Abstract
We report the diagnostic sensitivity of 3 EU-approved rapid tests (ELISAs; 1 from IDEXX and 2 from Bio-Rad) for the detection of transmissible spongiform encephalopathy diseases in goats. Ninety-eight goat brainstem samples were tested. All the rapid tests had 100% specificity and ≥80% sensitivity, with the IDEXX test significantly more sensitive than the 2 Bio-Rad tests. All tests detected 100% of samples from goats with clinical scrapie, but missed 8% (IDEXX) to 33% (Bio-Rad SG) of samples from preclinical goats. Importantly, only IDEXX picked up all samples from clinical bovine spongiform encephalopathy (BSE)-infected goats, whereas the other 2 rapid tests missed 15% (Bio-Rad SG) to 25% (Bio-Rad SAP). These results show that a fraction of preclinical scrapie infections are likely missed by EU surveillance, with sensitivity of detection strongly dependent on the choice of the rapid test. Moreover, a significant proportion of clinical BSE infections are underestimated by using either Bio-Rad test. Assuming that the same sensitivity on preclinical goats would also occur in BSE-infected goats, our data suggest that IDEXX is likely the most sensitive test for detecting preclinical field cases of BSE infection in goats, although with an 8% failure rate. These results raise some concerns about the reliability of current EU surveillance figures on BSE infection in goats.
Prion infection induces progressive and untreatable neurodegenerative diseases in humans, wild and farmed ruminants, and occasionally in other mammalian species. Prion or transmissible spongiform encephalopathy (TSE) diseases are characterized by the formation and accumulation of an abnormal isoform of the natural prion protein (PrPc) in the central nervous system (CNS) and, occasionally, in peripheral tissues. The pathologic prion protein associated with scrapie (PrPSc) differs from PrPc because it appears refolded, aggregated, and partially protease resistant. These unique features of PrPSc have been used for the development of most test methods currently used for the detection of TSE diseases.
Scrapie disease of sheep and goats has been endemic in Europe for >200 y, but has never been convincingly associated with any form of human TSE disease, although data based on experimental transmission of scrapie to humanized mice 4 or nonhuman primates 6 have reopened this issue. On the other hand, the epidemic of bovine spongiform encephalopathy (BSE) in the United Kingdom and other European cattle populations has been unequivocally linked to the appearance of variant Creutzfeldt–Jakob disease in humans.2,5,23 Because BSE is experimentally transmissible to sheep and goats 15 and these small ruminants were likely exposed to BSE-contaminated feed in the early 1980s, there is concern that the BSE agent may circulate in the sheep and goat population, representing a possible secondary risk to humans.7,16
In 2006, the European Commission Regulation (EC) 253/2006 8 approved 9 rapid postmortem tests to monitor the prevalence of scrapie and BSE in small ruminant populations. Sensitivity, based on the lowest detectable concentration of PrPSc above background noise, and specificity were assessed in classical scrapie cases. In addition, the performance of these tests for the identification of atypical scrapie and BSE in sheep was also evaluated.10–19 Only IDEXX, a Bio-Rad SAP, b and Bio-Rad SG c rapid tests (enzyme-linked immunosorbent assays [ELISAs]) were able to detect atypical scrapie, a result also confirmed by routine screening for scrapie in sheep and goats.3,22 In 2012, the European Food Safety Authority also recommended the PrioSTRIP SR d test (visual reading protocol) for the detection of TSE disease in small ruminants. However, a specific study on the suitability of rapid methods for the detection of TSE diseases in goats was never performed.
The goat population in Europe is considerably smaller than that of sheep, but these ruminants were likely heavily exposed to the BSE agent because of feeding of concentrate for dairy farming purposes. Thus, evaluation of surveillance for the goat population is crucial.
We compared the performance of 3 EU-approved rapid postmortem tests on brain samples from goats with “natural” scrapie or goats with experimental scrapie or BSE. These tests are stated to be 100% specific and 100% sensitive through the EU approval testing procedures on sheep brain samples.
Ninety-eight goat brainstem samples were included in our study. All samples were prepared as 50% tissue macerates in water, as described below. Thirty-one of these samples were sourced from goats with “natural” scrapie from 7 different EU countries (Table 1), 7 from clinically affected goats, and 24 from clinically healthy animals. Other samples (n = 32) from goats with experimentally induced scrapie or BSE were provided by the CVI, FLI, Roslin, INRA, and CEA (full names in Table 1). All samples from TSE-positive animals also tested PrPSc positive by Western blot or immunohistochemical analyses as required by EU Regulation (EC) No. 999/2001. 9 PRNP analyses revealed that 60% of goats carried the wild genotype, whereas polymorphisms I142M (11%), H143R (9%), R154H (2%), R211Q (23%), or repeats deletion (4%) were found in a few animals. Negative control samples were from clinically healthy goats slaughtered in Italy and the samples were, as expected, negative by Western blot analysis. 14 The whole brainstem sample was trimmed, pooled, and then minced with a scalpel blade, until the tissue appeared homogeneous. Sterile nuclease-free water was added in an equal amount (50% water/volume) to create a 1:1 dilution. The suspension was subjected to cycles of homogenization using a low-speed, handheld homogenizing unit until achievement of a homogeneous paste. The resulting homogenate was immediately stored at –80°C and kept frozen until tested. Samples were tested by the IDEXX, Bio-Rad SAP, and Bio-Rad SG ELISAs according to the manufacturer’s test instructions. The PrioSTRIP SR test was not included in this analysis. The 3 tests are based on semiquantitative ELISA methods that produce a qualitative result relative to a cutoff value. The 2 Bio-Rad tests include a proteinase K (PK) digestion step to unmask cryptic epitopes, whereas the IDEXX test relies on conformational detection technology using a specific proteinase-resistant binding dextran polymer. 17
Details and origin of goat samples used in the current study.*
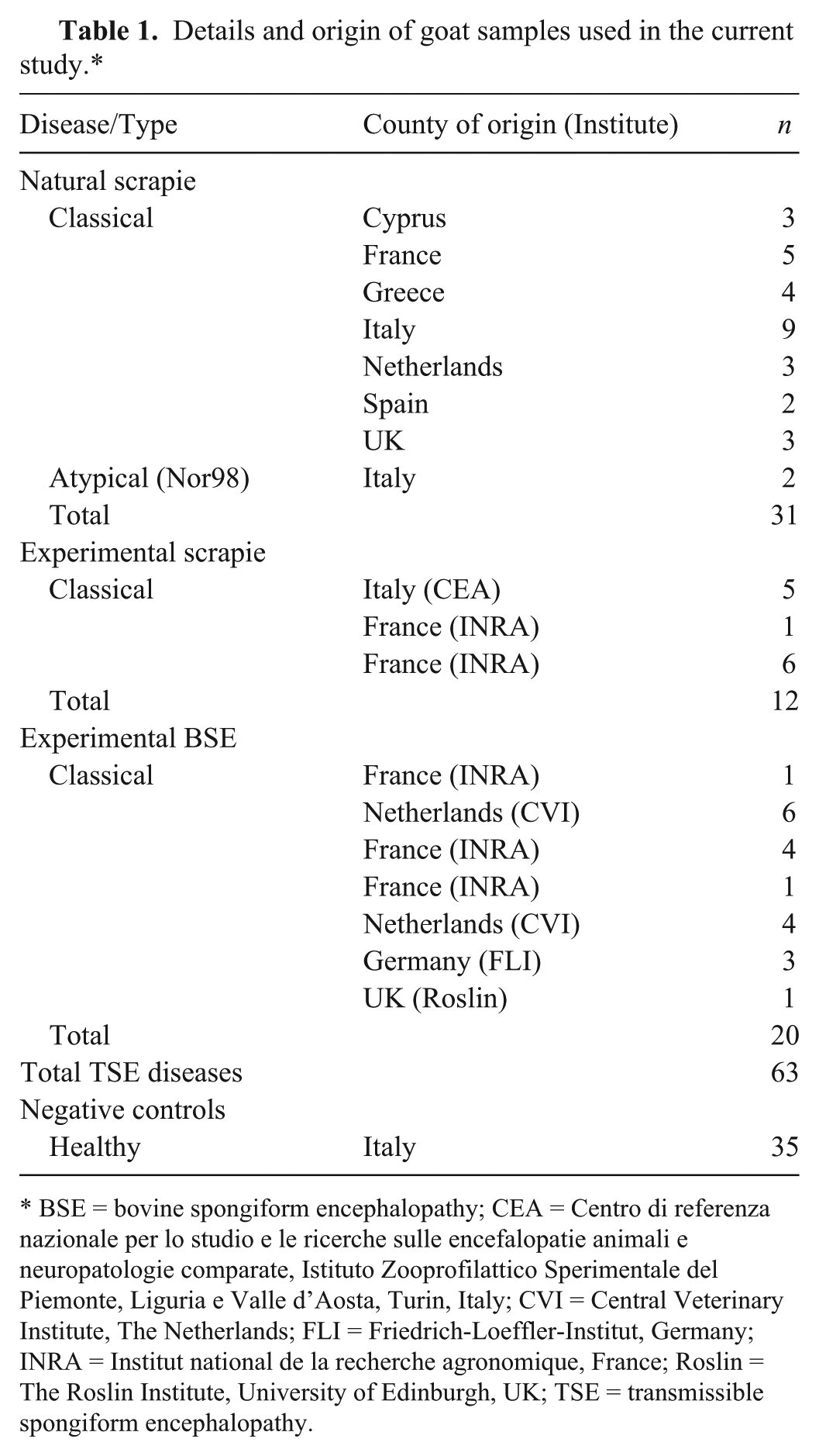
BSE = bovine spongiform encephalopathy; CEA = Centro di referenza nazionale per lo studio e le ricerche sulle encefalopatie animali e neuropatologie comparate, Istituto Zooprofilattico Sperimentale del Piemonte, Liguria e Valle d’Aosta, Turin, Italy; CVI = Central Veterinary Institute, The Netherlands; FLI = Friedrich-Loeffler-Institut, Germany; INRA = Institut national de la recherche agronomique, France; Roslin = The Roslin Institute, University of Edinburgh, UK; TSE = transmissible spongiform encephalopathy.
The manufacturers specifically provided us with a unique batch of each rapid test well before the expiration dates to avoid false results produced by old, although unexpired, batches. Samples were coded and then tested in duplicate, except for 3 samples from Greece and 1 from the United Kingdom because of insufficient material. The 3 rapid tests are semiquantitative ELISAs that produce qualitative results based on cutoff values. Samples with optical density lower than the cutoff value on both replicates were considered negative. Samples showing an optical density greater than or equal to the cutoff value on at least one replicate were considered positive. However, because the Bio-Rad specifications suggest a cautious interpretation of samples situated just below the cutoff value (cutoff value of 10%), we arbitrarily chose to consider these samples as positive. Environmental conditions that might influence testing, such as temperature and humidity, were strictly controlled and monitored during analytical sessions.
The efficiency of each rapid test was assessed by the receiver operating characteristic (ROC) curve analyses using STATA. e Nonparametric ROC curves analyzed TSE-infected goats versus healthy and unaffected goats. The area under the ROC curve and its 95% confidence interval (95% CI) indicate diagnostic efficiency.
Overall, the 3 EU-approved rapid tests analyzed showed 100% specificity and >80% sensitivity (Table 2). However, ROC curves showed that the IDEXX test was significantly more sensitive (97%) than the 2 Bio-Rad rapid tests (Tables 3, 4), which showed sensitivity just >80%.
Number and percentage of positive samples in goats with different forms of transmissible spongiform encephalopathy (TSE) diseases by different rapid tests.*
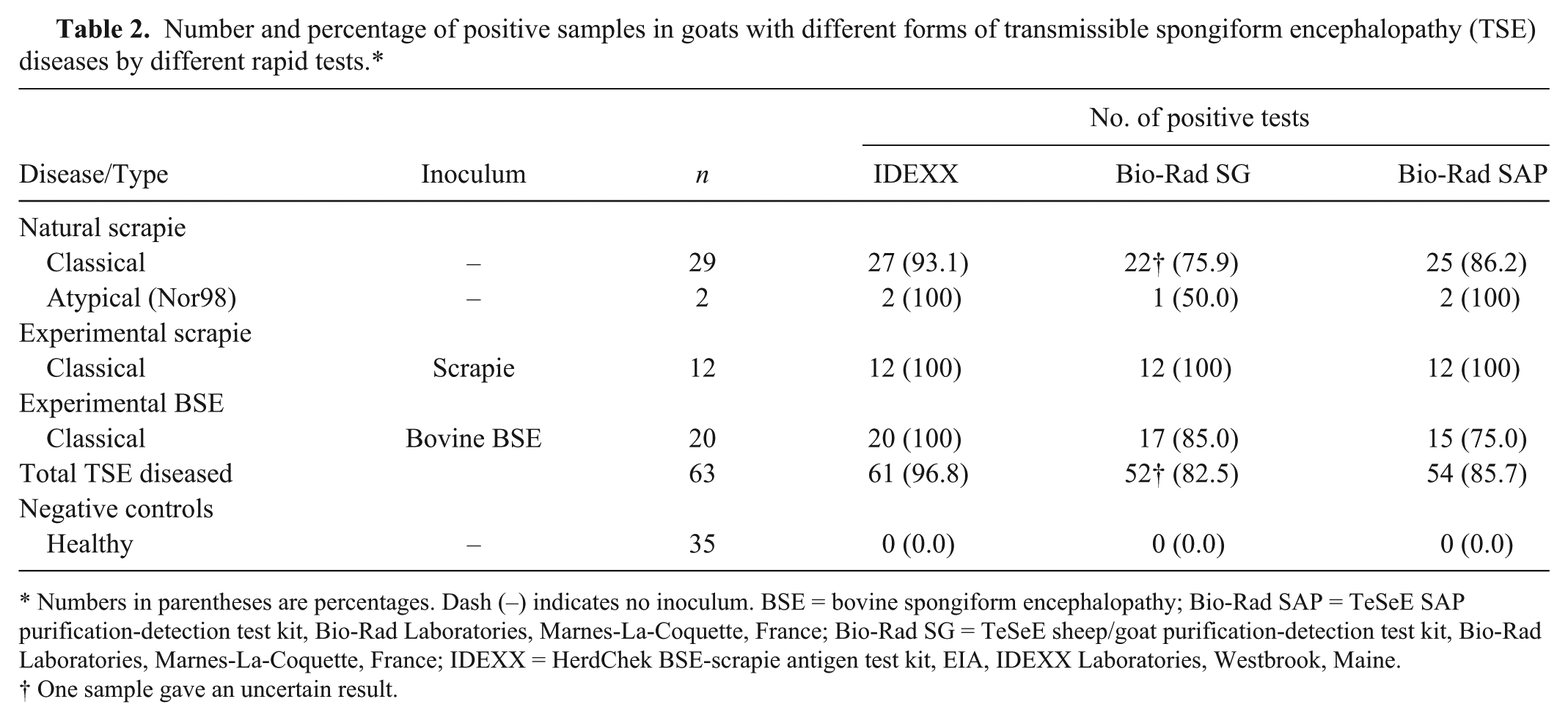
Numbers in parentheses are percentages. Dash (–) indicates no inoculum. BSE = bovine spongiform encephalopathy; Bio-Rad SAP = TeSeE SAP purification-detection test kit, Bio-Rad Laboratories, Marnes-La-Coquette, France; Bio-Rad SG = TeSeE sheep/goat purification-detection test kit, Bio-Rad Laboratories, Marnes-La-Coquette, France; IDEXX = HerdChek BSE-scrapie antigen test kit, EIA, IDEXX Laboratories, Westbrook, Maine.
One sample gave an uncertain result.
Number and percentage of positive samples by different tests on “natural” scrapie- affected goats.*
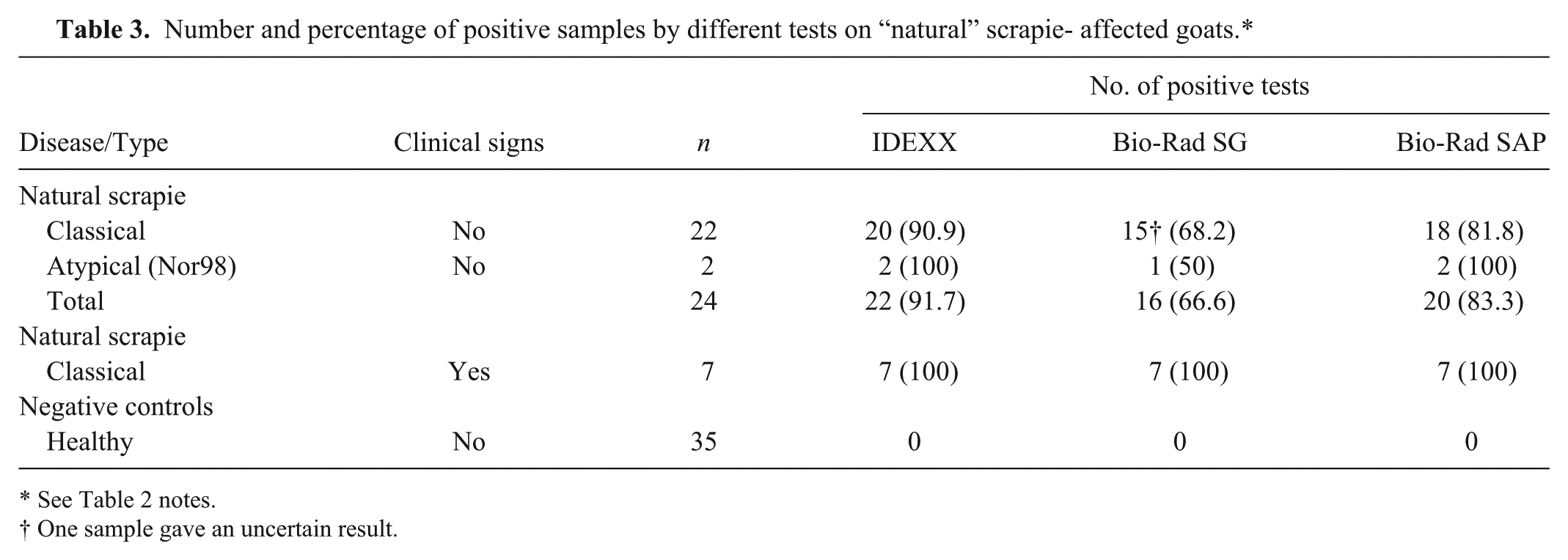
See Table 2 notes.
One sample gave an uncertain result.
Receiver operating characteristic curve analyses of rapid tests for transmissible spongiform encephalopathy (TSE) diseases in goats.*
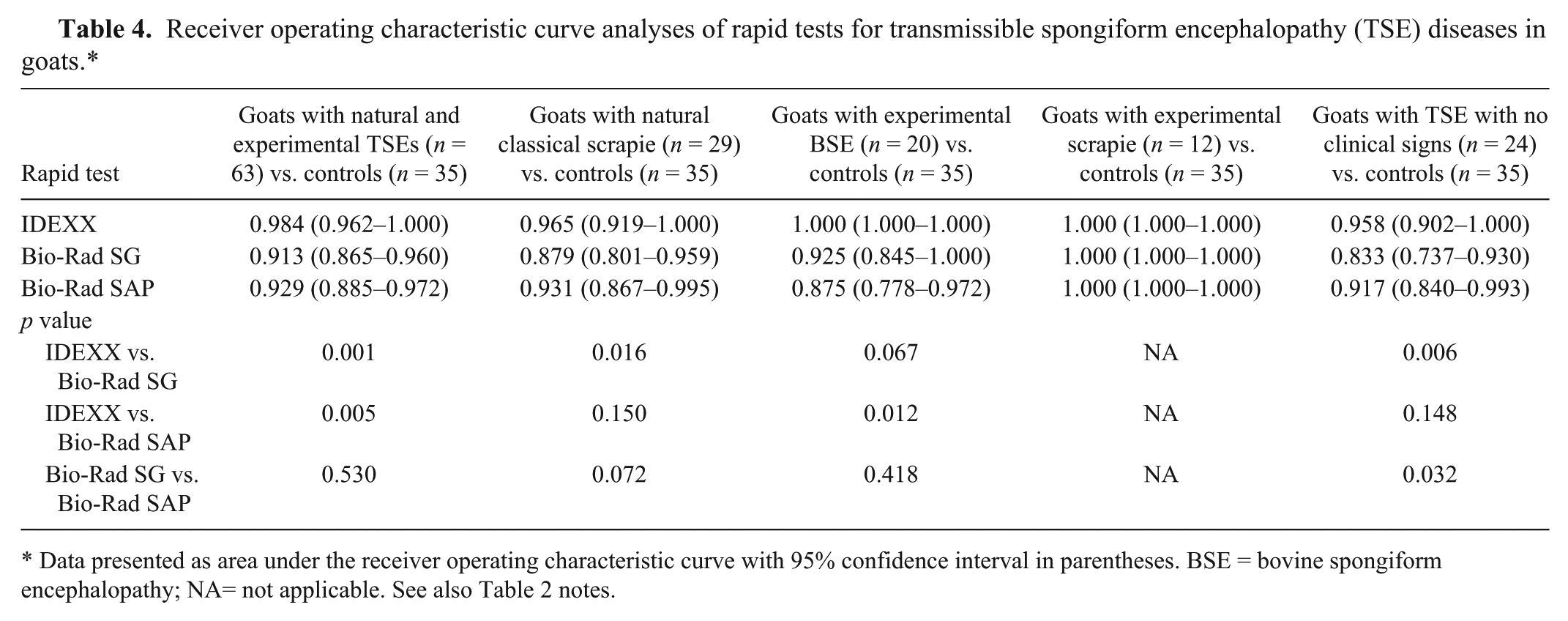
Data presented as area under the receiver operating characteristic curve with 95% confidence interval in parentheses. BSE = bovine spongiform encephalopathy; NA= not applicable. See also Table 2 notes.
A more detailed analysis showed that all 3 rapid tests recognized 100% of samples from goats with experimental scrapie regardless of the route of infection, but only the IDEXX test showed 100% sensitivity in detecting BSE-infected goats (Tables 2, 4). The other 2 rapid tests missed 3 (Bio-Rad SG) to 5 (Bio-Rad SAP) of the 20 BSE samples (Table 2) with differences that reached significance only between IDEXX and Bio-Rad SAP tests (Table 4).
In goats with natural “classical” scrapie, the IDEXX test missed 2 of 29 samples and none of the “atypical” scrapie-infected samples; Bio-Rad SAP missed 4 samples, and Bio-Rad SG missed 7 (a further sample gave an uncertain result, but was considered positive in the ROC curve analyses; Table 2). The 2 samples from asymptomatic goats were not recognized by the IDEXX test or by either of the Bio-Rad tests. ROC curves showed that the sensitivity of the IDEXX test was significantly higher only compared with the Bio-Rad SG test (Table 4). Other comparisons did not show any significant differences (Table 4).
Finally, we compared the sensitivity of rapid tests in recognizing goats with scrapie in the preclinical or clinical phase of disease. Although all rapid tests were systematically able to pick up both natural and experimental scrapie samples from symptomatic goats (Table 3), the IDEXX test missed 2 of 24 samples with “natural” scrapie in the preclinical phase of disease, the Bio-Rad SAP test missed 4 samples, and the Bio-Rad SG test missed 7 (Table 3). ROC curve analysis showed that IDEXX and Bio-Rad SAP tests were significantly more sensitive than the Bio-Rad SG test (Table 4) in detecting positive samples from preclinical animals.
Several important features of our study should be considered for the surveillance of TSE diseases in goats. All tests detected 100% of samples from goats with clinical scrapie, regardless of whether they were experimentally or naturally infected. In contrast, sensitivity was lower in goats with preclinical scrapie, and rapid tests missed 8% (IDEXX) to 33% (Bio-Rad SG) of these samples. A second important consideration is that only IDEXX detected all samples from clinical BSE-infected goats, whereas the other 2 rapid tests missed 15% (Bio-Rad SG) to 25% (Bio-Rad SAP) of samples. These results suggest that a consistent fraction of preclinical scrapie infection is likely missed by EU surveillance, mostly in areas where Bio-Rad tests are in use, and that BSE infection in goats may also be underreported in areas using the Bio-Rad rapid tests (Tables 2, 4). Assuming that the same sensitivity in preclinical goats would also occur in BSE-infected goats, our data show that the IDEXX test may detect eventual preclinical field cases of BSE infection in goats, although with a disappointing 8% failure rate. Although the analytical sensitivity of some TSE rapid tests might be reduced by the method used to prepare our samples,1,21 the results raise some concerns in relation to the current figures on BSE infections in goats deriving from EU surveillance.
In goats, the difference in performance of rapid tests between scrapie and BSE infection might depend on the use of PK digestion, the choice of the primary anti-PrP antibodies, or both. PK digestion is used by both Bio-Rad tests but not by the IDEXX test, and it is likely that antibodies used in each kit recognize different PrP epitopes. This last hypothesis, however, is purely speculative because the details on anti-PrP antibodies are covered by patents and are therefore not publicly available.
The other interesting result, although based solely on 2 samples, is that only the IDEXX and Bio-Rad SAP tests were able to fully recognize samples from goats with the atypical Nor98 scrapie infection, suggesting that surveillance in countries using the Bio-Rad SG test would miss a proportion of atypical scrapie infections in the goat population. The small number of samples, however, is too low to allow a firm conclusion.
All rapid tests in our study failed to recognize the same 2 samples of “natural” preclinical scrapie. This finding is of concern because it might indicate that there is a small subpopulation of “naturally” scrapie-infected goats (e.g., early preclinical animals) that would be missed by all available rapid tests, and thus by the surveillance system. PRNP polymorphisms might reduce the sensitivity of the assays in goats carrying specific genotypes by reducing antibody-binding epitopes.18,20 In our samples, however, statistical analysis did not show any association between failure of each test and goat genotypes (data not shown). The reason for this finding remains therefore unknown and might simply depend on low levels of PrPSc. Ultimately, none of the 3 rapid tests produced any false-positive results, showing a reassuring 100% specificity.
Footnotes
Authors’ contributions
D Meloni drafted the manuscript. D Meloni, E Bozzetta, and PL Acutis contributed to conception and design of the study. D Meloni, PL Acutis, JPM Langeveld, MH Groschup, O Andrèoletti, I Lantier, L Van Keulen, D Pitardi, T Sklaviadis, F Ingravalle, S Peletto, S Colussi contributed to acquisition, analysis, and interpretation of data. R Nonno, A Bossers, W Goldmann provided important information on samples. All authors critically revised the manuscript, gave final approval, and agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
HerdChek BSE-scrapie antigen test kit, EIA, IDEXX Laboratories, Westbrook, ME.
b.
TeSeE SAP purification-detection test kit, Bio-Rad Laboratories, Marnes-La-Coquette, France.
c.
TeSeE sheep/goat purification-detection test kit, Bio-Rad Laboratories, Marnes-La-Coquette, France.
d.
Check PrioSTRIP SR, Prionics, Zurich, Switzerland.
e.
Stata Statistical Software 2007 Release 10, StataCorp, College Station, TX.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grant GoatBSE–Specific Targeted Research Project (FOOD-CT-2006-36353).
