Abstract
The dog is the only species, other than humans, in which spontaneous prostatic cancer occurs; therefore, dogs are a valuable model for the study of factors that regulate tumor progression. Angiogenesis is important in the development and spread of a variety of cancers, including prostate cancer. To better define the role of cancer epithelial cells in prostate cancer neovascularization, immunohistochemical staining for angiogenic factors (vascular endothelial growth factor [VEGF], platelet endothelial cell adhesion molecule–1 [PECAM-1], Tie-2, and fibroblast growth factor–2 [FGF-2]) was performed on formalin-fixed, paraffin-embedded tissues from 10 normal prostates, 15 hyperplastic prostates, and 11 prostatic carcinomas from dogs. Normal and hyperplastic epithelial cells were negative for PECAM-1, VEGF, and Tie-2, while the same markers were expressed with a variable intensity of cytoplasmic staining by neoplastic cells. Mild to moderate FGF-2 staining was detected in all normal prostates with less than 10% of positive cells, mainly distributed in the basal layer. The percentage of FGF-2–positive hyperplastic cells was variable, with both basal and secretory cells exhibiting a perinuclear to diffuse cytoplasmic staining. The mean number of positive cells and the intensity of staining were higher in prostatic carcinomas than normal and hyperplastic prostates. Moreover, microvessel density analyzed on PECAM-1–stained slides was increased in prostate cancer compared with normal and hyperplastic prostates. Therefore, prostatic neoplastic cells are capable of simultaneous expression of various angiogenic factors and may increase tumor proliferation and angiogenesis in a paracrine and autocrine fashion.
Prostate cancer is the second leading cause of cancer-related death after lung cancer in men, and its incidence is steadily increasing in almost all countries. 9 Prostate cancer progression occurs in 2 stages, the first being androgen responsive and the second androgen independent with a high metastatic rate. 19 Dog prostates share anatomic and functional similarities with humans 28 and commonly and spontaneously develop benign prostatic hyperplasia (BPH), 29 high-grade prostatic intraepithelial neoplasia (HGPIN), and prostate cancer. 51 Thus, dogs are a highly relevant animal model of prostatic cancer.
The development of prostate cancer includes a multistep program of genetic alterations in pathways affecting cell cycle, apoptosis, adhesion, motility, and angiogenesis. It is well known that the angiogenic switch is vital for tumor growth and progression. 3,15 Cancer cells stimulate angiogenesis either by directly secreting growth factors and cytokines that act on the endothelial cells of adjacent vessels and microvessels or by activating angiogenic compounds in the extracellular matrix. 6
Different angiogenic proteins are overexpressed in solid tumors, therefore representing a potential target in the management of cancer. Vascular endothelial growth factor (VEGF), one of the most potent angiogenic factors, is expressed in normal human prostatic epithelium, 4,5,22,23 BPH, 20,22,50 and prostatic carcinomas 22,50,53 with variable intensity of staining. Nonneoplastic tissue has shown a low number of positively stained epithelial cells, 50,53 if not absence or weak immunoreactivity. 12,13 Tumor staining appears to correlate with degree of differentiation, with the highest expression in poorly differentiated human prostate cancers. 12,13
Basic fibroblast growth factor (bFGF or FGF-2) has attracted special attention for its angiogenic properties and for acting as a powerful mitogen stimulating cell differentiation and proliferation. 18 FGF-2 has broad biological functions that can play an important role in tumorigenesis, such as increasing motility and invasiveness, 25 as well as acting as an autocrine growth factor and stimulating the progression toward malignancy. 43 FGF-2 could also affect the cytotoxicity of chemical and other nonphysiological stresses inflicted on the cells, and this is particularly important for the interaction of cancer cells with chemotherapeutic drugs and other DNA-damaging agents. 56 In humans, results on immunolocalization of FGF-2 in nonneoplastic prostates are controversial, and the role of FGF-2 in prostatic carcinoma is still unclear, 16,37,46,49 although FGF-2 concentration has been shown to be elevated in the serum of affected patients. 31
The immunohistochemical expression of platelet endothelial cell adhesion molecule–1 (PECAM-1) or CD31 7,8 by endothelial cells has been extensively used for analyzing the microvascular density of human prostatic carcinoma, 41 while no reports are available on the potential expression of this marker in neoplastic epithelial cells.
Tie-2 is an endothelial cell-specific tyrosine kinase receptor expressed in the vascular system from the early stages of embryogenesis, 45 whose expression has been recently described in hematopoietic precursors and neoplastic cells in human cancers. 35,48
To better define the role of angiogenic factors in canine prostatic diseases, especially canine prostatic carcinoma, this study analyzes the immunohistochemical expression of VEGF, FGF-2, PECAM-1, and Tie-2 in canine normal, hyperplastic, and carcinomatous epithelial cells.
Materials and Methods
Study Samples
Thirty-six formalin-fixed, paraffin-embedded canine prostatic samples were selected from the archive of the diagnostic pathology service of the School of Veterinary Science, The University of Queensland. The hematoxylin and eosin–stained sections were reexamined by an experienced pathologist (C.P.) to confirm the histopathological diagnosis. Ten normal canine prostates from adult dogs, 15 samples of BPH, and 11 prostatic carcinomas were studied. BPH and prostatic carcinomas were derived from dogs ranging in age from 3 to 11 years and from 6 to 12 years, respectively. The most affected breeds were German Shepherd (3), Rottweiler (2), and Scottish Terrier (2) for BPH and German Shepherd (3) and Doberman Pinscher (2) for prostatic carcinoma.
Histology
Prostatic carcinomas were classified according to the growth patterns described by Lai et al: 26 micropapillary (papillary projections of neoplastic cells within extended ducts, which lack a distinct fibrovascular core), cribriform (ducts completely filled by neoplastic cells forming regular fenestrae with frequent central necrosis or comedonecrosis), solid (solid nests or individual anaplastic cells), sarcomatoid (neoplastic cells with a spindle morphology), small acinar/ductal (acinar structures within a scirrhous fibromuscular stroma), or tubulopapillary (dilated ducts with single to stratified layers of columnar cells).
Immunohistochemistry
The immunohistochemical procedure is provided in detail in Supplemental Material 1.
Evaluation of Immunohistochemistry (VEGF, PECAM-1, Tie-2, FGF-2) in Normal, Hyperplastic, and Carcinomatous Prostatic Epithelial Cells
A semiquantitative and a qualitative immunohistochemical assessment were performed as detailed in Supplemental Material 1.
Microvessel Density
Microvessel density (MVD) was analyzed in benign and malignant prostatic tissue by counting the average number of PECAM-1–positive vessels, as previously described 52 and detailed in Supplemental Material 1.
Statistical Analysis
Differences among the 3 groups were assessed by the χ 2 test and considered significant at P < .001.
Results
Histology
In the normal prostates, small to medium glands lined by a single layer of cuboidal to columnar cells forming occasional short papillary infoldings were present. BPH was characterized by epithelial hyperplasia, cystic dilation, and mild fibrosis. The most common types of prostatic carcinomas were cribriform (6 of 11; dog Nos. 26, 28, 33, 34, 35, 36) with comedonecrosis in 5 cases (dog Nos. 26, 28, 34, 35, 36) and small acinar/ductal (3 of 11; dog Nos. 29, 31, 32). A prevalent solid pattern with multifocal sarcomatoid areas (dog No. 27) and a micropapillary pattern (dog No. 29) were observed in 1 case each. Metastases were detected in 1 case (dog No. 27) in the lung, lymph nodes, and kidney.
Immunohistochemistry
In all samples, positive staining was observed in the fibrovascular stroma (VEGF, FGF-2) and blood vessel endothelium (VEGF, FGF-2, PECAM-1, Tie-2). The results of the immunohistochemical analysis are provided in the Supplemental Table S1.
Normal and hyperplastic epithelial cells were negative for PECAM-1 (Fig. 1), VEGF, and Tie-2. PECAM-1 was expressed in all 11 prostatic carcinomas with a weak (8 cases) to moderate (3 cases) cytoplasmic staining (Fig. 2) and a variable semiquantitative score. Tie-2 expression was detected in 9 of 11 prostatic carcinomas (Fig. 3), with a percentage of labeled neoplastic cells ranging from >0% to <10% (5 cases) to ≥10% to <25% (3 cases). The intensity of staining was mild to moderate. In prostatic carcinoma, VEGF-positive neoplastic cells (Fig. 4) were lower in number compared with Tie-2 and PECAM-1–positive cells, with a multifocal, randomly scattered cytoplasmic expression in less than 10% of cells in 8 of 11 samples and 10% to 25% of cells in 1 sample. Two cases were negative. Most of the neoplastic cells demonstrated a mild staining reaction.
Benign prostatic hyperplasia; dog No. 15. The immunohistochemical signal is limited to the blood vessel endothelium. Epithelial cells are negative. Inset: platelet endothelial cell adhesion molecule–1 (PECAM-1) immunostaining of endothelial cells. Immunohistochemistry (IHC) for PECAM-1.
FGF-2 was expressed in each group of prostate samples but differed in the semiquantitative and qualitative score. A mild to moderate cytoplasmic staining was detected in normal prostates (Fig. 5) with less than 10% positive cells, mainly distributed in the basal layer. In 2 of 10 normal prostates, occasional (1%–2%) randomly scattered FGF-2–positive cells were observed. A variable staining characterized BPH, with less than 10% (4/15), ≥10% to <25% (4/15), ≥25% to <50% (4/15), and ≥50% to <75% (3/15) cells. Both basal and secretory cell types were positive (Fig. 6), with a prevalent perinuclear pattern in the latter. The mean number of positive cells and the intensity of staining were higher in prostatic carcinomas (Fig. 7, 8) than in normal and hyperplastic prostates, with a semiquantitative score of 4+ in 6 of 11 cases, 5+ in 3 of 11 cases, and 3+ in 2 of 11 cases. The protein was diffusely distributed within the cytoplasm of neoplastic cells. Supplemental Figures S1 to S12 show the expression of the 4 different markers (PECAM-1, Tie-2, VEGF, FGF-2) in each prostatic carcinoma subtype.
The final immunohistochemical score of the FGF-2, PECAM-1, Tie-2, and VEGF expression in normal, hyperplastic, and carcinomatous prostatic epithelial cells is summarized in Table 1. The FGF-2 final score was significantly different between normal and hyperplastic prostates and between normal prostates and carcinomas (P < .001). The difference in PECAM-1, Tie-2, and VEGF expression was statistically significant between prostatic carcinomas and both normal and hyperplastic prostates (P < .001).
Immunohistochemical Score of FGF-2, PECAM-1, Tie-2, and VEGF Expression in Normal, Hyperplastic (BPH), and Carcinomatous (PC) Prostatic Epithelial Cells (Mean ± SD).
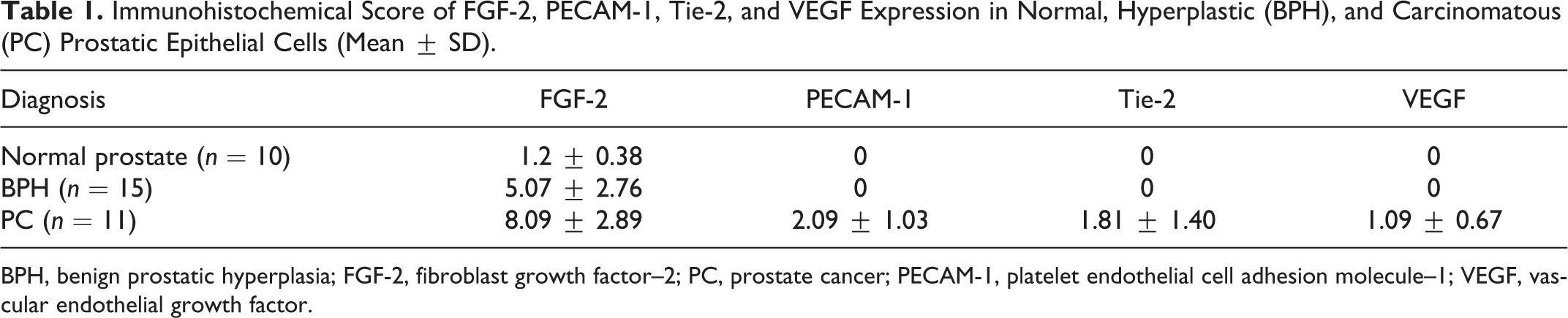
BPH, benign prostatic hyperplasia; FGF-2, fibroblast growth factor–2; PC, prostate cancer; PECAM-1, platelet endothelial cell adhesion molecule–1; VEGF, vascular endothelial growth factor.
Microvessel Density
The analysis of the microvessel density by PECAM-1 showed a statistically significant (P < .001) increase in the number of microvessels in prostate cancer (mean ± SD, 65.03 ± 6.63) compared with normal (11.02 ± 1.66) and BPH tissues (40.55 ± 69.23).
Discussion
This study suggests that canine prostatic carcinoma cells are capable of simultaneous production of various angiogenic factors (VEGF, PECAM-1, Tie-2) that are not expressed in normal or hyperplastic prostatic epithelial cells. Moreover, the increased expression of angiogenic factors by neoplastic cells is associated with an increased number of blood vessels.
VEGF is expressed by endothelial cells, tumor stromal cells, and prostate cancer cells, as already demonstrated in humans. 12,13,20 VEGF produced by neoplastic cells stimulates angiogenesis by acting in a paracrine fashion on adjacent endothelial cells. However, as vascular endothelial growth factor receptor (VEGFR)–1 and VEGFR-2 are also expressed by various human cancer cell lines, including prostate cancer cells, 14 VEGF may also act in an autocrine fashion to modulate tumor growth. Culture of the prostate cancer cell line LNCaP in the presence of recombinant human VEGF165 resulted in a 50% increase in [3H]thymidine uptake by these cells and recruitment of quiescent cells into the cell cycle. 23 Moreover, in humans, increased expression of VEGF in malignant cells during the transition from high-grade prostatic intraepithelial neoplasia to invasive carcinoma leads to an epithelial mesenchymal transition (EMT) through an autocrine loop that would promote tumor cell invasion and motility. 17 The author (C.P.) and others have recently described a redistribution of β-catenin in addition to an increased expression of mesenchymal markers in prostate cancer cells, 27,42 suggesting that EMT occurs during the progression of canine prostatic carcinoma. Therefore, the therapeutic blockade of VEGF could be a strategy for impairing tumor angiogenesis and early invasiveness of malignant cells.
The results of this study demonstrate that FGF-2 could be of importance in the induction of hyperplastic lesions and in the progression of prostatic carcinoma in dogs, while it is likely not involved in normal prostatic homeostasis. In humans, FGF-2 expression in normal and pathological prostates is controversial, with positive reactions demonstrated by some investigators in normal and hyperplastic prostates 1,10,34,36,40,44 and in rat and human prostate cancer cell lines, 37,46 and failed evidence of staining in prostatic neoplastic cells in other studies. 16,49 The different immunoreactivity of prostate cancer samples across different studies can be related to the neoplastic stage. Two research groups, using tissues from more advanced cancers, have detected the expression of FGF-2 in human prostate cancer epithelial cells in the majority of cases. 5,11 High levels of expression of FGF-2 are present in PC3 and Du145 prostate cancer cells, 5,37 and both of these cell lines are derived from metastatic prostate cancer. Thus, it may be suggested that FGF-2 is initially expressed as a paracrine factor by stromal cells in localized prostatic carcinoma but switches to an autocrine expression during tumor progression. This hypothesis is supported in the present study considering that prostatic carcinoma is a biologically aggressive neoplasm in dogs, and affected animals frequently have advanced cancer at the time of diagnosis. 2
PECAM-1 expression by cancer cells and lack of expression in normal or hyperplastic epithelia implies that this protein is acquired by epithelial cells during neoplastic transformation. The presence of PECAM-1 in malignant epithelial cells could be explained in the context of vasculogenic mimicry (VM). 21,30 Based on this theory, specific tumor elements form blood-filled channels and become intensely PECAM-1 positive at least in the experimental setting of uveal melanoma. 30 Vasculogenic mimicry has been seen in several human malignant tumors, such as breast cancer, liver cancer, glioma, ovarian cancer, melanoma, and prostate cancer, 55 and in 38% of canine mammary tumors. 10 Although still a topic of discussion in prostate cancer, this model is challenging. Channels formed by tumor cells were detected in human prostatic carcinoma (Gleason grade 5). 47 The precise mechanism triggering the upregulation of PECAM-1 in neoplastic cells is not known. PECAM-1 expression in prostate cancer might be the result of gene amplification, as suggested for human breast cancer, in which 17q23 (the region of the chromosome where CD31 gene is localized) is frequently amplified. 32 Not surprisingly, 17q23-24 amplification has been demonstrated in human prostatic carcinoma. 24
In humans, Tie-2 expression has been reported in different types of tumors, such as leukemia, prostate cancer, giant cell tumor of tendon sheath, and gastric, colorectal, and endometrial carcinoma, 33,35,38,39,48,49,54 although the precise function of Tie-2 expressed in tumor cells remains unclear. In 69% of human gastric carcinomas, the expression of Tie-2 was inversely correlated with the degree of histological differentiation, degree of stromal reaction, and tumor growth pattern. 39 However, the expression of Tie-2, as well as other angiogenic factors, was not correlated with tumor grade, degree of differentiation, and histological subtypes in the present study.
This study suggests the production and overexpression of angiogenic factors by neoplastic prostatic epithelial cells may increase angiogenesis and potentially tumor proliferation. However, many questions remain to be answered, including the mechanism by which expression of these factors is regulated in prostate cancer cells. Their expression can be modulated by transcriptional, posttranscriptional, and translational mechanisms, but little is known about what underlies the expression of them as autocrine growth factors in prostate cancer cells. Although many of these factors are pleiotropic and probably serve functions unrelated to angiogenesis, these cells appear to possess a redundant, overlapping capacity for inducing neovascularization. This finding of multiple angiogenic factors complicates the issue of how one could therapeutically control the angiogenic process in prostate carcinoma. Therefore, further studies on the identification of regulatory pathways are required to develop antiangiogenic strategies.
Footnotes
Acknowledgement
I thank Dr Rachel Allavena of the School of Veterinary Science (The University of Queensland) for invaluable assistance in reviewing and editing the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
