Abstract
Scrapie is a naturally occurring transmissible spongiform encephalopathy (TSE) or prion disease of sheep and goats. Scrapie is a protein misfolding disease where the normal prion protein (PrPC) misfolds into a pathogenic form (PrPSc) that is highly resistant to enzymatic breakdown within the cell and accumulates, eventually leading to neurodegeneration. The amino acid sequence of the prion protein and tissue distribution of PrPSc within affected hosts have a major role in determining susceptibility to and potential environmental contamination with the scrapie agent. Many countries have genotype-based eradication programs that emphasize using rams that express arginine at codon 171 in the prion protein, which is associated with resistance to the classical scrapie agent. In classical scrapie, accumulation of PrPSc within lymphoid and other tissues facilitates environmental contamination and spread of the disease within flocks. A major distinction can be made between classical scrapie strains that are readily spread within populations of susceptible sheep and goats and atypical (Nor-98) scrapie that has unique molecular and phenotype characteristics and is thought to occur spontaneously in older sheep or goats. This review provides an overview of classical and atypical scrapie with consideration of potential transmission of classical scrapie to other mammalian hosts.
Classical scrapie of sheep and goats is a transmissible spongiform encephalopathy (TSE) or prion disease that was first described nearly 300 years ago in the United Kingdom and other western European countries. Since then, classical scrapie has been reported throughout the world with the notable exception of Australia and New Zealand. Classical scrapie can spread within a flock. Within the United States alone, it brings economic losses due to decreased production, export loss, and increased cost for carcass disposal of up to US$20 million annually. 157 Other examples of prion diseases include bovine spongiform encephalopathy (BSE) in cattle, chronic wasting disease in cervids, and Creutzfeldt-Jakob disease (CJD) in humans.
The UK BSE epidemic in the 1990s and evidence that consumption of BSE-contaminated meat causes variant CJD in humans 169 greatly increased interest in the TSEs. More recently, recognition that prion diseases are protein-misfolding diseases akin to Alzheimer disease, Parkinson disease, amyotrophic lateral sclerosis (ALS), and Huntington disease has fueled continued interest in prion diseases. The protein-misfolding diseases share the general mechanism where misfolded proteins catalyze a refolding of normal cellular protein that accumulates and is associated with neural degeneration. 3,48,77,130,148,149 In the case of prion diseases, it is host-encoded cellular prion protein (PrPC) that is misfolded into a pathogenic form (PrPSc). 131 In the absence of PrPC, such as PRNP knockout 27,133 or PrPSc depletion, 107 there is no new formation of PrPSc. Prion diseases are distinct from other protein-misfolding diseases because they are capable of potentially transmitting to susceptible individuals after natural exposure.
Pathogenesis
Two factors that play a major role in pathogenesis of classical scrapie are (1) the amino acid sequence of the prion protein as encoded by the prion protein gene (PRNP) and (2) the tissue distribution of the abnormal prion protein in infected hosts. PRNP genotype, especially amino acids at specific codons affecting prion folding, has a major effect on susceptibility to the classical scrapie agent and incubation periods. Tissue distribution of PrPSc, especially widespread distribution within lymphoid tissues, plays a major role in potential environmental contamination, allowing classical scrapie infection to spread within a flock.
Role of the Host Prion Protein Gene (PRNP)
Polymorphisms in the prion protein gene (PRNP) determine the amino acid sequence of the host’s prion protein and play a major role in relative susceptibility or resistance to classical scrapie. The PRNP polymorphisms in sheep are well defined, and increasing the incidence of resistant alleles is the cornerstone to eradication programs in many countries. The US scrapie eradication program has decreased the number of sheep that are scrapie positive at slaughter by 90%. 157
Sheep have 3 major polymorphic sites in PRNP that influence susceptibility to classical scrapie: codons 136, 154, and 171. Susceptibility to classical scrapie is associated with valine (V) 136, arginine (R) 154, and glutamine (Q) 171 (VRQ haplotype). In contrast, alanine (A) 136, 54,82 histidine (H) 154, 102 and arginine (R) 171 55,119 confer resistance after natural exposure to classical scrapie. The ARR haplotype has the greatest benefit for scrapie resistance. Of the polymorphic sites, codon 171 has the most dominant influence where sheep with QQ171 are susceptible and RR171 are resistant. 55,119 It is of note that a small number of cases of classical scrapie have been detected in sheep with the RR171 genotype, 71,83 and sheep with the RR171 genotype are susceptible to the scrapie agent with a low attack rate and long incubation periods after experimental intracranial inoculation. 87 In a minority of breeds, a third polymorphism, lysine (K) 171, occurs at low frequencies. Similarities in charge and structure suggest that K may behave similarly to R. Sheep with a QK171 genotype have prolonged incubation times relative to QQ171 sheep after intracranial inoculation. 69 Studies recently completed at the National Animal Disease Center indicate that sheep with the KK171 genotype are susceptible to classical scrapie after intracranial inoculation but are resistant to the classical scrapie agent after oronasal inoculation (unpublished data).
Genotype-based breeding programs designed to increase the RR171 genotype in sheep populations have been used in conjunction with removing affected animals, to significantly reduce the number of classical scrapie cases in the United States and European Union. Indeed, study of infected sheep with the VRQ/ARR genotype shows that PrPSc is composed of over 90% Q171 prion protein. 84 Sheep with the ARQ/ARR genotype are considered to have a low susceptibility to the scrapie agent 64 and demonstrate mixed results in experimental studies where age and dose may play a role. Sheep with the ARQ/ARR genotype did not develop classical scrapie when orally inoculated as neonates with 0.1 g of brain material 67 or when inoculated with 5 g of brain material at 5 to 6 months of age. 60 However, 9 of 14 sheep with the ARQ/ARR genotype that were dosed with 5 g of brain material at 7 to 13 days of age had evidence of PrPSc accumulation in lymphoid tissues or brain. 64 Several other polymorphisms that occur with lesser frequency may also increase resistance to classical scrapie: substitution of methionine (M) by threonine (T) at codon 112 (M112 T) 60,100 or leucine (L) by phenylalanine (F) at codon 141 (L141F). 60 Furthermore, codon 141 polymorphisms affect the phenotype of classical scrapie, including clinical presentation and molecular phenotypes. 95
At least 50 PRNP polymorphisms have been described in goat breeds, 1,36 but how they affect incubation times and susceptibility to classical scrapie is less defined than in sheep. Amino acid substitutions that may affect susceptibility to classical scrapie include G127 S, 57 I142 M, 56 N146S/D, 124 H154 R, 10,20,125,158 Q211 R, 10 and Q222 K. 2,10,158 The M142 polymorphism appears to prolong incubation period in experimental studies 56 but not confer resistance to classical scrapie. 99 Of 556 goats tested from infected herds in Cyprus, all positive animals were NN146 with no cases of classical scrapie diagnosed in goats that were either heterozygous or homozygous for S146 or D146. 124 Goats with the H154, Q211, and K222 polymorphisms appear to be resistant to classical scrapie after oral exposure. 99 In 1 recent report of classical scrapie in goats, all affected goats had at least 1 R211 codon, whereas none of the animals were homozygous for Q211 and only 2 heterozygotes were affected, suggesting a strong protective effect after natural exposure. 152 Cases have been diagnosed in goats with 1 K222 allele but not in goats homozygous for the K222 allele. 10,35 Further study of goats heterozygous for the K222 allele that were diagnosed with classical scrapie indicates that the PrPSc produced is almost entirely composed of the Q222 variant, perhaps due to effect of the additional positive charge of lysine interfering with PrPC to PrPSc conversion. 108 A small number of goats homozygous for the K222 allele developed classical scrapie with extremely prolonged incubation periods after intracranial inoculation, suggesting that resistance to classical scrapie is not absolute. 99
Tissue Distribution of PrPSc
Oral exposure of the classical scrapie agent in a contaminated environment is the major route of entry. 6,45,65,134,161 After ingestion, PrPSc crosses the intact intestinal barrier at the level of the enterocytes and passes rapidly into lymph and blood, with M cells playing a critical role in prion uptake. 43 These initial steps of PrPSc entry are identical in sheep of susceptible and resistant genotypes; however, only in susceptible sheep has PrPSc accumulation been shown to subsequently take place in the follicular dendritic cells of lymphoid structures. 76,86 The first replication of the classical scrapie agent is within the gut-associated lymphoid tissues such as the ileal Peyer’s patches. 6,86 The greater prominence of gut-associated lymphoid tissues in young animals is likely a factor in the increased relative susceptibility of younger compared to older sheep. 129,153
After early replication of prions in gut-associated lymphoid tissues, PrPSc can be demonstrated in tonsil, spleen, and retropharyngeal and mesenteric lymph nodes 116,132 for months before there is evidence of PrPSc in the brain. Widespread accumulation of PrPSc in lymphoid tissues during the asymptomatic phase may allow infected sheep to serve as a source of environmental prion contamination long before clinical signs occur as they excrete prions through saliva 155 and feces. 136
Within infected flocks, the risk of natural transmission is greatest from the ewe to offspring of susceptible genotypes through the placenta and placental fluids, resulting in vertical transmission to susceptible offspring and the potential for horizontal exposure at the time of lambing. 156 PrPSc in the placenta is associated with the placentomes of genetically susceptible offspring 7 or occurs in lesser amounts in the cotyledons associated with lambs of intermediately resistant genotypes (ARQ/ARR) if they share the same uterine horn as a susceptible sibling. 4 Infectivity of fetoplacental tissues may also lead to in utero transmission. 150 Milk and colostrum represent an additional risk of transmission to susceptible offspring born to infected ewes. 93,96
Goats that are infected with classical scrapie commonly come from herds that are comingled with sheep, 117 but classical scrapie also spreads within herds containing only goats. 172 Similar to what is observed in sheep with classical scrapie, assessment of goats with classical scrapie suggests widespread involvement of the lymphoreticular system. 63 Accumulation of PrPSc in the brain of goats with classical scrapie appears to be affected by the PRNP genotype at codon 142, with M142 carriers showing earlier and more frequent accumulation of PrPSc in the brain as opposed to I142 homozygotes that accumulate PrPSc in the brain only after a large number of lymphoid tissues are affected. 62 One factor that may play a major role in the rates of transmission of natural scrapie is that goats with classical scrapie have lower amounts of PrPSc in cotyledons than sheep; 120 however, the amount present is adequate to support transmission to goats or sheep after oral exposure. 138
Diagnosis of Scrapie
Before 1998, routine diagnosis of TSEs relied on the presence of vacuolation in the brainstem (Fig. 1a). Current diagnostics are based on detection of PrPSc, which can be done by enzyme-linked immunosorbent assay (ELISA), immunohistochemistry, or western blot after a pretreatment such as proteinase K to allow for differentiation between PrPC and PrPSc. 110,121 Confirmatory diagnosis of classical scrapie by immunohistochemistry uses tissues from brainstem at the level of the obex (Fig. 1b), 122 which is the first area of the brain to become immunoreactive in preclinical cases of classical scrapie. 160 PrPSc is detectable by immunohistochemistry in both follicular dendritic cells and tingible body macrophages 89 of the lymphoid follicles (Fig. 7), which allows for antemortem diagnosis using biopsies from the third eyelid, 118 tonsil, 139 or rectal mucosa (Fig. 9). 59
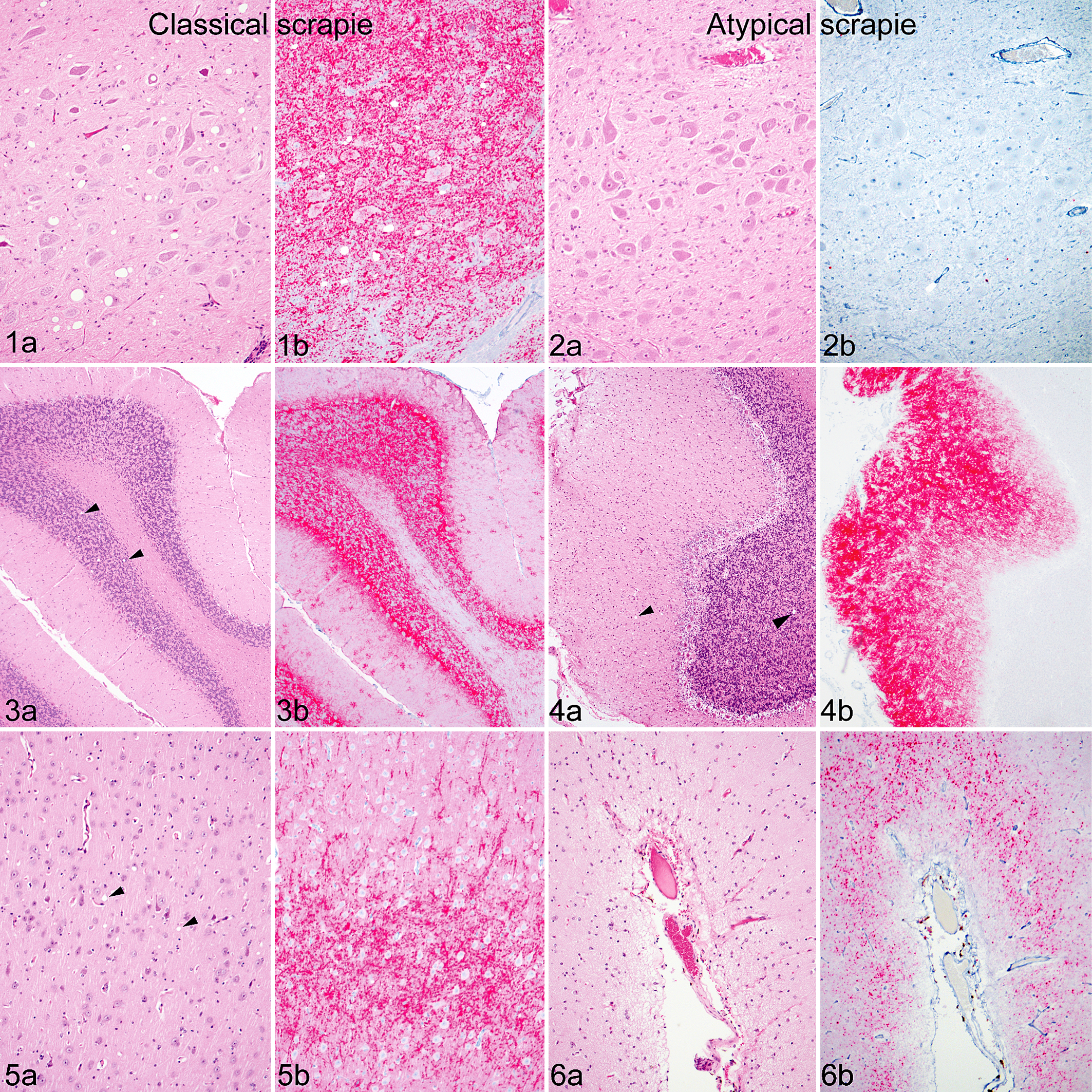
Scrapie, brain, sheep. Comparison of spongiform change and PrPSc immunoreactivity in various brain regions of a sheep (VRQ/ARQ genotype) clinically affected with classical scrapie after experimental oronasal inoculation (Figs. 1, 3, 5) and a sheep (ARQ/ARR genotype) clinically affected with atypical scrapie after experimental intracranial inoculation (Figs. 2, 4, 6). a: hematoxylin and eosin. b: immunohistochemistry for PrPSc.
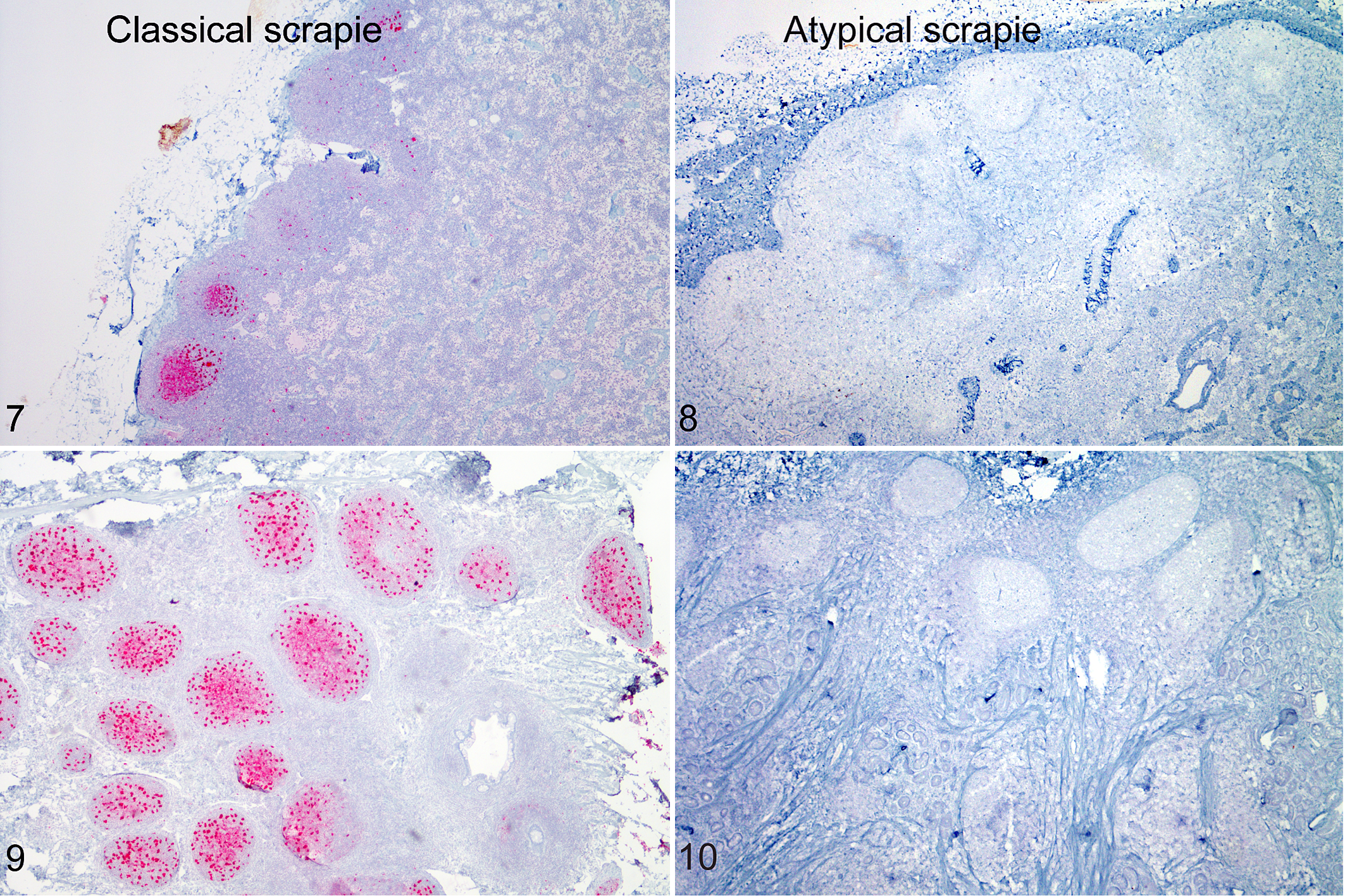
Scrapie, sheep. Comparison of immunoreactivity (red) for PrPSc in retropharyngeal lymph nodes and rectal mucosal biopsies of a sheep (VRQ/ARQ genotype) clinically affected with classical scrapie after experimental oronasal inoculation (Figs. 7, 9) and a sheep (ARQ/ARR genotype) clinically affected with atypical scrapie after experimental intracranial inoculation (Figs. 8, 10). Immunohistochemistry for PrPSc.
Strains
TSE agents can exist in multiple strains that may exhibit different disease phenotypes and pathogenesis. 30,143 Strains may be differentiated by clinical signs, 126 incubation periods and lesion profiles in mouse models, 23,51,52 cellular and neuroanatomical deposition of PrPSc, 24,61 molecular profile on western blot (the apparent molecular mass 18 and/or glycoform ratios 154 of PrPSc fragments), 18 or reactivity to antibodies binding to different regions of PrPSc. 101 Strain properties are maintained through conformational differences in PrPSc as there are no amino acid sequence differences between PrPC and PrPSc. 19,104 The hypothesis that strains arise from differences in protein structure 127 is supported by the reaction of different strain isolates to conformational antibodies 135 and stability assays. 163 It is not known how new strains arise, but it could be due to inherent conformational flexibility of the prion protein, presence of PRNP polymorphisms within a host species, or interspecies transmission events. 115
Many scrapie strains are difficult to differentiate by western blot 114 but can be differentiated by in-depth analysis of immunoreactivity patterns in multiple brain regions. 61,113 A rarely identified sheep scrapie isolate referred to as CH1641 is of note because of an appearance on western blot with some similarities to BSE, 80 such as a lower apparent molecular mass of the unglycosylated fragment. 154 With the use of a panel of antibodies, CH1641 can be differentiated from BSE by immunohistochemistry 85 or western blot. 11,12,154 Furthermore, BSE readily transmits to conventional mice, 26 whereas CH1641 does not. 49 The most important strain designation to recognize in scrapie is between classical scrapie strains (described above) and the more recently described atypical scrapie.
Atypical Scrapie
Atypical (Nor98) scrapie was first detected in Norway in 1998, 15,16 but retrospective studies indicate that this phenotype has been present since at least the 1980s. 25,166 Atypical scrapie is different from classical scrapie in clinical presentation, molecular characteristics and distribution of PrPSc within infected sheep, genotypes affected, and epidemiology. Atypical scrapie has been identified throughout Europe, 9,40,41,47,53 North America, 106,111 New Zealand, 91 and Australia. 34 The worldwide distribution with similar incidence rates where detected supports a separate etiology from classical scrapie 47 and that it is spontaneous 109 or transmits very poorly under natural conditions. 46 Thus, atypical scrapie is recognized as a separate, nonreportable disease by the World Organization for Animal Health (OIE).
The original report cited progressive ataxia as the predominant clinical sign, 16 but most often these cases are detected during routine diagnostic screening of older cull animals (active surveillance) where neurologic findings are absent or ill-defined. The spongiform change and PrPSc deposition in atypical scrapie cases occur predominantly in the cortices of the cerebellum (Fig. 4) and the cerebrum (Fig. 6) rather than the medulla oblongata, as seen in classical sheep scrapie. 113 Similarly, the immunohistochemistry pattern of PrPSc deposition for atypical scrapie is different from classical scrapie (Figs. 3, 5). PrPSc immunoreactivity in atypical scrapie is mild in the obex (Fig. 2) but more intense and widespread, especially in the cerebellum. 113 As opposed to classical scrapie, cases of atypical scrapie demonstrate only 4 morphologic forms of immunoreactivity: fine granular, aggregates, linear, and perineuronal and distinctive immunoreactivity in the white matter. 113 Fine granular and aggregate staining types are most common, 113 are not cell associated, and may represent PrPSc near synapses or on distal neuronal processes. 5 Intraneuronal staining does not occur in atypical scrapie, which may reflect the fact that atypical scrapie is more proteinase K sensitive than classical scrapie, 92 suggesting a greater ability of cells to digest the abnormal prion. Current diagnostic methods fail to demonstrate PrPSc in peripheral or lymphoid tissues of sheep affected with atypical scrapie (Figs. 8, 10); 8,16 however, infectivity has been demonstrated in ileum, spleen, skeletal muscle, lymphoid tissues, and peripheral nerves by bioassay. 8,146
A major diagnostic feature of atypical TSEs are short proteinase K–resistant fragments that are associated with poor transmissibility and immunoreactivity largely limited to synapses. 98 Cases of atypical scrapie are distinguished by a 5-band profile on western blot with a prominent lower band at approximately 11 to 12 kDa, 9,15,70,92 whereas classical scrapie has the nonglycosylated band at approximately 19 to 21 kDa (Fig. 11). 75,80,147 In addition, the PrPSc of atypical scrapie is relatively sensitive to proteinases. 92
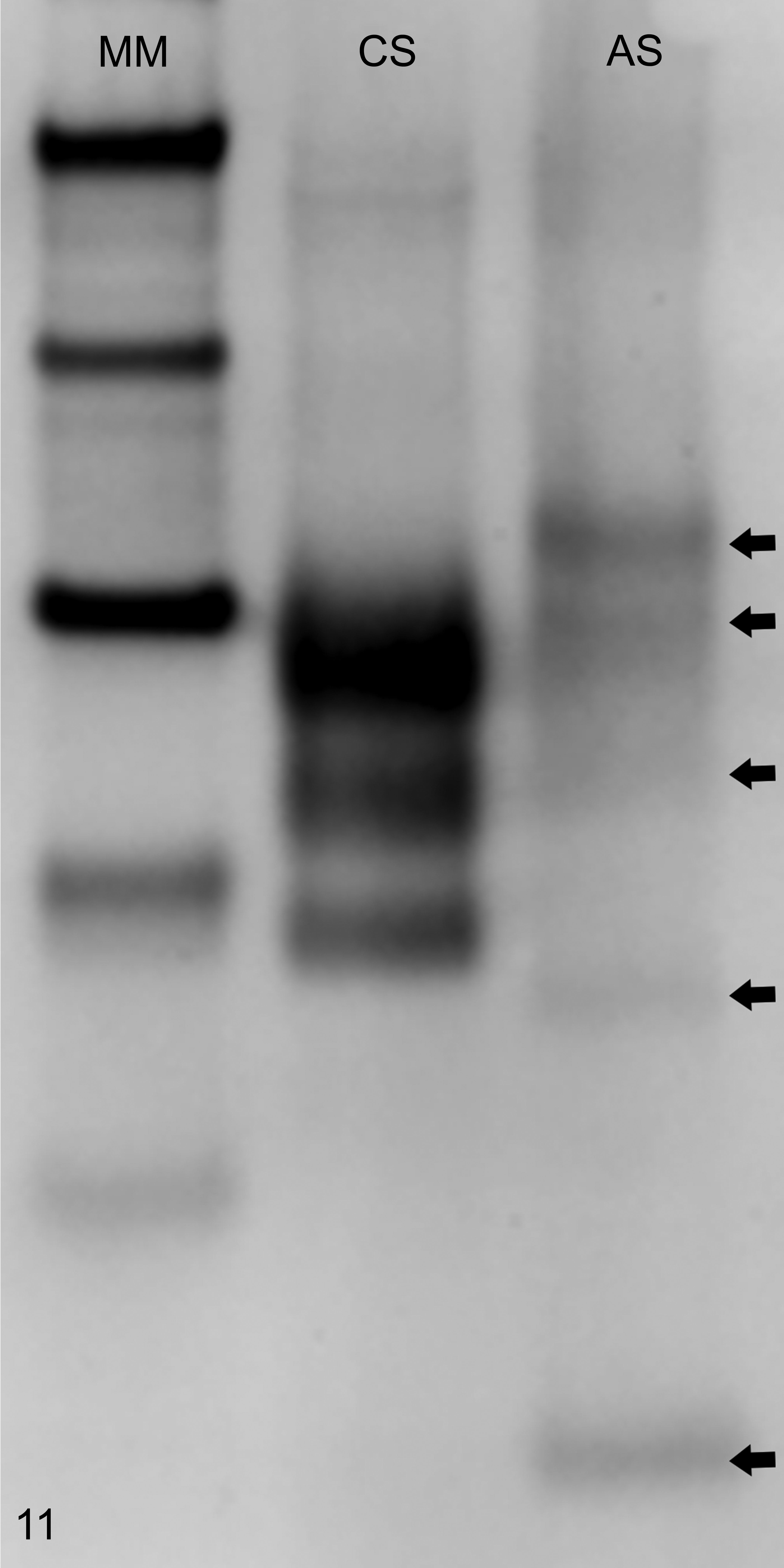
Western immunoblot of brain samples from sheep clinically ill with either classical (CS) or atypical sheep (AS) scrapie developed using monoclonal antibody P4 to detect PrPSc. After proteinase K digestion, a sample from the brainstem of a sheep with classical scrapie has 3 immunoreactive bands that migrate at approximately 33, 30, and 21 kDa, whereas a sample from the cerebellum of a sheep with atypical scrapie has 5 less intense bands (arrows) with the smallest migrating at approximately 11 to 12 kDa. MM, molecular mass.
PRNP genotype is a major factor in atypical scrapie cases with polymorphisms at codons 141 (F, phenylalanine) and 154 (H) being highly associated with identified cases. Sheep in the original report carried at least 1 AHQ allele. 16 Atypical scrapie has been transmitted experimentally to AHQ sheep by the intracranial 145 and oral 146 routes. An increased risk of atypical scrapie has also been identified in sheep with the AF141RQ haplotype. 137 Atypical scrapie does experimentally transmit to sheep with the AL141RQ haplotype but with very long incubation periods without clinical signs. 123 Furthermore, sheep with the ARR haplotype, which confers resistance to classical scrapie and is the cornerstone of genotype-based eradication programs, do not appear to be protected against developing atypical scrapie. 41,137
Atypical scrapie has also been reported in goats, 103,142 where the molecular profile on western blot is similar to atypical scrapie in sheep, but the distribution of lesions within the brain is more rostral (thalamus and midbrain) than atypical scrapie of sheep. 142 Similar to sheep with atypical scrapie, histidine substitution at PRNP codon 154 is a risk factor for atypical scrapie in goats, 32 and PrPSc has not been demonstrated in the lymphoid tissues of affected goats. 142
Interspecies Transmission
Experimental interspecies transmission of prion agents provides valuable information about potential host ranges. Unsuccessful attempts at interspecies transmission led to the concept of a species barrier, an influence on prion transmission due to mismatches between host and recipient prion amino acid sequence and the resulting structures and folding. 17,31,78,79,112,140,162 Species barrier can manifest as complete lack of susceptibility, incomplete attack rates, or prolonged incubation times. Interspecies transmission studies are done to fully assess potential risks to animal health, and potential risks to human health can be studied through the use of transgenic mice expressing human PRNP.
Classical scrapie has been experimentally transmitted to numerous omnivorous species after intracranial inoculation, including European bank voles (Myodes glareolus), 42,128 meadow voles (Microtus pennsylvanicus), 28 raccoons, 72,74 and pigs. 66 With the exception of European bank voles, which express PRNP that is permissive to many donor strains, 165 these studies suggest a substantial species barrier to infection by natural routes.
One hypothesis for the origin of BSE in the United Kingdom was that it resulted from the passage of a scrapie-like disease into the cattle population 168 through the feeding of ruminant-derived meat and bone meal (MBM). Experimental studies performed in the United States and United Kingdom demonstrated that classical scrapie does not transmit to cattle by the oral route of inoculation, 37,97 and successful transmission after intracranial inoculation results in a disease that is distinguishable from BSE by clinical signs, the molecular profile of PrPSc, and PrPSc deposition patterns in brain sections. 21,38,39,94 These studies, however, are far from exhaustive and leave untested the possibilities that prion protein genotype of the donor or transmission of another prion agent such as CH1641 scrapie or atypical scrapie to cattle could have been the origin of BSE.
The agent of BSE has been demonstrated to transmit to other species, including humans. 26,141 Small ruminants were likely exposed to the same sources of BSE infectivity as cattle, creating concern that the BSE agent could be misdiagnosed as scrapie 90 and represent an additional risk to human health. Experimental studies demonstrate that the agent of BSE transmits to sheep, results in a wide distribution of PrPSc in peripheral tissues and brain, 14,50,105 and can transmit horizontally between sheep. 13 Most cases of classical sheep scrapie appear to be invariant in western blot profile that is differentiable from BSE, 81 but BSE in sheep has some similarities to the CH1641 strain of scrapie (described above). Despite significant surveillance efforts, no natural cases of BSE have been described in sheep, but 2 natural cases of BSE have been identified in goats. 44,88,151 One hypothesis as to why these cases have only been identified in goats is that goats are more intensively managed and had higher exposure to contaminated feed concentrates, 151 similar to the explanation of why the incidence of BSE is higher in dairy herds relative to beef herds. 167
Chronic wasting disease (CWD) is a naturally occurring prion disease of cervids with strong similarities to classical scrapie, including widespread accumulation of PrPSc in the lymphoid and nervous tissues of affected animals. 114,144 Chronic wasting disease was first identified in captive cervids, and one hypothesis is that it originated as a cross-species transmission of the classical sheep scrapie agent. 170 Experimental studies in white-tailed deer lend support to this hypothesis: inoculation of white-tailed deer results in a 100% attack rate after either intracranial or oronasal inoculation. 68 Furthermore, PrPSc is distributed throughout the lymphoid tissues, and samples collected from brainstem have a CWD-like western blot pattern. 68 The classical scrapie agent was transmitted to Rocky Mountain elk (Cervus elaphus nelsoni) after experimental intracranial inoculation; however, the results suggest elk are not likely to be susceptible to the classical scrapie agent by more natural routes of exposure. After intracranial inoculation with the classical sheep scrapie agent, only 3 of 6 animals developed neurologic signs, and accumulation of PrPSc was limited to the central nervous system without spreading to lymphoid tissues. 73
Continued research is needed to clarify the potential risks of the scrapie agent to human health. There is no evidence from epidemiologic studies that the scrapie agent has ever transmitted to humans, 22,159 and studies using transgenic mice that express wild-type levels of human PRNP have failed to demonstrate transmission of the classical 58 or atypical 164,171 scrapie agents. However, the classical scrapie agent has transmitted to cynomolgus macaques (with a slightly different prion protein amino acid sequence than humans) 33 and mice overexpressing human PRNP. 29 Western blot analysis of brain tissues from these studies demonstrates a molecular profile similar to sporadic CJD, suggesting that ongoing surveillance for potential interspecies transmission events and further studies to clarify potential risks of scrapie transmission to humans are critical.
Footnotes
Acknowledgements
Mention of trade names or commercial products in this article is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the US Department of Agriculture (USDA). USDA is an equal opportunity provider and employer.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
