Abstract
Most cats infected with Bartonella henselae remain outwardly healthy carriers for years; however, self-limiting fever, transient anemia, neurologic dysfunction, lymphadenopathy, reproductive disorders, aortic valvular endocarditis, and neutrophilic myocarditis have been described in experimentally or naturally infected cats. Two cats in a North Carolina shelter died with pyogranulomatous myocarditis and diaphragmatic myositis. Bacteria were visualized in the lesions by Warthin-Starry silver impregnation and by B. henselae immunohistochemistry. B. henselae DNA was amplified and sequenced from the heart of 1 cat and from multiple tissue samples, including heart and diaphragm, from the second cat. This study supports a potential association between B. henselae and what has been historically described as “transmissible myocarditis and diaphragmitis” of undetermined cause in cats.
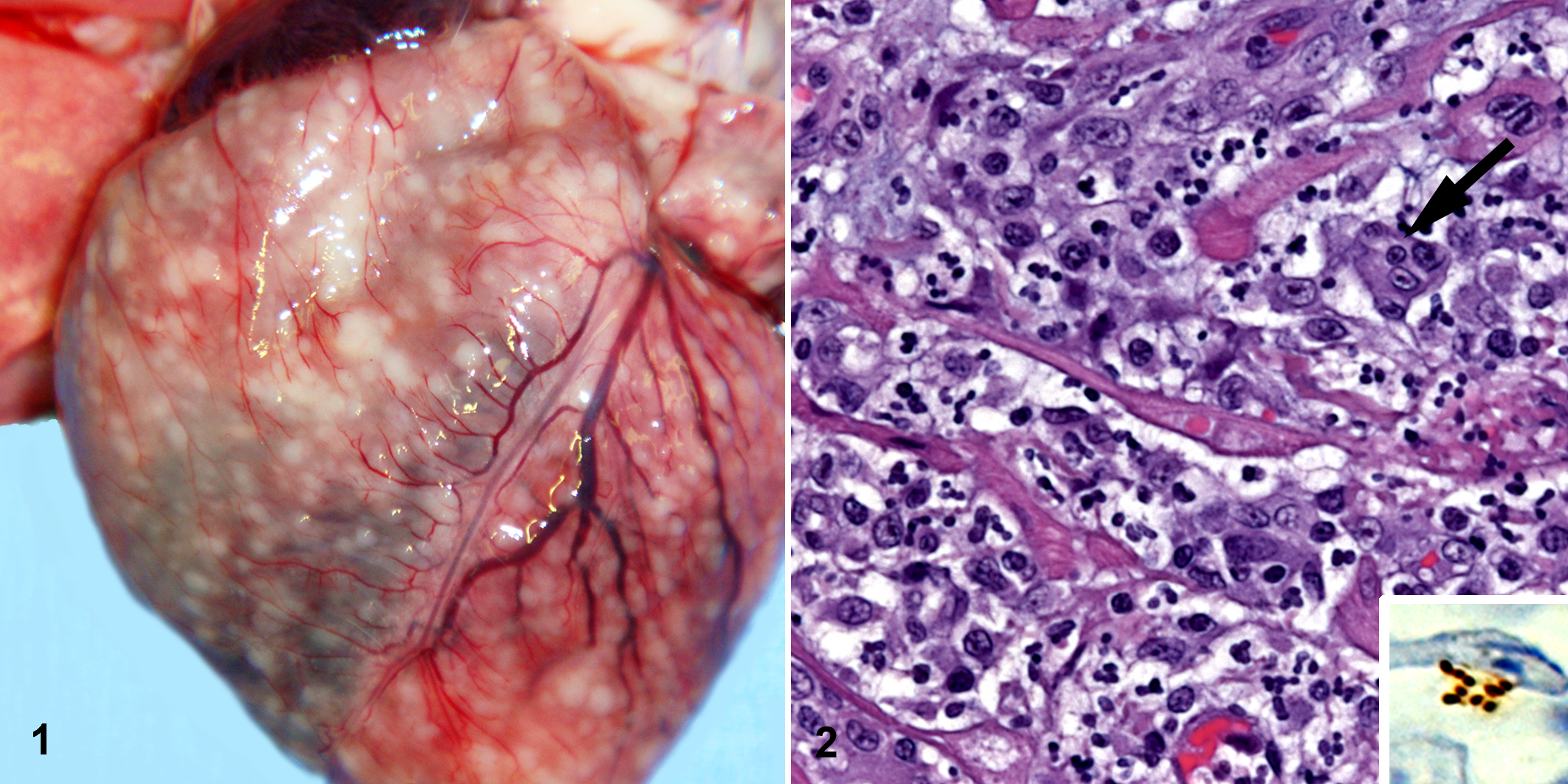
Fever, lethargy, and watery diarrhea developed in 10 cats, of which 2 died, after a litter of flea-infested kittens were introduced to the shelter. No helminth eggs or protozoa were detected by direct or floatation fecal tests. Blood samples tested negative for feline leukemia virus, feline panleukopenia virus, and feline immunodeficiency virus. An approximately 8-week-old female kitten (No. 1) was treated subcutaneously with dexamethasone, triamcinolone, and vitamin B12 (cyanocobalamin), and oral oseltamivir phosphate in a molasses suspension. The kitten had formed stools within 48 hours but developed acute respiratory distress and died about 1 week later. At necropsy by the attending veterinarian, pleural effusion and biventricular cardiac dilation were accompanied by numerous, discrete to coalescing, tan-white, pinpoint to 2-mm nodules in the myocardium (Fig. 1 ) and diaphragm. Only formalin-fixed heart was submitted for diagnostic evaluation. Histologically, the myocardial lesions were multifocal to coalescing pyogranulomas. Feline infectious peritonitis was suspected; however, immunohistochemistry was negative for feline coronavirus.
Two weeks after the death of cat No. 1, an 8-month-old castrated male domestic shorthair cat (No. 2), which had been cohoused with the flea-infested kittens, was examined because of anorexia of 2 days’ duration. Physical examination abnormalities were limited to lethargy and fever (40.1°C). Shortly after admission to the hospital, cat No. 2 died acutely. Necropsy findings included enlarged thoracic and mesenteric lymph nodes and myocardial and diaphragmatic nodules like those in cat No. 1. Gross lesions were not seen in other organs. Formalin-fixed specimens of heart, diaphragm, and thoracic and mesenteric lymph nodes—plus unfixed specimens of heart, diaphragm, mesenteric lymph nodes, and ileum—were submitted to diagnostic laboratories for histologic and microbiologic evaluation. Liver, spleen, kidney, pancreas, small intestine, lymph nodes, and lung were frozen at –18°C.
Histologic findings included lymphoid hyperplasia in the lymph node and pyogranulomatous inflammation of the myocardium and diaphragm. Immunohistochemistry was negative for feline coronavirus, Toxoplasma gondii, and Leptospira. No bacterial or viral pathogens were isolated from the tissues or feces of cat No. 2. We reviewed paraffin sections stained with hematoxylin and eosin from the hearts of both cats and from the diaphragm of cat No. 2. In both cats, the lesions consisted of aggregates of neutrophils and macrophages that ranged from microscopic clusters to partially organized pyogranulomas up to 2 mm in diameter (Fig. 2). The number of macrophages varied from few to many; multinucleated giant cells were rare. Myocardial lesions were disseminated through both ventricles, the atria, and the interventricular septum. Larger granulomas had hemorrhage and central necrosis with loss of cardiocytes. Only a few individual lymphocytes were found in the granulomas or in adjacent perivascular areas; plasma cells were rarely observed. The epicardium and endocardium, as well as the pleural and peritoneal surfaces of the diaphragm, were spared, except where granulomas had expanded to contact one of these surfaces. Vasculitis was not noted. Additional formalin-fixed tissues from cat No. 2 included liver, spleen, kidney, pancreas, lymph node, small intestine, and lung. Histologic lesions were not observed in the liver, kidney, lung, or pancreas. One microscopic pyogranuloma, like those in the heart and diaphragm, was in the tunica muscularis of the small intestine. Follicular hyperplasia was noted in the mesenteric lymph node and spleen. Gomori-Grocott methenamine silver histochemistry was negative for fungi. Clusters of short bacilli had been identified in the hearts from both cats and from the diaphragm of cat No. 2 by Warthin-Starry or Steiner silver histochemistry at the reference laboratories. In our laboratory, Steiner and Steiner silver histochemistry was negative for bacteria, whereas the Warthin-Starry silver technique delineated extracellular bacilli, 1 to 2 μm long, in the centers of myocardial and diaphragmatic granulomas. Bacteria were not detected with Warthin-Starry technique in sections of liver, lung, spleen, kidney, small intestine, lymph nodes, or pancreas.
Frozen lung, liver, kidney, duodenum, pancreas, spleen, and lymph nodes from cat No. 2 and paraffin blocks of myocardium (from both cats) and diaphragm (from cat No. 2) were tested for Bartonella sp DNA by polymerase chain reaction (PCR) amplification. Frozen and formalin-fixed, paraffin-embedded (FFPE) tissues were extracted using QIAamp DNA Blood Mini Kit per the manufacturer’s instructions (Qiagen, Valencia, California). Each tissue was sampled using a single-use sterile scalpel blade to avoid DNA carryover between samples. 13 A blank reagent negative control was included with each set of DNA extractions. As described, 5 Bartonella genus–specific PCR was performed using primers designed to amplify the 16S-23S intergenic transcribed spacer (ITS) region, pap31 and rpoB genes. DNA from blood of a healthy dog was used as the negative control; 0.001 pg/μl (equivalent to 2.5 genomic copies per reaction) of B. henselae (ITS Houston I strain) genomic DNA was used as the positive control. All amplicon products were sequenced to establish the Bartonella sp and genotype. Using the 16S-23S ITS primers, B. henselae DNA was amplified and sequenced from frozen tissues (kidney, lung, liver, duodenum, pancreas, spleen, and abdominal lymph node) and from FFPE heart and diaphragm from Cat No. 2. B. henselae DNA was also amplified and sequenced from the FFPE heart of cat No. 1.
Cat No. 2 was coinfected with 2 B. henselae ITS genotypes (SA2 and Cal I), whereas only the Cal I genotype was sequenced from the heart of cat No. 1. An identical pap31 gene sequence was amplified from both B. henselae Cal I strains (ie, cat Nos. 1 and 2), but there was no amplification from the 3 B. henselae SA2 PCR-positive FFPE tissues. A 652 base pair–fragment of the B. henselae rpoB gene was amplified and sequenced from 7 frozen tissue samples but from only 1 FFPE tissue sample. No rpoB amplicon was obtained from the FFPE myocardium of cat No. 1.
Immunohistochemistry was performed using a 1:100 dilution of a B. henselae mouse monoclonal antibody in phosphate-buffered saline (Abcam Inc., Cambridge, Massachusetts) in 5-μm sections of FFPE tissues, with diaminobenzidine as chromogen and Mayer hematoxylin as counterstain. Positive anti–B. henselae immunoreactivity labeled extracellular organisms that ranged in length from 1 to 2 μm (Fig. 2 inset) and were usually in the center of myocardial pyogranulomas in both cats and in the diaphragmatic granulomas of cat No. 2.
We provide molecular and immunohistochemical evidence of B. henselae infection in 2 cats from a North Carolina animal shelter. The pathologic findings of granulomatous myocarditis and diaphragmitis in these 2 cats resemble those described in naturally and experimentally infected cats by Pedersen and coworkers in the early 1990s. 10 As in the cats of this report, clinical manifestations were nonspecific in the “rag doll kittens” described by Pedersen et al and were limited to lethargy, fever, and decreased response to external stimuli. Based on high fever, generalized lymphadenopathy, and a seasonal (summer) occurrence, vector transmission of an infectious agent was suspected by Pedersen and colleagues. Transmissibility of a putative infectious agent was implicated by Pedersen and colleagues by intraperitoneal inoculation of blood from a naturally infected cat into specific-pathogen-free cats. Uniphasic and biphasic fever patterns of 1 to 3 days’ duration were documented in the experimentally infected cats. When necropsied during the febrile stage, 4 experimentally infected cats had a unique distribution of pyogranulomatous inflammation in the heart and diaphragm. Microscopic pyogranulomatous foci were infrequently observed in other organs, including the tongue, liver, and cecal lymph node. Two outwardly healthy experimentally inoculated cats were euthanized and necropsied 3 to 4 weeks after resolution of fever. No gross lesions were reported; however, 1 cat had multifocal aggregates of lymphocytes in the myocardium, and both cats had evidence of chronic lymphoid stimulation. Unfortunately, the blood from the cats and frozen or FFPE tissues from the naturally and experimentally infected cats were discarded.
Pedersen and colleagues were unable to define an infectious cause for transmissible granulomatous myocarditis. 10 No growth was obtained from aerobic, anaerobic, or fungal cultures of heart, diaphragm, spleen, or mesenteric lymph nodes. There were no cytopathic effects in cell culture, and viral particles were not seen by electron microscopy of the inoculated cell cultures. In addition, no organisms were observed in the heart or diaphragm from experimentally inoculated cats with Warthin-Starry silver or by electron microscopy. In our study, bacteria were visualized by Warthin-Starry silver in myocardial lesions from both cats in our laboratory and by Steiner and Steiner silver technique in other diagnostic laboratories. The reason for the discrepancy between our results and those of Pedersen’s group is unknown; however, silver impregnation, immunohistochemistry, and PCR are all relatively insensitive techniques for the identification of B. henselae in lymph node biopsy specimens from human patients with classic cat scratch disease, and only rarely do all 3 testing modalities generate positive results for a given sample. 3
Even though cats are usually considered to be healthy carriers of B. henselae, 2 feline disease has been documented in natural and experimental infections with this bacterium.4,6 Evolving evidence suggests that Bartonella sp can cause endocarditis and myocarditis in cats, as reported for dogs and human patients.4,8 Following experimental infection by fleas 1 or needle inoculation, 6 myocarditis has been reported in young specific-pathogen-free cats infected with B. henselae. Direct evidence is lacking to explain the apparent muscle tropism for B. henselae in the current study; however, Pedersen’s group reported increased creatinine phosphokinase values at the peak of fever in several cats, which could indirectly support a muscle tropism for strains of this bacterium. Experimentally, differences in pathogenicity among B. henselae strains have been reported; therefore, one or more virulence factors may affect disease expression. 14 In this study, the older of the 2 cats was coinfected with 2 ITS genotypes (SA2 and Cal I), whereas only the Cal I B. henselae genotype was sequenced from the index kitten. Both B. henselae Cal I strains contained identical bacteriophage pap31 gene sequences. Whether bacteriophages or various B. henselae ITS genotypes are associated with differences in virulence has not been determined; however, the combination of a potentially virulent strain of B. henselae, young age, poor nutrition, chronic diarrhea, exogenous corticosteroid administration, and stress may all have contributed to the development of severe disease and death in cat No. 1. Strain virulence or sequential infection with 2 B. henselae strain types might be an important consideration for cat No. 2, as this cat had been healthy and had resided in the shelter for several months before exposure to the flea-infested kittens. However, because other organs were not cultured (eg, liver, lung, spleen), other potential pathogens could have been missed and may have contributed to the disease in both cats in this study. Other infectious agents associated with feline myocarditis and myositis include Toxoplasma gondii, Streptococcus canis, feline immunodeficiency virus, and feline panleukopenia virus.7,9,11,12 Diagnostic testing failed to detect these agents or feline leukemia virus, feline coronavirus, or feline calicivirus in the cats in this study.
Our laboratory has documented the potential for DNA cross-contamination when using paraffin-embedded tissues for PCR detection of Bartonella spp. 13 Therefore, extreme care was taken to avoid DNA carryover while processing samples. Importantly, formalin fixation appeared to decrease the sensitivity of PCR amplification when results from frozen and fixed tissues were compared. In conclusion, our results suggest that B. henselae should be included in the differential diagnosis of myocarditis and/or diaphragmatic myositis in cats. Molecular and immunohistochemical studies are warranted to determine if B. henselae, other Bartonella spp, or other pathogens can be consistently found in cats with pyogranulomatous myocarditis and diaphragmatic myositis. Antemortem Bartonella culture is recommended to derive isolates of virulent B. henselae strains from cats with myocarditis. These isolates could be used to attempt to satisfy Koch’s postulates for this pathologic entity.
Footnotes
Acknowledgements
We thank Antech Diagnostics and the Animal Health Diagnostic Center, Cornell University, for initial diagnostic testing and for providing the paraffin-embedded tissue for polymerase chain reaction testing. We thank Tonya Lee for editorial assistance and Bayer Animal Health for supporting M. Varanat, a graduate student in the Intracellular Pathogens Research Laboratory.
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: With S. Sontakke and North Carolina State University, Dr Breitschwerdt holds US patent No. 7115385, media and methods for cultivation of microorganisms, issued October 3, 2006. Dr Breitschwerdt is the chief scientific officer and Dr Maggi is the scientific technical advisor and laboratory director for Galaxy Diagnostics, which provides diagnostic testing for Bartonella.
The authors declared that they received no financial support for their research and/or authorship of this article.
