Abstract
Scuticociliatosis is a disease of fish induced by ciliated parasites of the genus Scuticociliatida. It has been described in sea horses (Hippocampus sp.), flounders (Paralichthys olivaceus), and turbots (Scophthalmus maximus). Here we present a case study of a population of sea dragons chronically infected with scuticociliates identified as Philasterides dicentrarchi by histopathology and PCR. Beginning in 2004, over a period of 19 months, 10 sea dragons (Phycodurus eques and Phyllopteryx taeniolatus) were found dead in an aquarium of the Zoological Garden Basle, Switzerland. Clinically, the animals showed only faint symptoms of disease over a short period of time. At necropsy, macroscopic lesions were confined to the skin with multiple, often hemorrhagic, ulcerations. Histologically, epidermal ulcers were associated with necrosis and inflammation of the underlying dermis and musculature. Numerous ciliates, with a morphology consistent with scuticociliates, were present in these lesions. In several animals these ciliates had invaded blood vessels and were detected in gills and internal organs including kidney, thyroid gland, and central nervous system (CNS). In these organs, mild degenerative lesions and inflammatory reactions were evident. The ciliates were identified as Philasterides dicentrarchi based on small-subunit ribosomal RNA (SSUrRNA) gene sequences obtained by polymerase chain reaction (PCR) on DNA extracted from paraffin-embedded tissue sections. Our report shows that scuticociliate infections of sea dragons can develop into a systemic infection and that both species of sea dragons can be affected.
Keywords
The leafy sea dragon (Phycodurus eques) and the weedy sea dragon (Phyllopteryx taeniolatus) belong to the family Solegnathina, which also includes sea horses. Resembling floating pieces of seaweed, they are endemic to Southern Australia (http://www.marinebio.org, public database). Recently, they have become popular as aquarium fish. Since breeding remains difficult, the majority of the sea dragons originate from the wild population (http://www.marinebio.org, public database).
Fatal infections with histophagous ciliates of the genus Scuticociliatida have been reported repeatedly in ornamental fresh water fish. 2, 6 Scuticociliatosis is of increasing importance in marine aquaculture. 1, 3, 17 Scuticociliatosis is rarely described in sea dragons and so far reported to be limited to dermal infections. 7, 12, 19
This report describes the pathology associated with scuticociliatosis in a population of leafy and weedy sea dragons.
All sea dragons originated from the same merchant, but were imported as wild-caught animals at different time points. After arriving at the Zoological Garden Basle, animals underwent a period of quarantine of almost 2 months, where they were adapted to water and feeding conditions. After quarantine, animals were transferred to the public aquarium, where leafy sea dragons inhabited a 1,000-liter sea water tank without any other fish species. Weedy sea dragons were kept in a 1,000-liter sea water tank together with sea horses. In both aquaria, water temperature varied from 14°C to 18.5°C, salinity from 33 to 35 pm. Oxygen content was always >99%. NH3 and NO2 were not detectable, NO3 ranged from 26 to 49 mg/liter. Both aquaria were additionally stocked with sea anemones, sea snails, and sea stars. Sea dragons were fed with Artemia salina and floating crayfish. Prior to feeding, these live organisms were dipped for 1 hour in fresh water to eliminate potential salt water–adapted pathogens.
Starting in 2004, over a period of 19 months, 10 juvenile or subadult sea dragons from the 2 public aquaria—5 leafy sea dragons and 5 weedy sea dragons—died. Sea horses kept in the same aquarium were not affected. Four out of 10 animals died without clinical symptoms. The clinical signs of 6 animals were recurrent skin ulcerations and central nervous symptoms such as whirling and swimming on the water surface or in lateral position. Three leafy and 2 weedy sea dragons were treated with metronidazol without prevention of mortality (Table 1). Dead fish were immediately sent to the Centre for Fish and Wildlife Health for further investigation. A complete necropsy was performed on all animals. Fresh mounts of skin and gill samples and the intestinal content were examined microscopically for the presence of parasites. For bacteriologic examination, samples of skin, liver, spleen, and kidney were cultured on blood agar plates and bromothymol-blue lactose agar plates (Bio Merieux, Lausanne, Switzerland). Samples of skin, gills, liver, kidney, spleen, thyroid gland, intestine, and CNS were fixed in 10% buffered formalin, routinely processed for histology, and stained with hematoxylin and eosin (HE) and Ziehl-Neelsen (ZN). To verify the presence of Scuticociliatida and to determine the species, we performed a PCR on DNA extracted from formalin-fixed and paraffin-embedded material of 2 fish. Three 20-µm-thick sections of muscle and skin were deparaffinized. Lysis and DNA extraction were performed using the DNeasy Tissue Kit (Qiagen, Basel, Switzerland) following the manufacturer's instructions. The DNA yield was determined by spectrophotometry using the NanoDrop photometer (Witec AG, Littau, Switzerland). Polymerase chain reaction was performed with a HotStarTaq Kit (Qiagen, Basel, Switzerland) according to the manufacturer's protocol. Primers for the amplification of a 350 base pair fragment of the small-subunit ribosomal RNA (SSUrRNA) gene of Philasterides dicentrarchi were taken from Parama et al. 14 with the forward primer 5′-GAGAAACGGCTACCACATCTA-3′ (PSSU1) and the reverse primer 5′-CAAG GTAAAGAGCCTACTCCA-3′ (PSSU2). The annealing temperature was 58°C. As a positive control, an extract of DNA of cultured P. dicentrarchi trophozoites, kindly provided by Prof. José Manuel Leiro Vidal, University of Santiago de Compostela, Spain, was used. The PCR-amplification products were analyzed by agarose gel electrophoresis, and amplification products were visualized using SYBR Green I staining (Molecular Probes, Leiden, Netherlands). A single band with a molecular weight of 350 bp was detected in the samples of both diseased fish and the control DNA. The PCR products were purified using a PCR Clean-up Kit (Macherey-Nagel AG, Oensingen, Switzerland) followed by sequencing in an ABI PRISM 310 Genetic Analyzer (Applied Biosystems, Foster City, CA) and comparison with published GenBank sequence (EMBOSS-Align).
Clinical and pathologic observations of sea dragons of the Zoological Garden Basle.∗
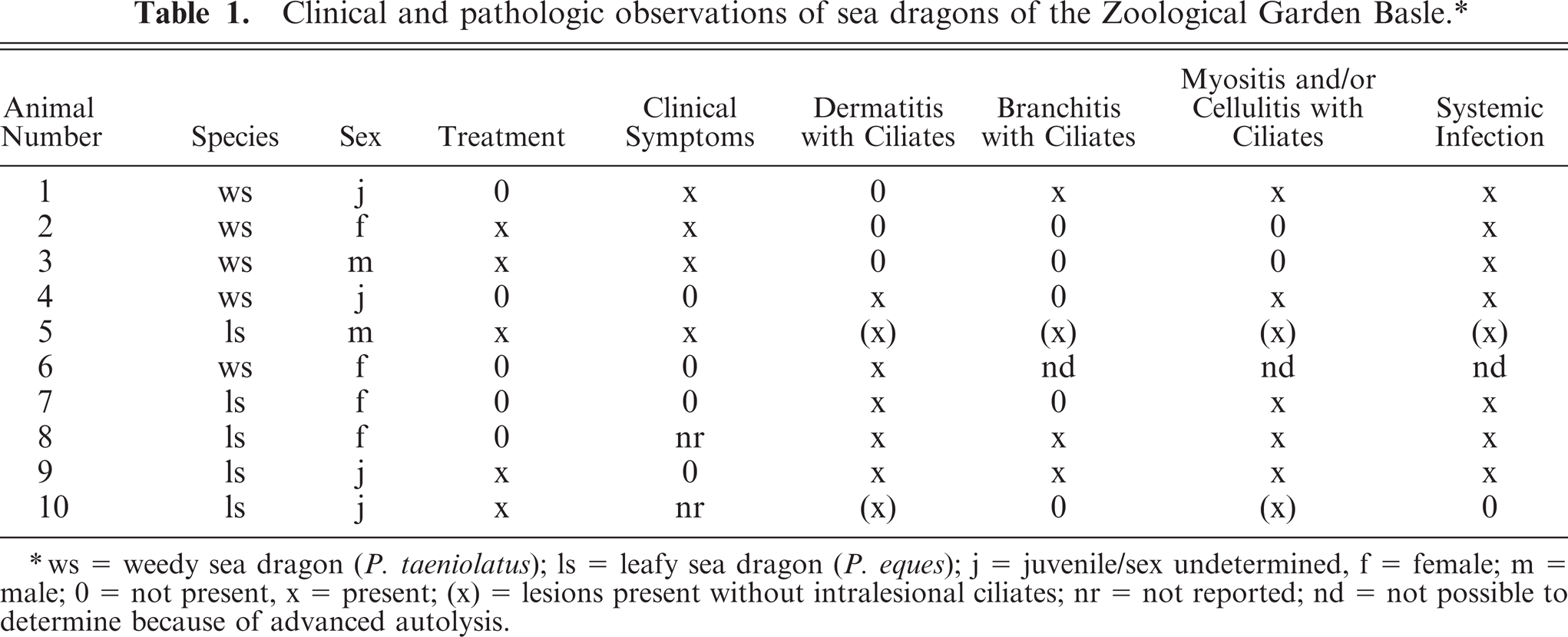
ws = weedy sea dragon (P. taeniolatus); ls = leafy sea dragon (P. eques); j = juvenile/sex undetermined, f = female; m = male; 0 = not present, x = present; (x) = lesions present without intralesional ciliates; nr = not reported; nd = not possible to determine because of advanced autolysis.
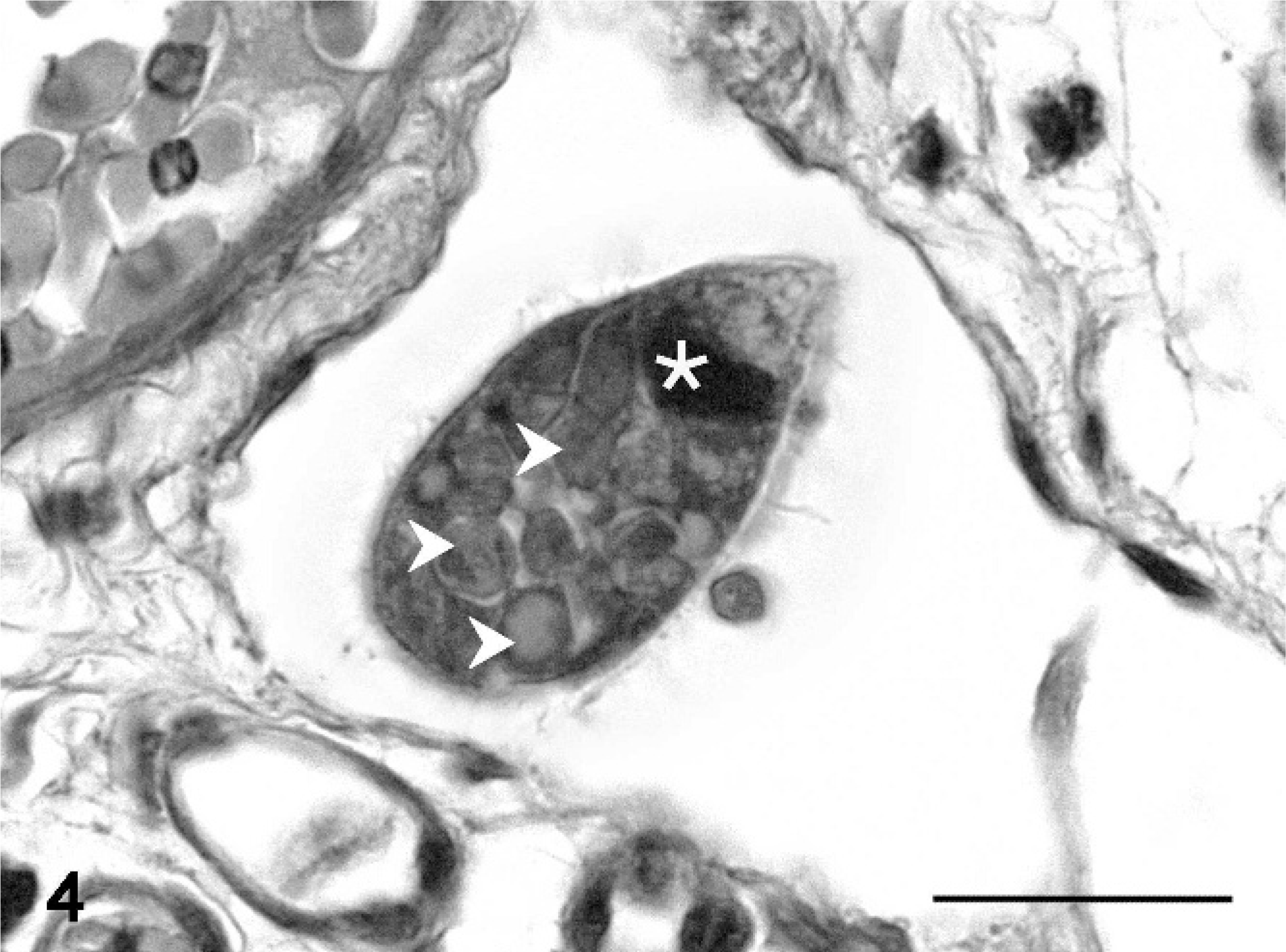
At necropsy, all fish were emaciated, and the length of the leafy sea dragons varied between 20 and 36 cm, the length of the weedy sea dragons between 10 and 32 cm. Direct microscopic investigation of skin smears revealed ciliates in 3 animals. No pathogenic bacteria were isolated. Macroscopically, 6 of 10 animals showed multifocal to diffuse hyperemia of the skin and multifocal epidermal ulcerations with hemorrhages in the underlying dermis (Fig. 1). Internal organs did not show macroscopically visible alterations. The most severe histopathologic changes were present in the skin, the skeletal musculature, the gills, and the brain. Skin lesions were characterized by ulceration of the epidermis with marked edema and necrosis of the underlying dermis and skeletal musculature (Figs. 2, 3). A marked inflammatory infiltrate consisting of large numbers of lymphocytes, macrophages, and scattered eosinophilic granular cells extended from the superficial dermis into the underlying musculature. Adjacent to these ulcerations, the epidermis was hyperplastic (Fig. 2). Numerous parasites measuring 15–25 µm × 20–50 µm in diameter (Fig. 4) were present in the affected dermis and musculature. Parasites were radially symmetric, round to pyriform, and covered with ciliate rows. They had a central globular, round, basophilic heterochromatic macronucleus (up to 10 µm in diameter) and a small micronucleus (up to 2 µm in diameter). Varying numbers of eosinophilic homogenous intracytoplasmic droplets most likely representing phagocytosed erythrocytes in a vacuole were present (Fig. 4). In the gills, the branchial epithelium was hyperplastic and hypertrophic, with multifocal intra- and subepithelial hemorrhages. The stroma was distended because of edema and a moderate to severe infiltration with numerous lymphocytes, fewer macrophages, and eosinophilic granular cells. Ciliates were present on the surface of the epithelium, subepithelial and intravascular (Fig. 5). In 1 animal, the brain was examined, and ciliate parasites were associated with infiltration of the meninges by numerous macrophages and lymphocytes (Fig. 6). In 7 animals the parasites were also found in the kidney, liver, intestine, thyroid gland, and coelomic cavity. In these organs, the presence of the parasites was associated with a scant inflammatory reaction and single cell necrosis or small necrotic foci (Table 1). Based on the morphology of the parasites and the distribution of organ lesions, a presumptive diagnosis of systemic scuticociliatosis was made. 4, 6, 17– 19 DNA extraction from paraffin-embedded tissue section, PCR amplification, and sequence analyses revealed a 98% identity of our samples to published SSUrRNA gene sequences of P. dicentrarchi and 85% identity to corresponding sequences of Uronema marinum, a closely related scuticociliate. From these findings we conclude that the sea dragons were infected with P. dicentrarchi.
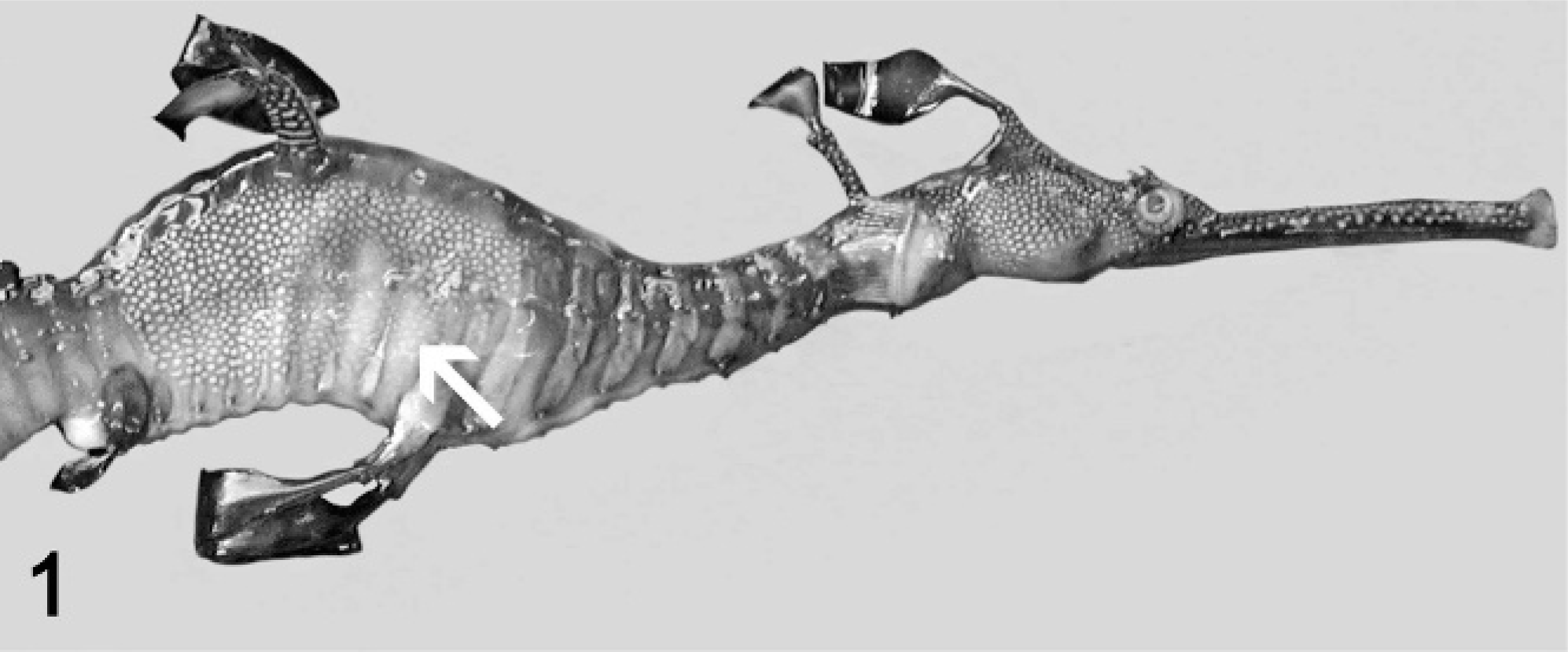
Weedy sea dragon (Phyllopteryx taeniolatus); animal No. 4. Well-circumscribed pale area and skin ulceration (arrow).
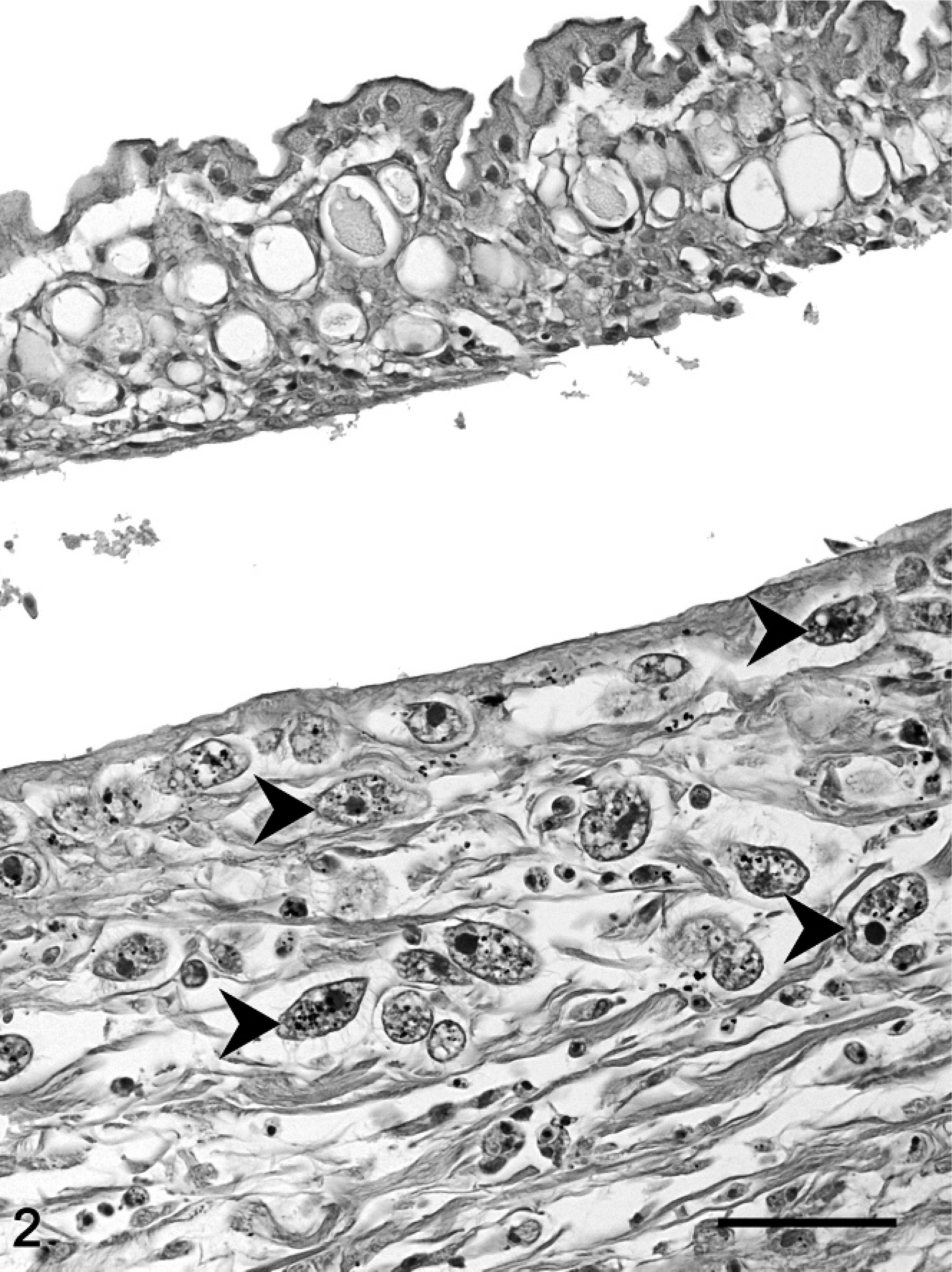
Skin, weedy sea dragon (P. taeniolatus); animal No. 4. Epidermis hyperplastic, underlying dermis is expanded by edema, infiltration with macrophages, lymphocytes and eosinophilic granular cells and multiple cross sections of ciliates (arrowheads). HE stain. Bar = 50 µm.
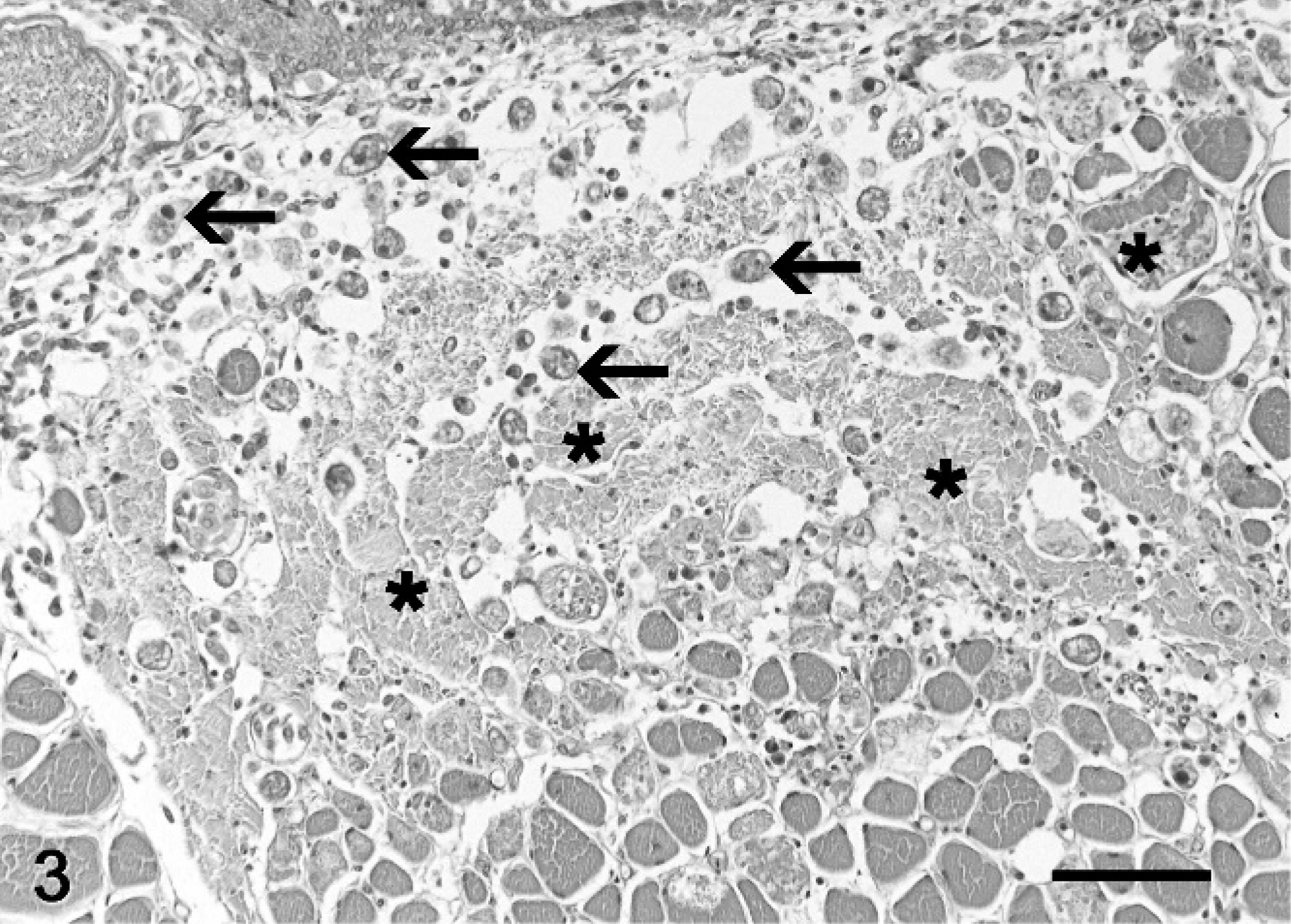
Musculature, leafy sea dragon (P. eques); animal No. 8. Degenerated myofibers (asterisks), expansion of the interstitium by macrophages and lymphocytes admixed with fibrin and translucent fluid (edema), erythrocytes, cellular debris, and numerous ciliates (arrows). HE stain. Bar = 100 µm.
Gill, weedy sea dragon (P. taeniolatus); animal No. 1. Intravascular ciliate with a central globular round, basophilic heterochromatic macronucleus (asterisk), varying numbers of eosinophilic homogenous intracytoplasmic droplets in the a contractile vacuole (arrowheads). Note no vascular alterations. HE stain. Bar = 20 µm.
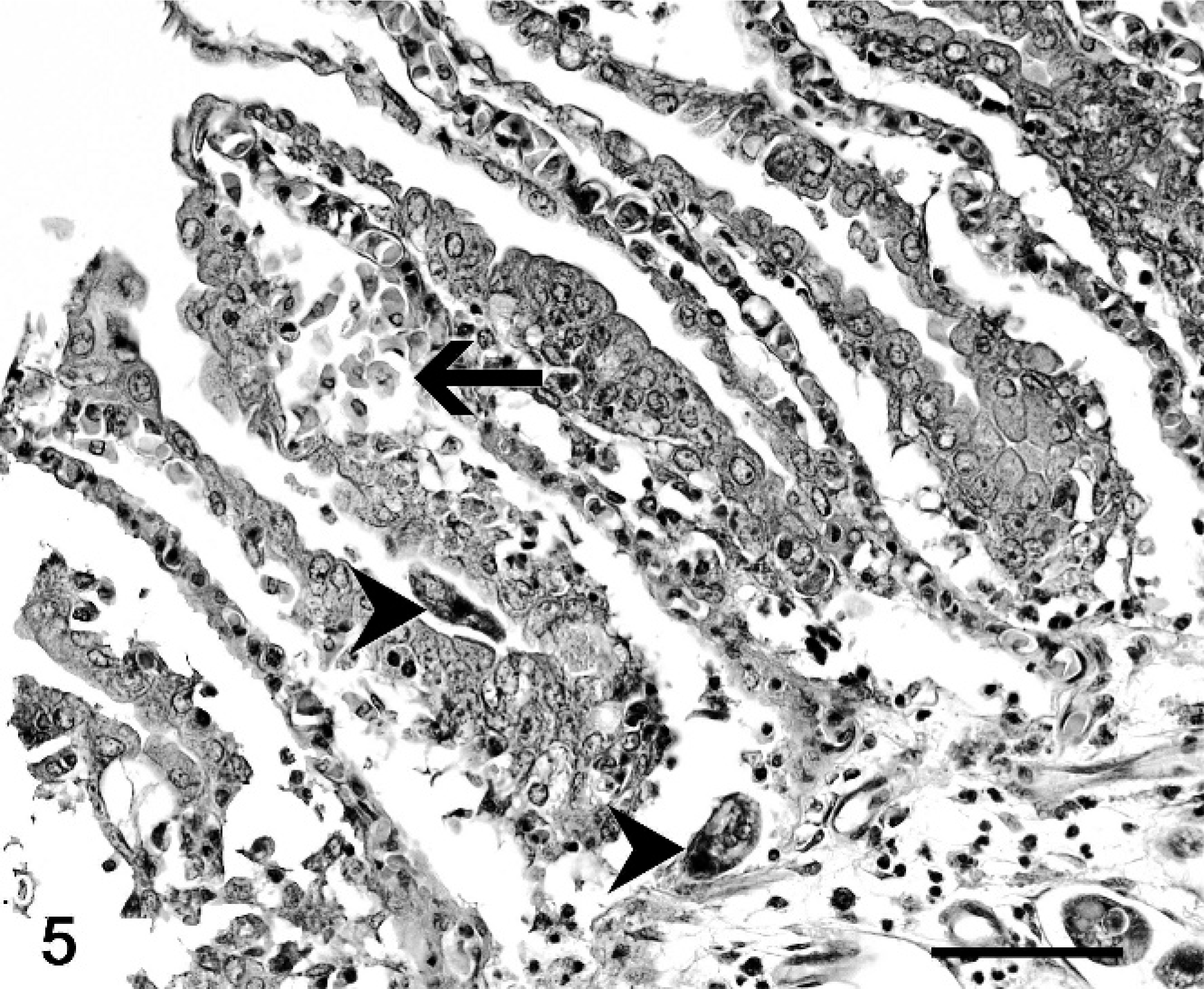
Gill, weedy sea dragon (P. taeniolatus); animal No. 1. Hyperplastic and hypertrophic branchial epithelium, subepithelial hemorrhages (arrow). Ciliates are present on the epithelial surface, in subepithelial tissues (arrowheads), or intravascularly. HE stain. Bar = 50 µm.
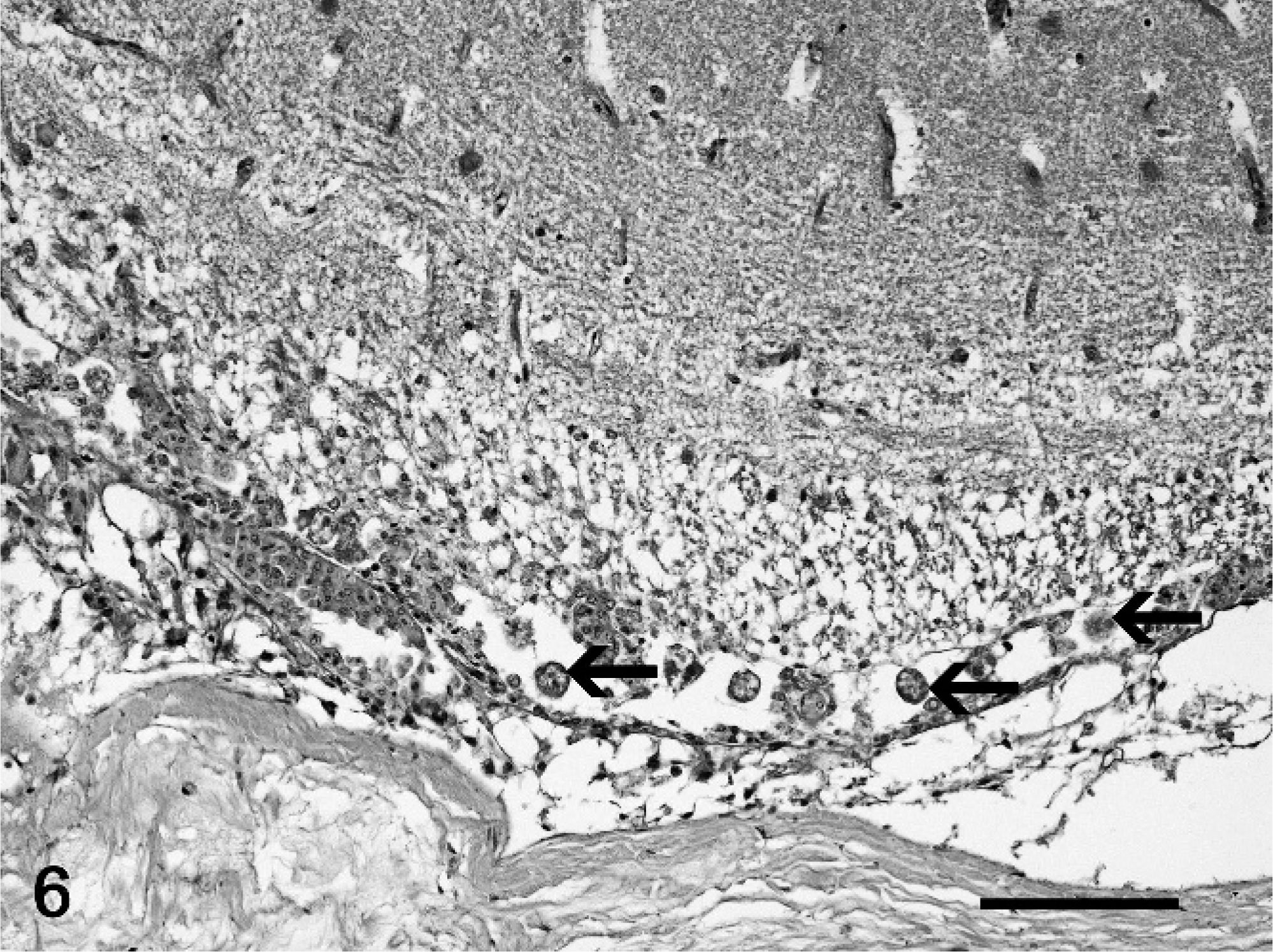
Central nervous system, meninges, leafy sea dragon (P. eques); animal No. 9. Infiltration of the meninges by mainly macrophages with multiple cross sections of ciliates (arrows). HE stain. Bar = 50 µm.
Scuticociliate infection is known to cause necrotizing dermatitis and myositis in turbots and flounders. 8– 10, 12– 15 Additionally, meningitis and encephalitis caused by these parasites were reported in tuna fish. 13 Tetrahymena corlissi, the fresh water equivalent of the scuticociliates, is known to cause severe ulcerative dermatitis and necrosis of the underlying muscle, leading to high mortalities in guppies. 11 In sea horses a fatal infection with scuticociliates similar to U. marinum with invasion of gills, muscle, connective tissue, kidney, urinary bladder, neural canal, and blood vessels leads to destruction of muscle and connective tissue. 4 Neither skin lesions nor inflammatory reaction associated with the parasites in other tissues were reported in these sea horses. 4 Infestations of sea dragons with dermal ciliates have been observed, however without clinical or pathological relevance (Stidworthy, personal communication). 7, 12 Umehara et al. 19 reported that dermal infestation with P. dicentrarchi in weedy sea dragons results in lesions restricted to the skin and low mortality rates. Our report demonstrates that P. dicentrarchi can also lead to severe ulcerative dermatitis and necrotizing myositis. A primary function of the skin in fish is osmoregulation and sustaining of a waterproof barrier. Severe lesions result in a systemic disturbance of osmolarity and death. 5 The extent of the skin lesions in our sea dragons, associated subcutaneous edema and ascites clearly indicate massive osmoregulatory disturbance and thus are the cause of death.
In our cases P. dicentrarchi additionally caused systemic infection in 2 species of sea dragons probably via hematogeneous spread because parasites were visible intravascularly. This is supported by our finding of a granulomatous meningitis in association with intralesional parasites in 1 animal. The involvement of the CNS was further supported by the clinical neurologic symptoms in several animals. In contrast to the findings in the CNS, lesions in other internal organs were generally mild. P. dicentrarchi was reported to express cysteine proteinases, which inhibit leukocyte migration and thus might be able to evade the host immune response. 15
The source of infection could not be determined in our cases. Most likely the parasite is endemic in the wild population of sea dragons and thus was already present in imported animals. Because of the stress of transport and the additional translocation from the quarantine into the public aquarium, the fatal outbreak was probably triggered. The parasite's route of entry is still unknown, but an infection via nostrils, gills, or skin lesions is hypothesized. 8, 14 The pathomorphologic findings in our cases also support that primary lesions are induced in the skin. After gaining access to the blood stream in these lesions hematogeneous spread might lead to lesions in distant organ systems, such as the CNS. Additionally, Puig et al. 16 discussed a spread of parasites via the connective tissue because of their preference to loose connective tissue.
In conclusion, scuticociliate infections may be an emerging disease in wild caught/captively maintained sea dragons. These animals are very fragile to handling and disease treatment options are lacking. Therefore, further research is required to ensure the maintenance of these increasingly popular and expensive animals in captivity.
Footnotes
Acknowledgements
We are grateful to Prof. José Manuel Leiro Vidal for providing material of P. dicentrarchi as a positive control for PCR, to Dr. Evangelia Kallivretaki for her investigations on PCR protocol, and to Marie-Louise Zahno for the sequencing of the PCR product. We also thank Rahel Lavater, who took care of the fish.
