Abstract
Bovine spongiform encephalopathy (BSE) had never been detected in Sweden until 2006, when the active surveillance identified a case in a 12-year-old cow. The case was an unusual form, because several molecular features of the protease-resistant prion protein (PrPres) were different from classical BSE. The differences included higher susceptibility for proteinase K, higher molecular weight of the PrPres bands, affinity to the N-terminus-specific antibodies 12B2 and P4, and peculiar banding pattern with antibody SAF84 showing an additional band at the 14 kDa position. The molecular characteristics were in accordance to previous descriptions of H-type BSE. This report shows that a range of Western blot techniques and antibodies can be applied to confirm H-type BSE and further describes that the ratio of the amounts of PrPres#1 and PrPres#2, after deglycosylation, depends on the antibody used during processing. Immunohistochemistry on sections of medulla at the level of the obex applying antibodies with epitopes covering a broad range of the PrP sequence showed accumulation of disease-specific PrP (PrP d ) in the gray matter. Fine punctate deposition in the neuropil was the most predominant type and was more severe in BSE target nuclei. The types of PrP d deposition are described in comparison with classical BSE. PrP-gene sequencing showed 6 copy octarepeat alleles and no abnormalities. It is postulated that the disease had a spontaneous origin, rather than having had been acquired in the BSE epidemic.
Keywords
Introduction
Transmissible spongiform encephalopathies (TSE) or prion diseases are infectious neurodegenerative diseases that are accompanied by accumulation of host-encoded prion protein (PrP c ) in an aberrant or disease-associated prion protein (PrPSc or PrP d ). Upon digestion with endoproteases, a partially resistant core known as PrPres remains.
Bovine spongiform encephalopathy (BSE) is a TSE primarily of cattle, but there is strong evidence that BSE has crossed the species barrier infecting humans and several species of animals. 10,14,20,28,44 BSE was first identified in the United Kingdom in 1986, 42 and its human form, variant Creutzfeldt-Jakob, was described in 1996. 10,14 Bovine spongiform encephalopathy is transmitted by the dietary route and was spread in cattle by recycling of meat and bone meal. The resultant epidemic within the United Kingdom, most European countries, the United States, Japan, and Canada has been a major food safety concern and has had devastating effects in the beef industry.
Until recently, all cases of BSE from different geographical origins were thought to be caused by a single prion strain. This was based on 1) an invariable phenotype of disease in cattle, 30,36,43 including consistent and homogenous patterns of immunostainings in the brain stem, 12 2) a unique and consistent lesion profile, incubation time, and PrP d distribution in the brain of mouse models in biological strain typing of BSE from various sources, including human with variant Creutzfeldt-Jakob, 7,9,18,29 and 3) similar biochemical features of the PrPres in natural and experimental BSE. 37
However, bovine TSE that differs from classical BSE (C-type BSE) has been reported, mainly in older cattle. Initially, a case of BSE with an unusually low amount of diglycosylated banding and low-molecular-weight migration of the unglycosylated was reported in Japan in a 23-month-old Holstein steer. 45 In Italy, 2 cows were reported to show PrP d deposition mostly as amyloid plaques, with different neuroanatomical distribution and aberrant molecular features, including an apparent predominance for the monoglycosylated form. The condition was named bovine amyloidotic spongiform encephalopathy. 13 In France, 3 cases were also reported to have different molecular features than classical BSE. These cases were essentially characterized by a higher apparent molecular mass (Mr) of the unglycosylated form associated with binding by a PrP N-terminus-specific monoclonal antibody (MAb). 6 The 2 unusual types of BSE were thereafter named L-type and H-type BSE, making reference to the lower and higher molecular weight of the unglycosylated band observed in the Italian and French cases, respectively. 1,11 Additional cases of atypical BSE were reported in cattle 1,11,21,32,34 and in a zebu (Bos indicus). 35
Panels of antibodies against different regions of the prion protein can be used in Western blot (WB) and immunohistochemistry (IHC) to characterize the properties of PrPres and PrP d . 5,24,41 Recently, it was shown that antibodies to PrP can be categorized into groups A, B, and C, which are thought to bind in the respective bovine PrP regions. Group A antibodies represent the 62–107 (N-terminal region), group B, the upstream amino acid sequence 109–164, and group C, 154–236 (C-terminal region). 5 Using WB analysis of proteinase K (PK)-digested brain homogenates, all group A, B, and C antibodies yielded similar banding and glycoprofile patterns in C-type and L-type BSE. L-type was characterized by a similarity in relative concentrations of diglycosylated and monoglycosylated PrPres and C-type by a relative high concentration of diglycosylated PrPres. 5,6,11,13,21 Group A antibodies 12B2 and P4 had a remarkable binding with H-type BSE but none or greatly reduced binding with C-type or L-type BSE. Of these 2 antibodies in group A, 12B2 is the most suitable for application in Bosgenus because its epitope is fully present in bovine PrP and perhaps for discrimination of BSE strains in all animal species. 21,27,33 Group B and C antibodies were reactive with all types of BSE, but these groups differed between each other when applied to H-type BSE in banding patterns and glycoprofiles. 21 Group B antibodies to H-type BSE showed 1–2 kDa higher Mr of the PrPres fragments, and with group C antibodies a complex Western profile was shown in H-type BSE. This was characterized by the presence of 4 bands I to IV, respectively, including an additional unglycosylated band of 14 kDa. It was proposed that PrPres from H-type BSE was formed by a mixture of 2 triplet band populations, PrPres#1 and PrPres#2. PrPres#1 is reactive with group A, B, and C antibodies, while PrPres#2 is reactive only with group C antibodies. Each of these 2 populations is supposed to occur in diglycosylated, monoglycosylated, and unglycosylated forms. 5
Bovine spongiform encephalopathy had never been detected in Sweden until the surveillance program in 2006 identified a case. The purpose of this report is to describe the clinical characteristics, to show the confirmatory WB results with a variety of techniques, and to show the results of further molecular investigation of the PrPres#1 and PrPres#2 using deglycosylation. This report also describes the first detailed study of the immunohistochemical PrP d deposition in an H-type case, in comparison to that found in C-type BSE.
Material and methods
Animal, tissue sample
A 12-year-old mixed-breed Charolais cow, born on the farm, within a herd of 50 cows, located in Västmanland, central Sweden, was sent for rendering. A sample of brain stem was submitted to the National Veterinary Institute (SVA) of Sweden for TSE testing as part of the active BSE surveillance. The material was partially autolytic. The brain stem was cut sagitally in 2 hemisections; 1 part was fixed in 10% neutral buffered formalin, and another portion was used for rapid enzyme-linked immunosorbent assay (ELISA) screening test (Platelia/TeSeE a ). The remaining unfixed tissue was used for WB and genotyping analysis according to methods previously described. 34
Western blot analyses
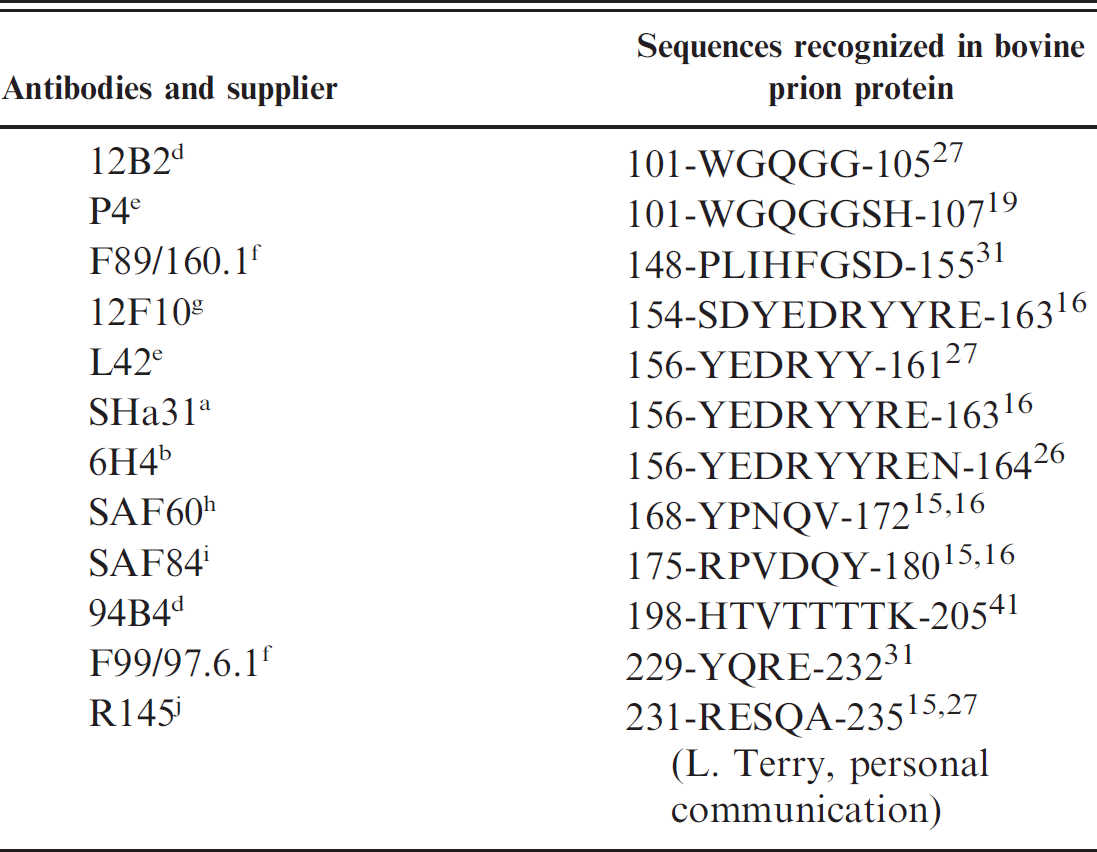
For WB analysis the SVA utilized a commercial kit (BioRad Bovine WB a ). Samples of other prions diseases, C-type BSE, classical scrapie, and Nor98 were analyzed in parallel for comparison. Further WB analysis was performed at Veterinary Laboratories Agency (VLA; Weybridge, UK), Agence Francaise de Sécurité Sanitaire des Aliments (AFSSA; Lyon, France), and Central Institute for Animal Disease Control (Lelystad, The Netherlands). The antibodies used for the WB analyses, their source, and the sequence they recognize in the bovine PrP are shown in Table 1.
The VLA applied 4 WB protocols: 1) the VLA hybrid discriminatory WB method, which discriminates between experimental BSE in sheep and natural sheep scrapie isolates using monoclonal antibodies (MAbs) 6H4 and P4 38,39 2) the OIE-SAFW B using MAb 6H4 and ultracentrifugation 37 3) the Bio-Rad TeSeE WB a using MAb Sha 31; 4) the VLA sodium phosphotungstic acid WB using MAb 6H4 and P4, which is based on the incorporation of a sodium phosphotungstic acid-precipitation step to enhance the PrPres band signals. 37 For comparison, a C-type BSE case from the UK was included in all of the analyses.
Methods employed for WB analysis at the AFSSA have been previously described in detail. 5 The following MAbs and concentrations were applied: P4 (0.2 μg/ml), SHa31 (1:10), 12B2 (0.3 μg/ml), and SAF84 (0.150 μg/ml). Additional WB analysis after enzymatic deglycosylation by peptide: N-glycosidase F (PNGase) treatment was conducted
Monoclonal antibodies employed in the Western blot and immunohistochemistry studies and the sequences recognized in the bovine prion protein.
as previously described. 5 This analysis was used to study the relative proportions of PrPres#1 and PrPres#2 shown by the MAbs and their respective concentrations: F89/160.1.5 (200 ng/ml), 12F10 (100 ng/ml), SAF60 (ascitic fluid 1/5,000), SAF84 (150 ng/ml), 94B4 (35.5 ng/ml), and F99.97.6.1 (540 ng/ml). The labeling signals were quantified using VersaDoc 5000 a digital capture and software Quantity One. a Methods used by Central Institute for Animal Disease Control for testing PK sensitivity of the case have been described in detail before. 21
Histopathology and immunohistochemistry
Caudal brain stem was routinely processed, embedded in paraffin blocks, sectioned at 4 to 5 μm, and stained with hematoxylin and eosin and Congo red for visualization of amyloid. Blocks were not treated with formic acid. For IHC the sections were mounted on Vectabond c -treated glass slides, deparaffinized in xylene, and rehydrated. Endogenous peroxidase activity was blocked with 3% hydrogen peroxide. Sections were incubated with normal horse serum (ABC Kit c ) to block nonspecific binding sites. The sections were pretreated in 96% formic acid for 5 minutes followed by hydrated autoclaving in citrate buffer at 122°C for 30 minutes. Afterwards, they were incubated at room temperature for 60 minutes with 1 of the following primary MAbs and dilutions: F89/160.1 (1:1,900), F99/97.6.1 (1:2,000), P4 (1:1,000), 12F10 (1:400), 12B2 (0.2 μg/ml), 94B4 (2 μg/ml), and R145 (1 μg/ml). The amino acid sequences of the bovine PrP recognized by the MAbs and their source are shown in Table 1. The detection was conducted with the Vectastain ABC System, c and color was developed with aminoethylcarbazole substrate c for all of the antibodies except for 12F10, which was developed with diaminobenzidine. c Sections were counterstained with hematoxylin. Each immunostain was accompanied by a positive reference control (obex of C-type BSE reference cases from Spain, 6 cases) and negative controls (obex of BSE negative cattle of matched age, 1 normal and 1 with listeriosis, and a primary antibody-omitted control of each section). Patterns of deposition of the PrP d and its intensity were assessed semiquantitatively in the reticular formation, dorsal motor nucleus of the vagus nerve (DMNV), nucleus of the solitary tract, cuneatus nuclei, nucleus of the spinal tract of the trigeminal nerve, and olivary nuclei.
Results
Clinical examination
The cow was in late pregnancy and showed sudden difficulties in rising. Clinical examination revealed lateral recumbency and tympany, and the rectal temperature was 37.0°C. Reactions to pain stimuli and muscle tone in both hind limbs were normal. There were no signs of fractures of the pelvis, and the calf and uterus were in normal position. General appearance was normal, and there were no signs of nervousness or abnormal behavioral reactions. The cow was treated with calcium and a nonsteroidal anti-inflammatory drug and was standing a few hours later. The next morning she was again in recumbence. A blood sample was obtained, the same treatment administered, and she was walking 30 minutes later. The veterinarian noted a slight lameness on the left hind limb but no ataxia. Serum calcium was normal, but there was increased creatinine kinase. On the third day, the cow was recumbent again, and she was treated with prostaglandins to initiate parturition. Three days later, when parturition did not occur, she was culled.
Rapid test
The ELISA screening test was positive in the first run, with an optical density (OD) value of 2.42, in a second run in 2 wells (OD values 2.77 and 2.88), and in a subsequent testing of a second homogenate (OD value of 3.22). The cutoff value was 0.22 in all of the runs.
Western blot
All of the WB analyses detected PrPres and therefore confirmed the case as a TSE. The WB test at the SVA using MAb Sha31 showed a higher molecular weight of the unglycosylated band (and the 2 glycosylated bands) than in the C-type BSE control (Fig. 1A). Compared with scrapie samples, the Swedish case also exhibited a higher molecular weight of the bands than classical scrapie and a different banding pattern than Nor98 atypical scrapie.
Results of the various WB methods revealed the following molecular characteristics, which discriminate the Swedish case from C-BSE: higher molecular weight of the unglycosylated band than in the C-type BSE when using the group B antibodies, such as
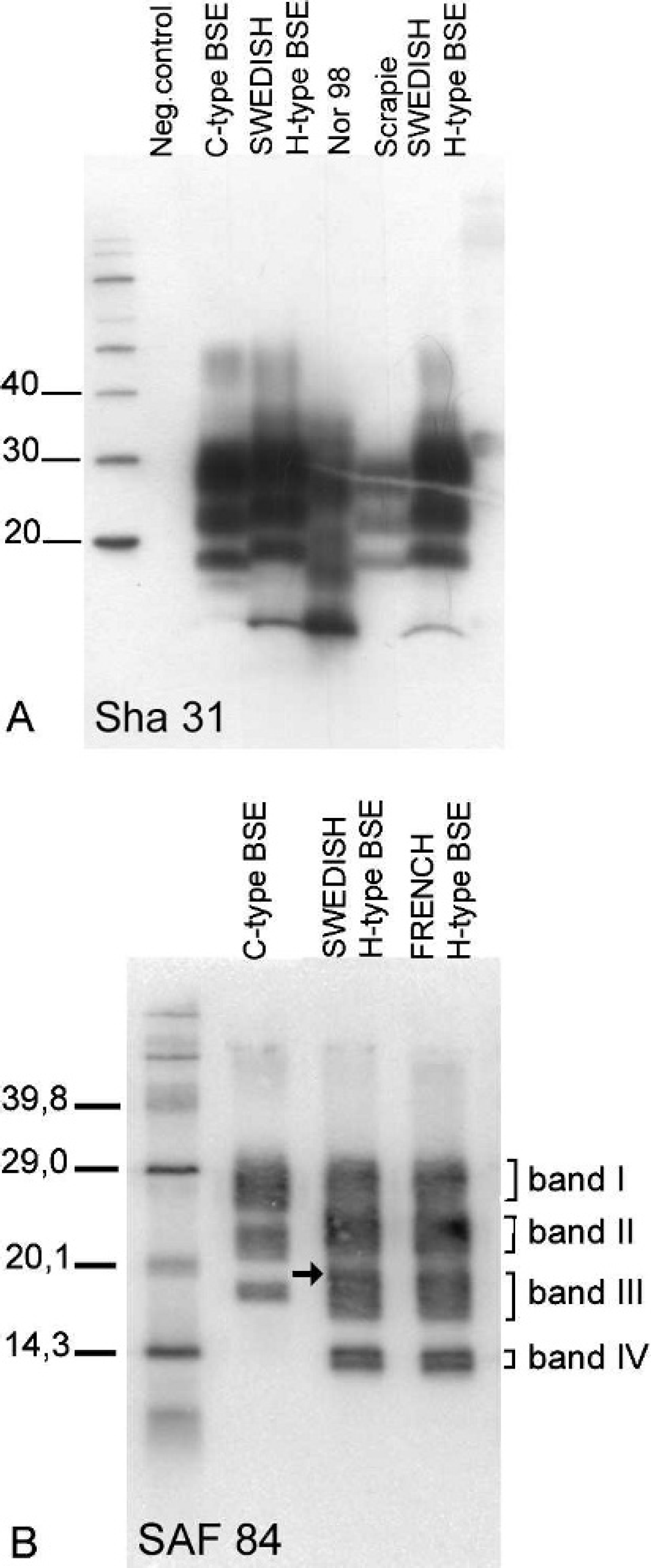
Western blot (WB) analyses of the Swedish H-type BSE case conducted on the brain stem. The bars indicate the position of molecular markers (kDa).
SHa31 (Fig. 1A), labeling with group A MAb P4 and 12B2, particularly strong with the latter, enhanced sensitivity for PK digestion, and a 4-banding pattern with a group C antibody (Fig. 1B). In addition to these previously described features of H-type BSE, the proportion of PrPres#1 to PrPres#2 in enzymatically deglycosylated samples was very different depending of the group C antibody used (Fig. 2). The ratio PrPres#1/PrPres#2 was 80/20 with SAF84 and F99/160.1.5, 56/44 with SAF60, and 39/61 with 94B4.
Histopathology and immunohistochemistry
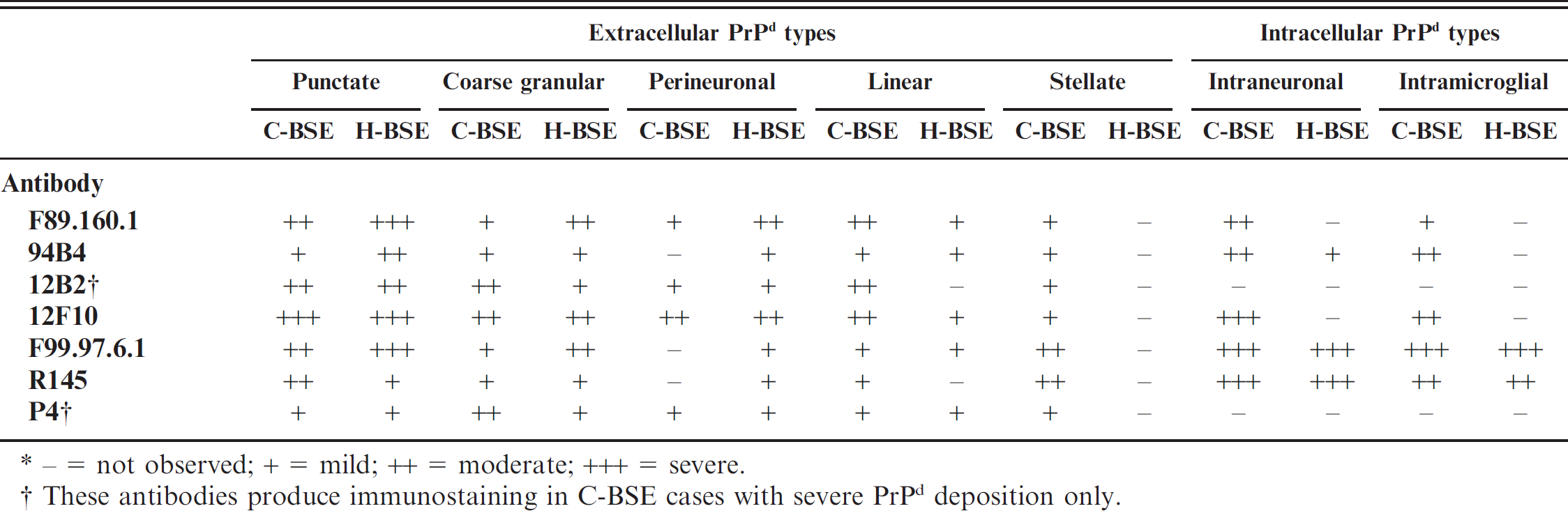
Samples from this case for histopathology and IHC were of poor quality; however, a few vacuoles in the neuropil in the solitary tract nucleus and the nucleus of the spinal tract of the trigeminal nerve resembled lesions described with BSE (Fig. 3A). No amyloid was observed. All the antibodies applied in IHC resulted in immunostaining, but the intensity of different PrP d types varied with the different antibodies. The average score for the neuroanatomical areas for each type of PrP d in the Swedish H-type case and in the C-BSE cases is shown in Table 2. The most frequent PrP d type in the Swedish case was thin punctate deposition in the neuropil (Fig. 3A, 3B), which sometimes coalesced into coarser granules (Fig. 3B). Staining was diffuse in the gray matter and was strongest with F89.160.1. Neuropil PrP d deposition was more intense, with all of the MAbs, in the lateral aspects of the nucleus of the spinal tract of the trigeminal nerve (Fig. 3C) followed by the solitary tract nucleus. Perineuronal (Fig. 3F) and to a lesser extent linear (Fig. 3E) depositions were also observed. The MAbs F99.97.6.1 and R145 produced the most concentrated intracellular PrP d deposits (Fig. 3E, 3G). Antibodies 94B4 showed it only in a few neurons, mostly in the reticular formation, while MAbs P4, 12B2, 12F10, and F89.160.1 did not show either intraneuronal or intramicroglial accumulations. In general, the accumulation of intraneuronal granules of PrP d were more evident in the large neurons of the cuneatus and olivary nuclei and of the reticular formation. Within the reticular formation, there was a high degree of variation in the intensity of intraneuronal PrP d deposition and in the number of neurons affected. No other PrP d types could be identified. In C-type BSE, the MAbs 12B2 and P4 produced extracellular immunostaining, but only in the cases with heaviest PrP d deposition. Antibodies 12F10 and F89.160.1 showed abundant intracellular PrP d accumulation.
Sequencing of the prion protein gene
The prion protein alleles of the cow were found to be normal (i.e., having unremarkable cattle-like PrP gene sequences). The animal had synonymous polymorphisms at codon 23 (L23L, CTC/CTT) and codon 78 (Q78Q, CAG/CAA), and both alleles contained the 6-copy octapeptide repeat region.
Discussion
Results reported here indicated that the Swedish cow was affected by a TSE different from epizootic classical (C-type) BSE. Several differences in biochemical features of PrPres included: the position of
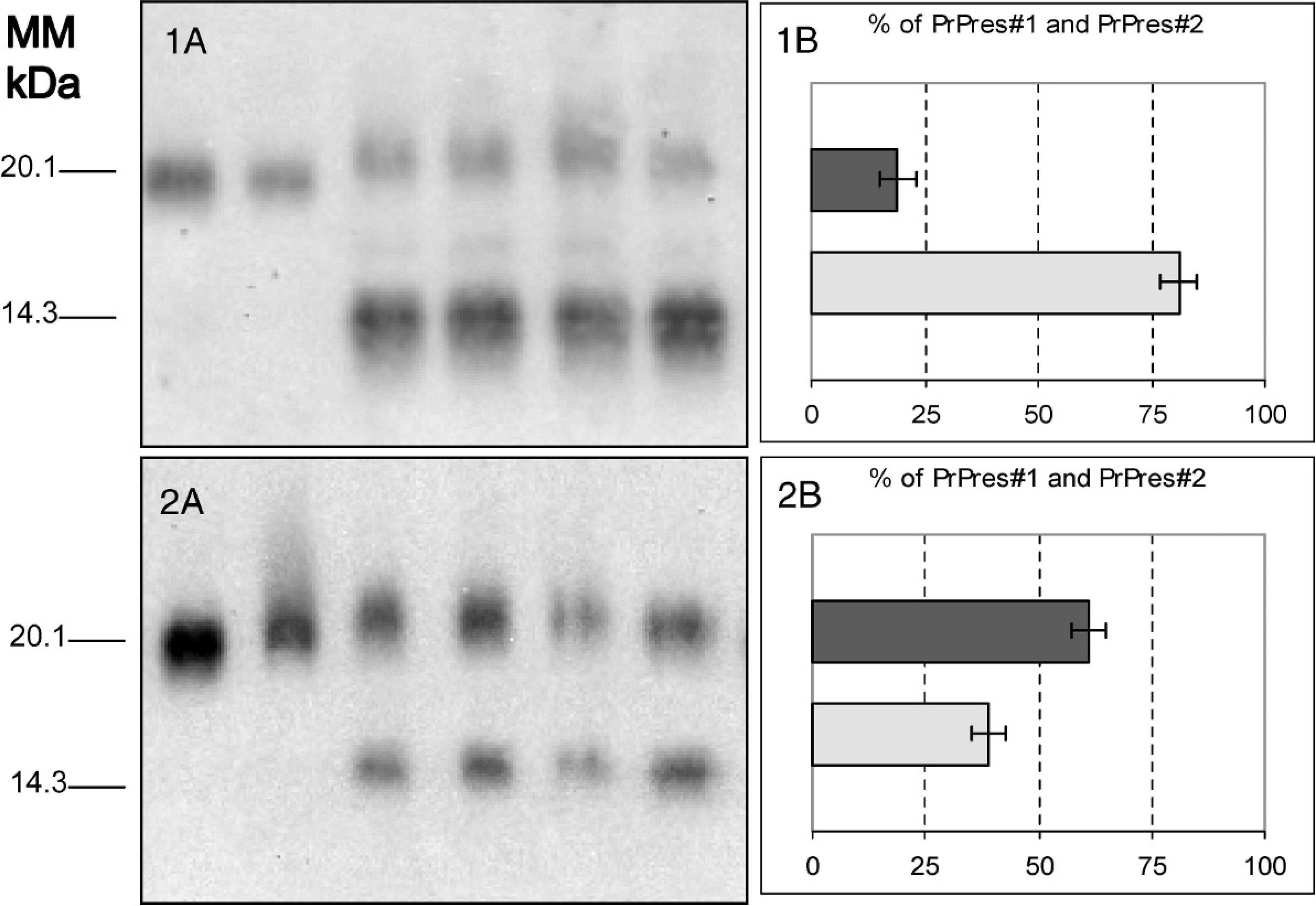
Western blot (WB) analysis of PrPres with antibodies F99/97.6.1 (1) or 94B4 (2) after PNGase F treatment.
the PrPres fragments, with a higher molecular weight of the monoglycosylated and the unglycosylated PrPres bands, the antibody affinity, with binding to group A, N-terminus-specific antibodies, the markedly higher susceptibility for PK under stringent conditions of pH and PK concentration, and the peculiar 4-band pattern (bands I to IV) with group C antibody, including an additional band (band IV) at about the 14 kDa position. In addition, the occurrence of 2 bands after enzymatic deglycosylation of PrPres with PNGase F was different from the C-type. The Swedish case closely resembles descriptions for H-type BSE in Europe 5,6,11,21 and the United States. 34 This report indicates that H-type BSE can be diagnosed and/or confirmed by several different WB methods.
In the present study, the complexity of WB features with C-terminal antibodies was in agreement to a previous description of H-type BSE. 5 The 4-band pattern can be best explained by the existence of 2 PrPres triplet-band populations (PrPres#1 and PrPres#2) reactive with antibodies specific for region 154–242 of bovine PrP. The WB investigations showed additionally that the apparent proportion of the 2 PrPres populations was dependant on the C-terminal antibodies used. With group C antibodies, the different PrPres#1/PrPres#2 ratios as shown after enzymatic deglycosylation (Fig. 2) can be attributable to differences in affinities between antibodies and epitope structure and cleavage by PK between the PrPres populations.
Histopathologic examination was hampered by autolysis and was only possible in the medulla region. Nonetheless, a few neuropil vacuoles typical for BSE were identified in BSE-target nuclei. The PrP d deposits
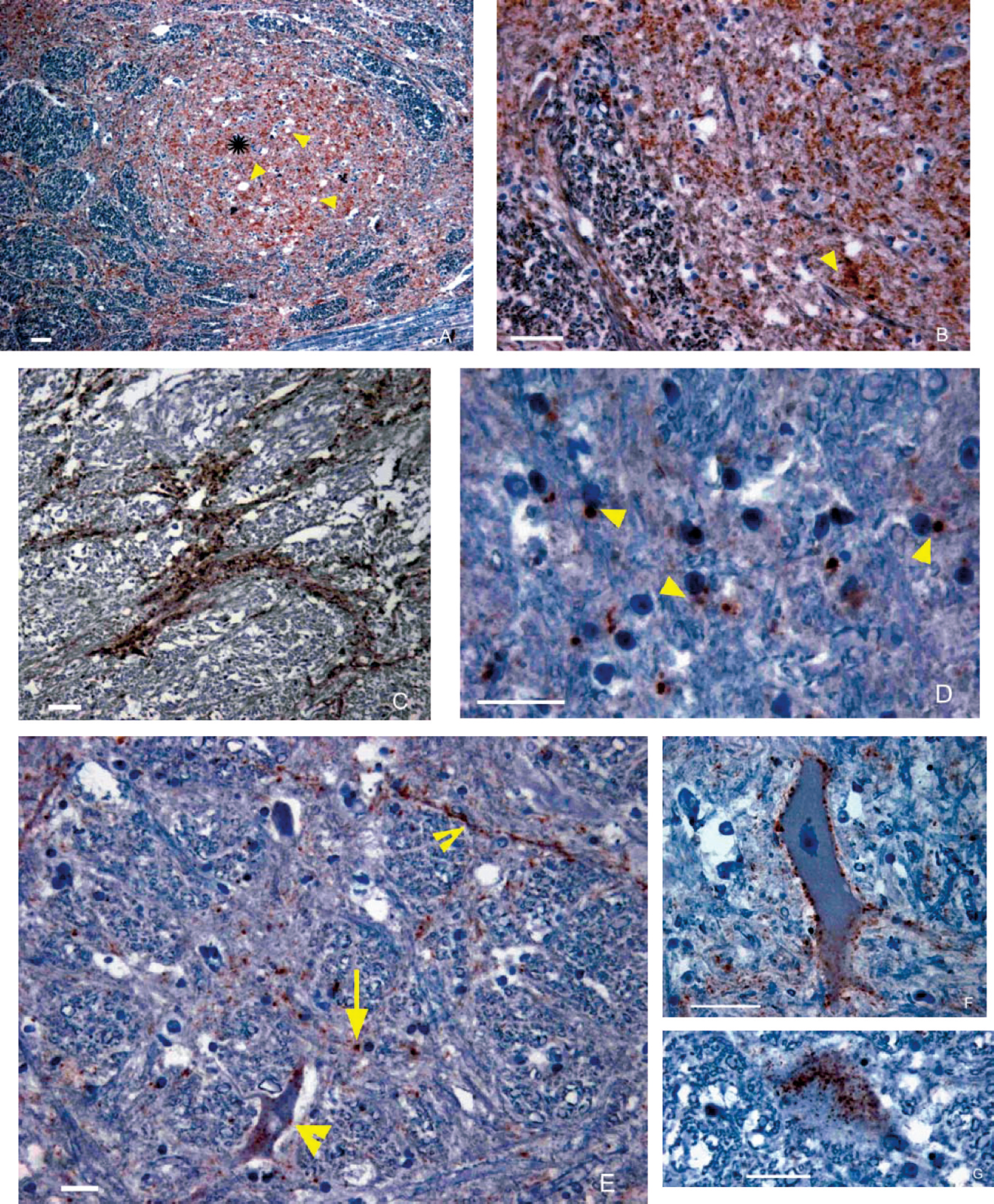
Immunohistochemistry of sections of medulla at the level of the obex of the Swedish atypical H-type BSE case.
− = not observed; + = mild; ++ = moderate; +++ = severe.
These antibodies produce immunostaining in C-BSE cases with severe PrP d deposition only.
observed by IHC confirmed the case as a prion disease. In well-preserved tissues, IHC allows a detailed evaluation of the cellular location and morphological characteristics of the PrP d accumulation. 17 In scrapie, particular patterns of intracellular and extracellular PrP d deposition have been described using IHC and electron microscopy. 22,23 Similar patterns were recognized in this study. Immunohistochemistry using a range of antibodies that bind different peptide regions of the prion protein shows differences in the recognition of intracellular PrP d accumulations between scrapie and BSE. 17 Differences between BSE and scrapie are attributed to variations in intracellular truncation at the N-terminus of PrP d , and are probably a result of differences in the conformation of the molecule. 17,40 In the present study, some information was obtained as to the epitope recognition of the PrP d in the Swedish H-type case by the various antibodies tested. A full description of cellular localization and of the PrP d distribution in the brain could not be performed because of the insufficient quality and quantity of the material. Little information is available about the PrP d deposition in C-BSE with a range of antibodies; therefore, for comparison purposes, this study included a description of the reference C-BSE control cases.
In general, the distribution and morphology of the immunostaining for most antibodies was similar in C-type BSE and the H-type. In both, the MAbs directed to epitopes located C-terminally, F99.97.6.1 and R145 showed marked intracellular (intraneuronal and intramicroglial) PrP d deposition, while antibodies directed to more N-terminal located epitopes, such as P4 and 12B2, did not reveal this type of staining. Nonetheless, some differences were observed in the immunostaining types and/or intensity between the C-type BSE reference sections and the H-type case. Perhaps differences were a result of the poor preservation and loss of tissue structure in the Swedish case. For example, in the H-type case stellate PrP d accumulation could not be identified, and there were antibody-dependent variations with respect to intraneuronal PrP d deposition. In C-type BSE sections, intraneuronal and intramicroglial accumulation was shown with core specific antibodies F89.160.1 and 12F10, but this was not evident in the atypical case. It is possible that the lack of demonstration of intracellular PrP d with these 2 antibodies in the Swedish case may reflect a truncation at a more upstream level than in C-type BSE. On the other hand, intracellular labeling with MAb 12F10 has been reported in an H-type BSE case in Germany. 11 Also intraneuronal PrP d with MAb P4 was reported in an H-type case in a zebu. 35 These different results may be attributed to the use of different protocols or the tissue quality. Alternatively, it is possible that different subtypes of H-type BSE exist, with possible variations in their immunohistochemical features. The cow in this report had signs that yielded no definitive clinical diagnosis, but symptoms of C-type BSE can vary, and other conditions may mask BSE. 25
Little information is available about clinical signs associated to H-type BSE. In this report it was not clear if the clinical signs in the cow were related to the TSE or hypocalcemia.
Experimental intracerebral inoculation of French and German H-type BSE in 2 different bovine transgenic mouse lines (TgXV and tg540) produced neurological disease in all of the mice following incubation periods as well as PrPres molecular features and brain lesions that were distinctly different from C-type BSE. These results suggest atypical H-type cases harbor a prion strain different from classical BSE. 4,11 Other transmission studies, in wild-type C57B/6 mice, classically used for strain typing of BSE, also indicated the isolation of a different strain, 2 and especially showed that the distinct PrPres molecular features, which are similar to those described in this report, 5 were maintained after transmission in mice. Transmission studies of the Swedish case into transgenic mice are currently in progress.
The origin of C-type BSE remains enigmatic, and the discovery of the atypical types of BSE has given rise to further hypotheses. 1,6,8,13,21 It has been proposed that unusual forms of BSE arise as a sporadic disease in cattle, perhaps related to old age, and comparable to sporadic Creutzfeldt-Jakob disease, or possibly to the Nor98 variant of scrapie in sheep and goats. 3 It can also be hypothesized that sporadic BSE has occurred in the past and that it initiated the BSE epidemic by contamination of feed. It should be noted, however, that this would have resulted in a change of the molecular and biological properties of the agent, from H-type to C-type. Genetic studies in the Swedish case do not support the possibility of H-type BSE being caused by a germ-line mutation in the prion gene. This supports previous reports in 3 French H-type cases. 6 To date no cases of C-type BSE have been detected in Sweden. The TSE Swedish case showed similarities with previous descriptions of H-type BSE cases. The screening methods for BSE surveillance in Sweden were able to identify an H-type BSE case. Uncertainties remain concerning the origin and potential infectivity of atypical BSE cases for humans and animals.
Acknowledgements
The authors acknowledge the support of NeuroPrion. They especially thank M. Simmons for the confirmation of the BSE case at the Community Reference Laboratory, VLA, Weybridge, R. Feinstein and M. Elvander, SVA, for their contribution to the initial diagnosis, and E. Westergren and L. Ottander for their technical help with the immunohistochemistry. The authors are grateful to B. Ekberg for his technical help with the illustrations, Linda Terry, VLA, for supplying the MAbs R145, and Sylvie Benestad for her helpful review of the manuscript. Thanks to S. Wallgren and A. Odhelius for providing detailed information on the clinical signs and for their examinations of the cow. The Dutch Ministry of Agriculture, Nature, and Food Quality financially supported this work (project 8041889000) as well as the NeuroPrion task group Eurostrain (EC FOOD-CT-2004-506579).
Footnotes
a.
Bio-Rad, Marnes, la Coquette, Paris, France.
b.
Prionics AG, Schlieren, Switzerland.
c.
Vector Laboratories, Inc., Burlingame, CA.
d.
Available at Central Institute for Animal Disease Control, Lelystad, The Netherlands.
e.
BioPharm Ltd., Darmstadt, Germany.
f.
VMRD Inc., Pullman WA.
g.
Cayman Chemical, Ann Arbor, MI.
h.
Jacques Grassi, CEA, Saclay, Gif sur Yvette, France.
i.
SPI-BIO, Montigny le Bretonneux, France.
j.
Kindly supplied by Dr. Linda Terry, Veterinary Laboratory Agency, Weybridge, UK.