Abstract
This report describes an uncommon case of nonamyloidotic fibrillary glomerulonephritis. A 5-year-old female European cat was presented with nephrotic syndrome. Serum biochemistry and urinalysis revealed a mild increase in cholesterol, low total protein, severe hypoalbuminemia, and high proteinuria with a high protein-to-creatinine ratio. An histologic examination revealed an interstitial nephritis and a diffuse glomerulonephritis, with multifocal thickening of the Bowman's capsule. Transmission electron microscopy showed widespread fibrillary deposits in the glomerular basement membrane and in the mesangium. These fibrils ranged between 18 and 26 nm in diameter and were Congo red negative, which allowed their differentiation from amyloid. Immunohistochemistry demonstrated expression for immunoglobulin M (IgM) and immunoglobulin G (IgG) within the mesangium. Renal deposits of Congo red-negative amyloid-like fibrils have been described in humans, horses, monkeys, and dogs. This is the first report of noncongophilic fibrillary glomerulopathy in a cat.
Glomerular diseases, at least those severe enough to result in the development of nephrotic syndrome, are uncommon in the cat. 13 In human patients, fibrillary glomerulopathies are a specific category of glomerular disease, confirmed only by ultrastructural investigations, which include amyloidosis; light chain deposition diseases; systemic lupus erythematosus; cryoglobulinemia; and diabetic, immunotactoid, collagenofibrotic, and fibronectin glomerulopathies. 5, 7, 8, 13 The first step indicated in the diagnostic algorithm is the separation of amyloidosis from the other fibrillary glomerulopathies by a specific histochemical test, such as the Congo red stain. 7, 8 Renal deposits of Congo red–negative amyloid-like fibrils have rarely been reported in animals such as the horse, 15 monkey, 2 and dog. 3 To the authors' knowledge, in the cat, only amyloid fibril deposits have been observed. 11 In this report, the clinical, biochemical, immunofluorescent, and light and electron microscopic observations of nonamyloidotic fibrillary deposits in feline glomerulonephritis were described.
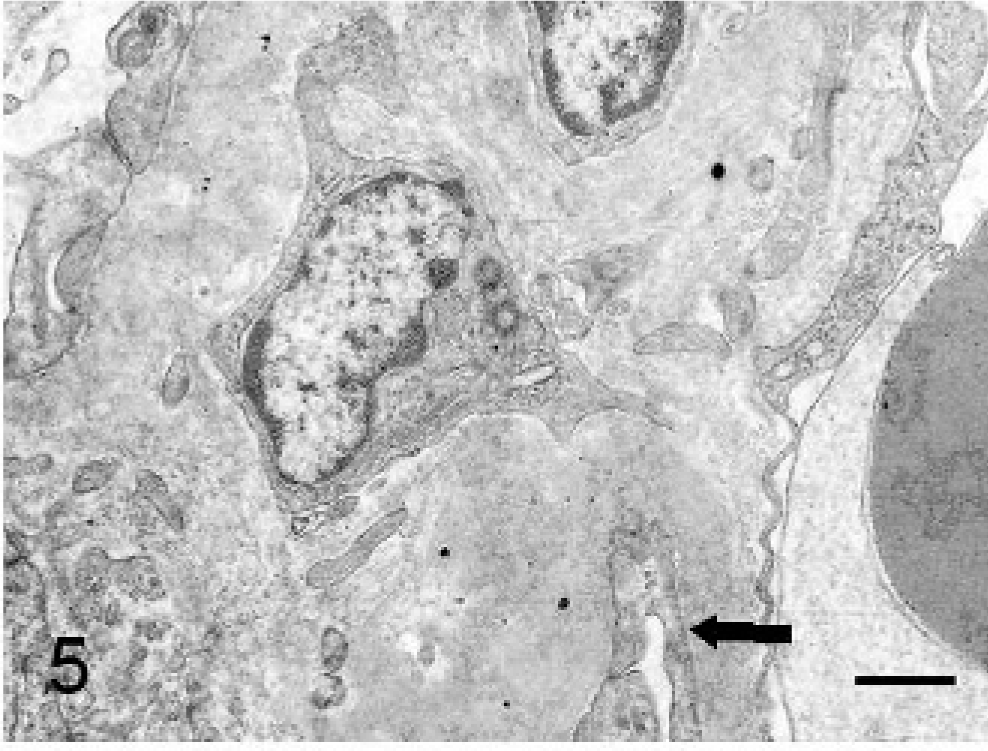
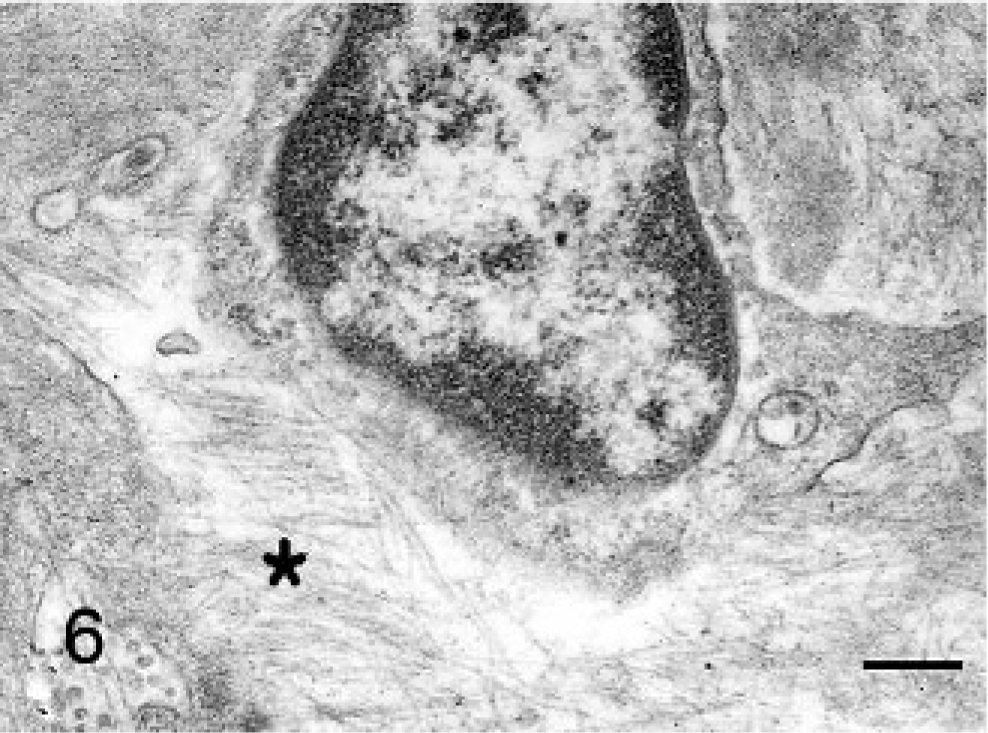
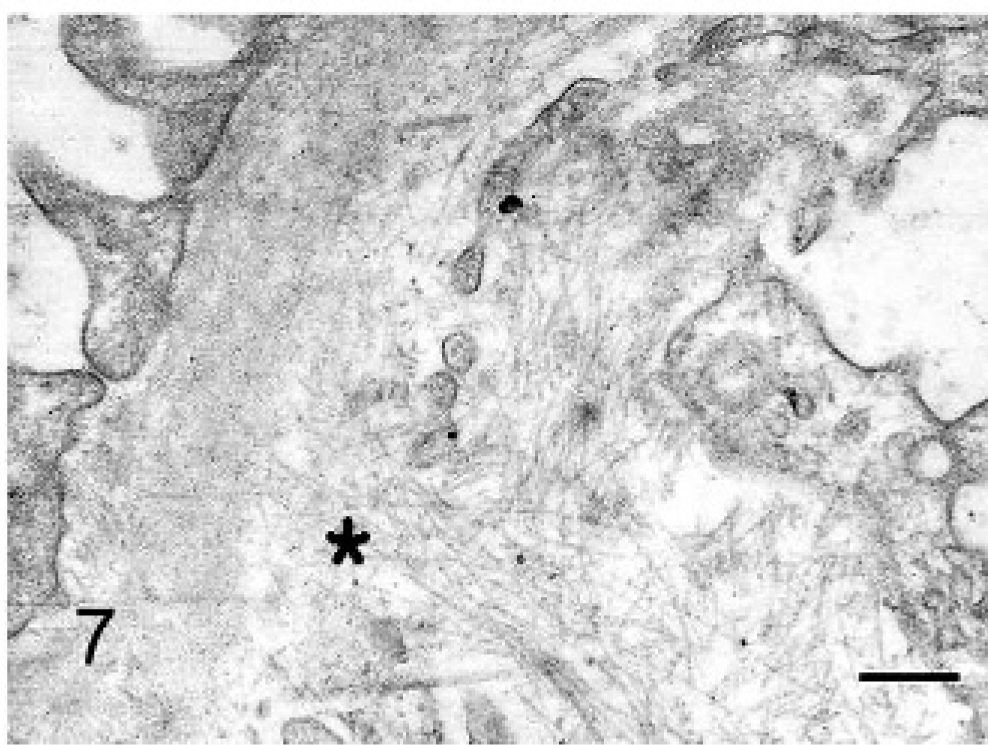
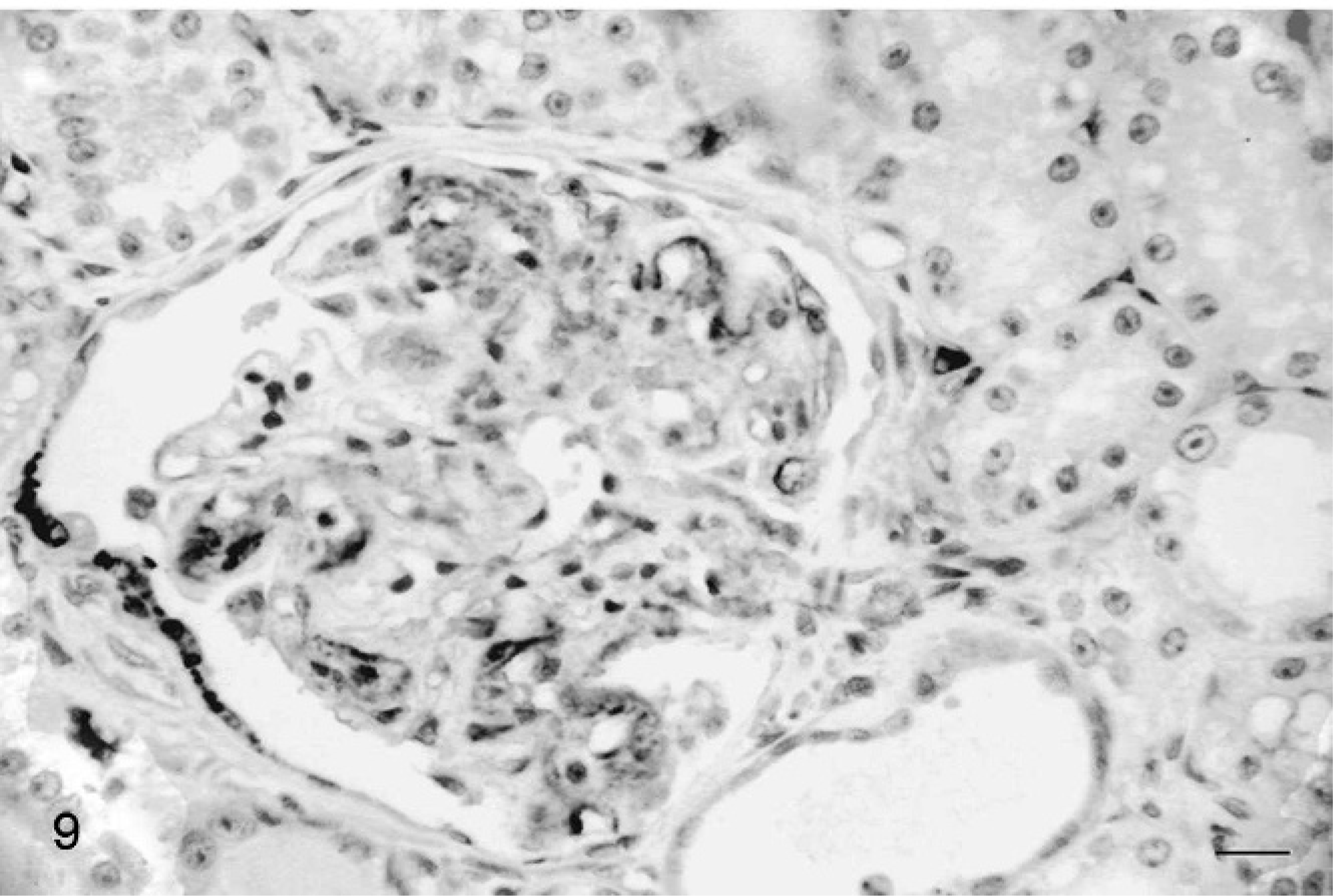
A 5-year-old neutered female European cat was presented with weight loss, weakness, dysorexia, polyuria, polydipsia, and recurrent vomiting and diarrhea, which lasted approximately 1 month. Physical findings included thinness, poor coat, ascites, and subcutaneous edema. Hematology revealed leukocytosis (32.7 × 103/μl; reference range, 6.3–19.6 × 103/μl) with neutrophilia (29.1 × 103/μl; reference range, 3.0–13.4 × 103/μl). Serum biochemistry showed a mild increase in cholesterol (160 mg/dl; reference range, 95–130 mg/dl), high triglycerides (311 mg/dl; reference range, 10–114 mg/dl), low total protein (4.3 g/dl; reference range, 6.0–7.5 g/dl), and hypoalbuminemia (0.65 g/dl; reference range, 2.2–3.2 g/dl). Other hematologic and biochemical values were unremarkable. Feline leukemia and immunodeficiency virus tests were both negative. Urinalysis revealed normal specific gravity (1.050; reference range, 1.035–1.060), high proteinuria (940 mg/dl) with high protein-to-creatinine ratio (UPC) (6.1; reference range, <0.43). Sediment abnormalities included numerous granular cylinders. Urine culture was negative. On the basis of these findings, a nephrotic syndrome was diagnosed. Urine electrophoresis and a percutaneous ultrasound-guided biopsy by using an 18-gauge Tru-cut type needle were performed. Sodium dodecyl sulphate-agarose gel electrophoresis (SDS-AGE) showed low molecular weight proteins (band at 14 kDa, band between 14 and 25 kDa, band at 25 kDa, 2 bands between 25 and 69 kDa), an albumin band, and high molecular weight proteins (2 bands between 69 and 150 kDa, proteins > 150 kDa) (Fig. 1). The biopsy specimen was fixed in 10% neutral buffered formalin for histologic investigation and in 2.5% glutaraldehyde for electron microscopy. The histology specimen was paraffin embedded, sectioned, and stained with hematoxylin and eosin, periodic acid–Schiff (PAS), periodic acid–silver methenamine, acid fuchsin orange G, and Congo red. For ultrastructure investigations, the tissue was post-fixed in 1% osmium tetroxide for 2 hours, dehydrated in acetone, and embedded in Spurr resin. Semithin sections were stained with toluidine blue and ultrathin sections were contrasted with uranyl acetate and lead citrate. On histologic examination, there was a membranous glomerulonephritis, with multifocal thickening of the Bowman's capsule (Fig. 2). The deposits observed in the glomeruli were PAS positive (Fig. 3), but Congo red–stained sections were totally negative. The transmission electron microscopy showed diffuse podocyte foot process effacement and thickening of glomerular basement membrane (GBM), with widespread fibrillary deposits in GBM (subepithelial and subendothelial regions) and in the mesangium. Fibrils were randomly scattered against a lucent background, which expanded the mesangium and the glomerular capillary wall. They were larger than amyloid fibrils, with diameters between 18 and 26 nm. Diffuse effacement and thickening of the pores of the fenestrated endothelial cells were also observed (Figs. 4–7).
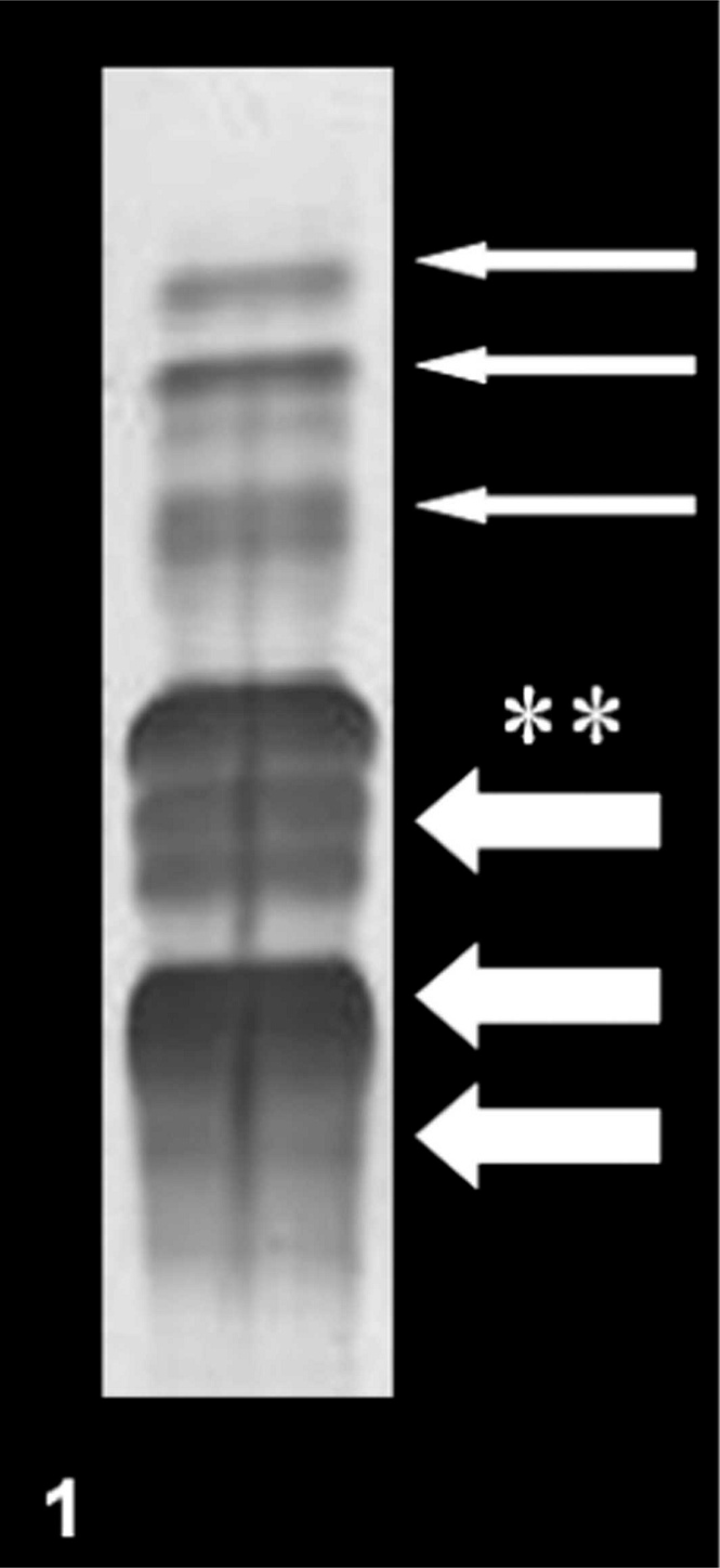
SDS-AGE of urine proteins; cat. Low molecular weight proteins (thin arrows), albumin (asterisk), high molecular weight proteins (thick arrows).
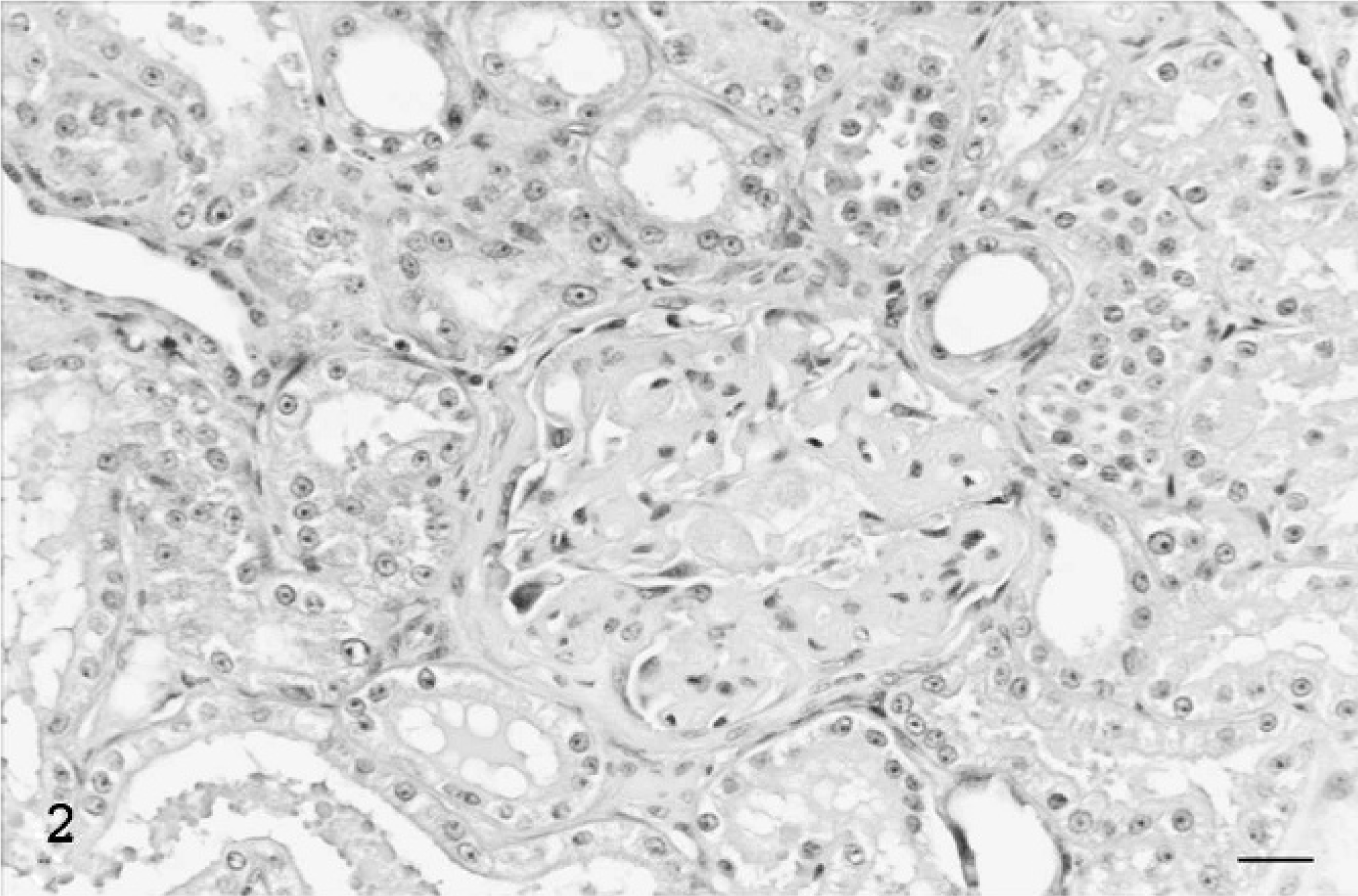
Kidney; cat. Diffuse and severe thickening of the GBM with multifocal involvement of the Bowman's capsule. HE. Bar = 20 μm.
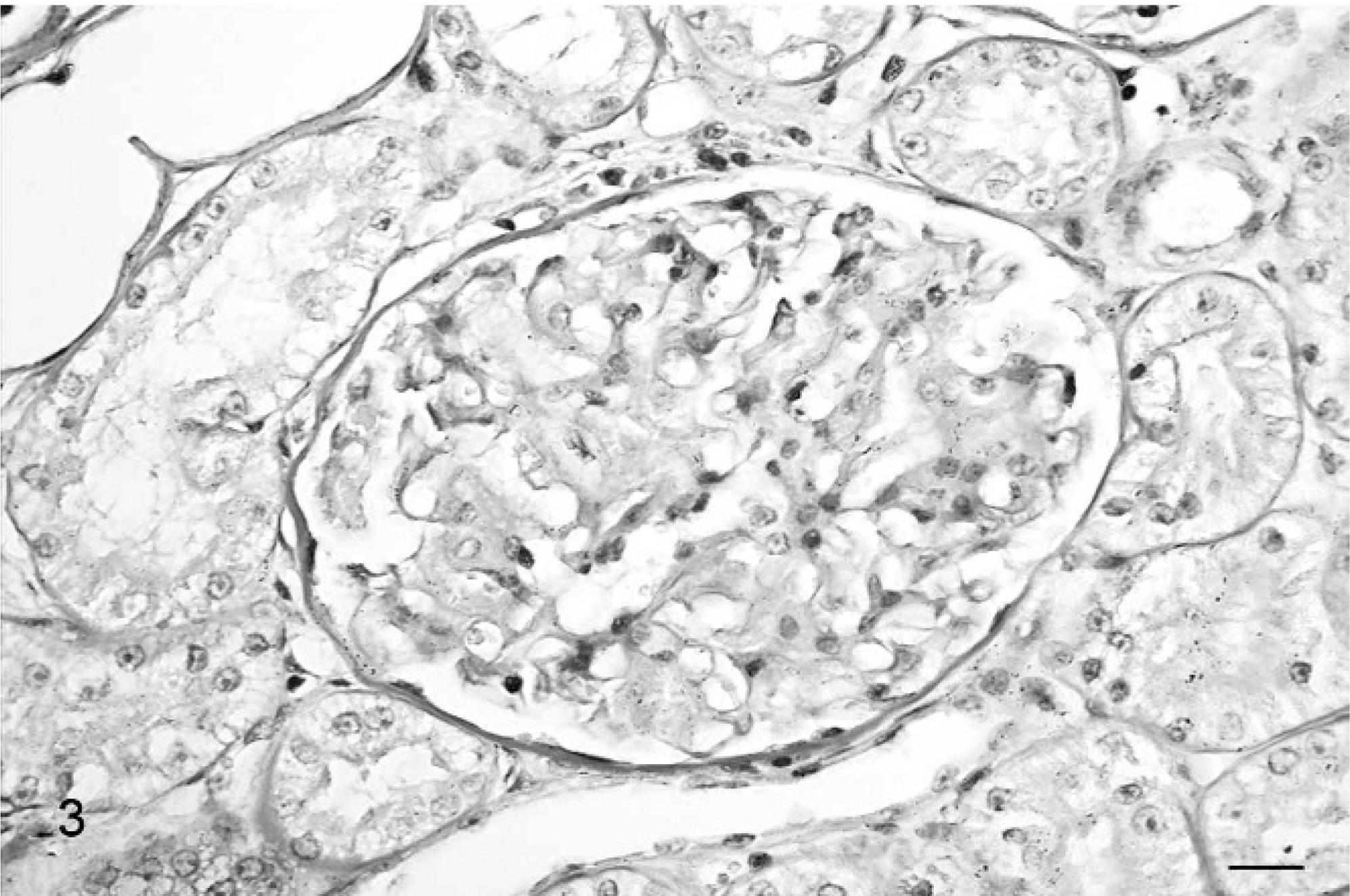
Kidney; cat. Diffuse and severe thickening of the GBM with multifocal involvement of the Bowman's capsule. Periodic acid–Schiff. Bar = 20 μm.
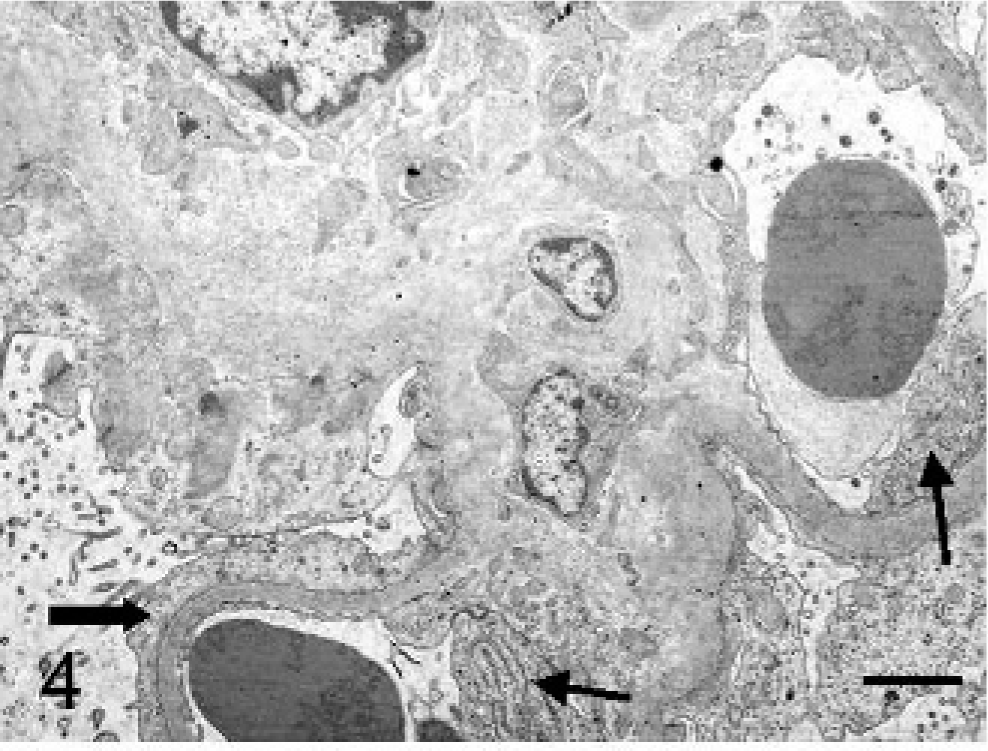
Kidney, ultrastructure; cat. Diffuse podocyte foot process effacement (thick arrow), thickening of GBM with widespread fibrillary (18–26 nm) deposits (asterisk) in GBM, subepithelial and subendothelial (thin arrow) regions and in mesangium. Uranyl acetate and lead citrate. Bars = 1.75 μm
Kidney, ultrastructure; cat. Diffuse podocyte foot process effacement (thick arrow), thickening of GBM with widespread fibrillary (18–26 nm) deposits (asterisk) in GBM, subepithelial and subendothelial (thin arrow) regions and in mesangium. Uranyl acetate and lead citrate. Bars = 0.75 μm.
Kidney, ultrastructure; cat. Diffuse podocyte foot process effacement (thick arrow), thickening of GBM with widespread fibrillary (18–26 nm) deposits (asterisk) in GBM, subepithelial and subendothelial (thin arrow) regions and in mesangium. Uranyl acetate and lead citrate. Bars = 0.26 μm.
Kidney, ultrastructure; cat. Diffuse podocyte foot process effacement (thick arrow), thickening of GBM with widespread fibrillary (18–26 nm) deposits (asterisk) in GBM, subepithelial and subendothelial (thin arrow) regions and in mesangium. Uranyl acetate and lead citrate. Bars = 0.26 μm.
The cat was hospitalized and treated with prednisone, ranitidine, amoxicillin/clavulanic acid, acetylsalicylic acid, and benazepril. Intravenous fluid therapy with lactated Ringer's and colloid solution was administered. Nutritional support was provided via a nasogastric tube, and a fresh plasma transfusion was carried out. The cat's clinical condition improved, and vomiting, diarrhea, subcutaneous edema, and ascites resolved. On day 5, the cat appeared adequately hydrated, stronger, and was able to eat on its own. One month later, the cat developed chronic renal failure, and its clinical condition rapidly worsened. Because of the poor prognosis, the cat was euthanatized at the owner's request.
At necropsy, no macroscopic lesions were observed. Samples of heart, lung, liver, spleen, pancreas, mesenteric lymph nodes, small intestine, colon, and kidney were fixed in 10% buffered formalin, routinely processed, and stained with hematoxylin and eosin. Samples from both kidneys and the liver were also fixed in 2.5% glutaraldehyde and processed for electron microscopy. Immunohistochemical investigations of the kidney samples were performed by using a polyclonal rabbit anti-human IgG (DakoREAL, Ely, UK) diluted 1 : 1600 in antibody diluent (DakoREAL), polyclonal rabbit anti-human IgM (DakoREAL) at a 1 : 2000 dilution ratio, and rabbit anti-human C3c complement (DakoREAL) at 1 : 200. An automated 2-layered, indirect technique was used and ChemMATE Detection Kit (DakoREAL). Endogenous enzymes were blocked with peroxidase and positive activity developed in diaminobenzidine (DakoREAL). All the tissues were pretreated by using a pronase digestion (Sigma Aldrich, Corp. St. Louis MO) and incubated at 37°C for 30 minutes. Canine subcutaneous tissue with diffuse and severe chronic inflammatory reaction was used as positive control. Kidney tissue of a clinically normal cat was used as a negative control. No lesions were observed in the heart, lung, pancreas, or spleen. Severe and diffuse lymphoplasmacytic enteritis associated with diffuse lymphoid hyperplasia, nonspecific reactive lymphadenitis, and severe hepatic steatosis, confirmed by ultrastructural investigations, were the most important lesions observed in other organs. Histologic and ultrastructural investigations on kidney samples confirmed the diagnosis performed on biopsy specimens. Severe diffuse membranous glomerulonephritis associated with the presence of irregularly arranged fibrils located in the mesangial matrix and in the GBM were observed in both kidneys. Interstitial nonsuppurative inflammation and fibrosis, as well as proteineous material in renal tubular lamina, were observed. No fibrillary deposits were noted in other organs. Immunohistochemical studies demonstrated strong expression of IgM and moderate expression of IgG associated with the mesangial thickening in many glomeruli and sometimes the GBM (Figs. 8, 9). No immunoreactivity was observed for C3.
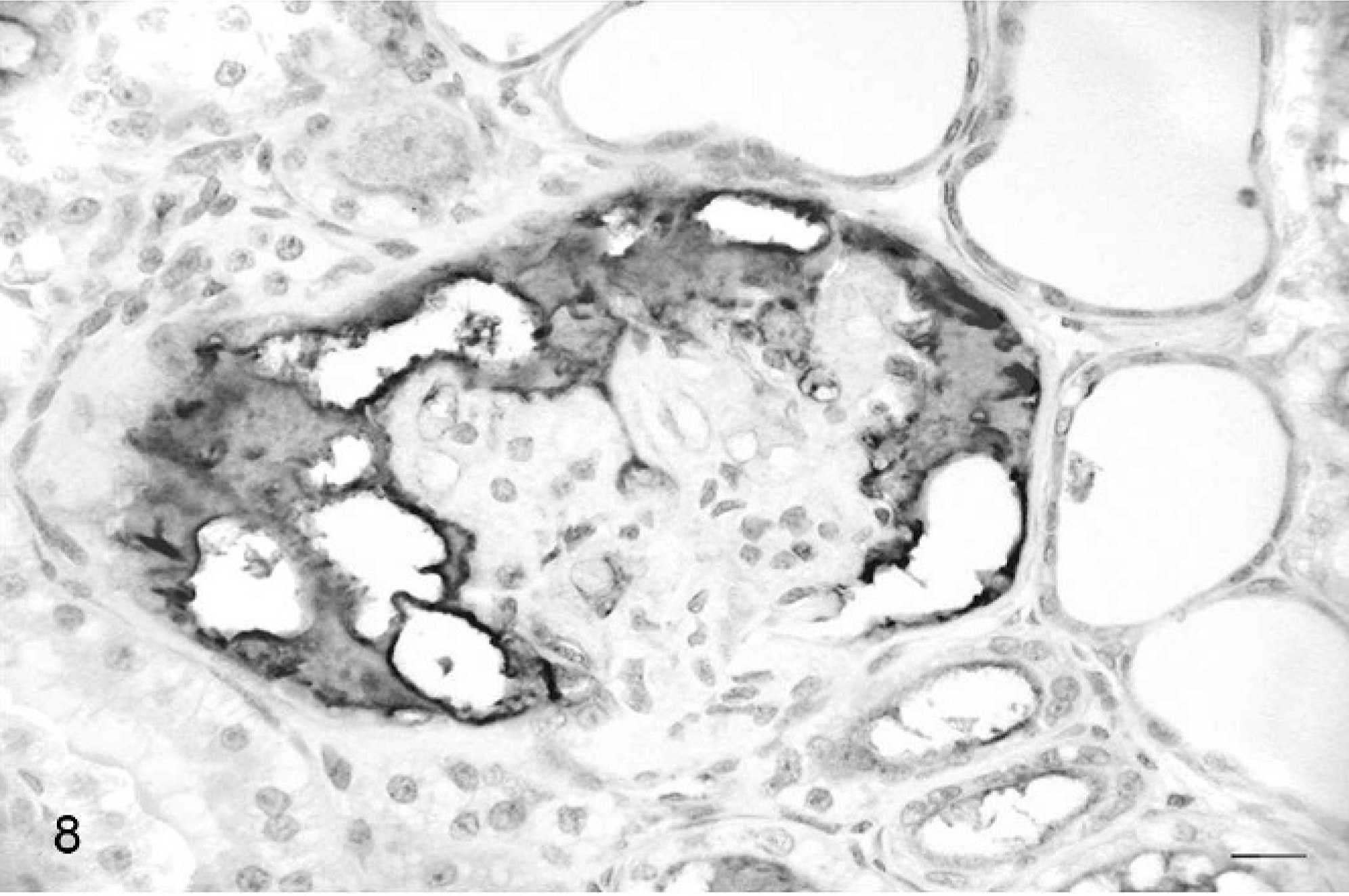
Kidney; cat. Strong immunopositivity for IgM associated with the mesangial thickening. ChemMATE Detection Kit. Bar = 20 μm.
Kidney; cat. Moderate immunopositivity for IgG associated with the mesangial thickening. ChemMATE Detection Kit. Bar = 20 μm.
Renal diseases that cause proteinuria with UPC values ≥1 rarely occur in cats and should prompt a high index of suspicion of glomerular disease. 9, 10, 14 In our case, characterization of proteinuria by using SDS-AGE, in which urinary proteins migrate based on molecular mass, 16 showed low-molecular-weight proteins, albumin, and high-molecular-weight proteins, which suggested tubular-interstitial and glomerular kidney damage, respectively. The ultrastructure of pathologic deposits in the extracellular matrix of the kidneys is rarely fibrillary. 5 In the cat, amyloid fibril deposition was reported mainly in the medulla. 4, 11 Amyloid deposits consist of nonbranching homogeneous microfibrils that measure between 7 and 15 nm in diameter. 8 The tertiary structure of the amyloid molecule results in the diagnostic green birefringence in Congo red–stained sections viewed with polarized light. 8, 10 This property is shared by all types of amyloid and has become the gold standard for the diagnosis of amyloidosis. 8 This case report provides the first description of fibrillary noncongophilic glomerular deposits in the cat. In humans, the diagnostic criteria and distinctions between various forms of fibrillary glomerulopathies are neither clear nor uniform. Once amyloidosis has been excluded, the next step is an immunohistochemical examination that separates the nonamyloid forms of fibrillary glomerulopathy into those with and without immunoglobulin molecules. 8 In our case, renal fibrillary deposits were immunoreactive for IgM and, to a lesser extent, for IgG. The glomerular deposits in this cat are similar to those in the human glomerular deposition disease called fibrillary glomerulonephritis (FGN). In FGN, glomerular deposits are composed of randomly arranged, Congo red–negative fibrils, 15 to 30 nm in diameter. The deposits are usually confined to the glomeruli, although occasional cases show peritubular deposits. The fibrils, which are thicker and more heterogeneous than amyloid fibrils, are most commonly detected in the mesangium or within the GBM with subepithelial or subendothelial distribution. 1, 7, 12 Transmission electron microscopy is necessary to distinguish FGN from other fibrillary glomerulopathies that are immunohistochemically positive for immunoglobulin. Lupus deposits generally display a fingerprint whorled, lamellated substructure, but sometimes have an organized tubulofibrillar substructure composed of parallel linear arrays that resembles those seen in cryoglobulinemia. Cryoprecipitates are morphologically heterogeneous but are usually randomly arranged as closely packed tubules that are up to 1-μm long and from 20 to 30 nm in diameter. The distinction between FGN and immunotactoid glomerulopathy (ITG) is sometimes unclear. 12 The diagnosis of ITG is usually reserved for cases with deposits of large tubules (9 to >50 nm in diameter and generally >30 nm) that are not randomly arranged but are closely packed in parallel arrays. However, the clinical presentations, immunopathology, prognosis, and response to therapy of ITG and FGN are generally the same, so the usefulness of this distinction is questionable. There exists a third group of Congo red–negative deposits characterized by nonimmunoglobulin-derived organized deposits. This group includes collagenofibrotic nephropathy, diabetic glomerulopathy, and fibronectin nephropathy. The diverse biochemical composition is the most important feature to discriminate these diseases from other fibrillar structures. 13
The pathogenesis of FGN has not been elucidated. Fibrils usually stain for IgG and C3, with more variable and weaker positivity for other immunoglobulins. 6, 7 In our case, the immunohistochemical demonstration of IgM and IgG in the glomeruli of the cat suggested a possible immune-mediated mechanism similar to that of human cases. The source of immune stimulation remains obscure, although some human patients with FGN had diverse underlying inflammatory, autoimmune, or neoplastic processes. 12 In this report, the cat had lymphoplasmacytic enteritis as a probable source of chronic inflammation. Prolonged dysorexia, lymphoplasmacytic enteritis, or both could be the cause of hepatic lipidosis. In human medicine, fibrillary glomerulonephritis presents diverse histologic patterns that correlate with clinical outcomes in terms of the percentage of patients who reach end-stage renal disease. Several patients have a transient response to immunosuppressive therapy, but most patients who experience significant renal failure have a poor prognosis. 12 In our case, the cat developed renal failure 1 month after diagnosis.
