Abstract
In 2005, a prion disease identified in a goat from France was reported to be consistent with disease from the bovine spongiform encephalopathy (BSE) agent. Subsequent retrospective examination of UK goat scrapie cases led to the identification of one potentially similar, but as yet unconfirmed, case from Scotland. These findings strengthened concerns that small ruminant populations exposed to the BSE agent have become infected. The lack of data relating specifically to scrapie in goats has been contributory to past assumptions that, in general, sheep and goats respond similarly to prion infections. In this study, brain material from 22 archived caprine scrapie cases from the UK was reviewed by histopathology and by immunohistochemical examination for accumulations of disease-specific prion protein (PrPSc) to provide additional data on the lesions of caprine scrapie and to identify any BSE-like features. The vacuolar change observed in the goats was characteristic of transmissible spongiform encephalopathies in general. PrPSc immunohistochemical morphologic forms described in scrapie and experimental BSE infections of sheep were demonstrable in the goats, but these were generally more extensive and variable in PrPSc accumulation. None of the cases examined showed a PrPSc immunohistochemical pattern indicative of BSE.
Scrapie is a chronic, progressive, and invariably fatal neurodegenerative disease that naturally affects both sheep and goats. It is classified as a transmissible spongiform encephalopathy (TSE) or prion disease, characterized primarily by the accumulation of an abnormal form of a host membrane glycoprotein (prion protein [PrP]), PrPSc, and, morphologically, by neuronal and neuropil vacuolation. 22, 30 Other examples within this group of disorders include bovine spongiform encephalopathy (BSE) in domestic cattle, 27 Creutzfeldt-Jacob disease (CJD) in man, 16, 21 transmissible mink encephalopathy in ranch-reared mink (Mustela vison), 15 and chronic wasting disease in elk (Cervus elaphus nelsoni), mule-deer (Odocoileus hemionus hemionus), white-tailed deer (Odocoileus virginianus), and moose (Alces alces). 20, 28, 29
Experimental evidence of the susceptibility of sheep and goats to BSE 9, 18, 23 and the demonstration that the prion agent responsible for a variant form of CJD in man is indistinguishable from that which causes BSE 3 created an urgency to obtain a more complete understanding of scrapie, the natural form of prion disease in domestic livestock. Recently, a prion infection in a goat in France was shown, by a range of discriminatory techniques, to have resulted from the BSE agent, 8 and a potentially similar case was identified in archived material in Scotland (http://www.defra.gov.uk/news/2005/050208a.htm; http://www.seac.gov.uk/papers/paper86-4.pdf). 17 These cases raise the possibility that past diagnostic criteria may not distinguish natural prion infections in small ruminant populations or identify cases of BSE in sheep and goats. The known exposure of goats to mammalian meat and bone meal 23 and their proven susceptibility to oral as well as intracerebral challenge with BSE 9, 10 also argues strongly for specific studies directed toward prion disease in goat populations.
The caprine population within the UK occupies a unique niche among agricultural livestock. It is both relatively small in number (http://statistics.defra.gov.uk/esg/default.asp) and of lower economic importance compared with the sheep sector. Goats are also subject to different husbandry practices than sheep, depending upon the purpose of the herd, although a considerable number are kept as “pets.” As a result of this varied industry makeup, precise population numbers are uncertain, and current scrapie disease status statistics can be hard to interpret (http://www.defra.gov.uk/animalh/bse/othertses/scrapie/index.html). At the time of this investigation, there had been no confirmed cases of caprine scrapie in the UK, through either the passive (i.e., clinical suspects) or active surveillance systems (i.e., healthy, fallen, or nonambulatory animals) since 2002, so archived material was sought to conduct a retrospective study of cases broadly contemporary with the BSE epidemic.
Histologic and immunohistochemical (IHC) methods were applied to the brains of animals affected by most of the natural TSEs to define and classify variations of the lesions within a given host species, and such studies were important in differentiating disease phenotypes. Morphologic studies concentrated particularly on the distribution patterns of the vacuolar changes. In some instances, this was extended to vacuolar lesion profiling, a semiquantitative approach to describe both the distribution and relative severity of vacuolar changes. Lesion profiling was first developed for the study of the biology of experimental murine scrapie. 11 It was subsequently adapted for the characterization of naturally occurring TSEs, initially for BSE, 24 and more recently for sheep, 1, 4, 19 and goat 4 scrapie. These previous applications of lesion profiling were used to address various hypotheses regarding similarities or common phenotypic features within the case sets examined. The same approach was used in this study, within the limitations of availability of cases and the range of brain regions archived.
IHC techniques also were recently developed to examine the distribution and severity of PrPSc accumulations in scrapie-affected sheep 7, 12– 14, 18 as an aid to distinction between different phenotypes and, more specifically, as a TSE discriminatory test to differentiate between scrapie- and BSE-infected animals (http://www.seac.gov.uk/papers/paper86-4.pdf). 17, 18 These methods were applied to the caprine cases in this study to establish morphologic features of PrPSc accumulation, PrPSc distribution, and severity patterns, and to identify the possible presence of disease because of infection with the BSE agent.
Materials and Methods
Case selection
A retrospective search of the Veterinary Laboratories Agency (VLA) and UK Veterinary Schools' Pathology Departments' archives identified a total of 28 histopathologically confirmed goat scrapie cases, from which hematoxylin and eosin (HE) stained sections and paraffin-wax-embedded tissue blocks of brain were retrieved from 22 (Table 1). These were from scrapie cases with dates of death in the period 1976–2002, with the majority in the 1980s and 1990s. Published Italian goat scrapie case data 4 was used for comparison of goat vacuolation profiles.
Summary of UK caprine scrapie cases and brain-level availability.∗
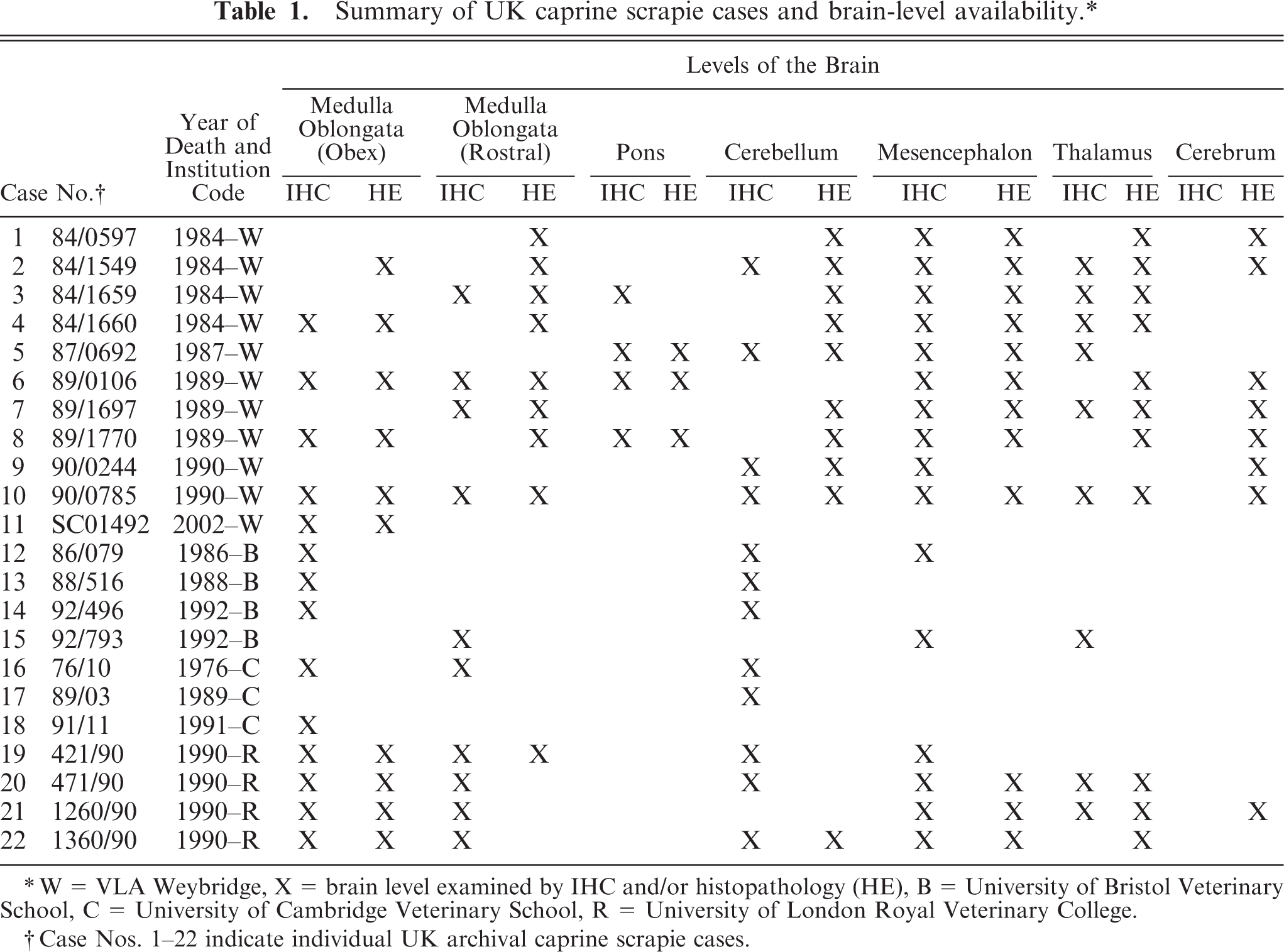
W = VLA Weybridge, X = brain level examined by IHC and/or histopathology (HE), B = University of Bristol Veterinary School, C = University of Cambridge Veterinary School, R = University of London Royal Veterinary College.
Case Nos. 1–22 indicate individual UK archival caprine scrapie cases.
Neuroanatomical areas examined
The levels of the brain selected were based on the availability and areas of brain in which scrapie lesions are most frequently encountered. Seven coronal brain blocks were selected according to availability: medulla oblongata at the level of the obex, rostral medulla oblongata at the level of the cerebellar peduncles, pons, cerebellum, mesencephalon, thalamus, and cerebrum at the level of frontal cortex and corpus striatum (Table 1).
Fixation, tissue processing, histology, and IHC
Because archived material was used, the tissues were already formalin fixed, trimmed, processed, and embedded in paraffin wax in a variety of different laboratories. The different gross trimming procedures used between laboratories limited the accuracy with which specific levels of brain regions could be selected for each case.
For histologic examination, sections were stained with HE. 2 For IHC preparations, a standard method was used (http://www.defra.gov.uk/corporate/vla/science/science-tse-rl-intro.htm). In brief, 4-µm-thick tissue sections were floated onto charged microscope slides and were dewaxed in 2 changes of xylene and 2 changes of absolute alcohol before epitope demasking (30 minutes in undiluted formic acid, washed in running tap water, then autoclaved at 121°C for 30 minutes in citrate buffer [pH 6.1]) to maximize antigen retrieval. Sections were immunolabeled for PrPSc by using 1 of 3 PrPSc monoclonal antibodies (mAb) (R145 [1/500] [VLA-Weybridge, Addlestone, UK], P4 [1/8000] [R-Biopharm, Darmstadt, Germany], or 2G11 [1/400] [Institute Pourquier, France]). These mAbs are directed against bovine PrP sequence residues 221–233 (R145), and ovine PrP sequence residues 89–104 (P4) and 146-R154-R171-182 (2G11). All incubations were performed at room temperature and all washes were with Tris-buffered saline solution Tween.
Vector Elite ABC (Vector Laboratories, Peterborough, UK) was used to amplify the immunodetection, by following the manufacturer's instructions. Diaminobenzidine (DAB:Sigma) was reconstituted with Mcllvanes citrate buffer (0.2 M disodium hydrogen orthophosphate, 0.1 M citric acid, 0.05% Tween-20 at pH 6.4) and applied to sections for 10 minutes at room temperature. Sections were counterstained with Mayer's hematoxylin, cleared in 3 changes of absolute alcohol, and 3 changes of xylene before mounting in di-n-butylphthalate-polystyrene-xylene (DPX:Sigma).
Vacuolation profiling
HE-stained sections were examined with light microscopy to assess the severity and distribution of vacuolation. Where detected, the intensity of neuropil vacuolation was scored by using a scale of 0 (no lesions) to 4 (numerous vacuoles, often coalescing). 4, 19, 24 Mean vacuolation scores were calculated for each neuroanatomic area and plotted according to an assigned central nervous system (CNS) level code to provide a profile of vacuolation severity.
IHC PrPSc profiling
By using sections immunolabeled with R145 and 2G11, morphologic forms of PrPSc deposition were identified based upon standard descriptions defined by the European Union Community Reference Laboratory (http://www.defra.gov.uk/corporate/vla/science/science-tse-rl-intro.htm), from published observations of ovine scrapie. 12, 13, 22 The intensity of each morphologic PrPSc deposit was described (mild, moderate, severe) in each available neuroanatomical area. Photographic standards were produced specifically for caprine material after a preliminary review of the case series.
PrPSc IHC discrimination of BSE and scrapie
A published IHC technique capable of discriminating between experimental BSE and scrapie in sheep was applied to each case (http://www.seac.gov.uk/papers/paper86-4.pdf). 17, 18 This approach proposes that BSE and scrapie differ in the amino-acid residues of the N-terminus of PrPSc that are processed by host lysosomal enzymes and that this phenomenon can be recognized by the relative distribution of immunolabeling by using antibodies directed toward the C-terminal and the N-terminal of the PrPSc protein. In experimental infection of sheep and goats with BSE, a marked reduction in the intracellular PrPSc signal occurs with the N-terminal antibodies, such as P4 (http://www.seac.gov.uk/papers/paper86-4.pdf). 17, 18 This method was applied to the immunolabeled medulla sections (n = 14), because this is the region required for small ruminant statutory TSE diagnosis (http://www.defra.gov.uk/corporate/vla/science/science-tse-rl-intro.htm).
Intraneuronal and intraglial PrPSc morphologic immunolabeling was scored by using a scale from 0 (no PrPSc intracellular forms) to 3 (intense and striking intracellular PrPSc forms present), by using both R145 (C-terminal) and P4 (N-terminal) antibodies. Scores with each antibody were averaged to the nearest half score for each brain level, and the relative score was compared within each case. All HE and IHC sections were examined by a single reader (BD), to maximize the consistency of observations by avoiding interoperative variation. 24
Results
Despite inevitable variation in tissue preservation and paraffin-block storage times spanning more than 30 years, histologic and IHC preparations provided interpretable observations of vacuolar changes and PrPSc immunolabeling in the majority of cases.
Vacuolation profiling
The scores for the 24 neuroanatomic areas examined are given in Table 2 and displayed graphically in Fig. 1. The overall intensity of vacuolar changes was generally bilaterally symmetrical.
Summary table of UK vacuolation profiling data by neuroanatomical location.
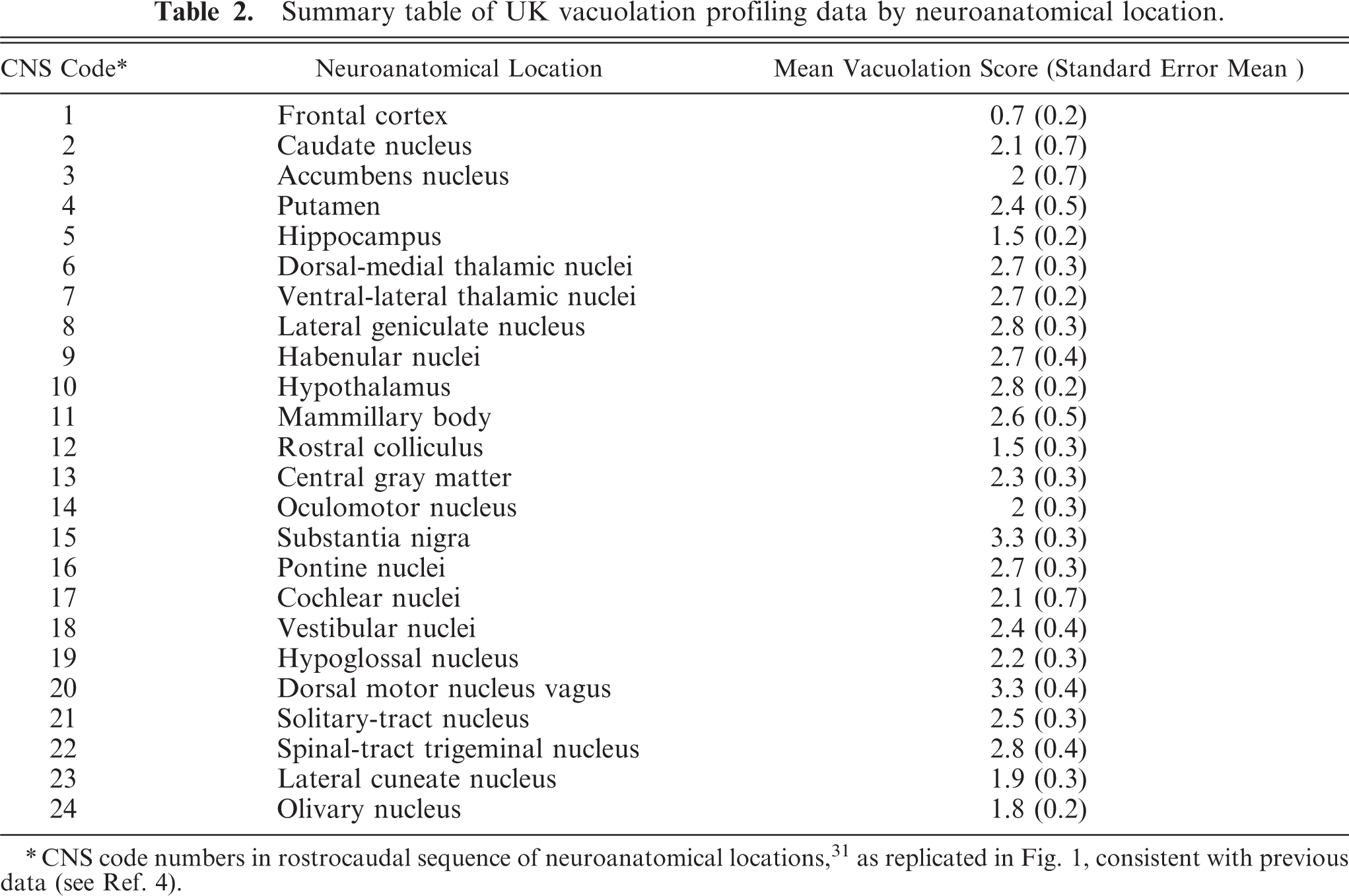
CNS code numbers in rostrocaudal sequence of neuroanatomical locations,31 as replicated in Fig. 1, consistent with previous data (see Ref. 4).
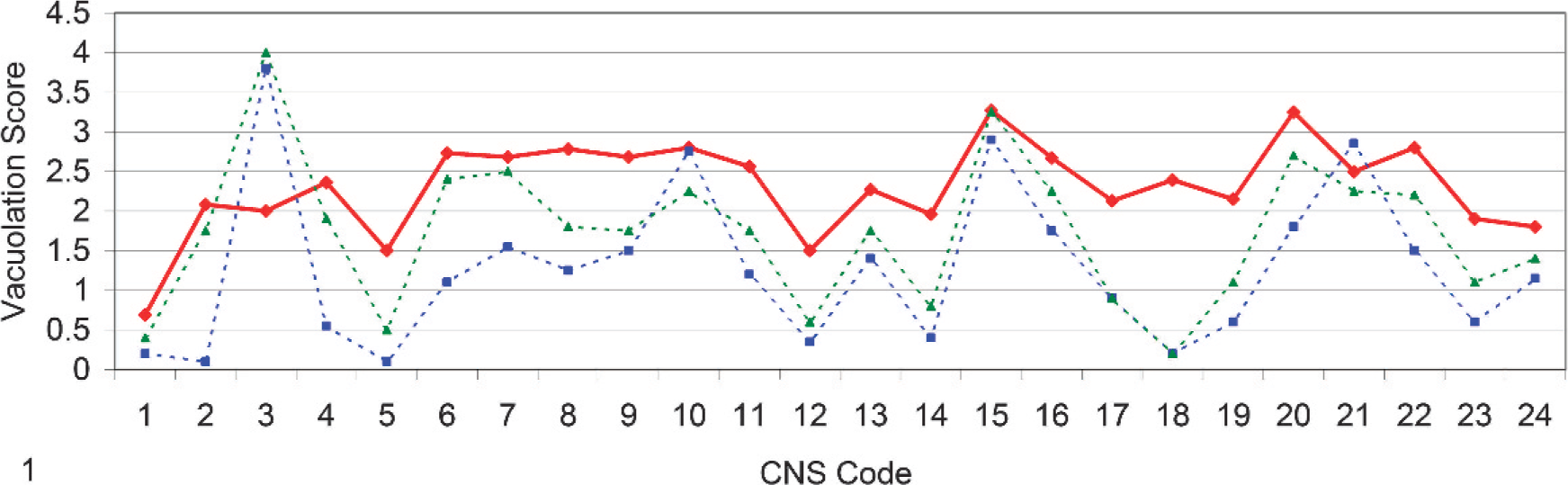
Line-graph representation of vacuolar profiles of UK natural scrapie (red), Italian natural scrapie (blue), and Italian iatrogenic scrapie (green) cases in goats. Mean vacuolar score plotted by neuroanatomical area code (Table 2) in rostrocaudal representation for consistency with previous data. 4 .
IHC PrPSc profiling
Ten morphologic forms of PrPSc deposition were identified: intraneuronal, intraglial, stellate, granular, fine punctate, perineuronal, perivascular, linear, coalescent, and plaque-like. These morphologic forms were observed irrespective of the antibody used and were consistent with those previously described for sheep scrapie. 12– 14, 24 The relative intensity and/or amount, when assessed subjectively, varied with antibody.
There was substantial PrPSc immunolabeling throughout the brain, and individual neuroanatomical nuclei frequently contained variable and numerous different PrPSc morphologic forms. General patterns of immunolabeling were apparent at each of the brain levels examined and are summarized as follows.
Medulla oblongata (obex)
The amount and intensity of PrPSc labeling at this level was variable but generally was of a high magnitude. Intracellular PrPSc signals appeared to have specific and reasonably conserved nuclear tropisms. Intraneuronal PrPSc was generally of low intensity within the dorsal motor nucleus of the vagus nerve (DMNV) (Fig. 2) and the nucleus of the hypoglossal nerve but was frequently intense and widespread in the lateral cuneate (Fig. 3) and olivary nuclei, while being typically variable in the nucleus of the spinal tract of the trigeminal nerve. Intraglial PrPSc was characteristically marked in the nucleus of the spinal tract of the trigeminal nerve (Fig. 4) and frequently also in the lateral cuneate nucleus but was often more variable in the olivary nuclei and of comparatively low intensity in the DMNV and nucleus of the hypoglossal nerve. Neuropil PrPSc deposits were variable but consistently associated with the DMNV and nucleus of the hypoglossal nerve. The DMNV typically showed a moderate-to-intense granular and sometimes fine punctate signal (Fig. 2), with the nucleus of the hypoglossal nerve usually containing a low-to-moderate amount of granular labeling.
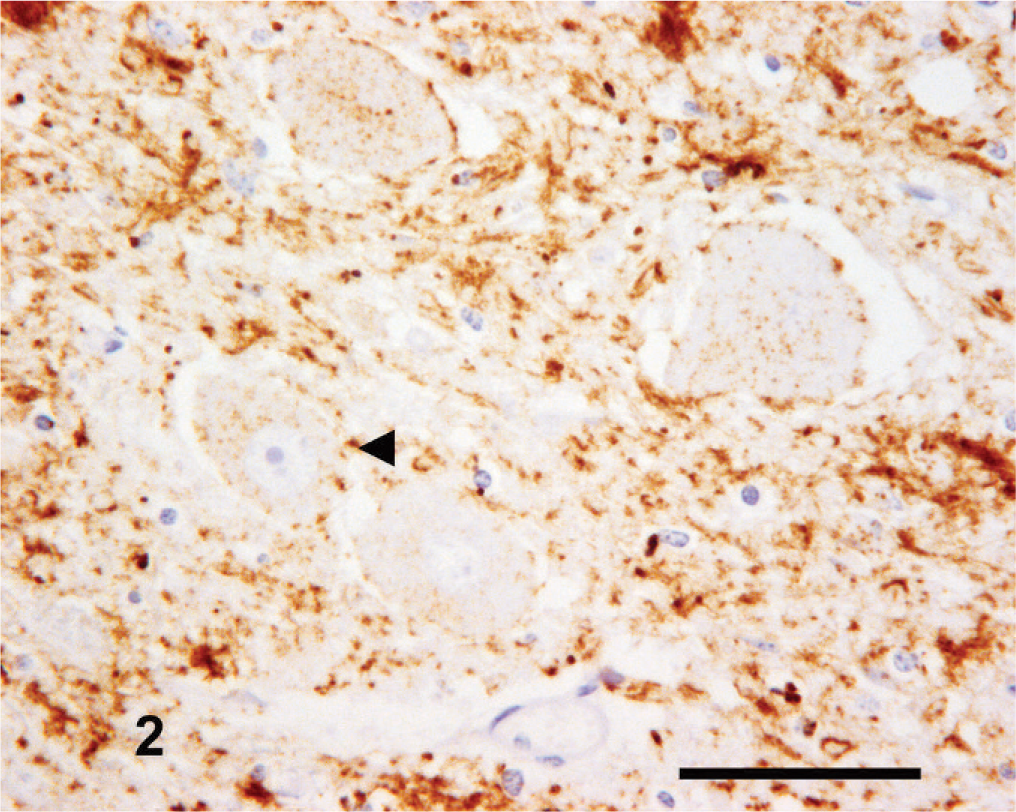
Medulla oblongata (obex); goat No. 4. Low-intensity intraneuronal PrPSc labeling (arrowhead) and moderate to high intensity neuropil-associated PrPSc labeling in the DMNV. IHC, monoclonal antibody R145. Mayer's hematoxylin counterstain. Bar = 50 µm.
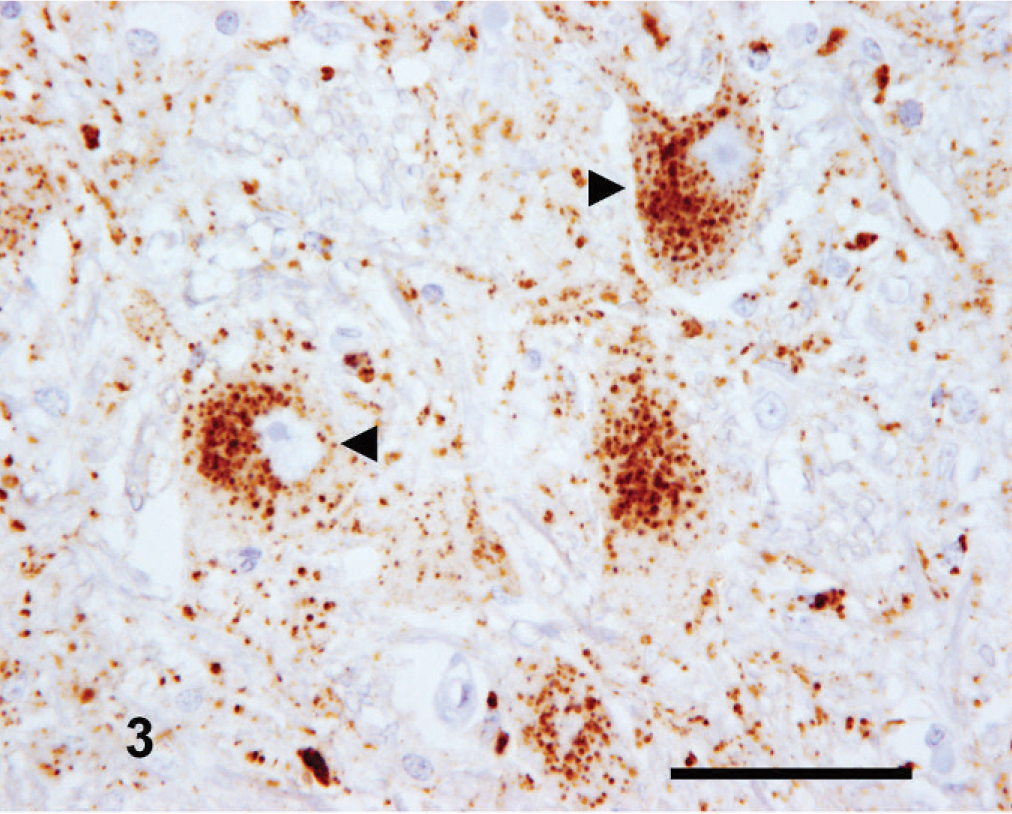
Medulla oblongata (obex); goat No. 16. Intense intraneuronal PrPSc immunostaining (arrowheads) within the lateral cuneate nucleus. IHC, monoclonal antibody R145. Mayer's hematoxylin counterstain. Bar = 50 µm.
Medulla oblongata (obex); goat No. 13. Intense intraglial PrPSc labeling (arrowheads) within the nucleus of the spinal tract of the trigeminal nerve. IHC, monoclonal antibody R145. Mayer's hematoxylin counterstain. Bar = 50 µm.
Medulla oblongata (rostral)
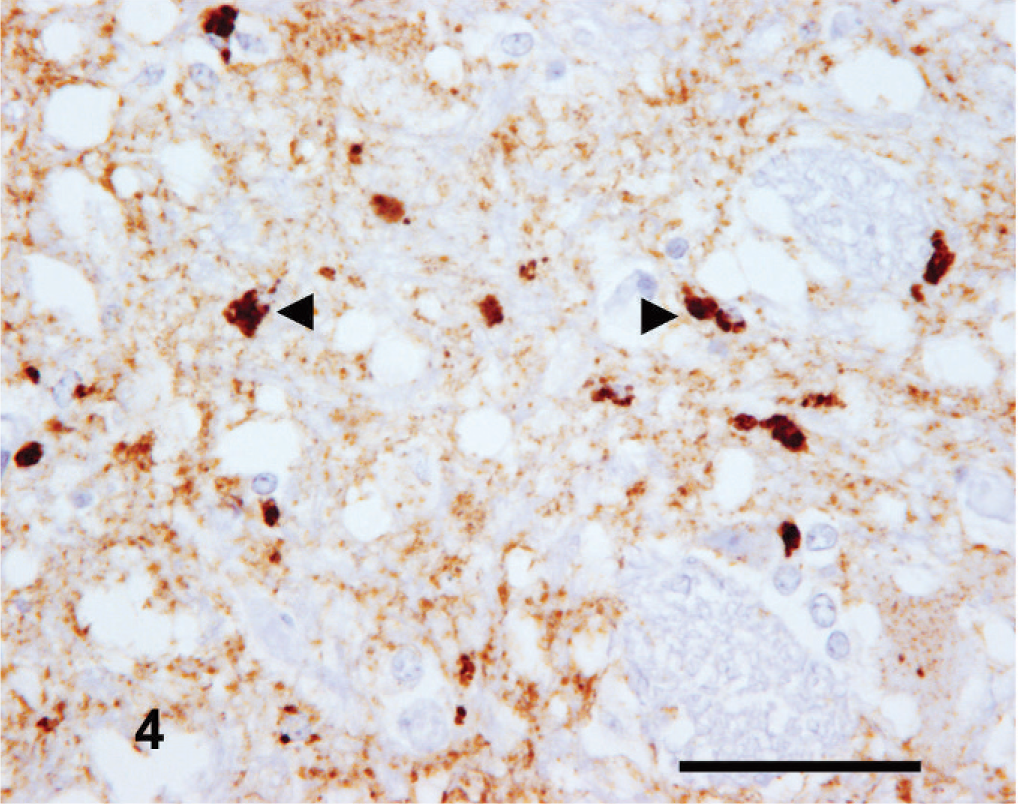
Intracellular PrPSc immunolabeling appeared to have neuroanatomical nuclear tropisms, as seen in the obex, with particularly striking intense intraglial PrPSc within the nucleus of the spinal tract of the trigeminal nerve and the nuclei of the trapezoid body. The intraneuronal signal, when present, was typically substantial in the vestibular nuclei and cochlear nucleus. A common finding throughout the cases, particularly at this level, was neuronophagia, with the formation of microglial nodules that featured intense cytoplasmic PrPSc labeling (Fig. 5).
Rostral medulla oblongata: goat No. 6. Microglial nodule with intense intraglial PrPSc immunolabeling (arrowheads). IHC, monoclonal antibody R145. Mayer's hematoxylin counterstain. Bar = 50 µm.
Pons
Consistent intracellular PrPSc immunolabeling of the pontine nuclei (medial, ventral, and lateral) was detected. Typically, the intraglial component was more variable than the intraneuronal labeling, and frequently the intracellular labeling was accompanied by a moderate intensity granular and/or fine punctate PrPSc neuropil signal.
Cerebellum
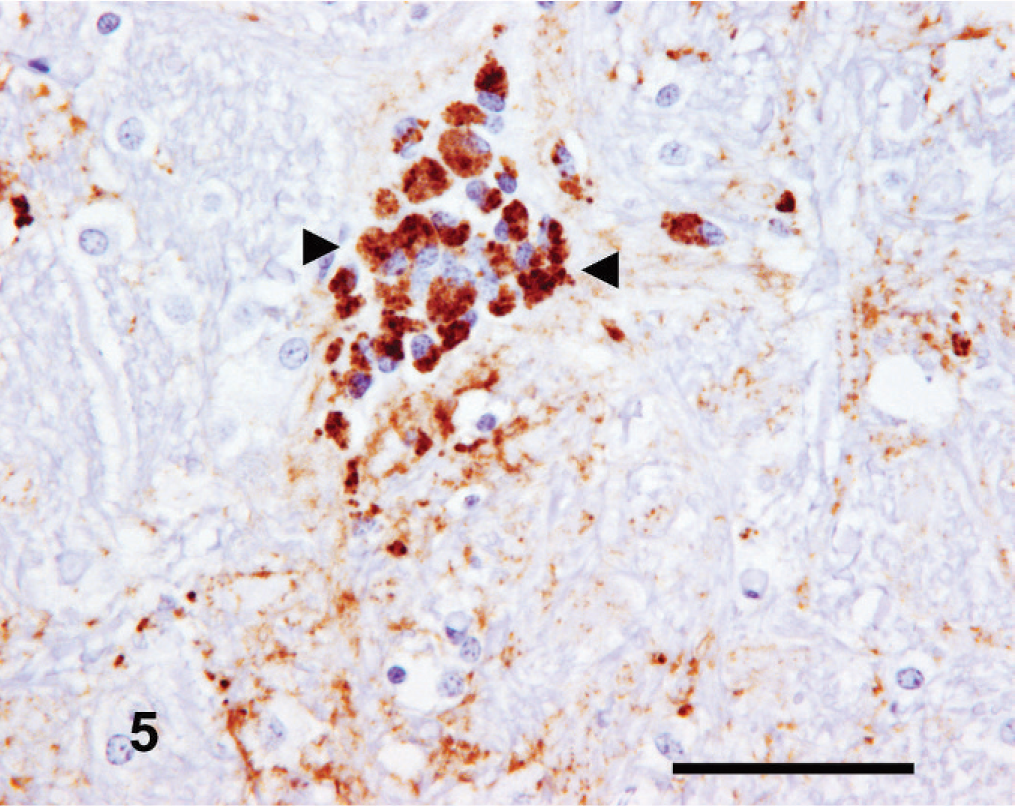
A consistent presentation of PrPSc labeling was observed in all available cerebellar sections. This comprised moderate intensity stellate and intraglial forms within the molecular stratum; moderate-to-intense intraglial, and, frequently, granular forms within the granular stratum; mild-to-moderate intensity intraglial, stellate, and often granular, perineuronal, and linear forms in the Purkinje cell zone (Fig. 6), and, finally, moderate-to-high intensity granular, mild-to-moderate stellate, intraneuronal, and intraglial labeling in the cerebellar white matter and cerebellar roof nuclei. Intraneuronal PrPSc labeling of the Purkinje cells was low grade.
Cerebellum; goat No. 5. Occasional focal intraglial PrPSc immunolabeling within the granular stratum (arrowhead) with widespread granular neuropil PrPsc immunostaining. IHC, monoclonal antibody R145. Mayer's hematoxylin counterstain. Bar = 100 µm.
Mesencephalon
This region commonly exhibited some of the greatest amounts and intensity of PrPSc immunolabeling. There was moderate-to-high intensity perineuronal labeling of the large neurons of the mesencephalic tract nucleus of the trigeminal nerve (Fig. 7). Variable intensity intraglial labeling was present in the rostral colliculus and nucleus of the oculomotor nerve, and there were stellate PrPSc deposits within the central gray matter (Fig. 8). Frequently, intense linear and fine punctate forms were found within the substantia nigra. Occasionally, coalescent and/or plaque-like forms were seen within the tectum or in regions ventral to the central gray matter but not in the oculomotor nerve nucleus.
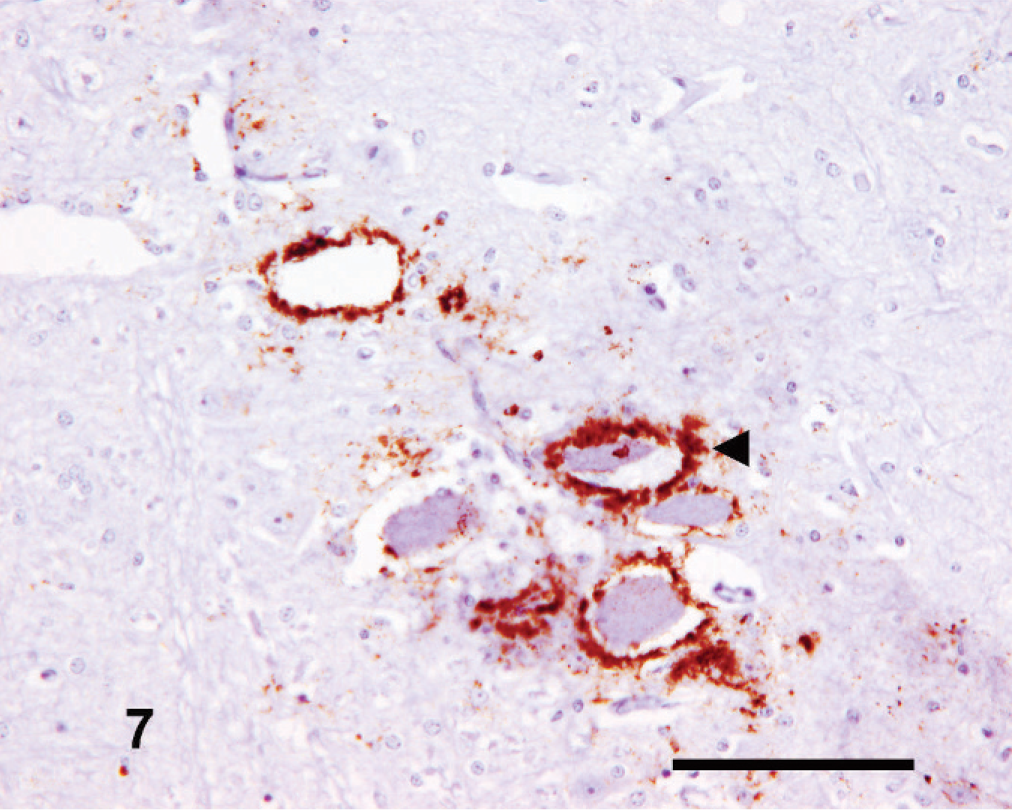
Mesencephalon; goat No. 3. Characteristic intense perineuronal PrPSc labeling (arrowhead) of the mesencephalic tract of the trigeminal nerve. IHC, monoclonal antibody R145. Mayer's hematoxylin counterstain. Bar = 50 µm.
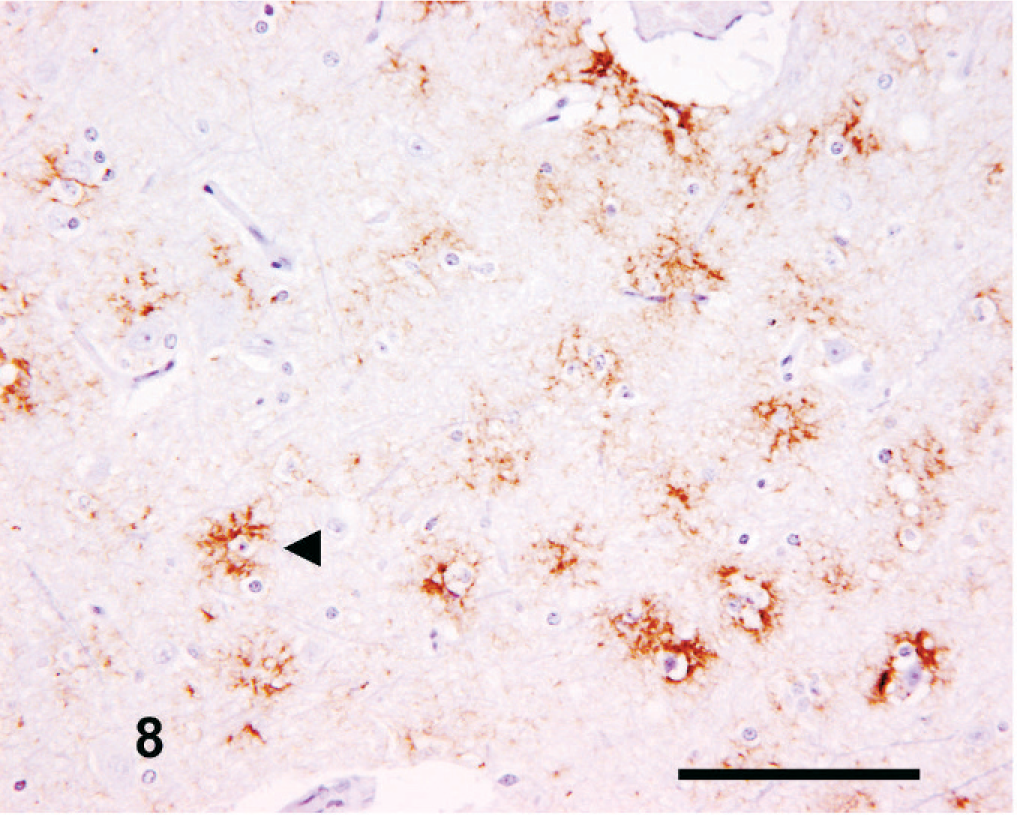
Mesencephalon; goat No. 6. Widespread stellate PrPSc immunolabeling in the central gray matter (arrowhead). IHC, monoclonal antibody R145. Mayer's hematoxylin counterstain. Bar = 50 µm.
Thalamus
The intensity and amount of PrPSc labeling was extremely variable. Common features included moderate-to-high–intensity focal perivascular labeling in the central gray matter, moderate intracellular immunolabeling distributed focally within the dorsomedial and ventrolateral thalamic nuclei, linear PrPSc immunolabeling in the substantia nigra, and focal coalescent and/or plaque-like forms throughout the thalamus. Fine punctate and perineuronal PrPSc forms were consistently infrequent or absent. An interesting observation at this level was the apparent differential targeting of the habenular nuclei, in which the medial (ventrally placed) habenular nucleus (Fig. 9) typically had greater neuropil-associated PrPSc immunolabeling than the lateral (dorsally placed) habenular nucleus (Fig. 10). Both nuclei also usually showed a moderate to intense intraneuronal PrPSc signal, with a more variable intraglial signal.
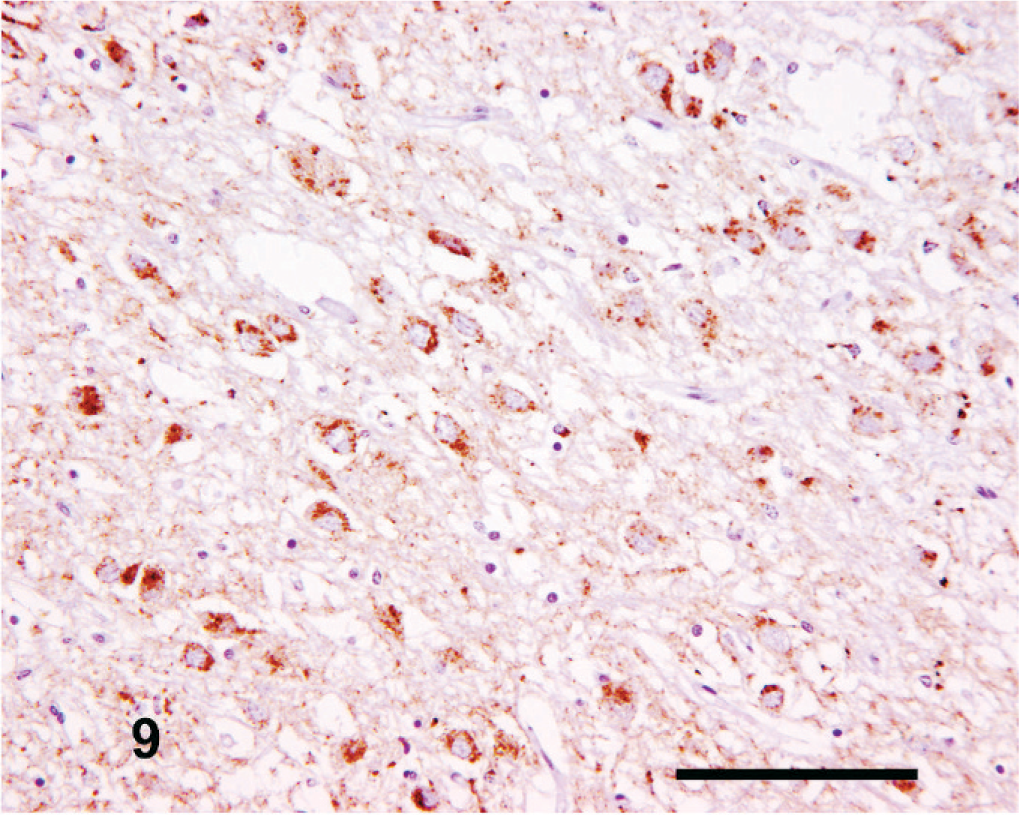
Thalamus; goat No. 4. Marked neuropil-associated PrPSc immunolabeling of the medial habenular nucleus. IHC, monoclonal antibody R145. Mayer's hematoxylin counterstain. Bar = 100 µm.
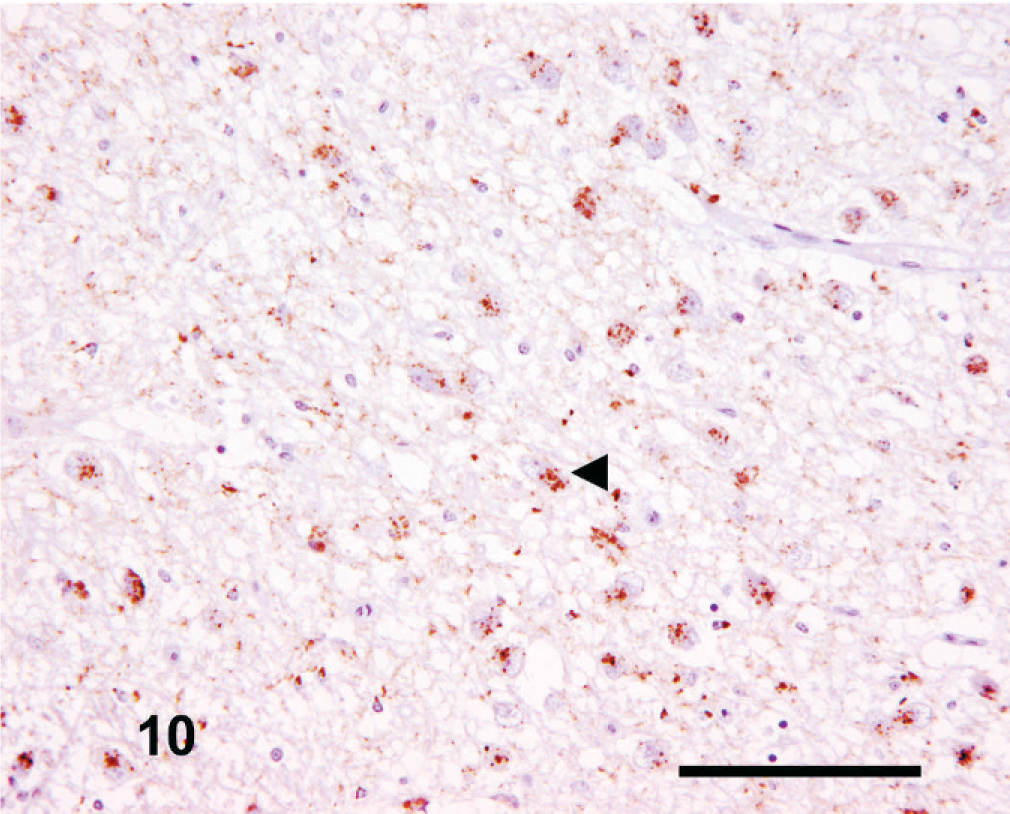
Thalamus; goat No. 4. Relatively sparse neuropil-associated PrPSc immunolabeling of the lateral habenular nucleus. Note the consistent moderate-to-intense intraneuronal PrPSc labeling (arrowheads), common to both habenular nuclei. IHC, monoclonal antibody R145. Mayer's hematoxylin counterstain. Bar = 100 µm.
Essentially, 3 general IHC PrPSc topographical distribution patterns could be defined at any given brain region but were not necessarily mutually exclusive within an individual case. Exceptions were found, and no clear distinction between cases on the basis of the patterns was possible. The patterns were most apparent in the mesencephalon and medulla oblongata (obex) regions.
The first comprised moderate-to-high intensity intracellular PrPSc immunolabeling of neuroanatomical nuclei throughout the brain areas examined. Areas dominated by intracellular PrPSc were generally associated with high-intensity aggregated PrPSc forms (e.g., coalescent, perineuronal, perivascular, and linear forms), and a commonly variable but weak neuropil-associated PrPSc signal (e.g., granular and/or fine punctate). Occasionally widespread, but not dominant, stellate PrPSc forms were also identifiable. The second was a pattern of moderate-to-high intensity, widespread stellate PrPSc forms, with a generally high neuropil-associated PrPSc signal. This pattern usually had a weak intracellular PrPSc signal, often with few or limited aggregated PrPSc forms identifiable. The third identifiable pattern was of moderate-to-high intensity intracellular PrPSc immunolabeling, with moderate-to-high intensity widespread neuropil-associated and stellate PrPSc signals, frequently accompanied by substantial aggregated immunolabeling. This group appeared a mixture of the other 2 patterns.
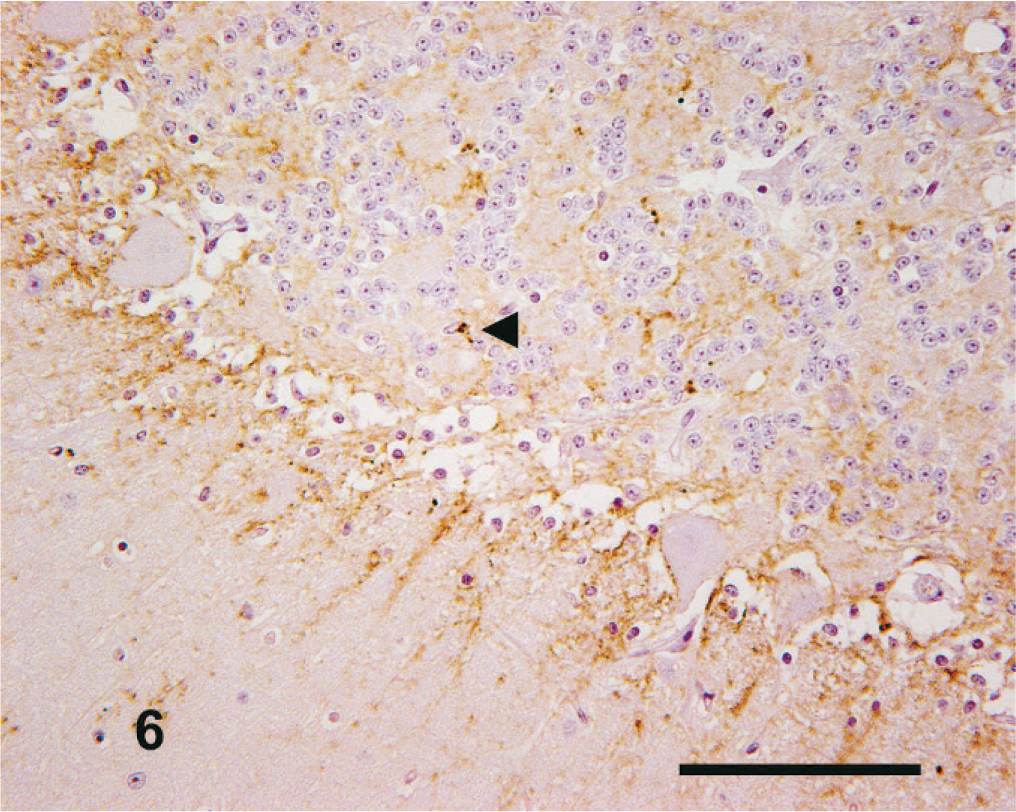
However, in one case (No. 11), the only one identified through active surveillance, a completely different pattern of immunolabeling was seen. This case had almost no intracellular PrPSc present but intense neuropil immunolabeling, confined principally to the DMNV and nucleus of the hypoglossal nerve in the medulla oblongata (obex), which was the only brain region available from this case. There were numerous, intense aggregated PrPSc forms, mainly coalescent, distributed multifocally, with granular PrPSc dominant in the DMNV, nucleus of the hypoglossal nerve, lateral cuneate nucleus and the nucleus of the spinal tract of the trigeminal nerve (Fig. 11).
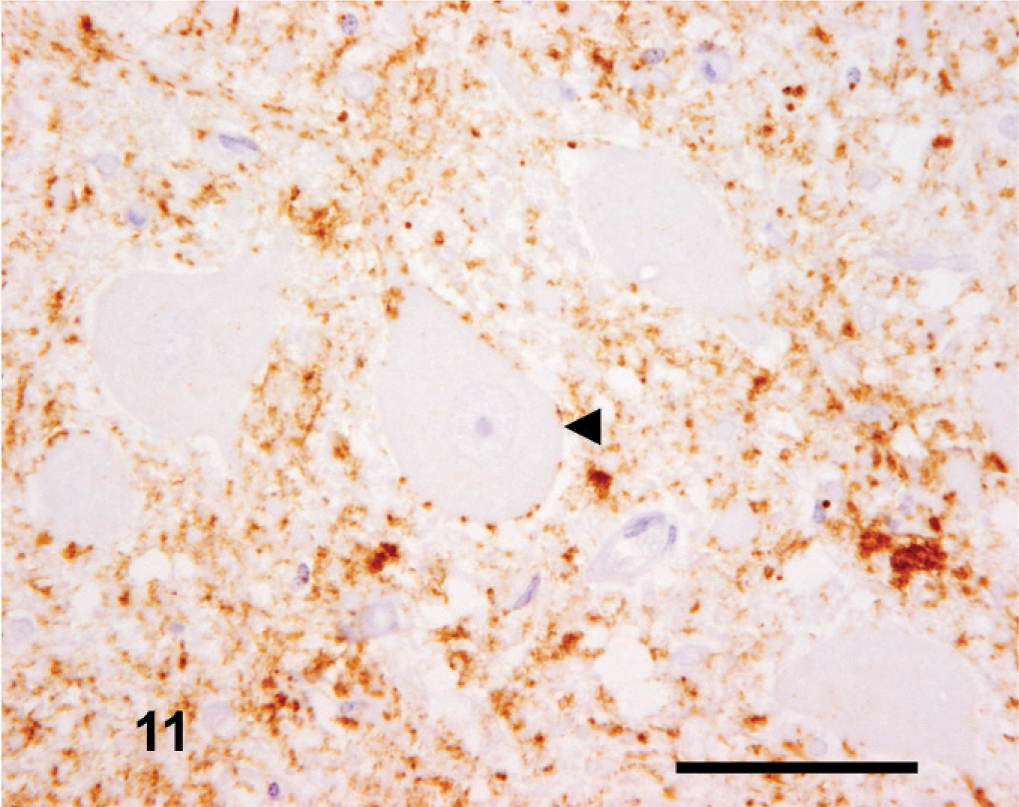
Medulla oblongata (obex); goat No. 11. Widespread neurophil-associated PrPSc immunolabeling within the DMNV but an absence of intraneuronal labeling (arrowhead). This is in contrast to the intense intraneuronal PrPSc labeling observed in the medulla oblongata (obex) of other cases in the series (see Figs. 2 to 4). IHC, monoclonal antibody R145. Mayer's hematoxylin counterstain. Bar = 50 µm.
PrPSc IHC discrimination of BSE and scrapie
In 13 of the 14 cases tested by this method, an intense intraneuronal (Fig. 12) and intraglial (Fig. 13) signal was detected within the medulla oblongata (obex) with both the C-terminal antibody R145 and the N-terminal antibody P4. This is a TSE discriminatory PrPSc IHC pattern highly suggestive of scrapie. None of the cases showed a significant reduction in intracellular PrPSc with the P4 antibody. Reduced P4 immunolabeling is suggestive of BSE infection. The application of this method to case No. 11 was not appropriate because of the virtual absence of intracellular PrPSc immunolabeling in the medulla oblongata (obex) of this case.
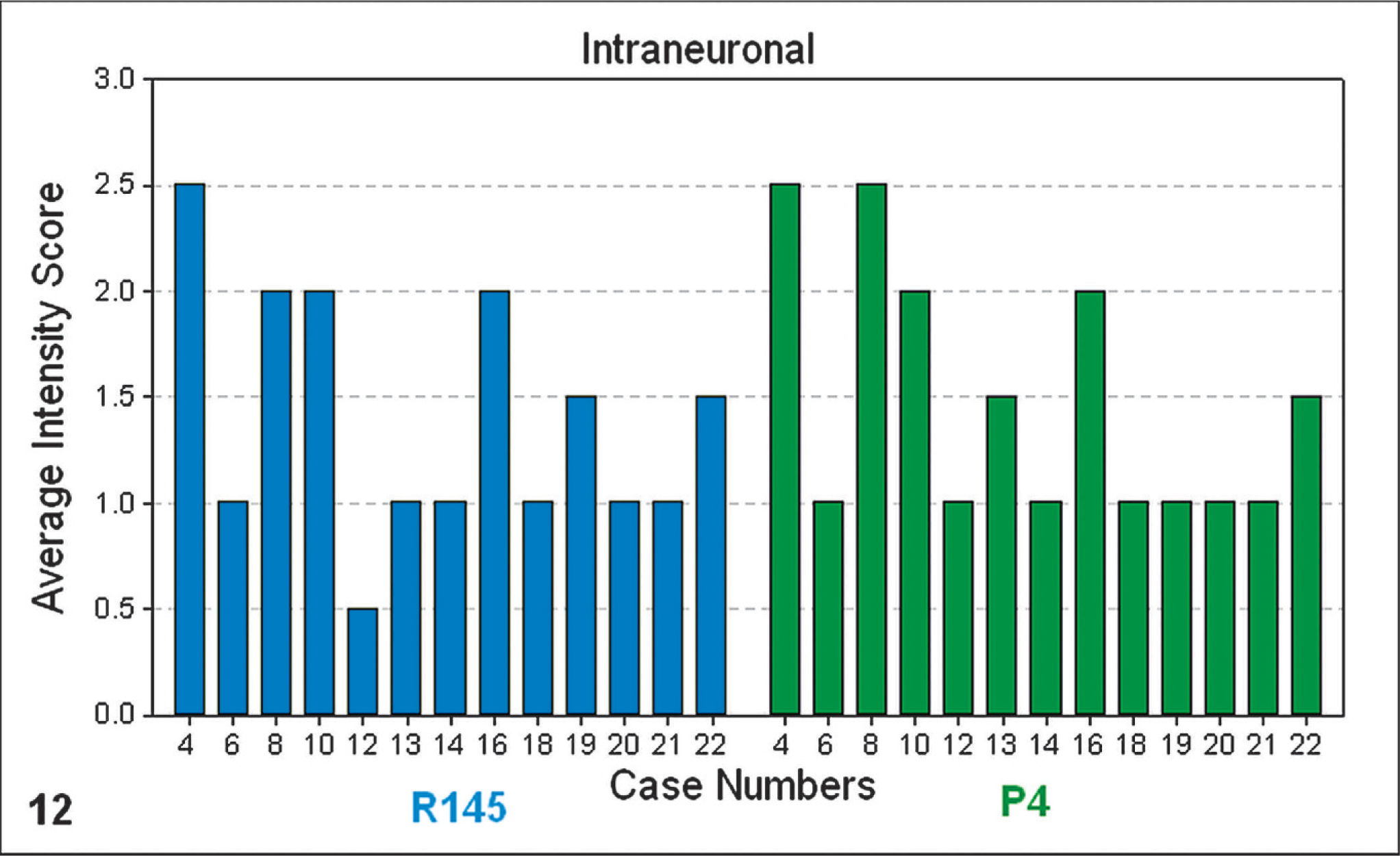
Bar-chart graphical representation of the average PrPSc intensity scores of intraneuronal PrPSc immunolabeling at the level of the medulla oblongata (obex), with PrPSc IHC monoclonal antibodies R145 (blue) or P4 (green). Note: numbered data labels indicate case number (Table 1).
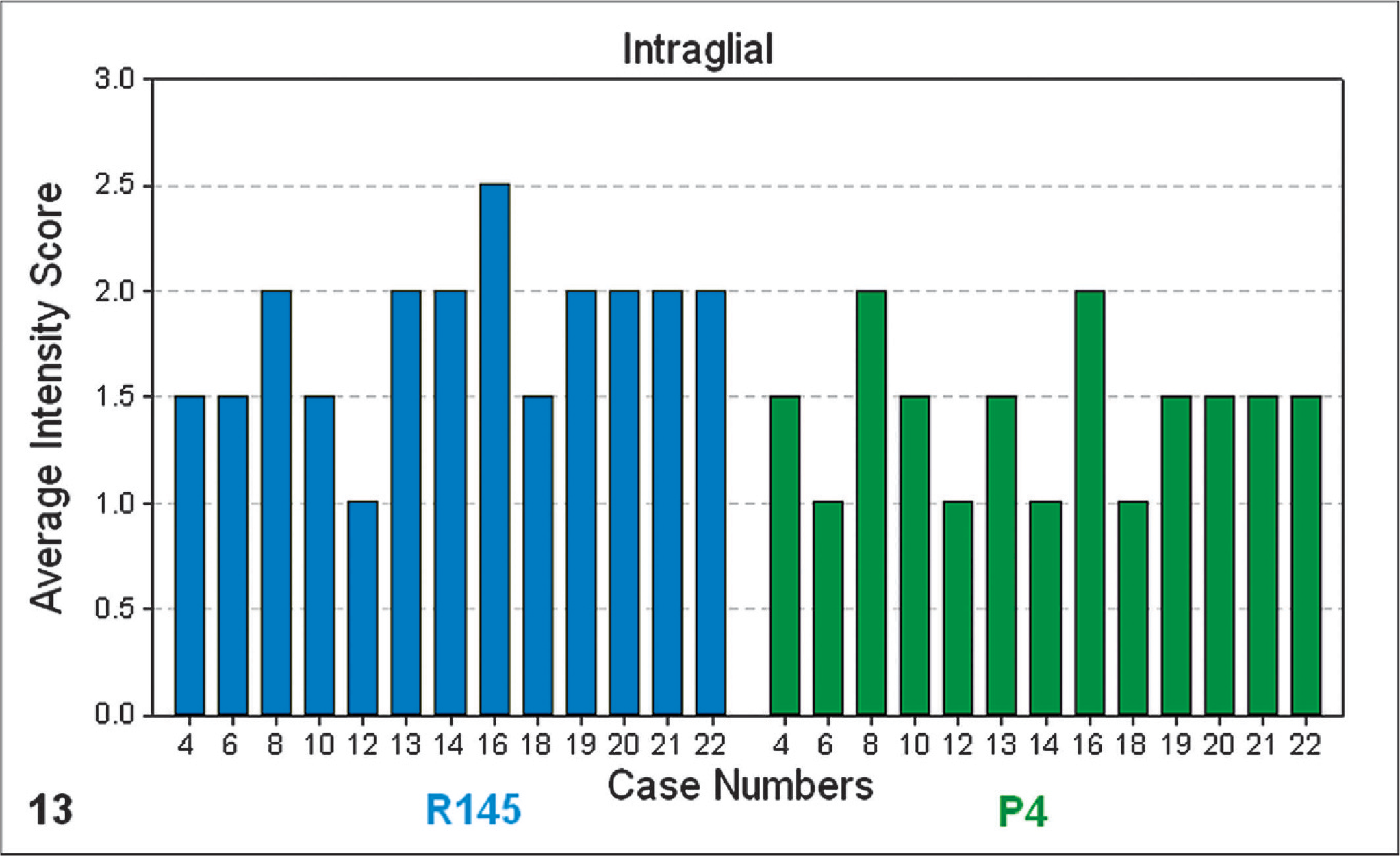
Bar-chart graphical representation of the average PrPSc intensity scores of intraglial PrPSc immunolabeling at the level of the medulla oblongata (obex), with PrPSc IHC monoclonal antibodies R145 (blue) or P4 (green). Note: numbered data labels indicate case number (Table 1).
Discussion
Contemporary ovine IHC and histopathologic techniques were successfully applied to confirm and further characterize an historical case set of caprine scrapie in the UK.
Vacuolar profiling
The vacuolar changes observed were characteristic of TSE infections, and the vacuolation profile was broadly similar to comparable published caprine data from studies conducted in Italy 4 (Fig. 1) and Greece. 25 The mean scores between the UK and Italian studies did not differ by more than approximately 1 score value at any given neuroanatomical location, with the exception of 2 neuroanatomical nuclei, the nucleus accumbens (CNS code 3) and the vestibular nuclei (CNS code 18) (Table 2, Fig. 1).
The UK data provided a mean for the nucleus accumbens that was approximately 2 scores lower than those in both Italian data sets, whereas, the UK data generated a mean score for the vestibular nuclei approximately 2.5 scores greater than those in both Italian profiles. However, in general, the overall degree of spongiosis was greater in the UK cases. This was confirmed by a parallel slide review of the UK archived caprine scrapie cases detailed and a sample of cases described in the Italian study (BD). 4
The significance of the profile differences between the studies is uncertain but clearly may reflect differences in the nature of the cases submitted. The UK cases were almost entirely submissions through the passive surveillance system (i.e., clinical suspects) and may possibly represent a more advanced disease state in which a more severe vacuolation profile would not be unexpected. In contrast, the Italian case series was a mixture of both passive and active surveillance submissions and could reasonably represent clinical and preclinical disease, with a resultant reduction in the mean overall severity of vacuolation.
The difference in the data sets for the nucleus accumbens (CNS code 3) could not be explained. Interestingly, published sheep data suggest that the nucleus accumbens is typically low-to-moderately vacuolated in sheep. 19 In sheep, it was possible to show that host PrP genotype may influence the severity of the vacuolation produced. 4, 19, 25 PrP genotype information was available for many of the goats in an Italian 4 and a Greek 25 study, but no such data were available for the UK cases.
Differences in infecting TSE strains within the different countries' goat populations should also be considered as a possible influencing factor on the vacuolation profile, but, with no information on strain(s), further interpretation is not possible. Also, some of the Italian data (Fig. 1) relates to scrapie that was transmitted to goats via a vaccine against Mycoplasma agalactiae 4 and, as such, does not represent only natural caprine scrapie infection. Indeed, it is likely that the vaccine was contaminated with potentially more than 1 circulating sheep scrapie strain, 32 and it is unknown what effect this may have on the vacuolation profile of infected goats. Such unknowns restrict the conclusions obtainable from these findings, but the observations do, nevertheless, serve as a reference from which it can be seen that broad similarities exist between vacuolation profiles in scrapie-affected goats from widely differing sources.
IHC PrPSc profiling
The morphologic forms of PrPSc immunolabeling observed, irrespective of the antibody used, were consistent with those previously described for sheep scrapie 12– 14, 22 but appear more extensive and variable in distribution in caprine scrapie, as was noted recently by other investigators. 17, 25 In sheep, strong correlations were demonstrated between host PrP genotype and the distribution of different morphologic forms of PrPSc within the medulla oblongata at the level of the obex. 26 No grouping of the UK goat cases was possible that might have suggested a clear influence of this or other host factors.
The overwhelming impression from the distribution mapping of PrPSc was that there was far greater variety of the defined PrPSc forms within a given nucleus when compared with sheep scrapie. This multiplicity and amount of PrPSc forms present made further comparison with sheep when using this method difficult. The differences between PrPSc labeling in sheep and goats with scrapie could imply differences in disease pathogenesis, and further species specific studies may be warranted.
Ovine PrPSc profile analyses were made based on the intensity of PrPSc morphologic forms at various brain levels. 12– 14 In the absence of crucial information on breed, PrP genotype, or strain of infecting agent, the present goat study could not include similar analyses. Initial attempts at arbitrary grouping (by sex, year of clinical onset, and geographical distribution), failed to identify individual or collective correlations, which, given the disparate nature of the case sample was not surprising. PrPSc deposition patterns observed in the mesencephalon of the cases examined, appeared particularly variable and complex, but the cerebellum in contrast did show a consistent single IHC PrPSc profile, as described and illustrated in Figs. 7 and 8.
Case No. 11 revealed an entirely different PrPSc morphologic form distribution, with a relative paucity of intracellular labeling. This was the first caprine scrapie case identified through the UK active surveillance system (i.e., abattoir-slaughtered animal sampled with no clinical suspicion of scrapie). 5, 6 It could, therefore, be speculated that the different PrPSc distribution related to a restricted, preclinical rather than end-stage disease pattern. However, the archive cases showed that intracellular PrPSc forms can occur as the sole or dominant form, as well as in conjunction with neuropil forms, so it is unlikely that this distribution truly indicates a preclinical stage of pathogenesis. It seems likely that the anomalous distribution pattern in this case may be linked to undetermined host or agent factors, but, in the absence of further brain tissue for this case, these observations cannot be further characterized. This study of scrapie in goats indicated a marked preponderance of intraglial PrPSc labeling, irrespective of the antibody used. However, the IHC PrPSc distribution profile of natural scrapie in goats appears highly variable and largely more intense and extensive than in sheep scrapie.
PrPSc IHC discrimination of BSE and scrapie
Several of the IHC PrPSc findings in this study, such as intense linear labeling of the substantia nigra in the mesencephalon and low-grade intraneuronal labeling of Purkinje cells in the cerebellum, were recently reported as being conspicuous in experimental BSE in both sheep and goats. 17 However, the discriminatory PrPSc IHC approach revealed that the archive cases tested in this study were not BSE-like and had a pattern indicative of scrapie.
A pilot study recently further substantiated that this discriminatory method (http://www.defra.gov.uk/corporate/vla/science/science-tse-rl-intro.htm; http://www.seac.gov.uk/papers/paper86-4.pdf) 17 can be used to differentiate between infections because of scrapie or BSE agents. The present study applied this technique to a greater number (n = 13) of historical caprine scrapie cases than previously examined, all of which were contemporaneous with the UK BSE epidemic. More variability in the detection of intraglial PrPSc accumulations with N-terminal antibodies were reported in experimental caprine BSE compared with experimental ovine BSE. 17 The period of preservation of the material examined in the present study was not generally observed to have an adverse effect on immunolabeling with the N-terminal P4 antibody compared with C-terminal antibody R145, in contrast with a recent report of less-predictable labeling with N-terminal antibodies in tissues retained in formalin for extended periods of time. 17
The application of the discriminatory method to case No. 11, which showed a different PrPSc morphologic form distribution than other cases, was not conducted because of the paucity of intracellular PrPSc immunolabeling with either R145 or P4 antibodies, because, if both antibodies fail to immunolabel the intracellular PrPSc component, then it is not possible to quantify any difference. Similar difficulties have arisen with the application of this method in a proportion of natural scrapie cases in sheep, (Y. I. Spencer and M. M. Simmons, unpublished). This is a limiting factor to the comprehensive use of this technique in field cases.
The findings of this study serve as a descriptive reference of caprine scrapie, which can now be concluded to be similar to published descriptions of ovine scrapie by using currently available methods for the pathologic characterization of TSEs in these species. Despite these similarities, the caprine cases consistently appeared to manifest more intense, diffuse, and variable neural lesions, which suggests that similarities between ovine and caprine scrapie should not be overemphasized.
Footnotes
Acknowledgements
We thank members of the Neuropathology and Histopathology Sections, VLA, Weybridge, especially Mr G. A. H. Wells, Dr P. Webb, Mrs A. Long, Mrs M. Denyer, and Miss S. J. Moore. We also thank the pathologists of the UK's veterinary schools, in particular, Dr G. Pearson of University of Bristol School of Veterinary Science, Division of Pathology Infection and Immunity, and Dr A. Palmer of University of Cambridge, Department of Veterinary Medicine for provision of material. This project benefited from constructive discussions with Drs M. Jeffrey and L. González, VLA—Lasswade, Edinburgh. The study was supported by the Wellcome Trust, the Royal Veterinary College, the EU TSE Community Reference Laboratory, and Defra.
