Abstract
Intranuclear coccidia and Mycoplasma spp. were identified from the nasal cavity of 5 Sulawesi tortoises (Indotestudo forsteni) affected by chronic rhinosinusitis and oronasal fistulae. This study provides the first antemortem diagnosis of intranuclear coccidiosis in tortoises and the first cytomorphologic descriptions of this disease. Histopathologic and ultrastructural morphology of the intranuclear coccidia were identical to those previously described in tortoises. Nucleic acid sequence data of a 1715 base-pair fragment of the 18S small subunit rRNA gene identify this coccidian as a novel species.
Introduction
The Sulawesi tortoise (Indotestudo forsteni) is a poorly studied chelonian endemic to Sulawesi, Indonesia. Several hundred of these tortoises have been imported into the United States for the exotic pet trade. 8 Rhinosinusitis is a common clinical syndrome in tortoises, often caused by Mycoplasma agassizii or herpesvirus. 4,5,14,16 This study documents the presence of intranuclear coccidia and Mycoplasma spp. in 5 Sulawesi tortoises affected by chronic rhinosinusitis and oronasal fistulae. In addition, this study provides the first antemortem diagnosis of intranuclear coccidiosis in tortoises and the first cytomorphologic descriptions of this disease. Nucleic acid sequencing of 1715 base pairs (bp) from the 18S rRNA gene indicates that this organism is a novel species of coccidia.
Materials and methods
Five Sulawesi tortoises from 2 private collections were examined. Case nos. 1–4 were from collection “A,” and case no. 5 was from collection “B.” Case nos. 1, 2, and 5 were adult females, and case nos. 3 and 4 were adult males. All animals had been purchased through the exotic pet trade and had been imported into the United States from Indonesia approximately 1 year before presentation. Case nos. 1 and 5 were presented for postmortem examination, with a history of several weeks of anorexia and nasal discharge. After the death of case no. 1, case nos. 2–4 were presented for clinical examination.
Nasal lavage samples were obtained from case nos. 1–5 by instilling 1 ml of 0.9% sodium chloride solution into each naris with a 1-ml tuberculin syringe (without a needle), then recovering the saline solution and nasal sample into the syringe. Nasal samples were submitted for mycoplasma culture, PCR testing, and mycoplasma nucleotide sequencing (case nos. 1–5) by using previously published techniques. 4,5 Nasal lavage samples (case nos. 3–5) were also submitted for aerobic bacterial culture to a commercial veterinary diagnostic laboratory, a and for fungal culture to a commercial mycology laboratory. b
Nasal cytology specimens were collected from case nos. 2–4 by inserting a cotton-tipped applicator swab within the choanae. The swabs were smeared onto glass slides and stained with modified Wright-Giemsa, periodic acid-Schiff (PAS), and Fite acid fast (AF) techniques.
Nasal mucosal biopsy specimens were obtained from case no. 4, while the animal was under general anesthesia by using a 5-French endoscopic cup biopsy forceps introduced per os into the choanae. c Comprehensive tissue sets were collected postmortem for case nos. 1 and 5. Tissues collected by nasal biopsy or at the postmortem were preserved in 10% neutral buffered formalin and frozen at −20°C. Fixed tissues were processed routinely, sectioned at 5-μm thickness, and stained with hematoxylin and eosin. Select sections were stained with Fite AF and PAS.
Antemortem nasal mucosal biopsy specimens from case no. 4 were extracted from the paraffin block in xylene, rehydrated through a descending ethanol series, transferred to one-half strength Karnovsky fixative, then washed in 0.2 M sodium cacodylate, followed by further postfixation in 2% osmium tetroxide reduced with 2.5% potassium ferrocyanide. 13,19 The tissue was washed in 0.2 M sodium cacodylate after osmification and dehydrated through a graded ethanol series, transitioned through propylene oxide, and infiltrated and embedded in Spurr epoxy formulation. 21 Thick sections were mounted onto glass slides, stained by toluidine blue O, and examined by light microscopy. Thin sections were cut, mounted onto 150-mesh copper grids, stained briefly by 6% methanolic uranyl acetate, and counter stained with Reynold lead citrate before examination by transmission electron microscopy at 60 kv accelerating voltage. d , 17 Fresh fecal samples and feces from case nos. 2–4 were preserved in potassium dichromate and examined by centrifugal flotation in zinc sulfate solution.
Virology
Nasal lavage samples (cases nos. 1–4) and frozen samples of brain, liver, tongue, choanal epithelium, and sinus epithelium (case no. 5) were evaluated for the presence of chelonid herpesvirus by PCR (case nos. 1–5) and virus isolation (case nos. 1–4). A nested PCR targeting the DNA polymerase gene was carried out for the detection of herpesviruses by using the following primers: DFA: 5′-GAYTTYGCNAGYYTNTAYCC-3′, ILK: 5′-TCCTGG-ACAAGCAGCARNYSGCNMTNAA-3′, KGI: 5′-GTCTTGCTCACCAGNTCNACNCCYTT-3′, TGV: 5′-TGTAACTCGGTGTAYGGNTTYACNGGNGT-3′, IYG: 5′-CACAGAGTCCGTRRTCNCCRTADAT-3′. e , 23 DNA was prepared from nasal flush material f ; 2 μl of viral template DNA was mixed with the mastermix (2 mM MgCl2; 2 mM each of dNTP and 1 × reaction buffer; 1.25 U Taq polymerase; and 1 μM each of primers DFA, ILK, and KGI). The first-round PCR was run as follows: 1 min of denaturation at 94°C, 45 cycles with 30 sec of denaturation at 94°C, 60 sec annealing at 46°C, and a 60-sec extension at 72°C. A final extension period of 7 min at 72°C was followed by cooling to 4°C. A nested PCR was carried out with 1 μl from the primary reaction as a template in a reaction mix and under the same conditions as above with 1 μM of each primer, TGV and LYG. All PCRs were carried out with 50 μl amplification mixtures. DNA prepared from Terrapene heart cells (TH-1, ATCC CCL-50) infected with 2 different tortoise herpesviruses (4295/7R/95 and 1301/B99R/97) 14 were used as positive controls in all PCRs. DNA prepared from uninfected TH-1 was used as a negative control.
For virus isolation, nasal flushes were further diluted in 3-ml tissue culture medium with Earle salt solution. g The samples were sonified and centrifuged, and supernatant was inoculated onto TH-1. Tissue cultures were incubated at 28°C and observed daily for 14 days for cytopathic effects.
Serologic testing for herpesvirus was performed by using frozen heparinized plasma (case nos. 1–4). Virus neutralization tests were carried out by using a mean tissue-culture infectious dose (TCID50) of 100. 14 Tests were carried out by using a type I tortoise herpesvirus (4295/7R/95) and type II tortoise herpesvirus (1301/B99R/97). 14 Titers of 16 or greater were considered positive. Plasma from a spurthighed tortoise (Testudo graeca) with a documented herpesvirus type I infection was used as a positive control for the tests with the type I herpesvirus and as a negative control for the tests with the type II herpesvirus. No positive controls were available for the tests with the type II herpesvirus.
Coccidia PCR
Nasal lavage samples and frozen nasal mucosa from case nos. 3–5 and fresh fecal samples from case nos. 3–4 were evaluated for the presence of coccidia via DNA extraction, h consensus PCR, and DNA sequencing, by using methods previously described for intranuclear coccidia of tortoises. 9 Initial primers were designed from conserved regions of various coccidian species in different genera, targeting an approximately 350-bp segment that corresponded to bases 1018 to 1377 of the small subunit ribosomal RNA gene (SSU) of Toxoplasma gondii (GenBank accession no. L37415). To obtain an additional sequence for phylogenetic clarification, 2 PCR protocols were designed to obtain approximately 1400 additional bp of the SSU. Primers 18F (CTGGTTGATCCTGCCAGTAGTC) and 1145R (GCAGGAGAAGTCGAGAATGACA) were used to amplify the 5′ end of the gene, and primers 1446F (TAAATAGGGTCGGGAACCTCTC) and 1898R (GATCCTTCYGCAGGTTCAC) were used to amplify the 3′ end. Amplification conditions were similar to those used in the first PCR, with extension times increased to 90 seconds. Bands were purified and sequenced as previously described. 9
Analysis of data
The sequences were compared with known sequences in GenBank (National Center for Biotechnology Information, Bethesda, Maryland), EMBL (Cambridge, United Kingdom), and Data Bank of Japan (Mishima, Shiuoka, Japan) databases by using BLASTN. 1
Predicted homologous 1642–1757 nucleotide sequences of representative coccidial 18S rRNA available from GenBank were aligned by the clustal algorithm with manual adjustment. i , 10 Bayesian Metropolis-Hastings coupled Markov chain Monte Carlo (MCMC) phylogenetic analyses of the predicted alignment were performed with MrBayes 3. 18 MCMC analysis was conducted with flat priors and 2 runs of 3 incrementally heated chains in addition to the cold chain, as per program defaults. A general time reversible model with invariant +4 gamma categories of rates was used. The MCMC was run for 106 generations and sampled every 100 generations. The first 100,000 generations were discarded as a burn-in; the standard deviation of split frequencies was used to determine that this was appropriate.
Maximum likelihood (ML) phylogenetic analyses of the predicted alignment were performed with the PHYLIP (Phylogeny Inference Package, version 3.61) program package. 7 A ML tree was generated by using DNAML global rearrangements and 5 jumbles. Adelina bambarooniae (GenBank accession no. AF494059), a noncoccidian apicomplexan, was used as the outgroup. The validity of the tree topology obtained was tested by using bootstrap analysis by starting with Seqboot with 100 resamplings from the aligned sequences, followed by ML calculations, and calculation of the most probable (consensus) tree. 6
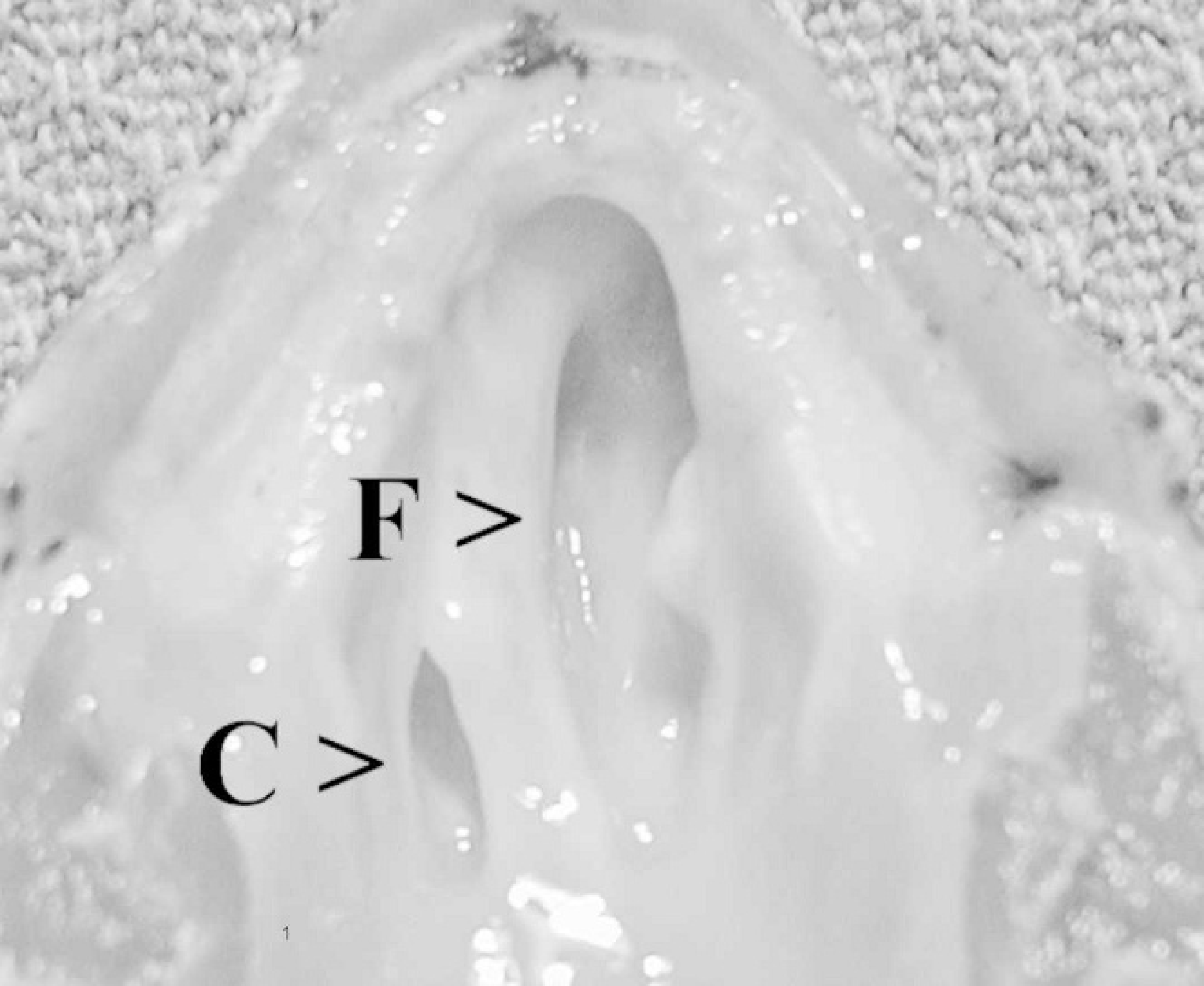
Ventral view of the palate, Sulawesi tortoise, case no. 1. The normal right choana (C) contrasts with the large oronasal fistula (F), affecting the left nasal cavity. The left side of the palate is obliterated, and the fistula has become contiguous with the left choana.
Results
Physical examination
Case nos. 2–5 had unilateral or bilateral, clear, gelatinous nasal discharge, accompanied by inflammation of the conjunctiva. Oral examination of all cases revealed variably extensive destruction of the vomer and palatine bones, resulting in oronasal fistulae anterior to the choanae, or coalescing with the choanae (Fig. 1). The internasal septum was lysed in most cases, resulting in communication of the right and left nasal cavities. Fistulae allowed food to become trapped within the nasal cavity. Despite these lesions, case nos. 2–4 maintained a normal appetite and activity level.
Cytology
Nasal cytology specimens from case no. 4 were moderately cellular and composed of respiratory epithelium, inflammatory cells, bacteria, and extra-cellular debris. Inflammation was predominantly lymphoplasmacytic, with fewer granulocytes and macrophages. Intranuclear coccidia were noted in the respiratory epithelium (Fig. 2), and extracellular coccidian oocysts that measured 8 to 10 μm were noted in the surface mucus. The organisms were intensely AF and had a positive reaction with PAS. The oocysts stained pale blue with Wright Giemsa stains. Surprisingly, cytologic detail of the parasites was better in slides that were not fixed in methanol at the time of collection. Wright Giemsa-stained cytologic specimens from nasal swab samples from case nos. 2 and 3 did not show intranuclear coccidia; however, a mononuclear inflammatory response was present, and coccidian oocysts were seen with Fite AF stain (Fig. 2).
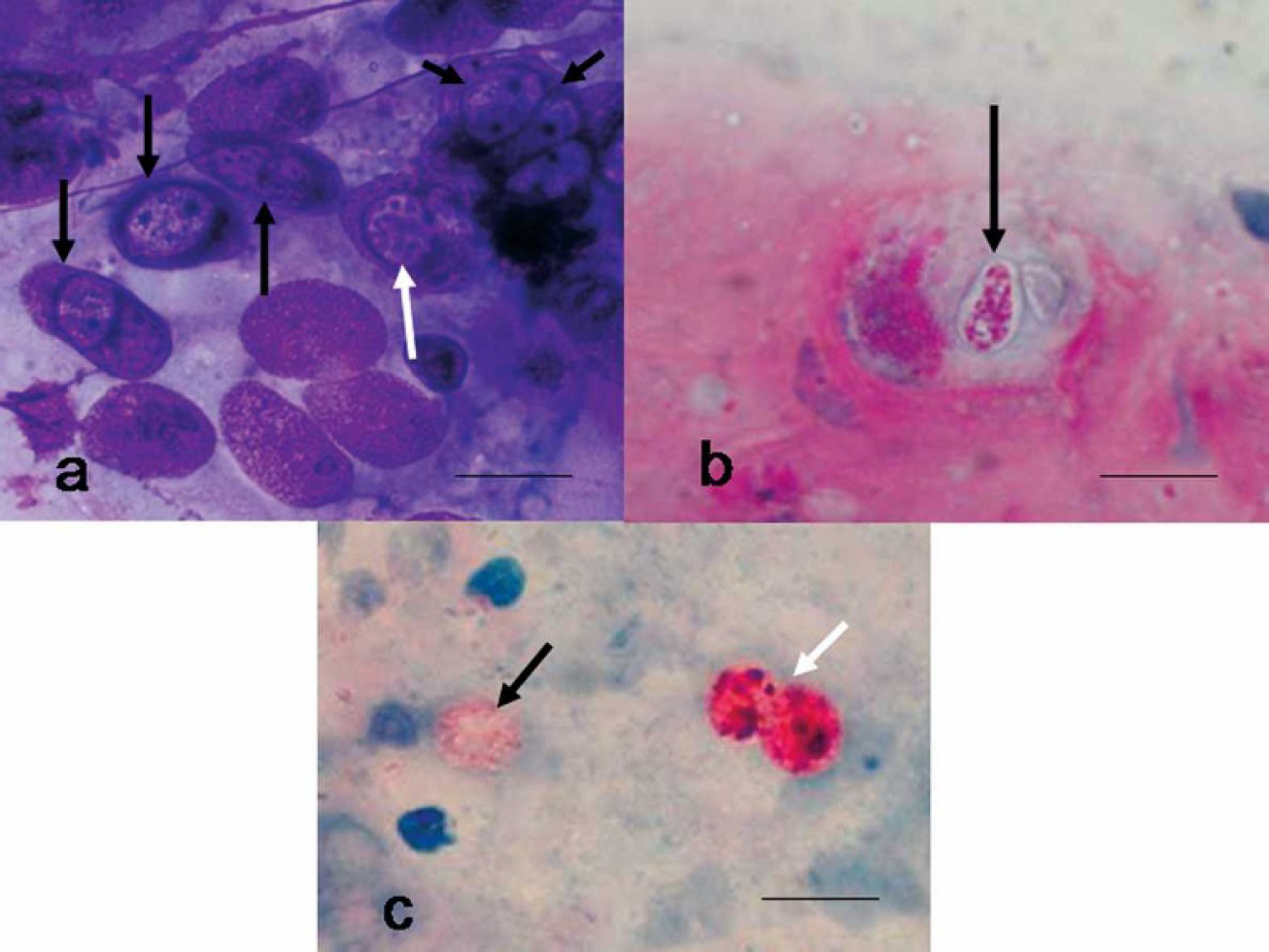
Nasal mucosal smear, Sulawesi tortoise, case no. 4.
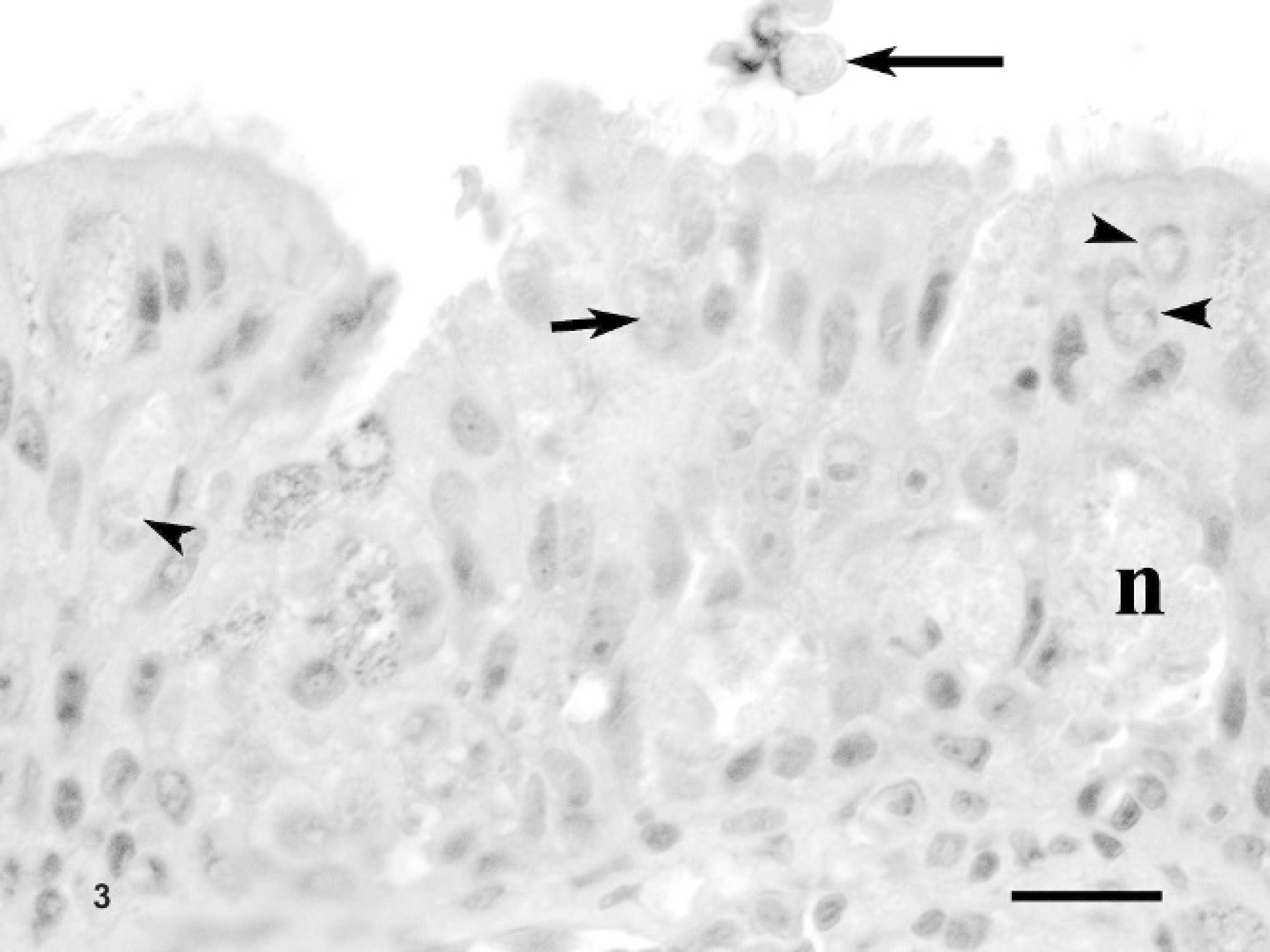
Nasal mucosal biopsy, Sulawesi tortoise, case no. 4. Note thick mucosal layer attributed to hyperplasia. Respiratory epithelial cells contain intranuclear gametes (arrowheads) or meronts (small arrow). An oocyst is on the mucosal surface (large arrow). Some foci of necrosis (n) are also present. Hematoxylin and eosin. Bar = 30 μm.
Histopathology
Histologic examination of the nasal mucosal biopsy samples (case no. 4) and postmortem specimens (case nos. 1 and 5) revealed moderate-to-marked hyperplasia of the respiratory epithelium. A lymphoplasmacytic inflammatory response with smaller numbers of granulocytes and macrophages was evident in the submucosa with occasional migration into the overlying epithelial layers. Focal necrosis was noted in the mucosal epithelial layers. Intranuclear coccidian gametes and meronts were detected in the apical respiratory epithelial cells; rarely, oocysts were noted in the surface mucus (Fig. 3). Squamous epithelium was histologically normal.
For case nos. 1 and 5, histologic examination of viscera revealed widespread nonsuppurative inflammation in most tissues, typical of the lesions seen with intranuclear coccidiosis in tortoises. 9 Few intranuclear coccidia were seen in the renal tubular epithelium and gut mucosa of case no. 1. No coccidia were seen in the visceral lesions of case no. 5. Serial sections of the decalcified heads for these 2 tortoises revealed inflammation, necrosis, exfoliation of epithelium, and luminal inflammatory cell exudate that involved the middle and inner ears, eustachian tubes, conjunctiva, and nasal mucosa. Many epithelial cells contained developing stages of the intranuclear coccidia. Inflammation was associated with fibrous thickening of the perichondrium of the nasal septum, resorption of the cartilaginous matrix, and sigmoid deviation of the septal cartilage. Similar cartilaginous changes were seen in the middle ear, and bone resorption and reactive bone formation were seen in the flat bones adjacent to areas of mucosal inflammation.
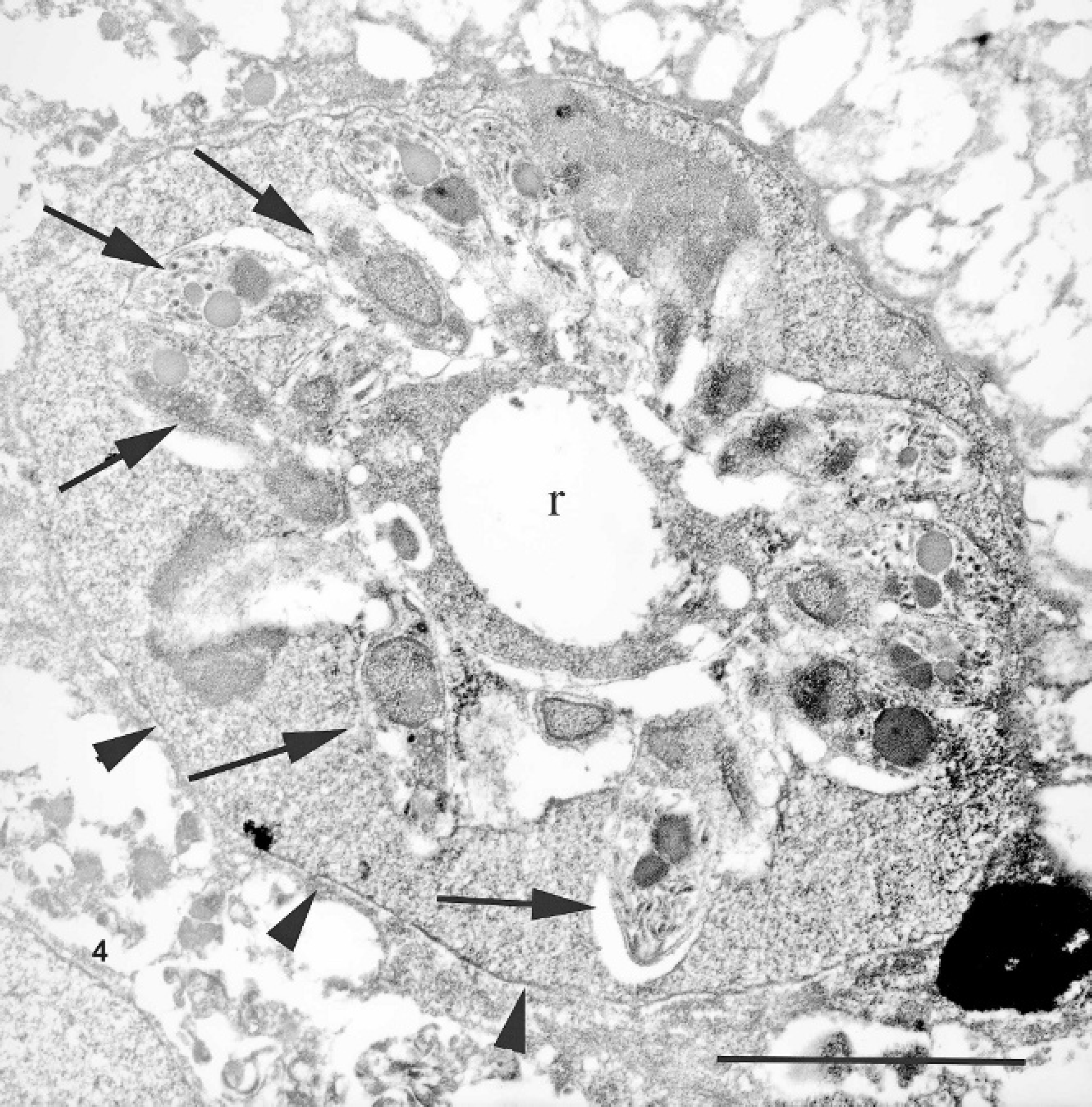
Nasal mucosal biopsy, Sulawesi tortoise, case no. 4. A membrane-bound (arrowheads) meront within the nucleus of a respiratory epithelial cell. Note the residual body (r) from which the merozoites (arrows) bud. Uranyl acetate and lead citrate. Bar = 5 μm.
Ultrastructure
Electron microscopic examination of nasal mucosal specimens from case no. 4 revealed much intracellular and extracellular artifactual vacuolar change attributed to the prior paraffin embedding. Infected nuclei were often enlarged and contained one or more organisms. Trophozoites were 2–5 μm in diameter. Meronts were up to 7 μm in diameter and contained numerous (>16) merozoites (Fig. 4). Merozoites were banana shaped, measured 4.0 × 1.5 μm, and budded from a residuum. The merozoites had apicomplexan features characterized by a distinct apical conoid apparatus, numerous rhoptries, and a single central to slightly eccentric nucleus (Fig. 5). Macrogametes and microgametes measured up to 6 μm in diameter. No Mycoplasma or other pathogens were seen by electron microscopy.
Coccidia PCR
PCR evaluation of nasal flush and fresh fecal samples from case nos. 3 and 4, and frozen nasal mucosa from case nos. 4 and 5 revealed positive results for a coccidian, which then sequenced. Sequence data from all cases was identical. This sequence was identical to that of all other cases of intranuclear coccidiosis in tortoises examined to date (Wellehan and Johnson, unpublished data). 9 The full extended product was 1715 bp once primer sequences were edited out. The BLASTN results for the sequence showed the highest score with the Eimeria arnyi 18S SSU (GenBank accession no. AY613853). 22 The Bayesian phylogenetic tree with posterior probabilities for branching is shown in Figure 6. The ML phylogenetic tree showed an identical branching pattern and similar branch lengths. The ML bootstrap values are shown on the Bayesian tree. Sequence data was submitted to GenBank; the accession number is AY728896.
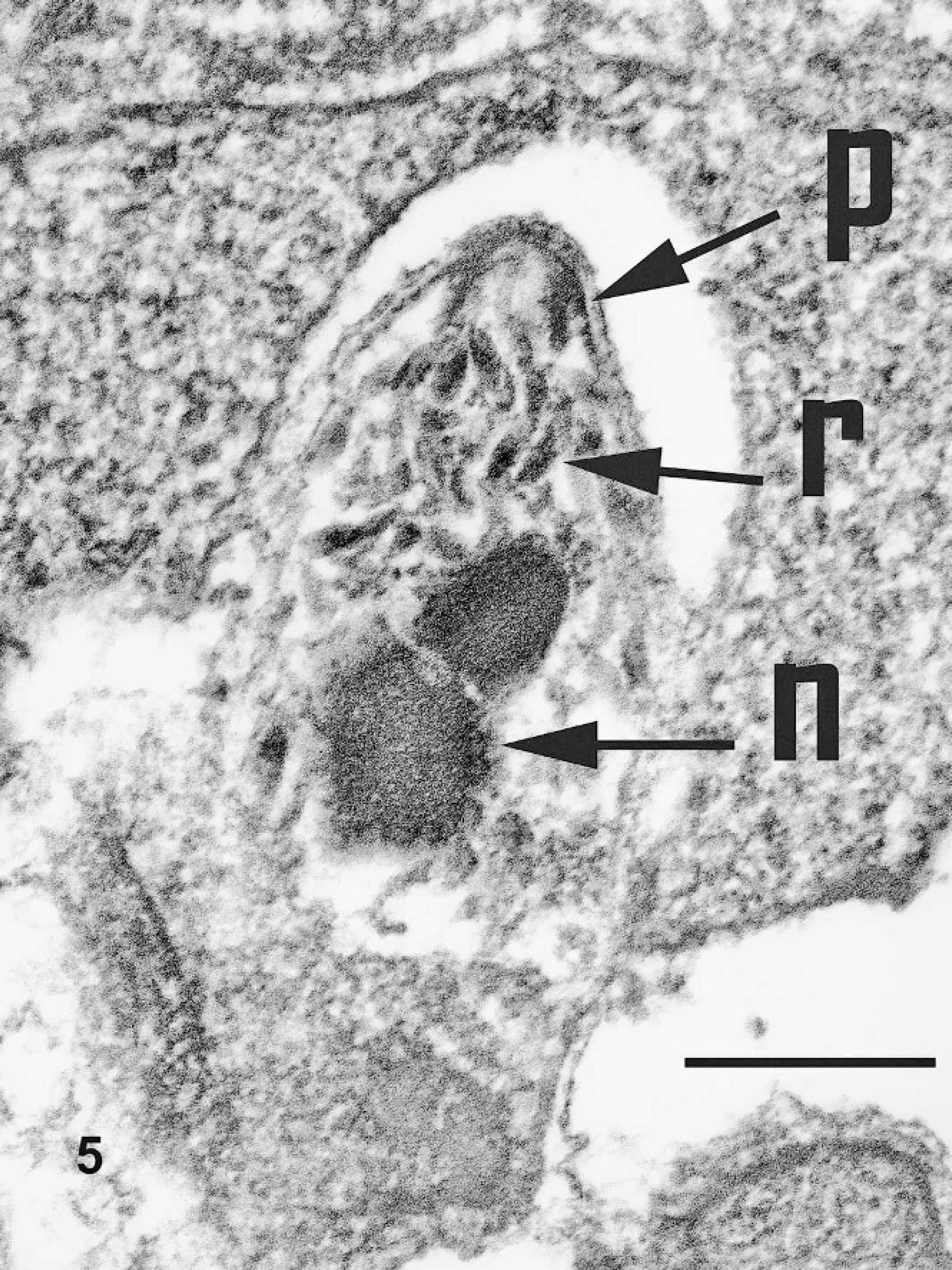
Nasal mucosal biopsy, Sulawesi tortoise, case no. 4. Structural features of a merozoite, including rhoptries (r), polar ring (p), and nucleus (n). Uranyl acetate and lead citrate. Bar = 1 μm.
Microbiology
Aerobic culture of nasal flush samples from 2 animals isolated a variety of organisms, including Klebsiella sp., methicillin resistant Staphylococcus intermedius, gamma (nonhemolytic) Streptococcus sp. (case no. 4), and Bacillus sp. (case no. 3).
Fungal culture of nasal flush samples from 2 animals isolated a variety of organisms, including Cryptococcus sp. (not Cryptococcus neoformans) (case no. 5), Penicillium sp. (case nos. 3 and 4), and Candida sp. (case no. 4).
Mycoplasma culture and PCR of nasal flush samples were positive in all 5 cases. DNA finger-printing revealed M. agassizii in case nos. 1, 2, and 4, and an unidentified Mycoplasma in case nos. 3 and 5.
Virology and fecal microscopy
Herpesvirus serology and herpesvirus PCR of nasal flush samples were negative for case nos. 1–4. Virus isolation from nasal flush samples was negative for case nos. 1–4. Herpesvirus PCR was negative for frozen brain, liver, tongue, choanal mucosa, and sinus mucosa from case no. 5. Positive controls showed the expected 235-bp amplicons in the PCRs. No parasitic organisms were detected in fresh fecal samples and feces preserved in potassium dichromate (case nos. 2–4).
Discussion
Before this report, 11 cases of intranuclear coccidiosis of chelonia were documented. 9,11 Species affected included the radiated tortoise (Geochelone radiata), impressed tortoise (Manouria impressa), leopard tortoise (Geochelone pardalis), Bowsprit tortoise (Chersina angulata), and Sulawesi tortoise. Organisms were widely distributed in previous cases and were most commonly found in the intestine, pancreas, liver, and kidney, with less frequent involvement of the eustachian tube, splenic macrophages, middle ear, lung, and stomach. Organisms were found within nasal respiratory epithelium of 1 previously reported Sulawesi tortoise, 1 radiated tortoise, and 1 leopard tortoise. However, only mild nasal lesions were noted, and oronasal fistulae were not present. All previous cases were diagnosed at postmortem. Case nos. 2–4 provide the first antemortem diagnosis of intranuclear coccidiosis in tortoises and provide the first cytomorphologic descriptions of this disease. Based on morphology, ultrastructure, and molecular data, the coccidia of this report are identical to intranuclear coccidia recently reported in 9 other tortoises. 9
The presence of intranuclear coccidia in the nasal respiratory epithelium of Sulawesi tortoises may indicate that these organisms are responsible for producing the chronic rhinosinusitis seen in these cases. However, Koch postulates were not fulfilled, and a clear causal role cannot be determined at this time. The role of this parasite in causing disease could be that of a primary pathogen or a secondary invader in an immunocompromised host. It is also possible that the intranuclear coccidia are an incidental finding. Oronasal fistulae have not been previously described in tortoises, but chronic inflammation secondary to an infectious agent could presumably produce such lesions. Case nos. 1 and 5 in this report had histologic lesions in the facial bones and cartilage associated with mucosal inflammation and intranuclear coccidia. These histologic lesions were severe enough to account for the gross lesions noted in the nasal septum.
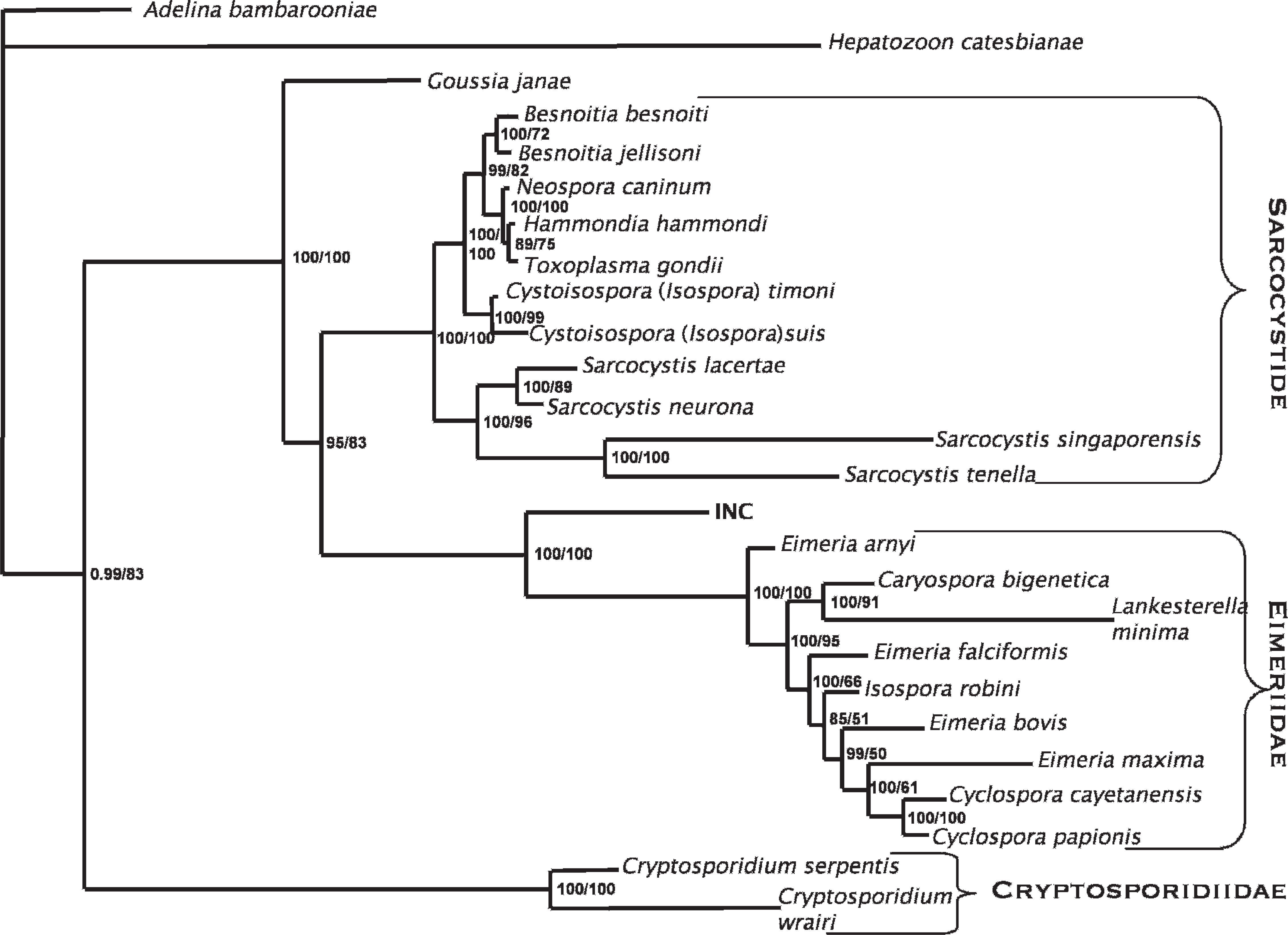
Bayesian phylogenetic tree of homologous 1642–1757 base pairs of coccidial 18S rRNA nucleotide sequences. Adelina bambarooniae (GenBank accession no. AF494059) was used as the outgroup. Confidence of the tree topology obtained is shown by Bayesian posterior probabilities to the left of the slash and ML bootstrap values to the right. Intranuclear coccidia (INC) of tortoises is in boldface. Other sequences were retrieved from GenBank. Besnoitia besnoiti (AF109678), Besnoitia jellisoni (AF291426), Caryospora bigenetica (AF060975), Cryptosporidium serpentis (AF151376), Cryptosporidium wrairi (AF115378), Cyclospora cayetanensis (AF111183), Cyclospora papionis (AF111187), Cystosospora suis (U97523), Cystosospora timoni (AY279205), Eimeria arnyi (AY613853), Eimeria bovis (U77084), Eimeria falciformis (AF080614), Eimeria maxima (U67117), Goussia janae (AY043206), Hammondia hammondi (AF096498), Hepatozoon catesbianae (AF130361), Isospora robini (AF080612), Lankesterella minima (AF080611), Neospora caninum (L24380), Sarcocystis lacertae (AY015113), Sarcocystis neurona (U07812), Sarcocystis singaporensis (AF434059), Sarcocystis tenella (L24383), Toxoplasma gondii (L37415).
All of the animals of this report tested positive for a Mycoplasma sp. by culture and PCR, but the same Mycoplasma sp. was not found in all tortoises. Mycoplasma agassizii has been found to be a primary cause of upper respiratory tract disease in free-ranging desert tortoises (Gopherus agassizii) and gopher tortoises (Gopherus polyphemus), however, it has not been reported to cause severe, chronic, oronasal fistulae, as noted in the present cases. 4,5 Additional research, including controlled transmission studies, will be required to elucidate the role of intranuclear coccidia and Mycoplasma as etiologic agents of chronic rhinosinusitis and oronasal fistulae in Sulawesi tortoises.
The presence of a variety of fungal and aerobic bacterial agents in the nasal discharge of Sulawesi tortoises likely results from secondary colonization and food contamination of the previously damaged sinus. No histologic evidence of a primary fungal or non-Mycoplasma bacterial infection was seen.
All virology testing in these cases was negative. Nasal flush samples were negative for herpesvirus by PCR, and animals were serologically negative for herpesvirus. No inclusions consistent with herpesvirus were seen histologically or ultrastructurally. However, this does not completely rule out herpesvirus as a possible cause of this syndrome. In some cases, tortoises experimentally infected with herpesvirus fail to seroconvert. 16 In addition, nasal flush samples from experimentally infected tortoises may not test positive by PCR. 16 The fact that frozen brain, liver, tongue, choanal mucosa, and sinus mucosa for case no. 5 were negative for herpesvirus by PCR is perhaps more convincing evidence that herpesvirus is not involved in rhinosinusitis in Sulawesi tortoises. These tissues are commonly found to harbor herpesvirus by PCR in infected tortoises. 16 Additional virology testing, such as immunohistochemistry and in situ hybridization, should be considered for future cases, because such methods have not yet been applied to the investigation of chelonian viruses.
In most previously reported cases of intranuclear coccidiosis of tortoises, fecal examination did not reveal coccidian oocysts. 9,11 This is consistent with the authors' findings that standard methods of fecal examination for coccidia are poorly sensitive for detection of the intranuclear coccidian organism of tortoises. In 1 previously reported case, postmortem fecal examination revealed an unidentified coccidia species; however, it was not confirmed whether the fecal organism was the same organism as seen in the tissues. 9 Examination of feces by PCR for coccidia was positive for 2 cases reported in the present study and yielded identical sequence to the coccidia in the nasal samples. The sequence data obtained in this investigation provide information for the further design of relatively simple nucleic-acid-based diagnostic tests for intranuclear coccidia infection of tortoises.
The 1715-bp SSU product described herein provides the most complete sequence data for intranuclear coccidia of tortoises to date and allows confident establishment of the phylogenetic position of this organism. The authors' phylogeny is in agreement with other recent coccidian phylogenies, finding 2 main clusters that correspond roughly to the families Sarcocystidae and Eimeriinae. 15,20 By using molecular data, the genus Isospora was found not to be monophyletic and was divided into Isospora and Cystoisospora, showing that the presence or absence of Steida bodies, and avian or mammalian host specificity, is more phylogenetically informative than the number of sporocysts or sporozoites for these genera. 3 The genus Eimeria is also clearly not monophyletic. Goussia, previously considered possibly synonymous with Eimeria, does not cluster within the families Sarcocystidae and Eimeriinae. 12,20 Another coccidia genus found in reptiles, Choleoeimeria, is also basal in the Eimeriinae. 12 Choleoeimeria is characterized by endogenous development in reptile biliary epithelium, a characteristic that may or may not be phylogenetically informative, and the generic status is not universally accepted. 2 Choleoeimeria was not included in this analysis because of the lack of a full-length sequence for comparison. However, analysis of a shorter available sequence unequivocally establishes that this organism is far basal to known Choleoeimeria and other Eimeriinae (Wellehan, unpublished data). 9 Phylogenetic analysis of the intranuclear coccidian sequence does not provide support for inclusion of the intranuclear coccidian of this report in either the Sarcocystidae or the Eimeriinae, and supports this organism as a novel species, likely of a novel genus. However, a formal description of a novel coccidian species requires identification of sporulated oocysts, and the failure to isolate sporulated oocysts of the intranuclar coccidian organism of tortoises prevents formal taxonomic description at this time.
The ecology and the life cycle of the intranuclear coccidian organism of tortoises are unknown. The coccidian life cycle is variable among species and may involve fecal transmission, vector transmission, and intermediate hosts. Both sexual and asexual reproduction may occur at various stages of the life cycle. Based on the authors' findings, the intranuclear coccidian of tortoises shows several typical apicomplexan life stages, including oocysts, macrogametes, microgametes, meronts, merozoites, and trophozoites. The distinguishing organelle of the Apicomplexa, the apical complex, was noted ultrastructurally; and the apical complex contained a polar ring and rhoptries, as expected for this phyla.
All reported cases of intranuclear coccidiosis in tortoises were in captive animals, which have often been through extensive networks of local collectors, regional wholesalers, importers, and pet stores, and have likely been exposed to many other reptile species and pathogens. Diagnosis of intranuclear coccidiosis in free-ranging tortoises would be informative.
Despite intermittent signs of rhinosinusitis and the persistence of oronasal fistulae, case nos. 2–4 remain alive, with normal appetite and activity level, 5 years after initial presentation. However, the tortoises have not been reevaluated for the presence of infectious agents since the initial diagnosis.
Acknowledgements
The authors wish to acknowledge the work of the late Dr. Barbara Bonner, who first noted rhinosinusitis and oronasal fistulae of Sulawesi tortoises. Dr. Lori Wendland and Dr. Deborah Gordon provided laboratory services, and E. J. Ree provided digital imaging expertise. Export of diagnostic samples from the United States to Germany was permitted under CITES Permit 02US065537/9. The authors appreciate the editorial comments of Dr. Ellis Greiner and Dr. Steve Upton.
Footnotes
a.
Antech Diagnostics, New York, NY.
b.
Gordon Mycology Laboratory, Littleton, MS.
c.
Karl Storz Veterinary Endoscopy, Goleta, CA.
d.
Zeiss 906E, Zeiss Electron Microscopy, Thornwood, NY.
e.
MWG Biotech, GmbH, Ebersberg, Germany.
f.
DNEasy tissue kit, Qiagen, Hilden, Germany.
g.
Basal Medium Eagle, Seromed, Biochrom KG, Berlin, Germany.
h.
DNEasy tissue kit, Qiagen, Valencia, CA.
i.
MegAlign, DNAstar, Madison, WI.
