Abstract
We identified 20 cases of feline lymphadenopathy that conform to many clinical and histologic manifestations of human Hodgkin's disease. Histologic subtypes encountered included lymphocyte predominance (nine cases), mixed cellularity (nine cases), and nodular sclerosis (two cases). Two cases were not easily classified; fibrous bands were present, but the absence of nodules supported a subclassification of mixed cellularity Hodgkin's disease. Immunohistochemical staining of the tissues using antibodies against the pan T-cell antigen CD3, the human B-lymphocyte antigen 36 (BLA.36), the pan B-lymphocyte and plasma cell marker CD79a, and a myeloid antigen (MAC387) confirmed the phenotypic heterogeneity of the tumor. Classic Reed–Sternberg (RS) cells and mononuclear, multinucleate, and lacunar cell variants did not stain with any of the antibodies used. In contrast, lymphohistiocytic RS variants (L+H cells) reacted positively to BLA.36 and CD79a B-cell markers. Eighteen of 20 affected cats were ≥6 years of age (range, 1-14 years). A sex predilection could not be identified. These findings support the existence of Hodgkin's-like lymphoma in the cat. Proper identification of this disease in the cat will enable further characterization of clinical features and biologic behavior to determine whether there are significant differences in the treatment and prognosis of feline Hodgkin's-like lymphoma compared with non-Hodgkin's lymphoma.
Hodgkin's lymphoma in humans is unique among lymphoid cancers in both its clinical and morphologic presentations. In humans, Hodgkin's disease is described as a slowly progressive, even curable neoplasm of lymphoid tissue that arises in a single node or chain of nodes and spreads via contiguous nodes. Splenic and hepatic involvement succeed nodal disease, and only in the late stages of the disease is the marrow infiltrated. The disease presents histologically as a heterogeneous population of lymphoid and inflammatory cells with a minority of malignant Reed–Sternberg (RS) cells (and/or variants) scattered throughout the tissue. 1,6 The presence of RS cells and their variants is not pathognomonic of the disease but is essential for a histopathologic diagnosis.
Hodgkin's disease has variable manifestations, and consequently subclassification schemes have been developed. 1,6 Currently, of the three subtypes, two are termed classic Hodgkin's disease. The third subtype, lymphocyte predominance Hodgkin's disease (LPHD), is considered a B-cell lymphoma, and there is debate as to whether it should be reclassified as a non-Hodgkin's lymphoma. 11,17 Although still controversial, studies favor a B-lymphocyte origin for the transformed RS cells in classic and lymphocyte predominant Hodgkin's disease. 5,7,8,20
Reports of Hodgkin's-like lymphoma in the veterinary literature encompass nondomestic species such as skunks, killer whales, rats, and mice; 10,18,22,23 cases in companion animals have been described for the horse, dog, and cat, but many are poorly documented or are single cases. 9,14,16,22 Here, we present 20 cases of feline lymphoma that conform to many clinical, histologic, and immunohistochemical criteria for Hodgkin's disease; slight variations in histologic presentation of feline versus human Hodgkin's disease are noted.
Materials and Methods
Selection of cases
Cases were identified on the basis of histopathologic examination of feline lymph node biopsies submitted to the surgical biopsy service of the Laboratory of Pathology and Toxicology, University of Pennsylvania School of Veterinary Medicine for the period 1992–1999. Signalment, clinical signs, outcome, and feline leukemia and immunodeficiency virus status were obtained from information reported on the biopsy report forms and from medical record information supplied by the referring veterinarians when available. The information supplied on biopsy report forms was verified by interview with the referring veterinarian when possible.
All of the biopsies selected were reviewed by one of the authors (M. J. Hendrick) and classified into Hodgkin's subtype according to established criteria. 1 The cases were classified into one of three major subtypes (nodular sclerosis, mixed cellularity, and lymphocyte predominance) based upon the presence or absence of RS cells, lacunar cells, and lymphohistiocytic variants (L+H cells), the composition of the cellular background, and the presence or absence of fibrosis and nodules. RS cells were identified according to accepted morphologic criteria as large binucleate or bilobed cells with mirror image nuclei and large, inclusion-like, “owl's eye” nucleoli. Mononuclear and multinucleate variants of RS cells were defined as cells consistent with RS cells but containing a single nucleus or multiple nuclei, respectively. Lacunar cells, yet another RS variant, were recognized by a folded or multilobated nucleus within a lacunar-like space; the lacuna is produced by cytoplasmic retraction in formalin-fixed sections. The L+H cell variants were defined as cells with vesicular, polylobated nuclei resembling exploded popcorn kernels (popcorn cells).
Mixed cellularity Hodgkin's disease was characterized by a diffuse or vaguely nodular pattern without band-forming sclerosis, although fine interstitial fibrosis could be present. 1,6 In contrast, nodular sclerosis was defined as possessing at least a partially nodular pattern, with fibrous bands circumscribing the nodules. Both mixed cellularity and nodular sclerosis subtypes were characterized by the presence of RS cells (classic or mononuclear or multinucleate variants) and/or lacunar cells. LPHD was defined by the predominance of L+H variants and paucity of classic, mononuclear, or multinucleate RS cells.
Cases with previous diagnoses of Hodgkin's-like lymphoma that did not conform to histologic criteria of Hodgkin's disease were excluded.
Histologic and cytologic preparation
All tissues for histologic evaluation were fixed in 10% neutral buffered formalin and routinely prepared for hematoxylin and eosin (HE) staining. Cytology preparations for cat Nos. 15 and 18 were made from fine-needle aspirates of the affected lymph node; slides were stained with buffered Wright's–Giemsa.
Immunohistochemical staining
Eighteen of the 20 cases representing lymphocyte predominance (cat Nos. 1, 6–9, 11, 16), mixed cellularity (cat Nos. 3–5, 10, 12, 14, 17, 19, 20), and nodular sclerosis (cat Nos. 13, 15) were evaluated immunohistochemically. Paraffin-embedded tissue blocks were not available for cat Nos. 2 and 18.
Immunohistochemical staining of paraffin-embedded 4-µm tissue sections was accomplished with commercially available antibodies (Dako Corporation, Carpinteria, CA) to the pan T-lymphocyte marker CD3, human B-lymphocyte antigen 36 (BLA.36), B-cell antigen receptor complex (CD79a), vimentin, and human myeloid antigen MAC387 using an avidin–biotin–peroxidase method (Zymed, San Francisco, CA) as previously described. 3,19,21 To facilitate labeling with CD3, tissues were pretreated by incubation in 0.1% porcine trypsin. Antigen recovery was accomplished for MAC387, glial fibrillary acid protein (GFAP), and vimentin by exposure to microwaves. Staining with BLA.36 and CD79a did not require pretreatment. Normal tissues containing the antigen of interest were stained in parallel for use as positive controls; feline spleen and lymph node were used as positive controls for the MAC387 and BLA.36, and CD3 antibodies, respectively. As a negative control, tumor sections were stained with an unrelated antibody (GFAP). Staining was visualized with diaminobenzidine; hematoxylin was used as a counterstain.
Statistical analyses
To determine whether there was a sex predilection for Hodgkin's-like lymphoma in these cats, the data were analyzed using a chi-square test statistic with a Yates correction. Feline biopsy submissions to the surgical pathology service of the Laboratory of Pathology and Toxicology served as a control population; 129 hundred twenty nine biopsies with a diagnosis of neoplasia (benign or malignant) were selected from a random 4-week period.
A Pearson's chi-square test was performed with respect to subclassification and sex to assess whether a sex predilection existed within Hodgkin's subtypes.
Results
Clinical summary
Signalment, clinical presentation, treatment, and outcome are summarized in Table 1. Affected cats ranged in age from 1 to 14 years; 18 of 20 cats were ≥6 years of age. There were 12 male and eight female cats. A variety of breeds were represented: domestic shorthair (15/20), Himalayan (1/20), Siamese (1/20), Maine Coon (1/20), and domestic longhair (2/20).
Clinical summary of cats with various forms of Hodgkin's-like lymphoma.
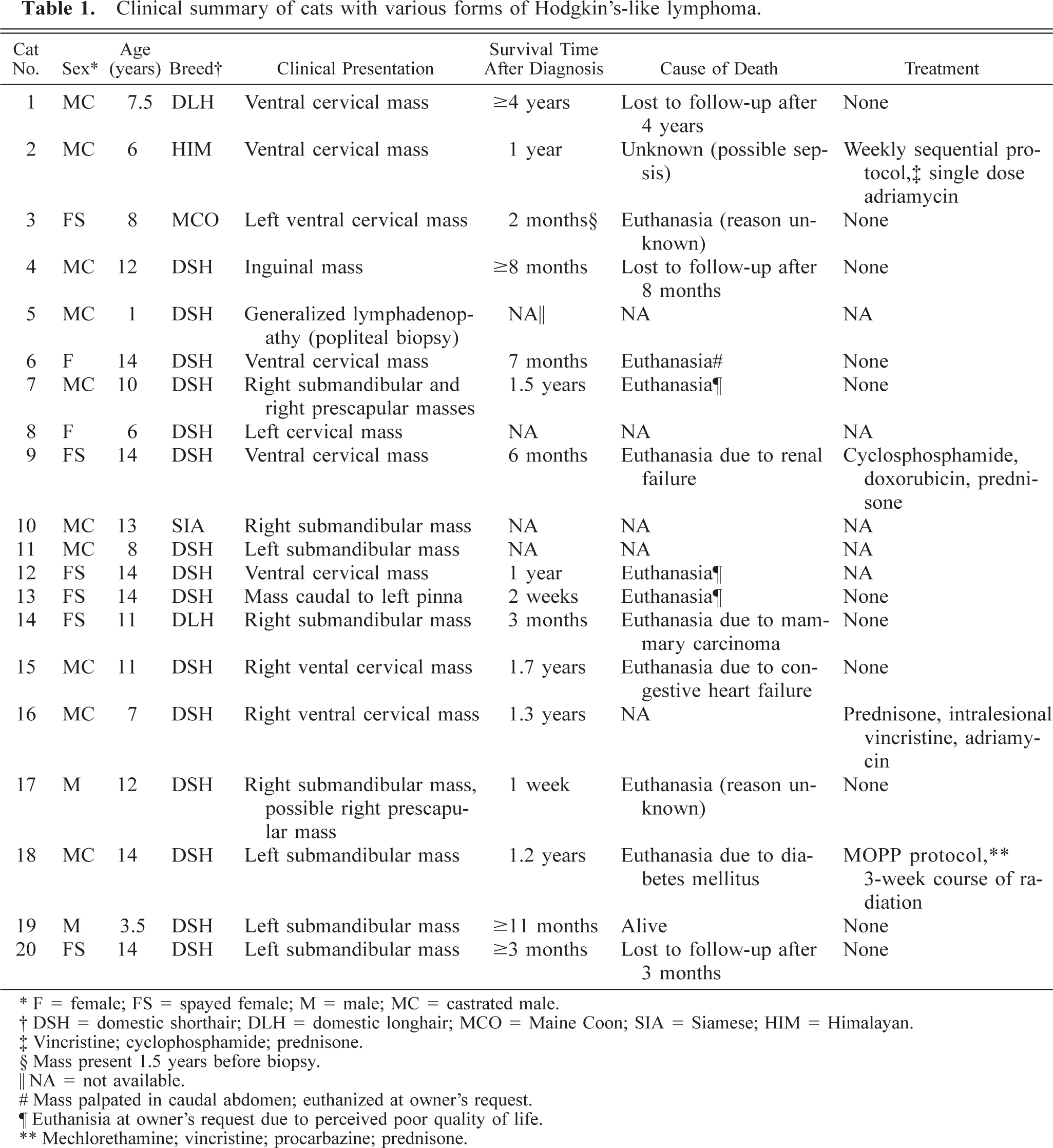
∗ F = female; FS = spayed female; M = male; MC = castrated male.
† DSH = domestic shorthair; DLH = domestic longhair; MCO = Maine Coon; SIA = Siamese; HIM = Himalayan.
‡ Vincristine; cyclophosphamide; prednisone.
§ Mass present 1.5 years before biopsy.
‖ NA = not available.
# Mass palpated in caudal abdomen; euthanized at owner's request.
∥ Euthanisia at owner's request due to perceived poor quality of life.
∗∗ Mechlorethamine; vincristine; procarbazine; prednisone.
Clinical presentation was very distinctive for the majority of cases. Eighteen of 20 cats were presented with unilateral mandibular or cervical lymphadenopathy. In two of these animals, the ipsilateral prescapular lymph node was also thought to be enlarged at the time of presentation, however, histologic evaluation of these nodes was not performed. Of the remaining two cats, one was presented with inguinal (cat No. 4) and the other with generalized peripheral (cat No. 5) lymphadenopathy. For cat No. 5, generalized lymphadenopathy was reported on the biopsy form, but could not be verified in follow-up interviews with the referring veterinarian; only the popliteal lymph node was assessed histologically.
Clinical staging
Additional diagnostics for staging purposes were performed for six cats (Nos. 2–4, 18–20). For cat No. 2, thoracic radiographs performed 5 months after the initial diagnosis showed no abnormalities. Abdominal ultrasound was performed 10 months postdiagnosis to evaluate a liver mass. Evaluation via ultrasound and fine needle aspirate cytology showed the mass to be consistent with a liver cyst. Cat No. 3 had thoracic radiographs taken just prior to the histologic diagnosis. No evidence of thoracic involvement was detected at this time. Cat No. 4 had a bone marrow aspirate evaluated at the time of diagnosis. No abnormalities were reported. Abdominal ultrasound evaluations at 4 and 6 months postdiagnosis were within normal limits. For cat No. 18, a complete staging was performed 6 months after the original diagnosis. At this time, thoracic radiographs, bone marrow aspirate cytology, and abdominal ultrasound examinations were performed. There was no evidence of internal lymphadenopathy, and the liver and spleen appeared within normal limits. The only abnormality detected via ultrasound was diffuse intestinal wall thickening consistent with inflammatory bowel disease, a hypersensitivity reaction, or lymphoma; an intestinal biopsy was not performed. Bone marrow aspirate cytology revealed only a mild plasmacytosis. Thoracic radiographs were also evaluated for cat Nos. 19 and 20 at the time of biopsy. No abnormalities were detected on the radiographs.
Clinical outcome
Eleven out of 20 cats received no chemotherapy after diagnosis of Hodgkin's-like lymphoma (cat Nos. 1, 3, 4, 6, 7, 13–15, 17, 19, 20; Table 1). Survival times ranged from 1 week (cat No. 17) to at least 4 years (cat No. 1). Although cat No. 2 was euthanatized 2 months following diagnosis, the referring veterinarian reported the mass to have been present for 1.5 years prior to biopsy. For five of these cats, death was the result of euthanasia at the owner's request. Two cats (Nos. 14, 15) were euthanatized due to unrelated disease (mammary carcinoma and congestive heart failure, respectively). One cat was still alive 11 months after diagnosis with no recurrence of lymphadenopathy following surgical excision of the node for biopsy.
No information was available regarding treatment or outcome for five of the nine cats (Nos. 5, 8, 10–12). The four remaining cats received chemotherapy (cat Nos. 2, 9, 16, 18; Table 1). Four different protocols were employed in each of the cases.
Feline leukemia/immunodeficiency virus testing
Information pertaining to feline leukemia virus (FeLV) and feline immunodeficiency virus exposure was available for only four of the 20 cats (Nos. 2, 5, 11, 20). Three of these cats were negative for both viruses using an enzyme-linked immunoassay, and cat No. 5 was positive for FeLV antigen.
Sex predilection
There was no significant difference in sex representation between the 20 cases of Hodgkin's-like lymphoma and a randomly selected sample of feline biopsy submissions diagnosed with neoplasia (129 biopsies; P < 0.5). Evaluation of subtypes with the Pearson's chi-square test resulted in no significant difference between subtypes with respect to sex (P = 0.836).
Histologic subclassification
Nine of the cats (Nos. 1, 2, 6, 7–9, 11, 16, 18) were classified as having LPHD. Five of these cats had diffuse involvement of the node, and four (cat Nos. 1, 2, 9, 18) presented as a well-circumscribed, somewhat compressive mass within the node (Fig. 1). This classification was due to the predominance of L+H variants and paucity of classic or mononuclear/multinucleate RS cells (Fig. 2). Two of the nine cats (cat Nos. 9, 11) had some lacunar cells in addition to the L+H cells (Fig. 3). In most cases, the background cell population was composed of small lymphocytes with or without macrophages. Cat No. 16 had some eosinophils and plasma cells, and in cat No. 6, the background consisted mainly of medium-size rather than small lymphocytes.
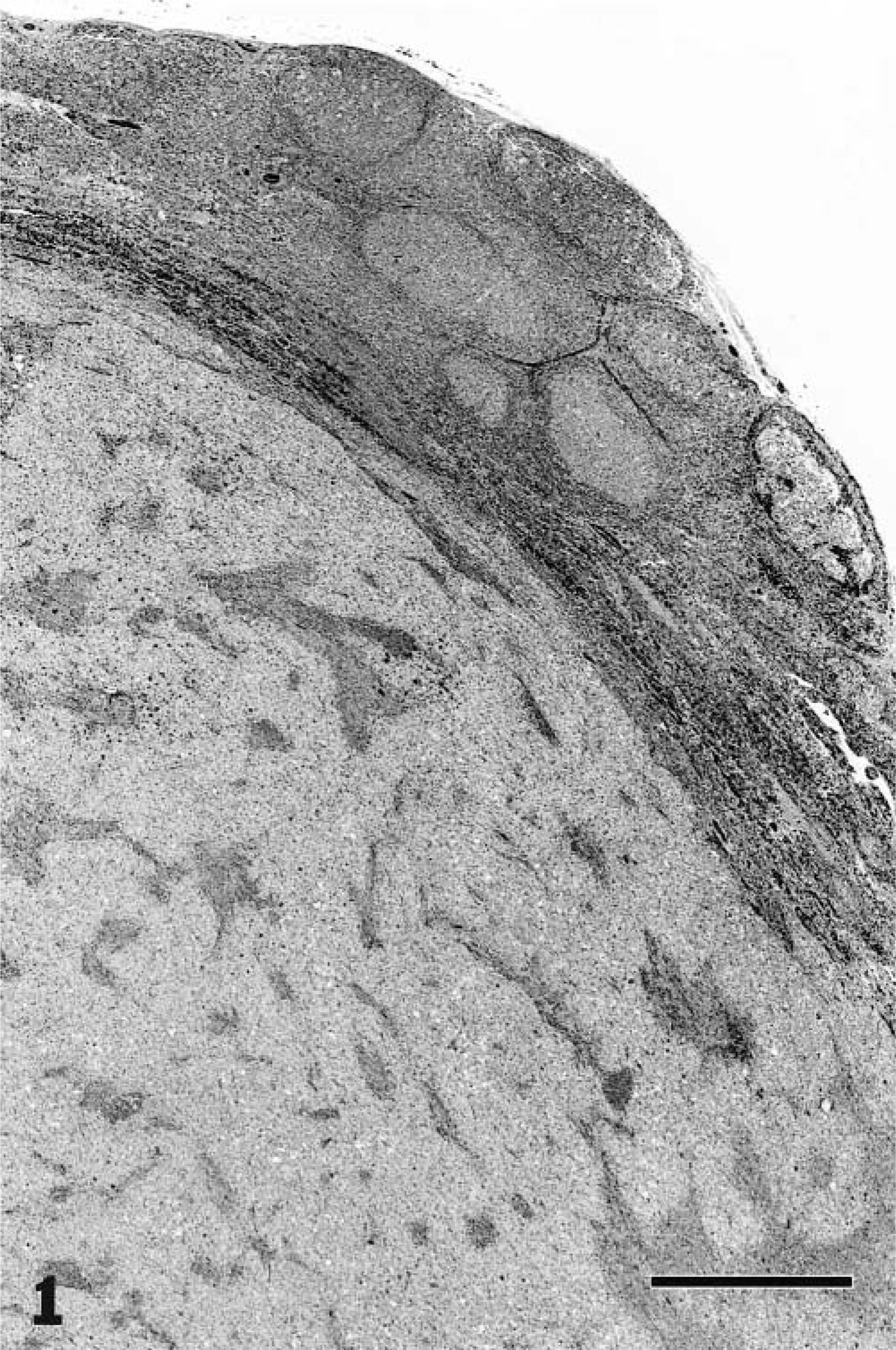
Mandibular lymph node; cat with lymphocyte predominant Hodgkin's disease. Most of the nodal architecture is replaced by a well-circumscribed compressive mass. HE. Bar = 750 µm.
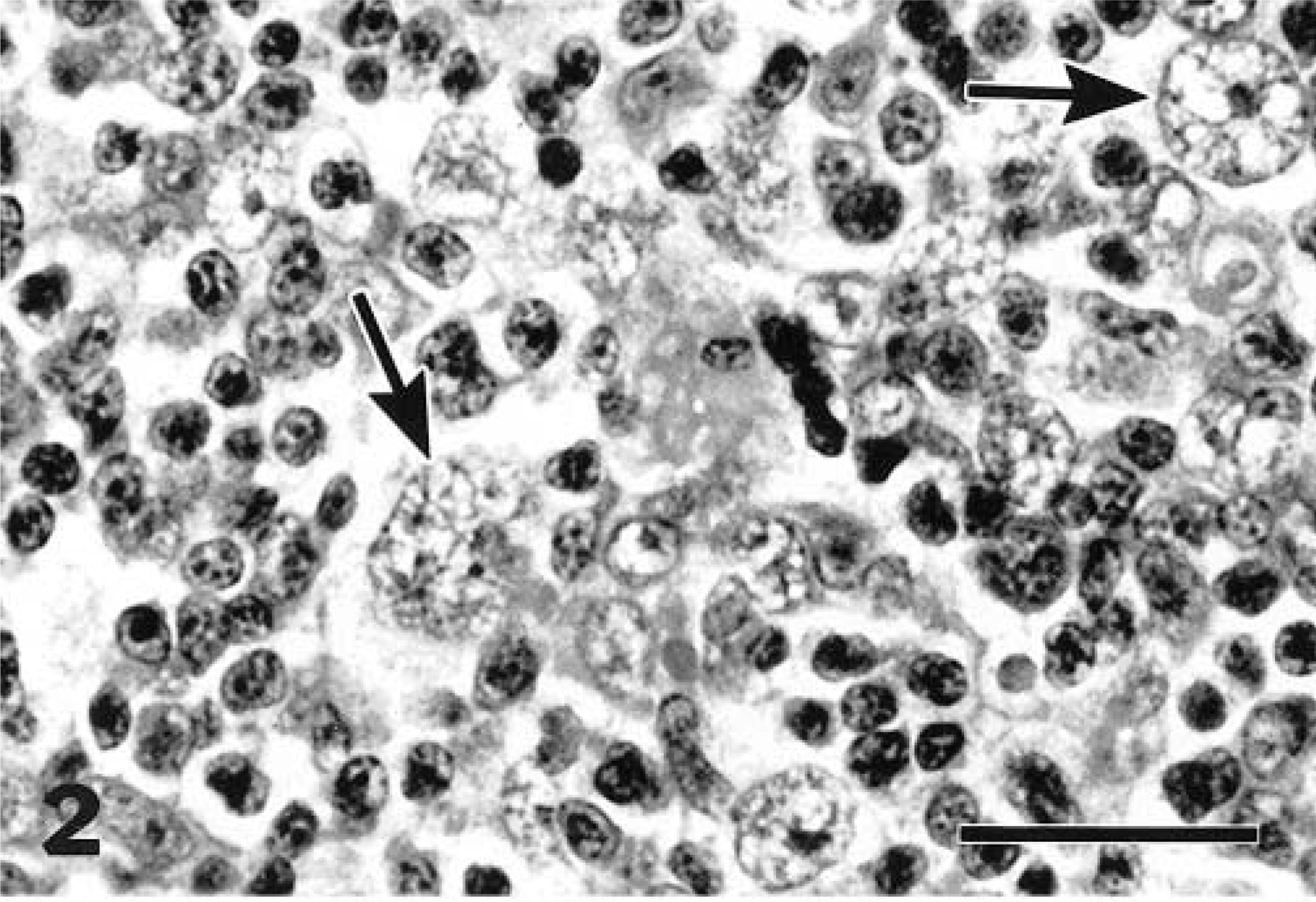
Mandibular lymph node; cat with lymphocyte predominant Hodgkin's disease. This disease form is characterized by the presence of lymphohistiocytic (L+H) cell variants of Reed–Sternberg cells (arrows) on a background of lymphocytes. The unique “popcorn” appearance of the nuclei of the L+H cells is apparent. HE. Bar = 20 µm.
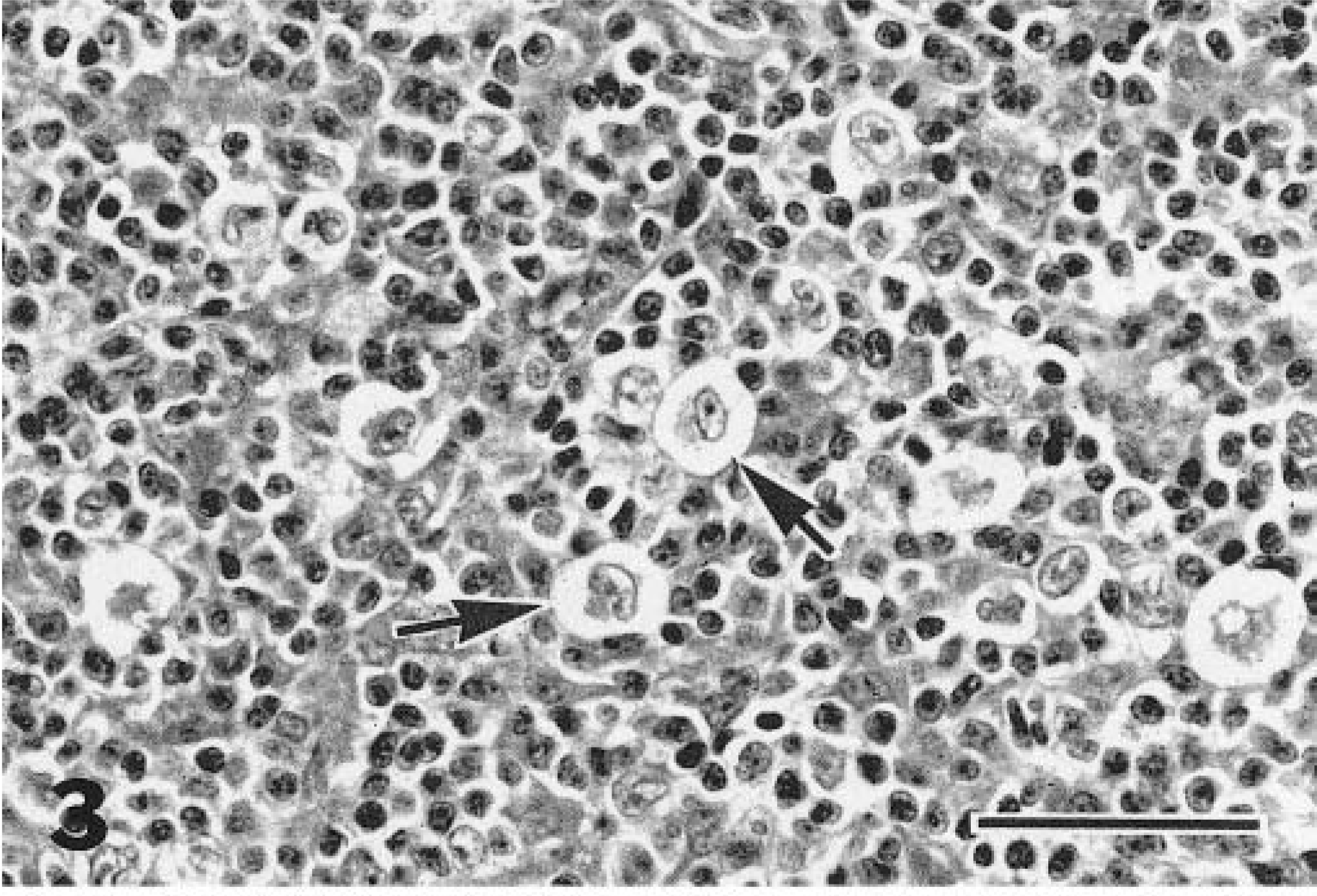
Mandibular lymph node; cat with lymphocyte predominant Hodgkin's disease. Lacunar cells (arrows), another variant of Reed–Sternberg cells, were present in this form as well as in the mixed cellularity and nodular sclerosis forms of feline Hodgkin's-like lymphosarcoma. HE. Bar = 50 µm.
Nine of the cases (cat Nos. 3–5, 10, 12, 14, 17, 19, 20) were classified as mixed cellularity due to the presence of RS cells (mononuclear, binucleate, and/or multinucleate) and/or lacunar cells (Fig. 4). As the subtype implies, the neoplastic RS cells and variants were accompanied by a mixed cellular background of small lymphocytes, plasma cells, eosinophils, neutrophils, and macrophages in varying proportions. The absence of fibrosis excluded seven of these cats from a classification of nodular sclerosis. By the established criteria, cat Nos. 17 and 20 were less easily classified. These cases were consistent with mixed cellularity, but there was moderate fibrosis. The absence of nodules precluded classification as nodular sclerosis, however.
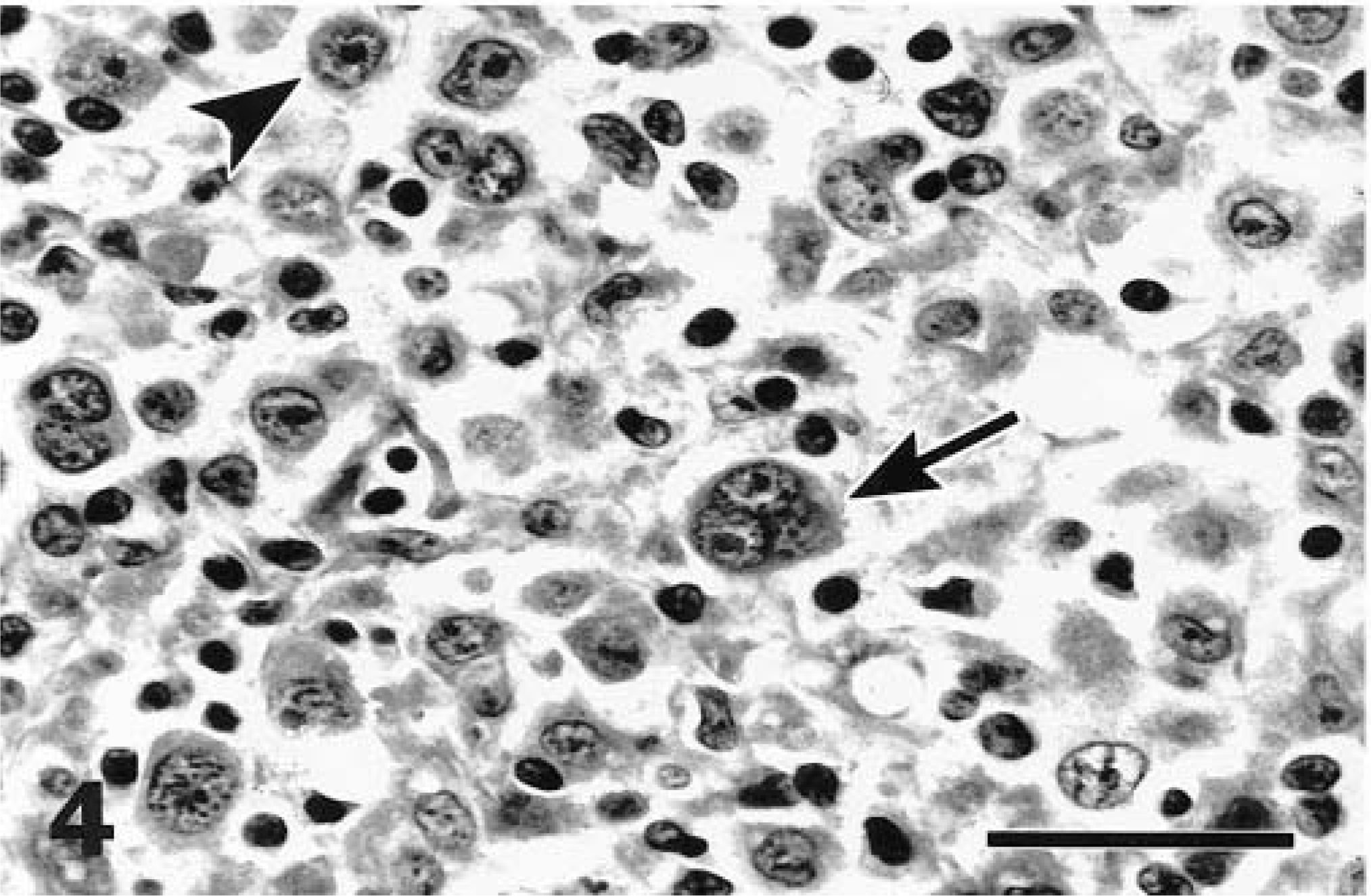
Mandibular lymph node; cat with mixed cellularity Hodgkin's disease. Classic binucleate (arrow) and mononuclear (arrowhead) Reed–Sternberg cells were found in this form and in nodular sclerosis cases. HE. Bar = 29 µm.
In contrast, cat Nos. 13 and 15 had bands of fibrosis as well as some nodules (Figs. 5, 6). The presence of nodules resulted in a classification of nodular sclerosis. The cases were otherwise similar to the mixed cellularity subtypes.
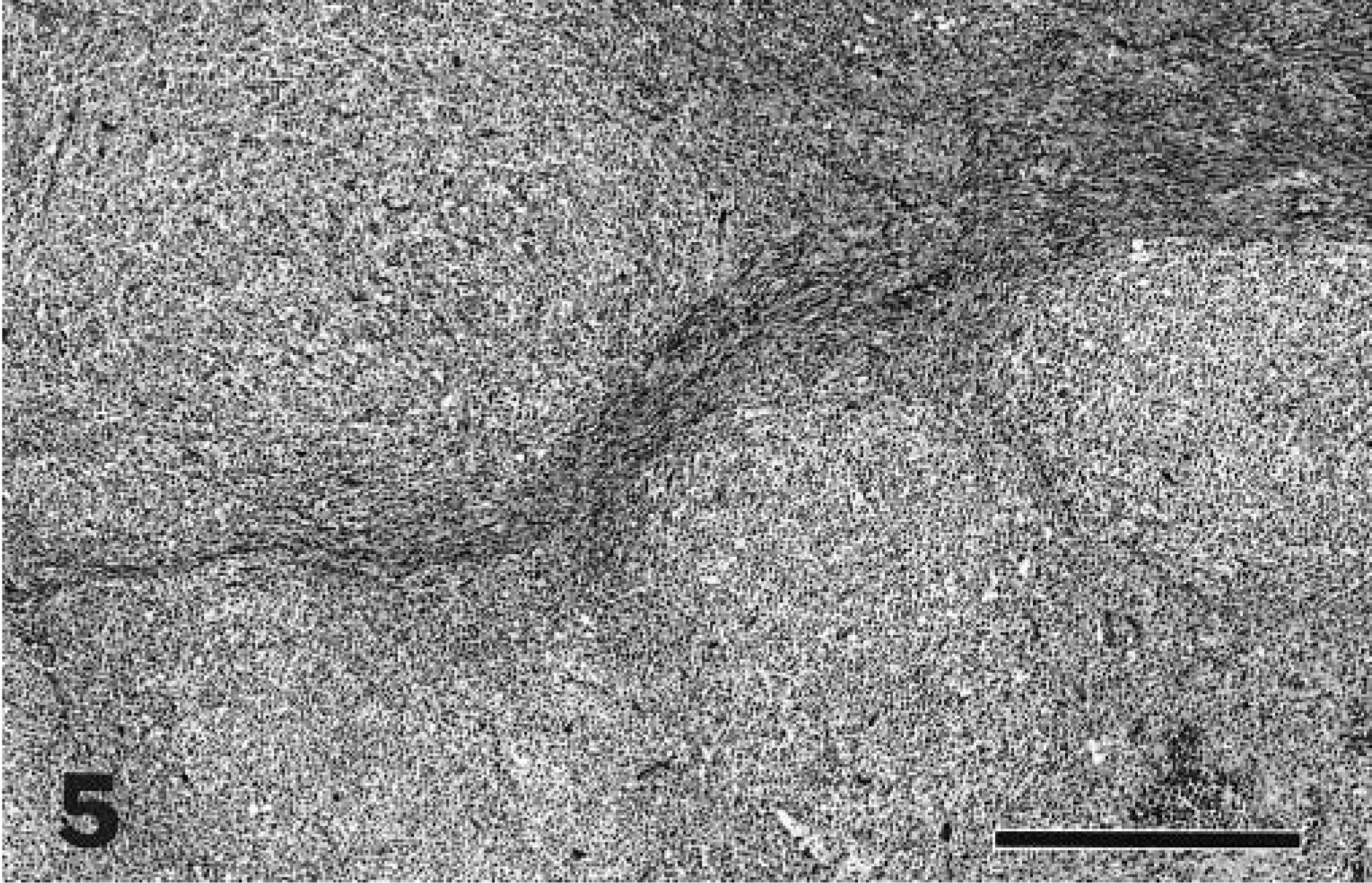
Mandibular lymph node; cat with nodular sclerosis form of Hodgkin's disease. Bridging bands of fibrosis give a multinodular appearance to the neoplasm. HE. Bar = 640 µm.
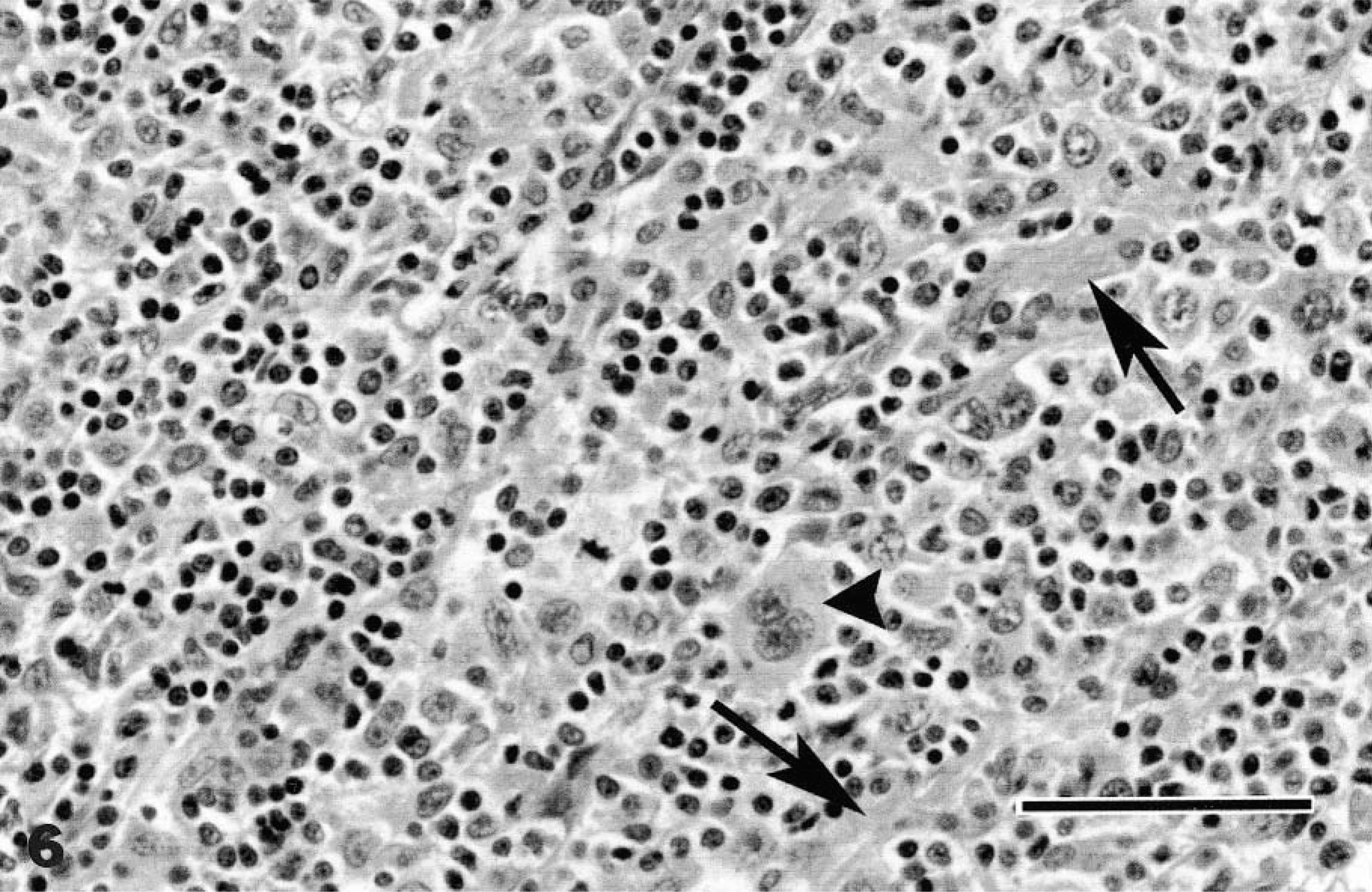
Mandibular lymph node; cat with nodular sclerosing form of Hodgkin's disease. Bands of fibrosis (arrows) are interspersed with a mixed population of small lymphocytes and putative neoplastic cells. Note the trinucleate Reed–Sternberg cell (arrowhead). HE. Bar = 50 µm.
Cytology
Cytology was evaluated for cat Nos. 15 and 18. The results for cat No. 15 were interpreted as reactive lymphoid tissue. Occasional histiocytic cells were noted throughout the smears, but no atypical cells were seen. Cytology for cat No. 18 was assessed on two separate occasions and revealed reactive lymphoid tissue with a moderate to large number of large histiocytic cells dispersed throughout the smears; no atypical cells were detected.
Immunohistochemical staining
Immunohistochemical staining confirmed the pleocellular populations in feline Hodgkin's-like lymphoma. In LPHD, most of the diffuse background small lymphocytes were CD3+. These cells were mixed with fewer small to midsize lymphocytes that were BLA.36+ and CD79a+. In all cats, variable but usually low numbers of L+H cells were BLA.36+ and CD79a+. Mononuclear RS cells, as well as the rare classic binucleate RS cells and lacunar cells, were negative for all markers used in this study. Small numbers of infiltrating neutrophils and histiocytes stained positively with MAC387.
In cases of mixed cellularity, mononuclear and classic RS cells were negative for all markers used, as were the rarely seen lacunar cells. Occasional L+H cells were positively stained with BLA.36, and even fewer L+H cells were stained with CD79a. Background small lymphocytes were CD3+, and midsize lymphocytes were BLA.36+ and CD79a+. Occasional to numerous infiltrating neutrophils and histiocytes were MAC387+.
In cases of nodular sclerosis, there was a mixture of CD3+ small lymphocytes and midsize to larger BLA.36+/CD79a+ lymphocytes throughout the lesion. Lacunar cells and mononuclear and classic RS cells were negative for all markers used. Rare L+H cells were BLA.36+ and CD79a+. Numerous MAC387+ histiocytes and neutrophils were seen in clusters at the periphery of fibrous bands or infiltrating the cellular areas.
Discussion
In general, Hodgkin's disease (HD) in humans differs clinically from non-Hodgkin's lymphoma in several ways. 1,6 In contrast to non-Hodgkin's lymphoma, HD typically involves a single axial group of nodes and spreads contiguously, and extranodal involvement is uncommon. The nature of HD is manifest as a minority of neoplastic cells, approximately 1–5% of cells in affected tissue, 1,17 against a background of lymphocytes, macrophages, neutrophils, and eosinophils. Diagnosis and subclassification of HD therefore necessitates histologic evaluation of whole affected nodes and fine needle aspirate cytology is of limited use.
In humans, the importance of properly diagnosing HD lies in treatment and prognosis. Treatment modalities differ from conventional chemotherapeutic protocols for non-Hodgkin's lymphoma. Dependent upon clinical staging, surgical excision and/or radiation therapy may be primary treatment strategies. 6 In contrast to most types of non-Hodgkin's lymphoma, HD is currently considered a curable neoplastic disease depending upon the stage at which it is diagnosed. 1
The cases in this report conform to many of the histomorphologic criteria of HD as elaborated for humans. The tumors all consisted of a minority population of putative neoplastic cells within a background of small lymphocytes, histiocytes, and granulocytes. Subclassification schemes of human HD are predicated upon the presence of classic RS cells or L+H cells. 1,6 The L+H cells are present only in the LPHD subtype, whereas classic binucleate RS cells and mononuclear or multinucleate variants are found in mixed cellularity HD, and lacunar cells and smaller numbers of classic RS cells or RS nuclear variants are characteristic of nodular sclerosis HD. For the most part, our cases conformed to these criteria. However, the feline tumors diverged from HD subclassification schemes in the following ways: 1) classic RS cells were scarce in cases of feline mixed cellularity and nodular sclerosis HD relative to human HD, 2) L+H cells were found in conjunction with classic RS cells/RS cell variants in mixed cellularity and nodular sclerosis, and 3) lacunar cells were noted in small numbers in LPHD.
The clinical presentation is unusual for feline lymphoma and appears very similar to that of human HD. In 95% (19/20) of cases, the cats were presented with either a single enlarged lymph node (17/20) or two contiguous enlarged lymph nodes (2/20). However, in all except six cats (Nos. 2–4, 18–20), internal/extranodal involvement was only assessed via palpation. No necropsies were available for any of the cats that were euthanatized, precluding accurate assessment of clinical progression. Further work is necessary to better characterize the clinical progression of feline HD to determine whether disease progression and biologic behavior is indeed similar to that of human HD.
In the 20 cats evaluated, differences existed with respect to subtypes and sex predilection as compared with human HD. In humans, the nodular sclerosis subtype comprises 65–75% of HD cases, whereas 90% (18/20) of feline Hodgkin's-like disease cases fell into mixed cellularity (9/20) or lymphocyte predominance (9/20) categories. Unlike our findings for feline HD, males are overrepresented in all human HD subtypes except nodular sclerosis. However, the absence of a sex predilection in these cats is based upon a relatively small sample size.
Except for L+H cell variants, immunohistochemical staining of RS cells and RS variants in human HD has yielded diverse and inconsistent results. 7 The emerging consensus of the identity of the RS cell and variants has largely been derived from genotypic analyses of T-cell receptor (TCR) and immunoglobulin (Ig) gene rearrangements, which suggest a cell of lymphoid origin. 5,7,8,11,20 Immunophenotypic and immunohistochemical studies of L+H cells consistently indicate a B-cell identity, 11 but RS cells and RS variants in the mixed cellularity and nodular sclerosis subtypes are of less certain origin. 4,7,8 Using a limited panel of markers, our findings suggest a B-cell phenotype (BLA.36+ and CD79a+) for the L+H cells in feline LPHD. None of the classic RS cells or mononuclear, multinucleate, or lacunar variants stained with CD79a, BLA.36, CD3, or MAC387 antibodies, a finding analogous to that for human HD.
A primary differential diagnosis that should be considered for HD is T-cell-rich B-cell lymphoma (BCL), a variant of BCL characterized by a minority of neoplastic B-cells (≤10%) in a reactive T-cell background. 2,15 In humans, the importance of identifying T-cell-rich BCL is to distinguish it from HD or T-cell lymphoma, which are treated differently and have different prognoses. In one report, reclassification of 5% of LPHD diagnoses to T-cell-rich BCL was made on the basis of absence of characteristic L+H cells and transformed germinal centers. Furthermore, these patients presented with an aggressive and advanced stage of disease, which is very unusual for LPHD. 2 Misdiagnosis of T-cell-rich BCL as HD occurred in 5% and 26% of cases in two studies, respectively. 2,15 This diagnostic dilemma appears to present confusion in the veterinary literature as well. A series of eight cases of feline lymphadenopathy whose “clinical, histological, and immunophenotypic findings … were identical with those of ‘nodular lymphocyte predominance (lymphocytic and histiocytic/L+H) Hodgkin's disease’ in man” was reported. 3 However, the report was entitled, “T-cell-rich B-cell lymphoma in the cat.” Similarly, the first report of T-cell-rich BCL in a cat has a clinical presentation and histologic features that might suggest HD rather than T-cell-rich BCL. 19 The authors argued that the absence of classic RS cells precludes a diagnosis of HD; however, the description of the histology would suggest an LPHD subtype, characterized by the presence of L+H cells and marked paucity or absence of classic RS cells.
Other differential diagnoses to be considered for peripheral lymphadenopathy in the cat would include distinctive peripheral lymph node hyperplasia of young cats 13 (DPLH) and generalized lymphadenopathy resembling lymphoma. 12 DPLH may be distinguished clinically from HD by the age of onset (≤2 years) and the transient nature of the adenopathy. Histologically, the nodes are characterized by a mixture of histiocytes, lymphocytes, plasma cells, and “immunoblasts” that encroach upon and even efface lymphoid follicles; however, atypical lymphoid cells conforming to RS cells or RS variants have not been reported. Six of nine cats with DPLH tested positively for FeLV. 13 As the name implies, the clinical presentation of generalized lymphadenopathy resembling lymphoma is not consistent with HD; the adenopathy also proves to be transient. Furthermore, histologic evaluation of the affected nodes reveals variable loss of normal architecture by a homogeneous population of lymphoid cells and lack of atypical RS cells or RS variants. 12
In this report, preliminary clinical information suggests that feline Hodgkin's-like lymphoma may be a less aggressive neoplasm than non-Hodgkin's feline lymphoma. Eleven of the cats for which information was available received no treatment. Survival times in untreated cats were at least 7 months and as long as 4 years with the exception of three cats that were euthanatized at the owner's request or as a result of another disease and one cat that was lost to follow-up after 3 months. These findings are similar to those reported previously in which there was no recurrence of disease in three of eight cats after 6 months with no treatment, and one of eight cats survived for 12 months with no treatment other than excision of the affected node. 3 One cat survived for at least 28 months with no treatment other than surgical excision of the affected node. 19 To establish whether Hodgkin's-like lymphoma behaves less aggressively than non-Hodgkin's lymphoma in cats, statistical analysis of survival data is needed and results should be compared with published survival data for feline non-Hodgkin's lymphoma.
Feline Hodgkin's-like lymphoma may prove to be a useful spontaneous animal model of Hodgkin's disease. However, there is much more work to be done to further characterize Hodgkin's-like disease in cats. Prospective studies to elucidate the biologic behavior of the disease are necessary. Further study of the origin and clonality of feline RS cells and RS variants via genotypic and immunohistochemical analyses is warranted.
Footnotes
Acknowledgements
We are grateful to Dr. A. Newton and M. Villar for their work on immunohistochemical staining.
