Abstract
Primary pulmonary tumors are less common in dogs than secondary (metastatic) tumors. Most primary tumors are malignant and of epithelial origin. Pulmonary large-cell carcinoma is considered extremely rare in domestic animals, and some of the few reported cases actually may have been cases of malignant pulmonary histiocytosis. An 8-year-old female Wire Fox Terrier with diffuse alveolar pattern radiographically was euthanatized when it failed to respond to antibiotic therapy. Histologically, pulmonary alveolar spaces contained clusters of large round anaplastic cells with ample eosinophilic cytoplasm and large irregularly shaped nuclei with prominent nucleoli. Immunohistochemistry was used to distinguish large-cell carcinoma from malignant pulmonary histiocytosis. Tumor cells had strong immunoreactivity for cytokeratin, consistent with epithelial origin. However, a substantial percentage of the neoplastic cells co-expressed vimentin and MHC-II. The type II alveolar epithelial cell was considered the cell of origin of the neoplasm based on the presence of lamellar bodies in some neoplastic cells and immunoreactivity for surfactant protein A and thyroid transcription factor-1.
Primary lung tumors are uncommon in dogs, with a prevalence of 1% in the overall canine population, in contrast to the situation in humans. 21 According to the World Health Organization (WHO) classification, malignant pulmonary epithelial neoplasms in domestic animals are classified into 9 types: adenocarcinoma (with 2 subtypes: papillary or acinar adenocarcinoma and bronchioloalveolar carcinoma), squamous carcinoma, adenosquamous carcinoma, small-cell carcinoma, large-cell carcinoma, neuroendocrine tumor, pulmonary blastomas, and combined carcinoma. The most prevalent types in veterinary medicine are adenocarcinoma and squamous cell carcinoma. 3 Large-cell carcinoma has been defined as extremely rare and poorly documented. 20 Its prevalence in retrospective studies is variable: 0/40 (0%), 7 18/218 (8.2%), 10 33/210 (15.7%), 11 or 2/120 (1.6%). 13 This variability is probably because of the fact that several previously reported cases were actually malignant histiocytosis, a neoplasm with similar histologic features: round cells with abundant eosinophilic or foamy cytoplasm and the presence of multinucleated giant cells. 3 Large-cell carcinomas have not been well characterized, in contrast with other uncommon pulmonary tumors, such as adenosquamous carcinoma 15 and small-cell carcinoma. 5 The possibility of confusing pulmonary large-cell carcinoma with malignant histiocytosis, because of their morphologic similarities, increases the interest in furthering our knowledge of the biologic behavior and cellular origin of this uncommon neoplasm. This report describes the clinical, pathologic, and immunohistochemical findings in a pulmonary large-cell carcinoma in a dog.
An 8-year-old intact female Wire Fox Terrier was presented with a 3-week history of anorexia, depression, and episodes of nonproductive coughing. Serum biochemistry results were unremarkable. In thoracic radiographs, the lungs had an alveolar pattern with fluid density, especially in the cranial lobe of the right lung. The cardiac silhouette was normal. Based on the radiographic appearance, a presumptive diagnosis of pneumonia was considered, and parenteral antibiotic therapy (cefalexin and gentamicin) was administered. Three weeks later, the owner reported no response to the antibiotic therapy and the development of dyspnea with minimal activity. Radiographs were repeated and the alveolar pattern was more evident, affecting both lungs. A diagnosis of diffuse pulmonary tumor was considered. Because of the worsening state of the dog and poor prognosis, the owner requested euthanasia.
At postmortem examination in our laboratory, both lungs were firm and failed to collapse. The dorsal area of the right cranial lobe had homogeneous yellowish-white discoloration (Fig. 1); scattered grayish areas of consolidation were observed in remaining lung lobes. A few milliliters of pleural effusion were found. No macroscopic signs of metastases in other organs were observed. Lung samples were fixed in 10% buffered formalin for histopathologic analysis. After routine embedding in paraffin, 5-µm sections were cut and stained with hematoxylin and eosin (HE). Samples were also embedded in Epon resin for electron microscopy of ultrathin sections, stained with lead citrate and uranyl acetate.
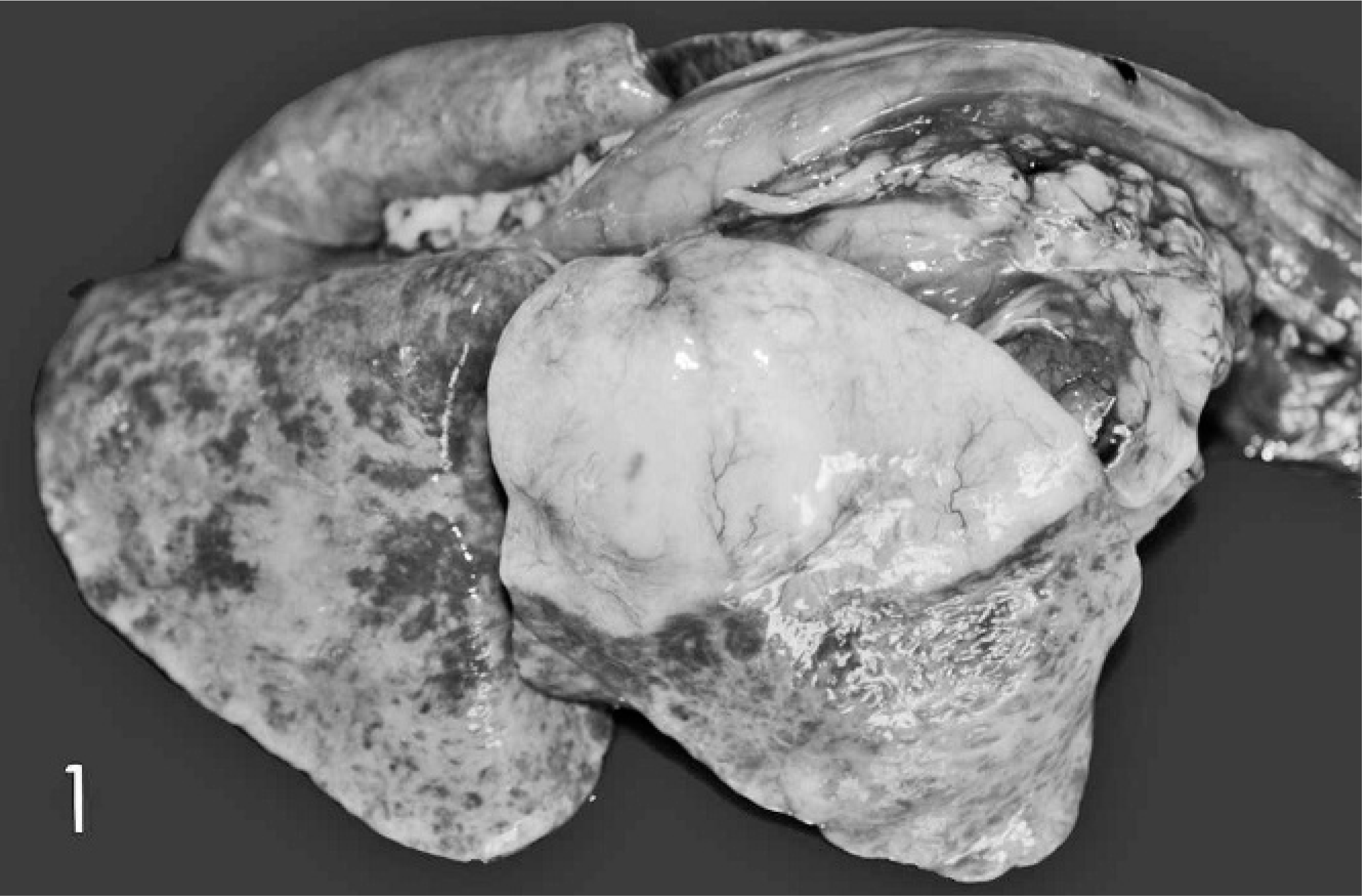
Large-cell carcinoma, lung; dog. Macroscopic image of the lung, with pallor in the dorsal area of the cranial lobe and mottling of other lobes.
Histopathologically, neoplastic cells filled alveolar spaces throughout the lung (Fig. 2). The tumor had multifocal necrosis with cholesterol clefts. Neoplastic cells were arranged in small groups occupying the alveolar spaces; in some areas they infiltrated the alveolar wall. The neoplastic cells were large and round or polyhedral with well-defined cell outlines. Cytoplasm was abundant and eosinophilic or foamy to vacuolated. Nuclei were pleomorphic (round to oval or cleaved), and vesicular with prominent nucleoli; binucleated and multinucleated cells were observed (Fig. 3). Anisocytosis and anisokaryosis were marked and abnormal mitotic figures were observed. In agreement with Griffey et al., 6 mitotic figures were counted in 20 random 400× fields to calculate the mitotic index.
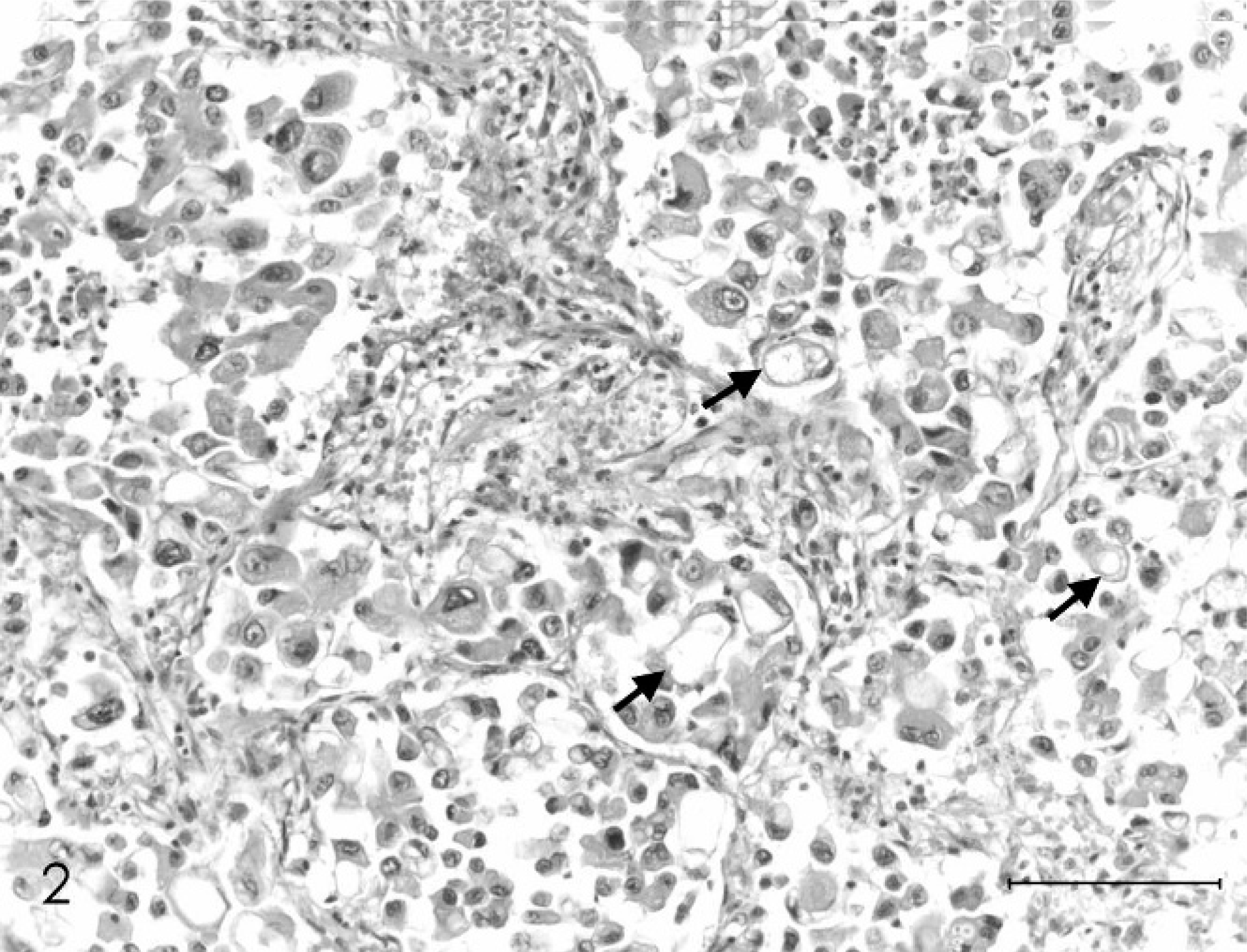
Large-cell carcinoma, lung; dog. Alveolar space contains neoplastic cells with ample cytoplasm and distinct borders. Note large cytoplasmatic vacuoles in some neoplastic cells (arrows). HE. Bar = 100 µm.
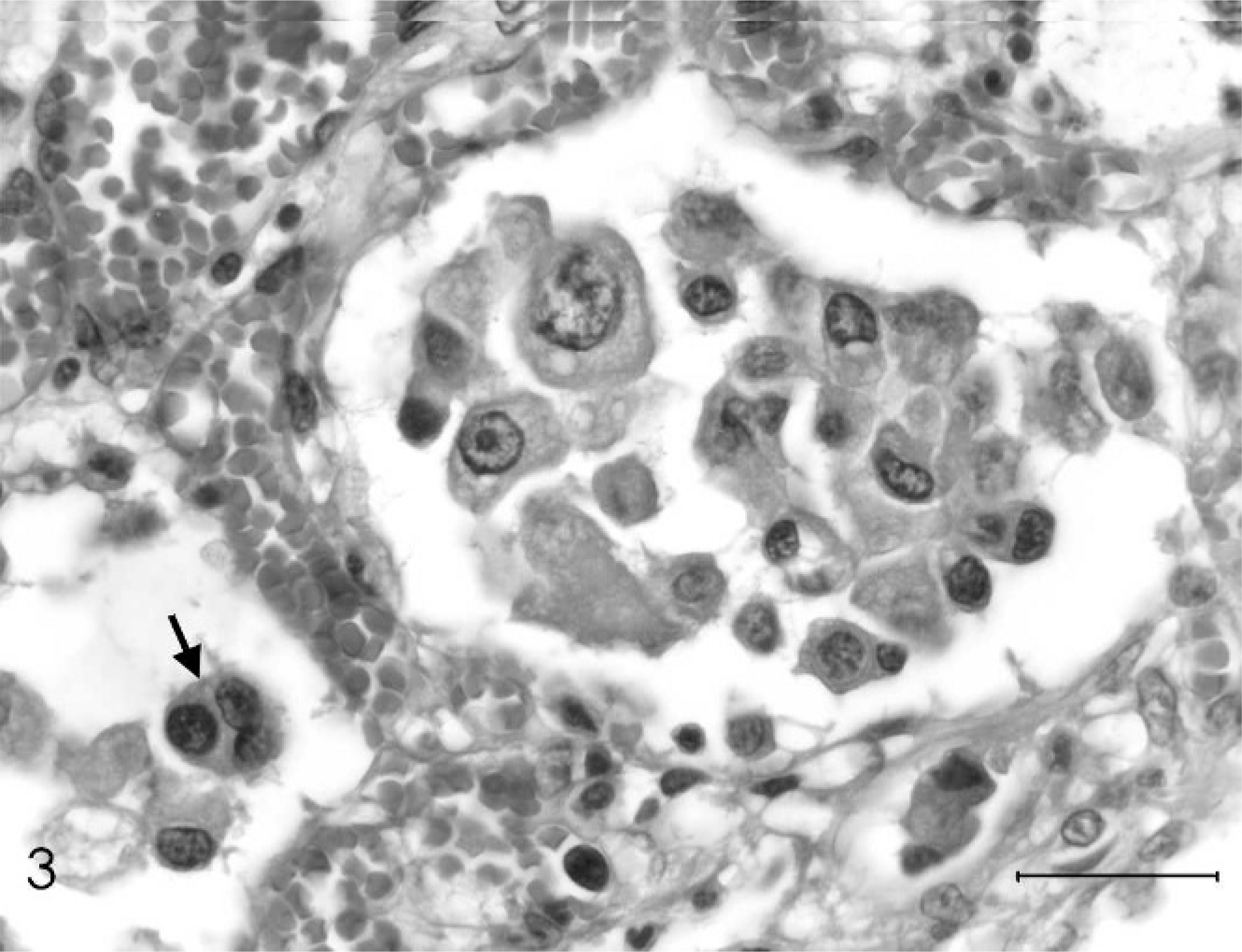
Large-cell carcinoma, lung; dog. Detail of the anaplastic tumor cells with marked anisokaryosis and presence of multinucleated cells (arrow). HE. Bar = 30 µm.
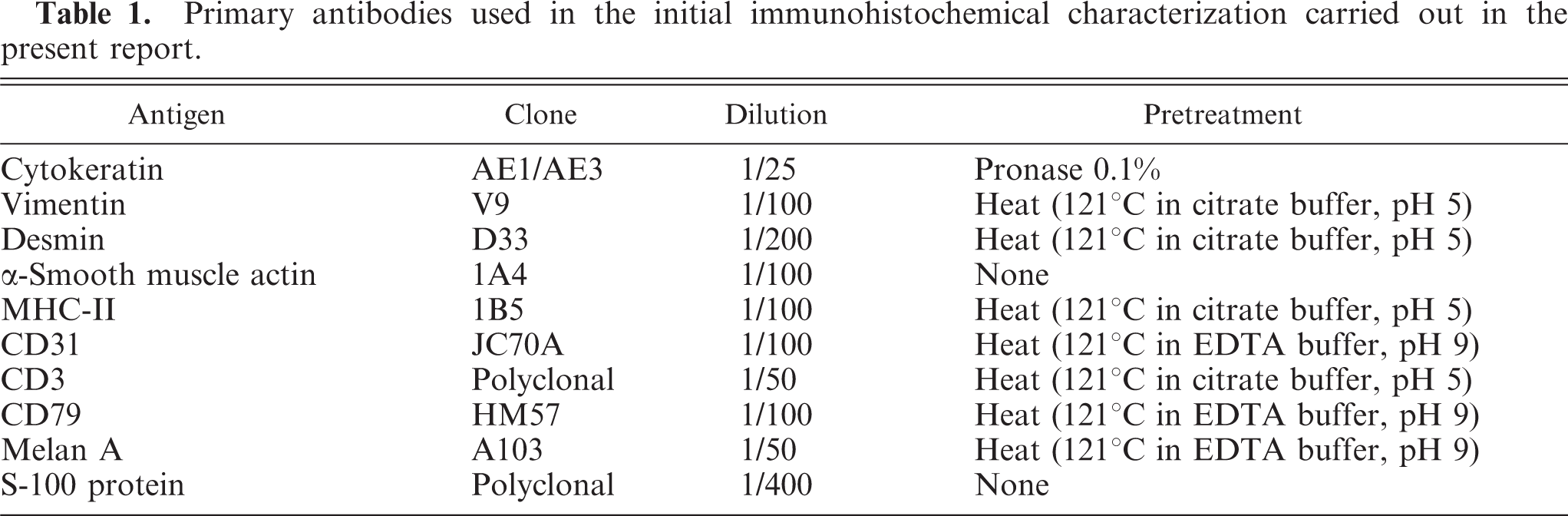
Preliminary immunohistochemistry was carried out by the avidin–biotin–peroxidase complex method (ABC) with 10 primary antibodies; clone, dilution, and pretreatment for antigen retrieval are summarized in Table 1. This panel of antibodies has been used as initial screening in the characterization of neoplasms in our laboratory. 14 All antibodies were purchased from DakoCytomation (Carpinteria, CA). Sections were counterstained with Mayer's hematoxylin; positive and negative control sections were included for each antibody. Tumor cells had strong and uniform immunoreactivity for cytokeratin (Fig. 4), and moderate patchy reactivity for vimentin (Fig. 5) and for MHC-II (Fig. 6). Neoplastic cells were negative for expression of desmin, α-smooth muscle actin, CD31, melan A, S-100, CD3, and CD79. Ultrastructurally, some neoplastic cells had cytoplasmic lamellar bodies and microvilli in the apical cell membrane (data not shown); both features are characteristics of the alveolar type II cells. Regrettably, autolytic changes and the initial fixation with formalin precluded detailed ultrastructural evaluation.
Primary antibodies used in the initial immunohistochemical characterization carried out in the present report.
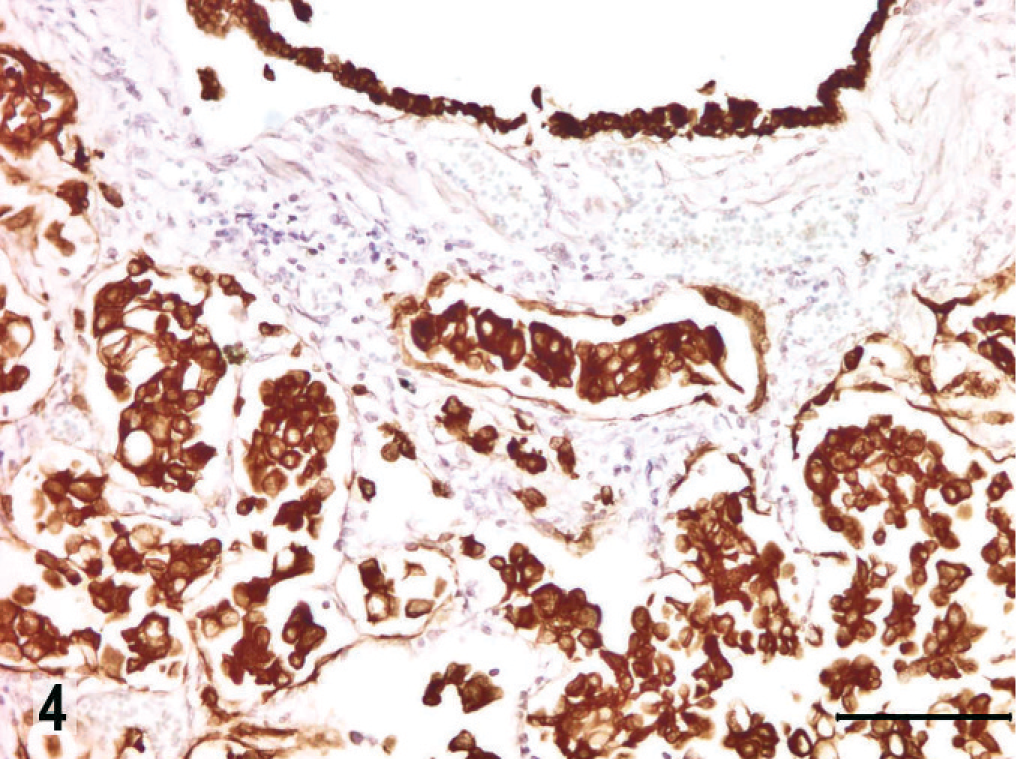
Large-cell carcinoma, lung; dog. Immunolabeling for cytokeratin. Note the strong expression of cytokeratins in all neoplastic cells. ABC, Mayer's hematoxylin counterstain. Bar = 100 µm.
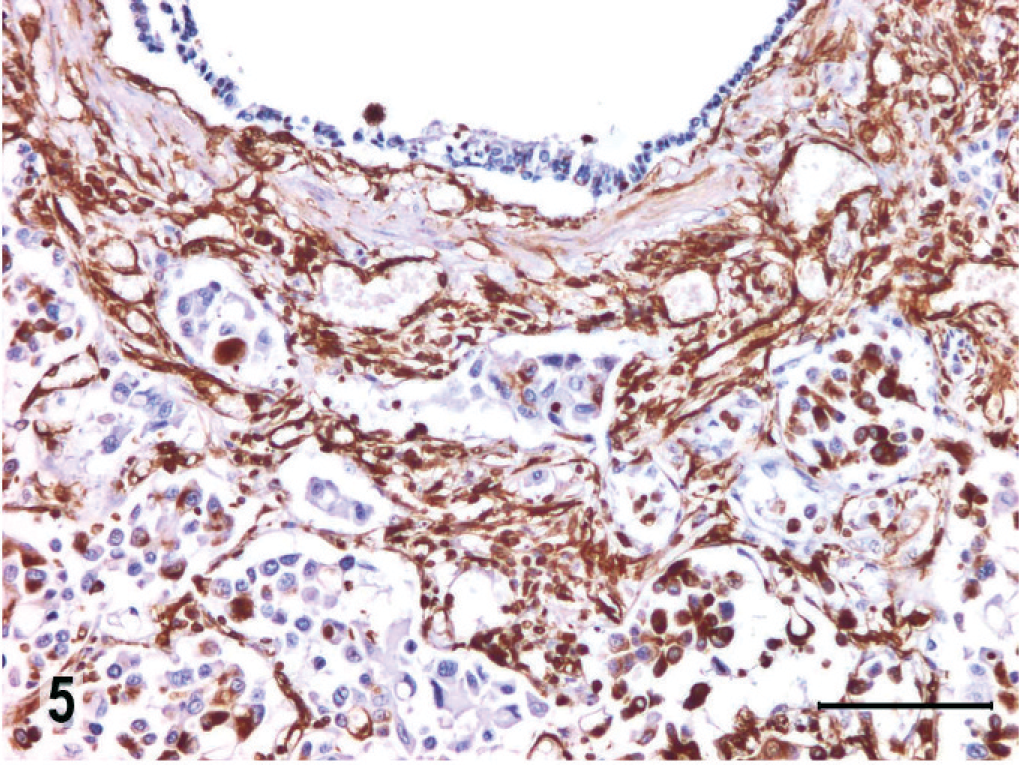
Large-cell carcinoma, lung; dog. Immunolabeling for vimentin. Vimentin expression is observed in the pulmonary interstitium and in some intra-alveolar tumor cells. ABC, Mayer's hematoxylin counterstain. Bar = 100 µm.
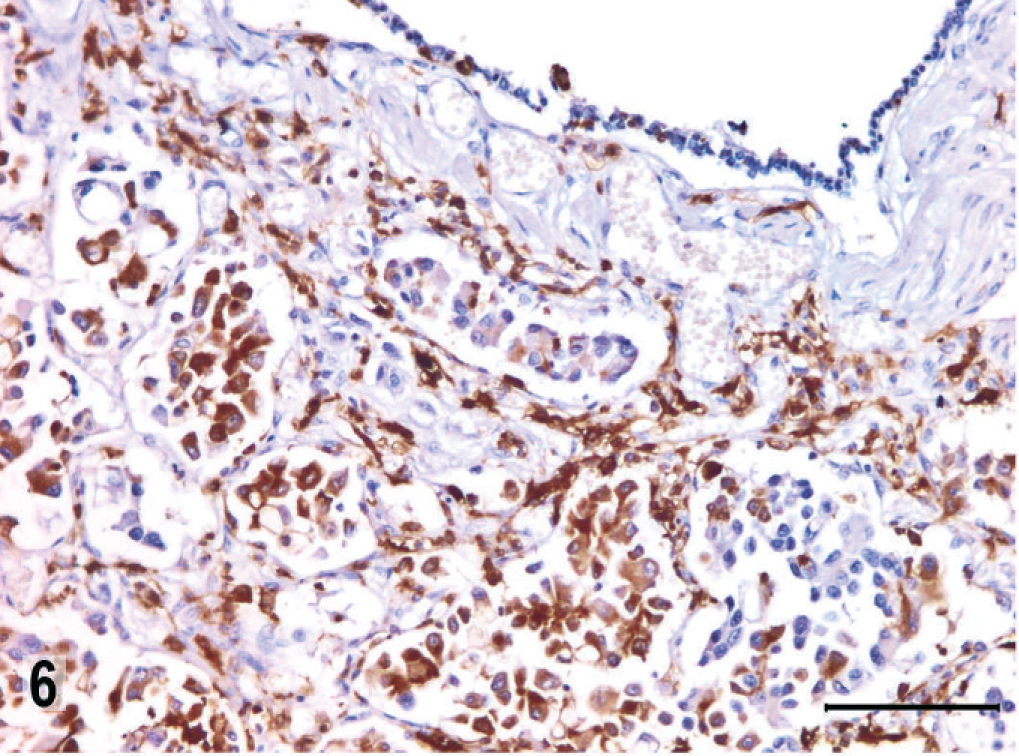
Large-cell carcinoma, lung; dog. Immunolabeling for MHC-II. Note substantial number of intra-alveolar tumor cells with MHC-II expression. ABC, Mayer's hematoxylin counterstain. Bar = 100 µm.
To confirm the cellular source of the tumor, sections were subsequently immunolabeled with a mouse monoclonal antibody against thyroid transcription factor-1 (TTF-1, DakoCytomation, clone 8G7G3/1), which is a reliable marker for canine primary pulmonary carcinomas, 13 and with a goat polyclonal antibody against the surfactant apoprotein A (N-19; Santa Cruz Biotechnology, Santa Cruz, CA) using an ABC technique and citrate buffer antigen retrieval in the case of surfactant marker. Immunoreactivity in the neoplastic cells for surfactant apoprotein A (Fig. 7) and for TTF-1 (Fig. 8) supported our conclusion that the neoplasm was a primary pulmonary carcinoma derived from alveolar type II cells.
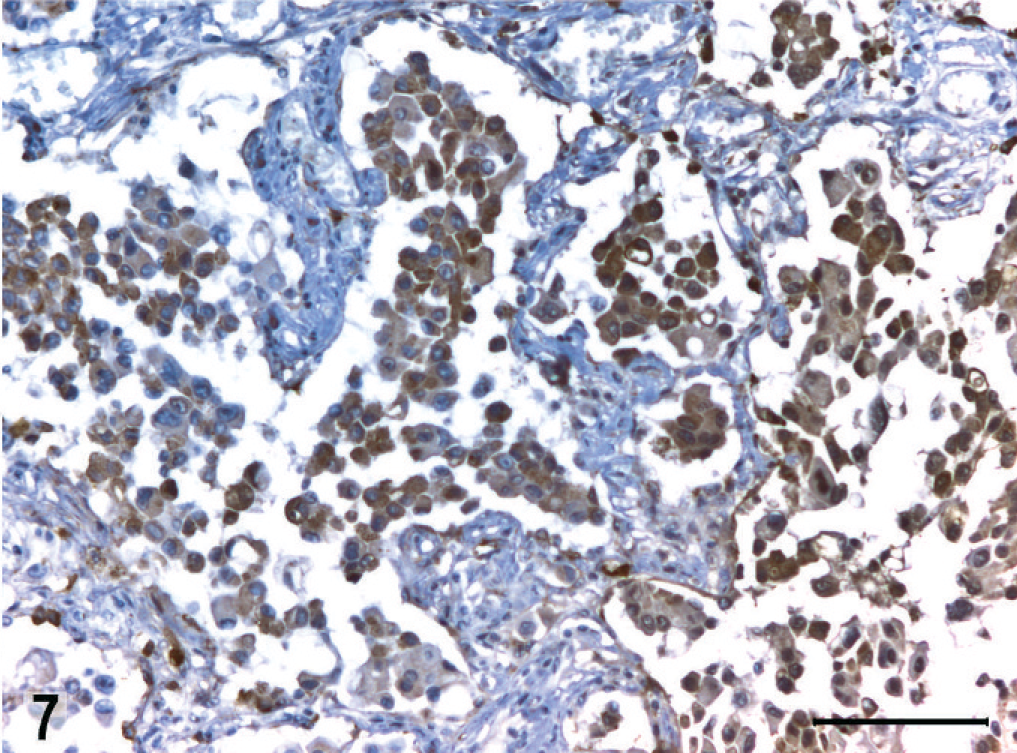
Large-cell carcinoma, lung; dog. Immunolabeling for surfactant protein A. Note substantial number of intra-alveolar tumor cells with surfactant protein A expression. ABC, Mayer's hematoxylin counterstain. Bar = 100 µm.
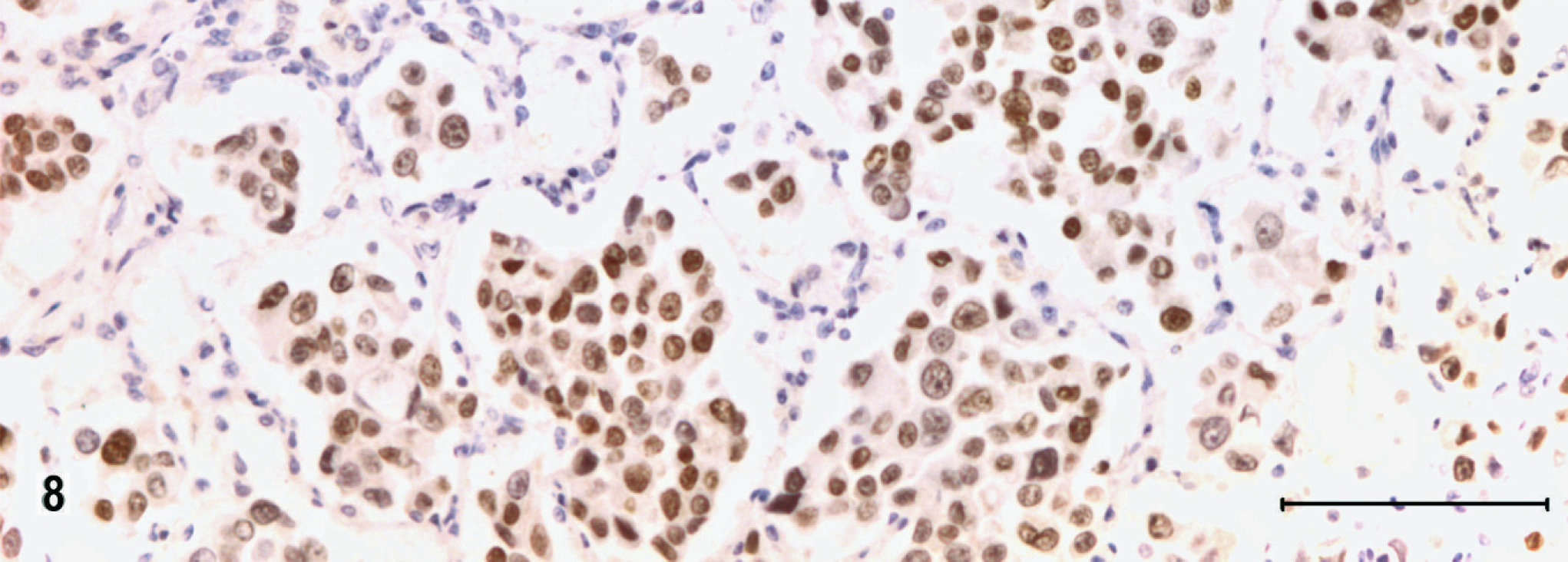
Large-cell carcinoma, lung; dog. Immunolabeling for thyroid transcription factor-1 (TTF-1). Note substantial number of intra-alveolar tumor cells with TTF-1 expression. ABC, Mayer's hematoxylin counterstain. Bar = 100 µm.
Proliferation indices, defined as a measure of the number of cells in the S-phase, in canine pulmonary carcinomas vary by histologic type. 6 Immunohistochemistry for the proliferation marker, Ki-67, was performed by the ABC technique with a monoclonal antibody (clone MIB-1, DakoCytomation). The proportion of neoplastic cells that expressed Ki-67, calculated following the methodology of Griffey et al., 6 was 524/1,000.
The present study describes a case of large-cell pulmonary carcinoma with a diffuse pattern of growth that involved the entire lung. The tumor did not produce a defined mass, but apparently spread via airways, probably from the dorsal area of the right cranial lobe, filling alveolar spaces and maintaining the gross structure of the lung. The radiographic appearance of the tumor mimicked that of pneumonia, leading to the initial misdiagnosis. This large-cell carcinoma is atypical, because bronchioloalveolar carcinomas, derived from Clara cells or alveolar type II cells, usually present as discrete masses; nevertheless, diffuse patterns have been described in dogs. 1, 3
The strong immunohistochemical expression of cytokeratins confirmed the epithelial origin of the tumor. However, some neoplastic cells coexpressed the nonepithelial markers, vimentin and MHC-II. The constitutive expression of MHC-II by alveolar type II cells has been well established. 2 A possible explanation for the unexpected expression of vimentin in this carcinoma is the capacity of alveolar type II cells to undergo transition to a mesenchymal phenotype in response to certain cytokines. 19 One sign of epithelial-mesenchymal transition is the expression of vimentin. It could be hypothesized that poorly differentiated (anaplastic) neoplastic alveolar type II cells would express not only cytokeratins and MHC-II, but also vimentin. Coexpression of vimentin and cytokeratin has been described in veterinary medicine in carcinomas from several locations (kidney, urinary bladder, genital tract, and liver) but is unusual in the lung. 12 However, expression of vimentin has been reported in poorly differentiated human pulmonary large cell carcinomas. 17 Coexpression of both cytokeratins and vimentin intermediate filaments has been associated with increased invasiveness in several kinds of nonmesenchymal tumors, 8 because the coexpression of epithelial and mesenchymal markers confers a selective advantage to tumor cells in their reaction to cytokine signals from extracellular matrices.
Analysis of the results for the Ki-67 marker showed a high percentage (524 positive cells per 1,000 neoplastic cells) of proliferating cells in the tumor. The proliferation index in this tumor, as estimated by immunohistochemistry for Ki-67, was higher than that reported for other more differentiated pulmonary carcinomas, such as bronchioloalveolar carcinoma or adenocarcinoma. 6 The Ki-67 proliferation index has been proposed as an unfavorable prognostic indicator for human pulmonary carcinomas 9 and in several types of malignant neoplasms in veterinary medicine. 4, 18 Similarly, the mitotic index of 18 mitotic figures in 20 400× fields, was also higher that described for bronchioloalveolar carcinomas or adenocarcinomas. 6 The high proliferation together with the advantages of coexpression of epithelial and mesenchymal markers could explain the rapid dissemination and aggressive course of this neoplasm. Indeed, vimentin expression and high Ki-67 expression have been associated with a more aggressive phenotype in human breast carcinomas. 16
In conclusion, our results suggest that this particular pulmonary large-cell carcinoma was derived from alveolar type II cells. This neoplasm could be considered as an anaplastic subtype of adenocarcinoma within the classification established by Dungworth et al. 3 Cytokeratin, TTF-1 and surfactant protein A immunoreactivity were required to distinguish the tumor from the morphologically similar malignant histiocytosis. The rarity of pulmonary large-cell carcinoma in domestic animals may explain why classification has been based historically more on the histologic appearance than on the histogenesis. Additional cases should be characterized to compare the clinical behavior of this anaplastic tumor with that of more differentiated pulmonary adenocarcinomas.
