from the WL-M/O (C/O) chicken strain used for the CEF and Japanese bantam hens that had been reared in isolators in our animal facilities were used in the animal infection experiment. All fertile eggs were incubated at 37.5 C and hatched in our laboratory. The bantams were inoculated intracerebrally at 1 day of age with 0.05 ml of the brain homogenate or with 0.05 ml of the viral suspension. SPF chicken embryos were inoculated through the yolk sac on day 6 of incubation with 0.1 ml of the viral suspension. Japanese bantams intracerebrally inoculated with a brain homogenate of normal bantam or culture supernatant of uninfected CEF and SPF chickens inoculated through the yolk sac with the culture supernatant were used as control group. The hatched chickens were reared in isolators of the animal facilities of our school. All experiments were performed humanely according to the guidelines set by the Hokkaido University Veterinary Medicine Animal Care and Use Committee. No chickens received any vaccinations or medication.
Histology and immunohistochemistry
The PNs (lumbosacral plexus, brachial plexus, sciatic nerve, and femoral nerve) and other organs, including the liver, spleen, kidney, heart, lung, bone marrow, gastrointestinal organs, pancreas, bursa of Fabricius, thymus, gonad, brain, spinal cord, eye, skeletal muscle, and skin, were fixed in 20% neutral-buffered formalin, embedded in paraffin wax, sectioned at 4 μm, and examined by light microscopy after staining with hematoxylin and eosin, Masson's trichrome, reticulin silver impregnation, or Bodian-Luxol fast blue double stain. For the immunohistochemical examination, selected sections were deparaffinized and treated with 0.3% hydrogen peroxide to inhibit endogenous peroxidase. Indirect immunoperoxidase staining was performed using a commercial streptavidinbiotin kit (Nichirei Corp., Tokyo, Japan). After blocking the nonspecific reaction, the sections were incubated with the specific primary antibody. These included antibodies to S-100α/β proteins (diluted 1 : 1,800, DAKO, Glostrup, Denmark), neuron-specific enolase (NSE; 1 : 200, DAKO Corp., Carpinteria, CA), neurofilament protein (NFP; 1 : 100, DAKO), cytokeratin MNF116 (1 : 10, DAKO Corp.), vimentin (1 : 100, DAKO), desmin (1 : 100, DAKO Corp.), muscle actin (HHF35; 1 : 3, ENZO Diagnostics Inc., Syosset, NY), or epithelial membrane antigen (EMA; 1 : 100, DAKO). The staining was carried out with a biotinylated secondary antibody and peroxidase-labeled streptavidin. The antigen localization was observed by incubation of the sections with 3,3′-diaminobenzidine-H2O2(DAB-H2O2) solution. The sections were counterstained with hematoxylin.
Polymerase chain reaction
Polymerase chain reaction (PCR) was conducted on DNA extracted from the paraffin-embedded enlarged PNs. To prepare DNA lysates, three 7 μm sections were cut from formalin-fixed, paraffin-embedded blocks, and these sections were extracted using DEXPAT™ (Takara Bio, Shiga, Japan) according to the manufacturer's instructions. Total cellular DNA samples were extracted from the virus-inoculated and uninoculated CEF, respectively, as the positive and negative controls. Forward and reverse primers designed to amplify the 135-bp sequences corresponding to the 5′ part of the 3′ noncoding region, which is specific for the fowl glioma-inducing ALV sequence (GenBank accession number AB112960), were as follows: 5′-TGT AGG CGG GCT CTT GTA TT-3′ (forward primer); 5′-TTG TCG GTC AAG CCT TGC CTT-3′ (reverse primer). 35 The PCR mixture (50 μl) contained 25.75 μl of MilliQ water, 5 μl of 10× buffer, 4 μl of deoxynucleoside triphosphate (dNTP) blend (2.5 mM each of four dNTPs), 2.5 μl of each primer (10 pmol), 10 μl of the template DNA, and 0.25 μl of Taq DNA polymerase (Perkin-Elmer Applied Biosystems Inc., Foster City, CA). This mixture was heated at 94 C for 2 minutes and then subjected to 40 cycles of PCR amplification (94 C for 30 seconds, 56 C for 30 seconds, 72 C for 45 seconds) using a PCR Thermal Cycler Dice TP600 (Takara Bio). The expression of the housekeeping gene GAPDH was analyzed on the same samples to confirm whether the formalin-fixed, paraffin-embedded tissues conserved DNA. Forward and reverse primers that amplified the 320-bp sequences spanning from exon 5 to exon 6 (forward: 5′-GTG ACC CCA GCA ACA TCA-3′; reverse: 5′-GCT GAG ATG ATA ACA CGC TT-3′) were constructed according to the chicken GAPDH gene sequence (GenBank accession number K01458). 29 The PCR mixture was prepared as above and heated at 94 C for 2 minutes and then subjected to 40 cycles of PCR amplification (94 C for 30 seconds, 50 C for 30 seconds, 72 C for 1 minute). These PCR products were electrophoresed through 2% agarose gels.
The expression of Marek's disease virus serotype 1 (MDV1) was also analyzed on frozen tissues of PNs, brain, kidney, or feather pulp by PCR as described previously. 7 Total DNA samples from the frozen tissues were extracted by standard phenol-chloroform extraction and ethanol precipitation methods. The positive and negative controls used were DNA from CEF inoculated with RB1B strain of the MDV1 and DNA from uninoculated CEF, respectively. Forward (5′-AGT TGG CTT GTC ATG AGC CAG-3′) and reverse primers (5′-TGT TCG GGA TCC TCG GTA AGA-3′) were used to amplify the 583-bp MDV1-specific meq gene fragment. The PCR mixture (50 μl) contained 34.75 μl of MilliQ water, 5 μl of 10× buffer, 4 μl of dNTP blend, 2.5 μl of each primer, 1 μl of the template DNA, and 0.25 μl of Taq DNA polymerase and was heated at 94 C for 30 seconds and then subjected to 35 cycles of PCR amplification (94 C for 45 seconds, 55 C for 45 seconds, 72 C for 1.5 minute). The PCR products were electrophoresed through 1.2% agarose gels.
Ultrastructural study
The enlarged lumbosacral plexus from seven affected bantams (bird Nos. 1–5 and 8) was diced into 1-mm cubes for electron microscopy, fixed in 2.5% glutaraldehyde buffered with 0.1 M phosphoric acid (pH 7.4), postfixed in 1% osmium tetroxide buffered with 0.1 M phosphoric acid (pH 7.4), dehydrated, and embedded in epoxy resin. Semithin, 0.5-μm-thick sections were stained with toluidine blue, and appropriate areas were selected for ultramicrotomy. Ultrathin sections were stained with lead citrate and uranyl acetate and examined with a transmission electron microscope (JEM-1210; JEOL, Tokyo, Japan).
Results
All 11 chickens showed leg paralysis and two (bird Nos. 4 and 8) concurrently had torticollis (Table 1). One bird (bird No. 3) was severely weakened because it could not feed itself adequately and was found dead at 118 days of age. At necropsy, in all the birds, there were multiple mild to severe cylindrical enlargements and yellow discoloration of PNs, especially in the lumbosacral plexus, brachial plexus, and sciatic nerves (Fig. 1). Both plexi were consistently affected, and the lesions were often bilateral. In severely affected birds, most spinal ganglia were spherically swollen and glossy, resembling small pearls.
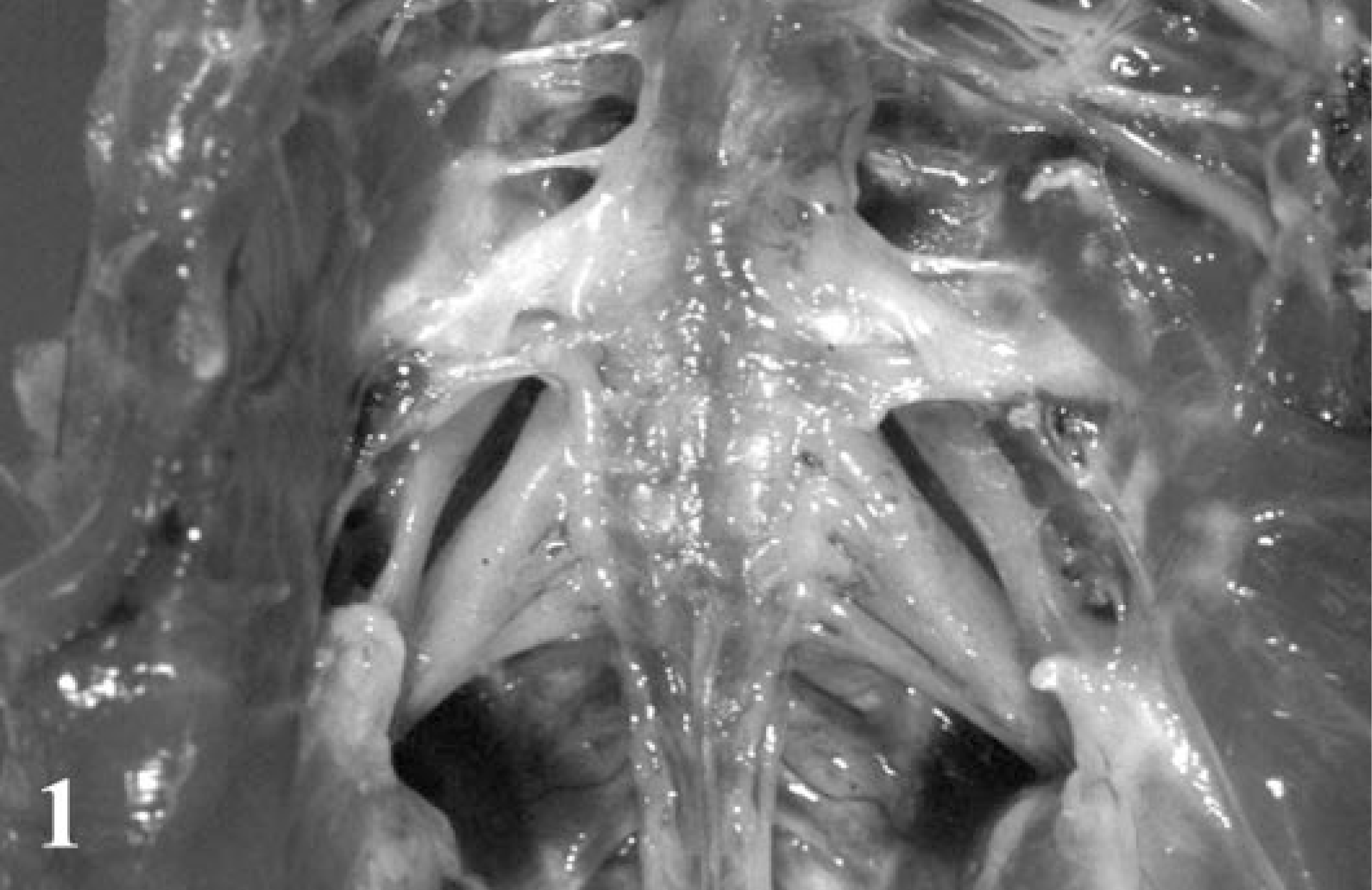
Lumbosacral plexus; Japanese bantam No. 7. Bilaterally cylindrical enlargement of each nerve fascicle.
The common histologic findings in the affected PNs of all examined birds were diffuse proliferations of spindle cells in the lumbosacral plexus, brachial plexus, and/or spinal ganglia (Fig. 2). The most common site was the lumbosacral plexus (Table 1). Cross sections of the affected nerve fascicles revealed numerous characteristic onion bulb–like structures that were composed of multiple layers of spindle cells surrounding a central axon (Fig. 3). These spindle cells were characterized by a bipolar, elongated, eosinophilic cytoplasm, hyperchromatic oval nuclei, and an indistinct cell border. Mitotic figures were extremely rare. Lymphocytes, plasma cells, and macrophages mildly to moderately infiltrated around blood vessels or among PN fibers.
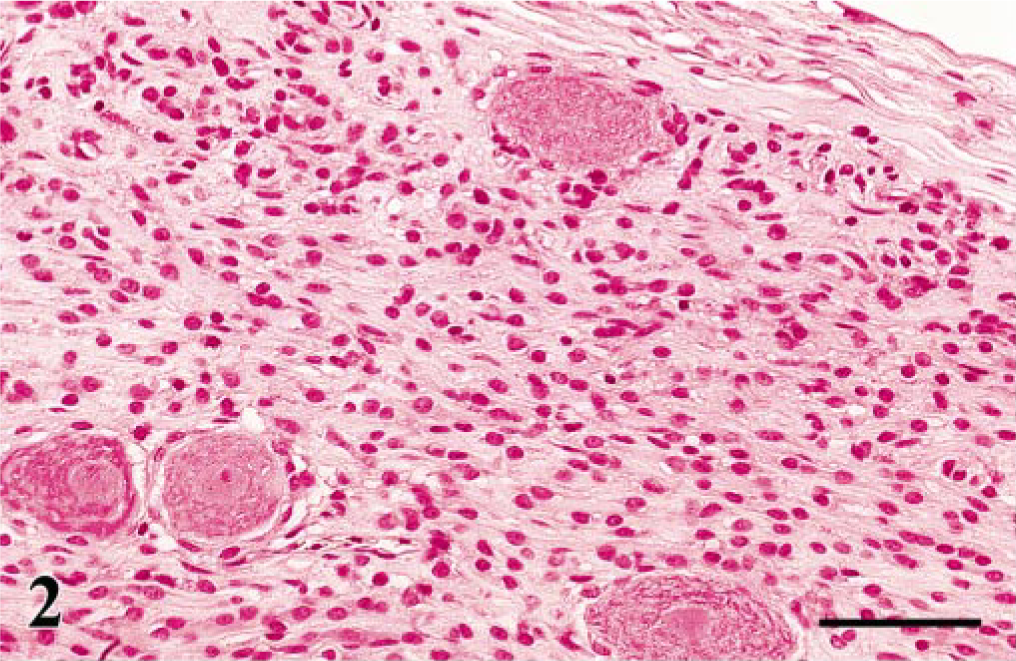
Spinal ganglion; Japanese bantam No. 6. Spindle cells showing a diffuse proliferation among ganglion cells. HE. Bar = 50 μm.
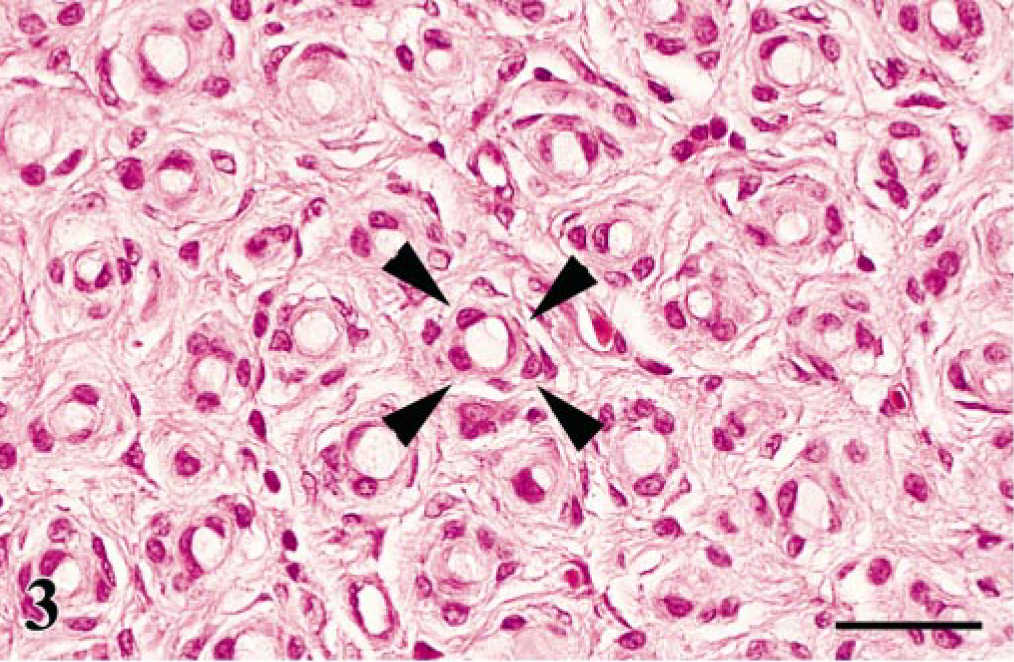
Lumbosacral plexus; Japanese bantam No. 7. Characteristic onion bulb–like structures (arrowheads) composed of mild spindle cell proliferation surrounding residual axons. HE. Bar = 25 μm.
The Bodian-Luxol fast blue double stain demonstrated that axons and myelin sheaths often remained intact among spindle cells in the milder lesions. In the moderate to severe cases, axons and myelin sheaths were compressed by the spindle cells and often degenerated (Fig. 4). Scattered digestion chambers containing degenerated myelin were also found. Masson's trichrome and silver impregnation method revealed that loose collagenous fibers proliferated in the endoneurium of the moderate and severe lesions, suggesting destruction of nerve fibers.
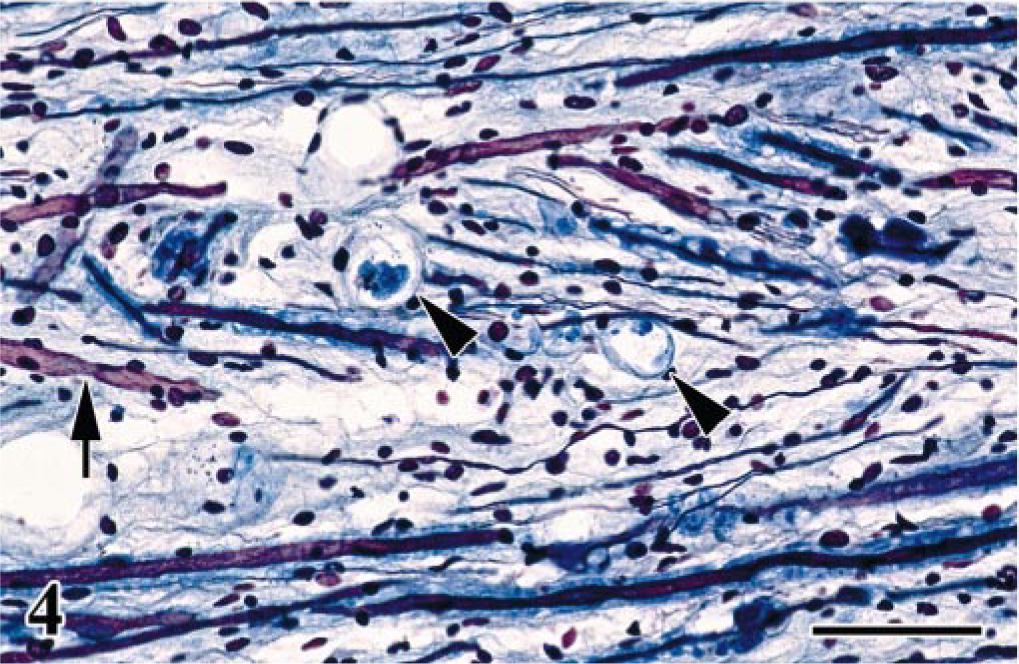
Lumbosacral plexus; Japanese bantam No. 7. Longitudinal section. Myelinated nerve fibers occasionally with digestion chambers containing myelin debris (arrowheads) and axons are occasionally degenerated (arrow). Bodian-Luxol fast blue double stain. Bar = 50 μm.
Reactivity for S-100α/β protein was detected only in the residual or degenerated Schwann cells (Fig. 5), whereas axons were positive for NSE and NFP. In contrast, the spindle cells were negative for S-100α/β and also for NSE, NFP, cytokeratin, vimentin, desmin, and muscle actin. In the cross section of the PNs, one cell layer of S-100α/β protein–positive Schwann sheaths was recognized at the center of each of the onion bulb–like structures (Fig. 6). No positive reactivity for EMA was detected in any PNs of the control tissue and the affected PNs.
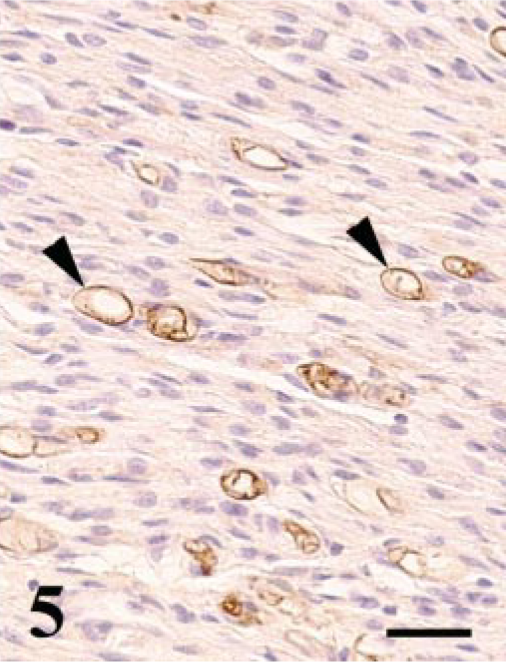
Lumbosacral plexus; Japanese bantam No. 5. Immunohistochemistry for S-100α/β protein. Residual Schwann cells are positive for this antigen. Some Schwann sheaths show vacuolar degeneration (arrowheads). Streptavidinbiotin (SAB)–DAB-H2O2, hematoxylin counterstain. Bar = 25 μm.
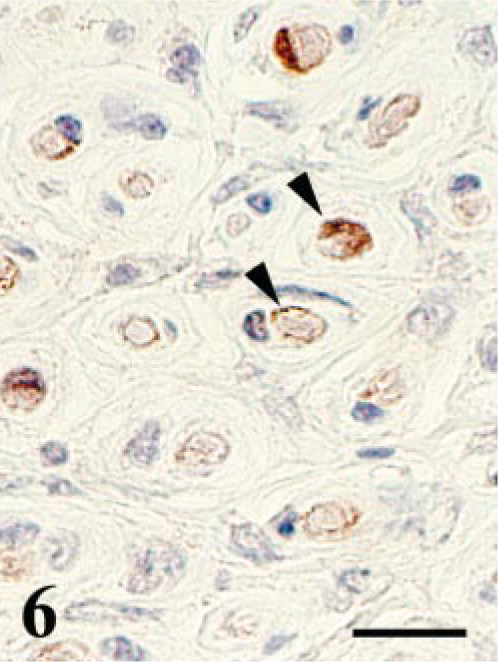
Lumbosacral plexus; SPF chicken No. 10. Cross section. Immunohistochemistry for S-100α/β protein. Residual Schwann sheaths near the center of the onion bulb–like structure are positive for S-100α/β protein (arrowheads), whereas proliferating spindle cells are negative. SAB-DAB-H2O2, hematoxylin counterstain. Bar = 15 μm.
Nine (bird Nos. 2–5 and 7–11) of the 11 chickens showed mild to moderate nonsuppurative encephalitis. Perivascular lymphoplasmacytic infiltration was noted in seven birds (bird Nos. 1, 3, 4, and 8–11), microgliosis in two (bird Nos. 1 and 6), and gliosis and neuronophagia in one (bird Nos. 2 and 4, respectively). Two SPF chickens (bird Nos. 10 and 11) had multiple nodular proliferation of astrocytes in the cerebrum and mild to moderate nonsuppurative myocarditis with matrix inclusions, which are intracytoplasmic inclusions in the infected cardiac myocytes. 16, 20 One bird (No. 10) also had microscopic renal cell carcinoma. There were no significant lesions in the other organs and tissues examined. None of the control chickens showed clinical signs, and no significant gross and microscopic lesions were seen.
The specific 135-bp DNA fragment of the ALV causing fowl glioma was amplified from eight (bird Nos. 1, 3, 5–7, and 9–11) of 11 enlarged PNs by PCR (Fig. 7). The GAPDH-specific fragment was detected in the same eight samples. The 135-bp fragment was not amplified from any PNs, kidney, or brain of control chickens examined. The MDV1-specific meq gene sequences were detected in the positive control but not in any affected birds.
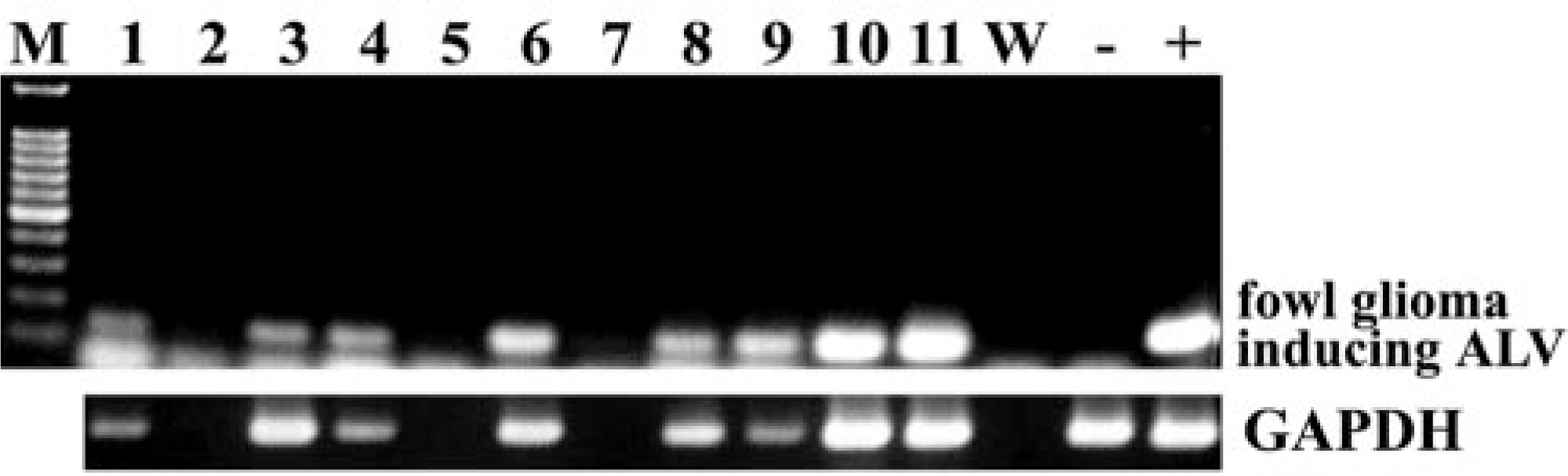
PCR analysis of the specific sequence of the ALV causing so-called fowl glioma and housekeeping gene GAPDH in DNA from formalin-fixed, paraffin-embedded enlarged peripheral nerves. M, molecular marker of 100-bp ladder; number, bird number; W, negative control of PCR using distilled water; −, negative control of PCR using DNA from uninoculated chicken embryo fibroblasts (CEF); +, positive control of PCR using DNA from CEF inoculated with the ALV-A causing fowl glioma.
Ultrastructurally, the spindle cells exhibited concentric proliferations surrounding the residual axons and Schwann cells (Fig. 8). These spindle cells showed bipolar, short cell processes and central nuclei, with occasional nuclear cleavage. The cell membrane was covered with a discontinuous basal lamina (Fig. 9). The cytoplasm contained few organelles, and pinocytotic vesicles were occasionally noted adjacent to the cell membrane. No distinct retrovirus particles were observed in the PNs examined.
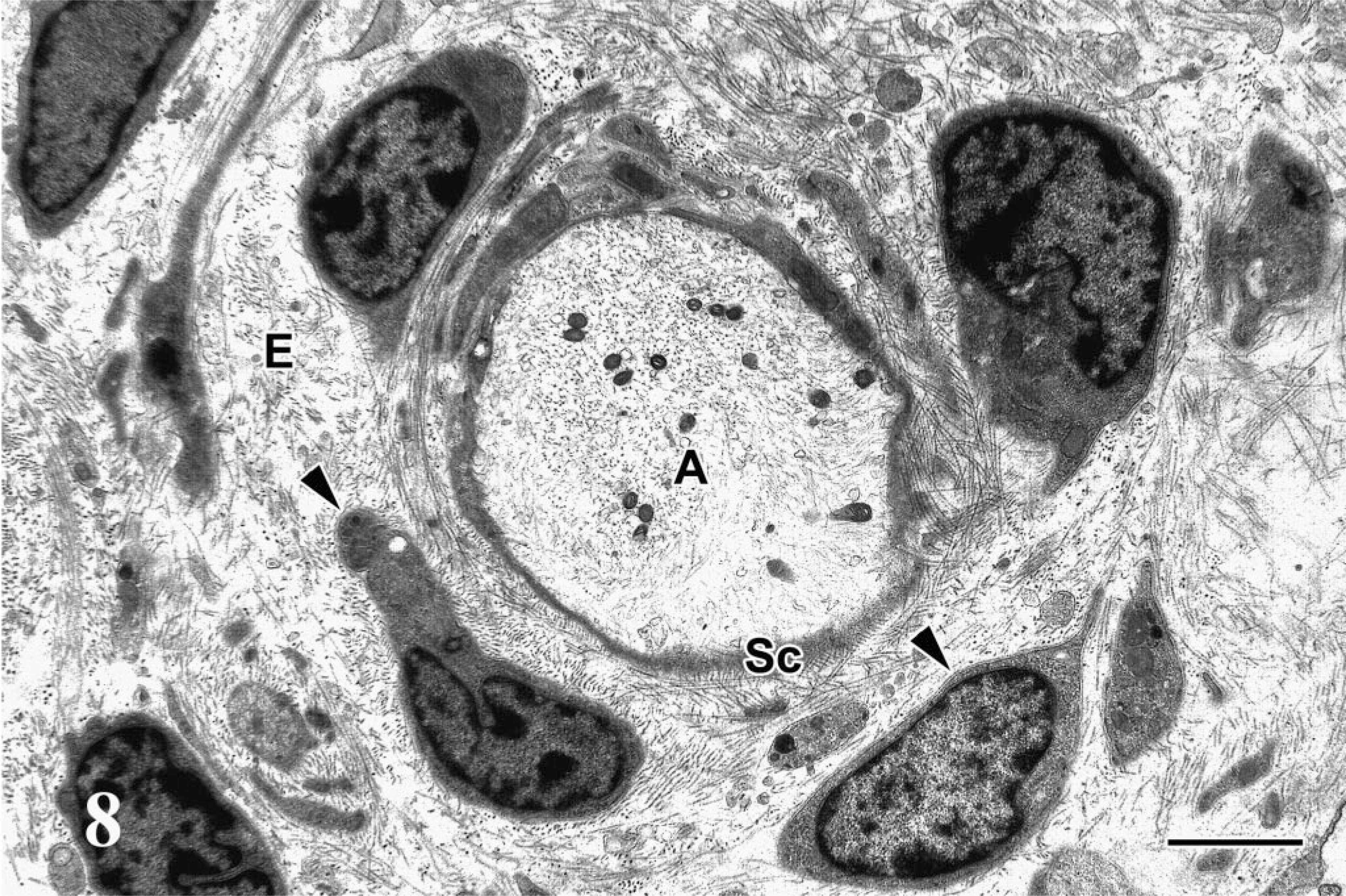
Electron micrograph; lumbosacral plexus; Japanese bantam No. 5. Onion bulb–like structure composed of short spindle cells (arrowheads) around residual axon (A) and flattened Schwann cells (Sc) in endoneurium (E). Bar = 2 μm.
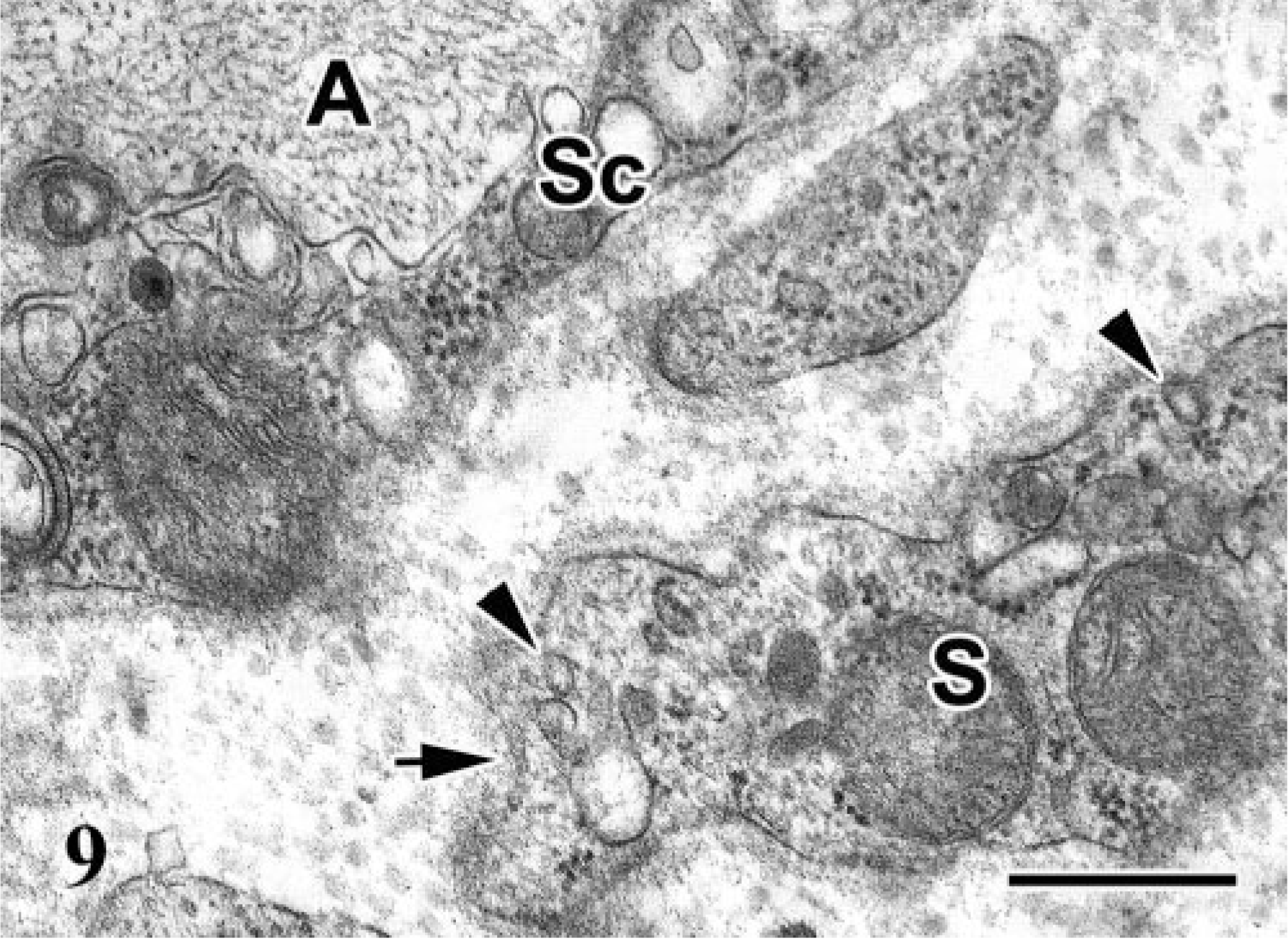
Electron micrograph; lumbosacral plexus; Japanese bantam No. 8. Spindle cell (S) around axon (A) and Schwann cell (Sc). The spindle cell has pinocytotic vesicles (arrowheads) and is covered with discontinuous basal lamina (arrow). Bar = 400 nm.
Discussion
All examined birds showed progressive leg paralysis resulting from degeneration and loss of PN fibers caused by an intraneural proliferation of spindle cells. The spindle cells formed characteristic onion bulb–like structures that surrounded central axons and accompanying Schwann cells, especially in the milder cases. The spindle cells were negative for S-100α/β and vimentin. Ultrastructurally, there were bipolar processes of various lengths, and the cytoplasm of spindle cells occasionally contained pinocytotic vesicles and was often covered with a discontinuous basal lamina. From these findings, the spindle cells in the present cases were considered to originate from perineurial cells rather than Schwann cells or endoneurial fibroblasts.
The origin of the perineurium is still unclear although fibroblasts, Schwann cells, or arachnoid cells have been proposed as candidate progenitor cells in humans. 11 Normal perineurial cells of chickens resemble those of mammals in their ultrastructural characteristics: elongated, bipolar cell processes, abundant pinocytotic vesicles, and a discontinuous basal lamina. 9 In the present cases, most of the proliferating spindle cells were ultrastructurally characterized by short cell processes and a few pinocytotic vesicles. Such ultrastructural differences may reflect morphologic diversity resulting from transformation. In fact, perineurial cells that proliferate in various disorders may show morphologic variety. 12, 18 They are thought to be capable of transforming into perineurial fibroblasts, which are characterized by a well-developed rough endoplasmic reticulum and discontinuous external lamina, or into transitional Schwann-perineurial cells with a continuous external lamina and a few pinocytotic vesicles. 11
The differential diagnosis includes Schwannoma and neurofibroma, which are common PN sheath tumors. The former consists almost exclusively of Schwann cells, whereas the latter is defined as a tumor with a proliferation of fibroblasts or perineurial cells in addition to Schwann cells. 30, 38 Schwann cells are immunohistochemically positive for S-100α/β protein, ultrastructurally are covered with a continual basal lamina, and have complicated cell processes. They contain few pinocytotic vesicles. Fibroblasts ultra-structurally contain an abundant endoplasmic reticulum without an association with a basal lamina. EMA is a useful antigen for identifying perineurial cells in humans. 32 However, the specificity of the anti-EMA antibody could not be determined in this study because no positive reaction was recognized even in any epithelial cells of normal control tissues.
Swelling of PNs, especially in lumbosacral plexus and sciatic nerves, is a characteristic gross finding in the chickens affected with classic Marek's disease (MD). 6, 27 Histologically, the PN lesions in MD are currently classified into three types: 1) type A characterized by a massive infiltration of neoplastic T lymphocytes, 2) type B by inflammatory lesions with infiltration of lymphocytes and plasma cells, demyelination, and Schwann cell proliferation, and 3) type C by such milder lesions. 31 Idiopathic polyneuritis, which is a rare hereditary disease in specific breeds of chicken that has histologic lesions similar to MD, is also reported. 2 Experimentally, PN lesions similar to those of MD occurred in some broiler chickens inoculated with ALV subgroup J. 28 Recently, Bacon et al. 1 reported peripheral neuropathies with leg paralysis in commercial layers that were suggested to be caused by an autoimmune reaction to the nerve tissue associated with a combination of common vaccines. The lesions were composed of inflammatory changes characterized by PN fibers separated by edema.
In contrast, the examined PN lesions consisted of proliferations of S-100α/β protein–negative spindle cells with an onion bulb–like arrangement. The histology and systemic distribution of the lesions was apparently different from that of these previously described PN disorders. Although there were scattered lymphocytes in the affected PNs, no atypism or mitotic figures were observed in these lymphocytes. DNA samples from PNs (bird Nos. 1, 4, and 8), brain (bird No. 2), kidney (bird Nos. 6 and 9), or feather pulp (bird Nos. 10 and 11) were screened by PCR using specific primers for MDV1, but the specific viral sequence was not amplified in all cases examined.
The progression of the PN lesion in the present cases could be hypothesized as follows. First, perineurial cells show a concentric proliferation surrounding axons and myelin sheaths, forming multiple onion bulb–like structures. The nerve fibers are gradually compressed by the cell growth, resulting in the degeneration or collapse of axons and myelin sheaths. Macrophages and lymphocytes occasionally infiltrate the affected PNs to various degrees. As the perineurial cells extensively proliferate, axons and Schwann cells are destroyed because of the compression. These pathomorphologic features are almost consistent with those of human intraneural perineurioma. 3, 10, 36 In the perineurial neoplasm in humans, however, the affected site is noted as a focal segmental cylindrical PN enlargement. The lesions of the present cases could be designated as multiple perineuriomas or even perineuriomatosis because most of the affected birds had multiple and bilateral distribution.
This study has not completely demonstrated the cause of multiple perineurioma in chicken. Although the serum antibody titers against the inoculated ALV in these affected birds were examined by enzyme-linked immunosorbent assay, we failed to detect them. Because the ALV was inoculated in ovo or intracranially at 1 day of age, immunotolerance toward the inoculum may be induced in the experimental birds. However, the multiple perineuriomas occurred in 9 of 203 bantams (4.4%) and 2 of 27 SPF chickens (7.4%) inoculated with the ALV-A causing fowl glioma and none of the control birds. The specific sequences of the ALV and GAPDH were identified by PCR in the paraffin-embedded tissues of enlarged PNs of 8 of 11 birds. Neither of them was amplified in the samples of another three birds, suggesting that these samples failed to conserve the DNA by formalin fixation. These results suggested a possibility that the ALV-A causing fowl glioma was associated with perineurioma of chicken.
This report represents the first cases of perineurioma in chicken or other domestic animals, except in dogs. Further virologic and serologic investigations are essential to confirm the relationship between perineuriomas in chicken and the ALV-A causing fowl glioma.
Footnotes
Acknowledgements
We thank Dr. B. A. Summers for critical suggestions. This work was supported in part by Grantsin-Aid for Scientific Research (B:15380207) from The Ministry of Education, Culture, Sports, Science, and Technology of Japan.
