Abstract
Rhabdoid tumor is a highly aggressive neoplasm of unknown cellular origin in humans, usually occurring in the kidney and central nervous system of infants or children. In older patients, it occurs rarely in other organs, including the skin and soft tissues. A subcutaneous mass in a 13-year-old male mixed-breed cat was composed of nests or sheets of round to polygonal cells with glassy eosinophilic cytoplasmic inclusions. Immunohistochemically, many neoplastic cells expressed vimentin, localized to the cytoplasmic inclusions, whereas the cytoplasm of some neoplastic cells was diffusely positive for neuron-specific enolase, neurofilament, or S-100 protein. By electron microscopy, the cytoplasmic inclusions were found to be composed of aggregates of intermediate filaments. These findings are quite similar to the histologic, immunohistochemical, and ultrastructural features of human rhabdoid tumors and the few rhabdoid tumors reported in animals.
Rhabdoid tumor (RT) is a rare, highly aggressive neoplasm in the kidney of human infants or children. 14 RT was initially described as a “rhabdomyosarcomatoid variant of Wilm's tumor” and has subsequently been defined as a distinct pediatric renal tumor. Histologically, RT is characterized by sheets of highly pleomorphic “rhabdoid” cells with eccentric vesicular nuclei, prominent centrally located nucleoli, and abundant eosinophilic cytoplasm with glassy eosinophilic intracytoplasmic inclusions. These inclusions are composed of paranuclear whorls of intermediate filaments.
Tumors histologically similar to renal RTs have been reported in various anatomic sites, including the skin,
6
soft tissues,
5
and, particularly, the central nervous system, where they are classified as atypical teratoid/rhabdoid tumors (ATRTs).
8
Cytogenetic studies have revealed biallelic inactivation of
The cells of RTs express a variety of antigens of mesenchymal (vimentin), myogenic (α-smooth muscle actin [α-SMA], desmin), epithelial (cytokeratin, epithelial membrane antigen), and neuroectodermal (neuron specific enolase [NSE], S-100 protein, glial fibrillary acidic protein [GFAP], neurofilament) derivation. 5, 8, 14, 15 Because of this immunophenotypic heterogeneity, the cellular origin of RT is still undetermined.
In nonhuman species, spontaneous RTs have been reported only in the gastric wall of an aged orangutan, 9 in the brain of a young dog, 10 and in the orbit of a young horse. 3 The pathologic findings in these cases resembled those in human RTs. Here we report histopathologic and ultrastructural features of a cutaneous RT in a cat.
A 13-year-old male mixed-breed cat developed a small mobile nodule in the dorsal truncal skin that gradually increased in size, became fixed to the skin, and appeared painful. After 4 months, the mass was surgically removed at the Veterinary Teaching Hospital of Osaka Prefecture University and sent to the laboratory for histopathologic examination. Grossly, it was a 4 × 2 × 1.5 cm, firm, whitish nodule and was confined to the subcutis (Fig. 1). No chemotherapy was given after surgery. The animal died of renal failure 11 months after surgical resection with elevated blood urea nitrogen (81 mg/dl) and serum creatinine (9.9 mg/dl). Although necropsy was not performed, no recurrence of the tumor was apparent at the surgical site and no metastatic lesion was detected radiographically.
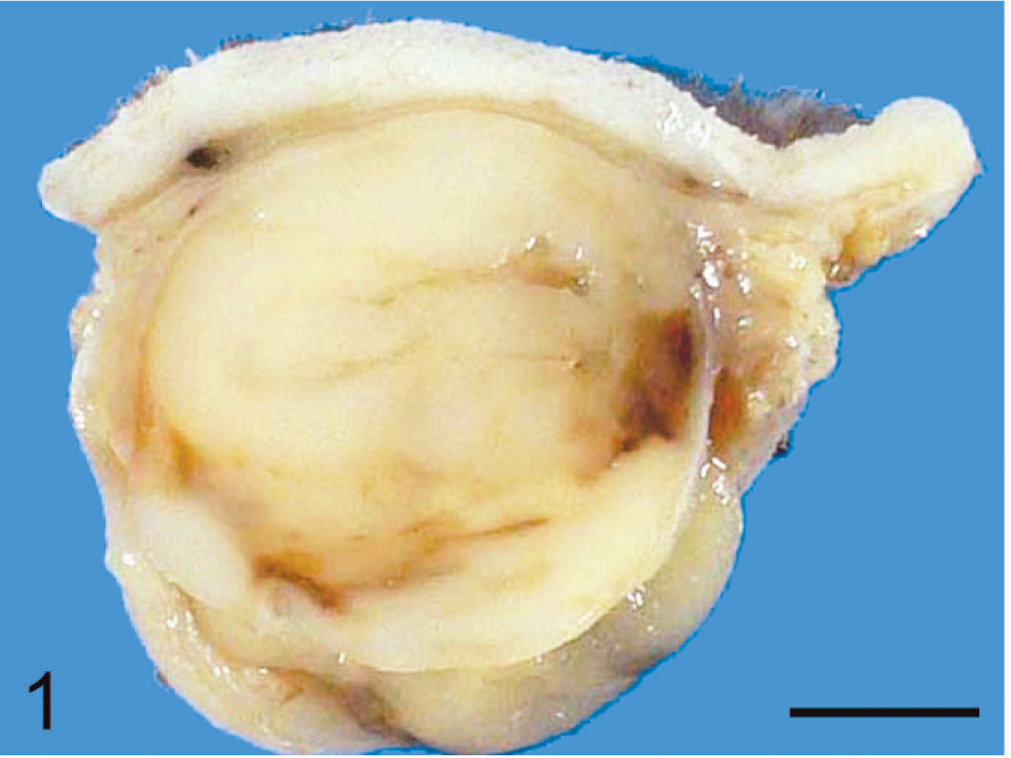
Subcutaneous rhabdoid tumor; cat. The tumor is a 4 × 2 × 1.5 cm, firm, whitish nodule, well demarcated from surrounding tissue but not clearly encapsulated. Bar = 0.5 cm.
The surgical specimen was fixed in 10% neutral-buffered formalin, embedded in paraffin, cut at 3 µm, and stained with hematoxylin and eosin (HE). For immunohistochemistry, the following primary antibodies were used: mouse anti-vimentin (1 : 400, clone V9, Dako, Carpinteria, CA, USA), mouse anti-pan-cytokeratin (ready-to-use, clone MNF116, Dako), mouse anti-desmin (1 : 200, clone D33, Dako), mouse anti-α-SMA (1 : 100, clone 1A4, Dako), mouse anti-NSE (ready-to-use, clone BBS/NC/VI-H14, Dako), mouse anti-Melan-A (1 : 200, clone A103, Dako), rabbit anti-GFAP (1 : 1,000, Dako), rabbit anti-S-100 protein (1 : 1,000, Dako), and rabbit anti-neurofilament (200 kDa; 1 : 1,000, Serotec, Kidlington, Oxford, UK). Bound antibodies were detected with horseradish peroxidase-conjugated anti-mouse or anti-rabbit secondary antibody (Histofine Simplestain MAX-PO; Nichirei, Tokyo, Japan) and 3,3′-diaminobenzidine as chromogen (Vector Laboratories, Burlingame, CA, USA). Adjacent cutaneous tissue provided an internal positive control for vimentin, cytokeratin, desmin, α-SMA, NSE, GFAP, S-100 protein, and neurofilament. A feline cutaneous melanoma was used as a positive control for melan-A immunohistochemistry. For ultrastructural examination, formalin-fixed tissue was postfixed in osmium tetroxide, dehydrated through a graded ethanol series, and embedded in epoxy resin. Ultrathin sections were stained with uranyl acetate and lead citrate and examined with a Hitachi H-7500 electron microscope (Hitachi, Tokyo, Japan).
Histologically, the mass was a well-demarcated, partially encapsulated neoplasm in the subcutis. Neoplastic tissue was arranged in nests or sheets of round to polygonal cells, 5 to 18 µm in diameter, with abundant eosinophilic cytoplasm, ovoid vesicular nuclei, and prominent nucleoli (Fig. 2). Many neoplastic cells contained glassy, lightly eosinophilic cytoplasmic inclusions. Nests of neoplastic cells were surrounded by a fine, fibrovascular stroma. Focally, spindle-shaped neoplastic cells were arranged in interlacing fascicles. The neoplastic cells were pleomorphic with scattered binucleated or multinucleated cells. The mitotic rate was 1–2 per 400× field. There were extensive necrosis and hemorrhage within the tumor. Surrounding tissue was compressed by the tumor, and the overlying skin was ulcerated.
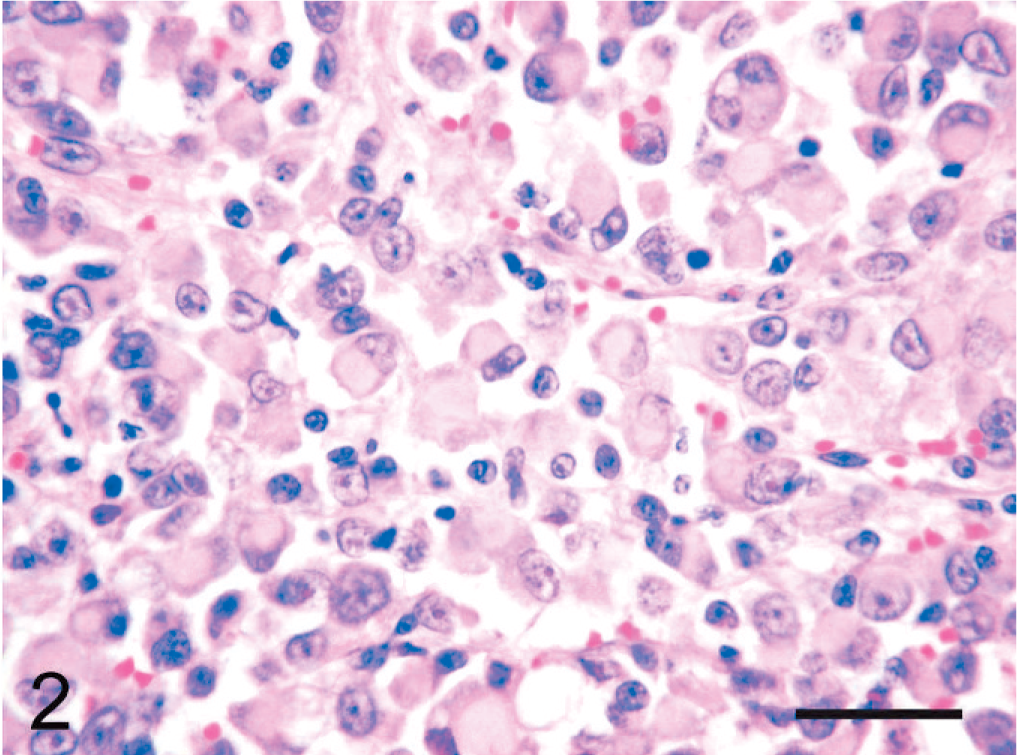
Subcutaneous rhabdoid tumor; cat. Many neoplastic cells contain glassy, lightly eosinophilic cytoplasmic inclusions. HE. Bar = 30 µm.
Immunohistochemically, the cytoplasmic inclusions of numerous neoplastic cells were strongly positive for vimentin (Fig. 3). The cytoplasm of many neoplastic cells with cytoplasmic inclusions was diffusely positive for NSE (Fig. 4), whereas the cytoplasm of some neoplastic cells without inclusions was diffusely positive for neurofilament (Fig. 5). The neoplastic cells that expressed neurofilament were angular; some had cytoplasmic processes. Less commonly, the cytoplasm and nucleus of some neoplastic cells was immunoreactive for S-100 protein (Fig. 6). The neoplastic cells did not react with antibodies against cytokeratin, desmin, α-SMA, melan-A, or GFAP. Ultrastructurally, the cytoplasmic inclusions were composed of aggregates of intermediate filaments (approximately 10 nm in diameter), which entrapped cytoplasmic organelles such as mitochondria and rough endoplasmic reticulum (Fig. 7). No melanosomes were detected in the cytoplasm of the neoplastic cells.
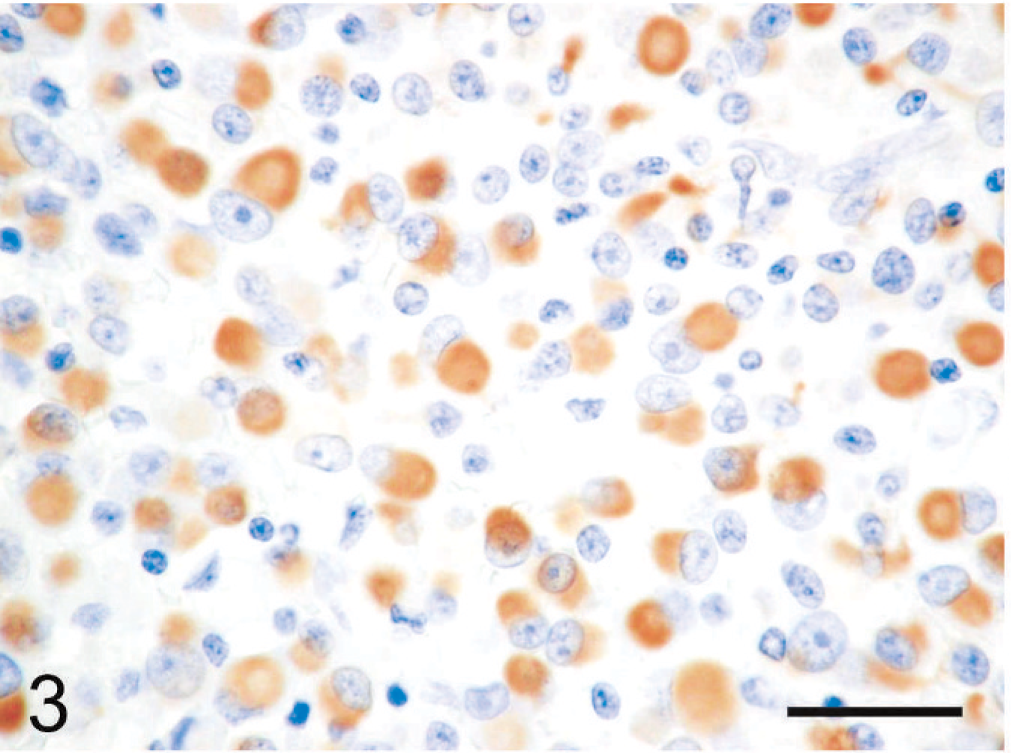
Subcutaneous rhabdoid tumor; cat. The cytoplasmic inclusions of many neoplastic cells are strongly positive for vimentin. Immunoperoxidase method for vimentin, hematoxylin counterstain. Bar = 30 µm.
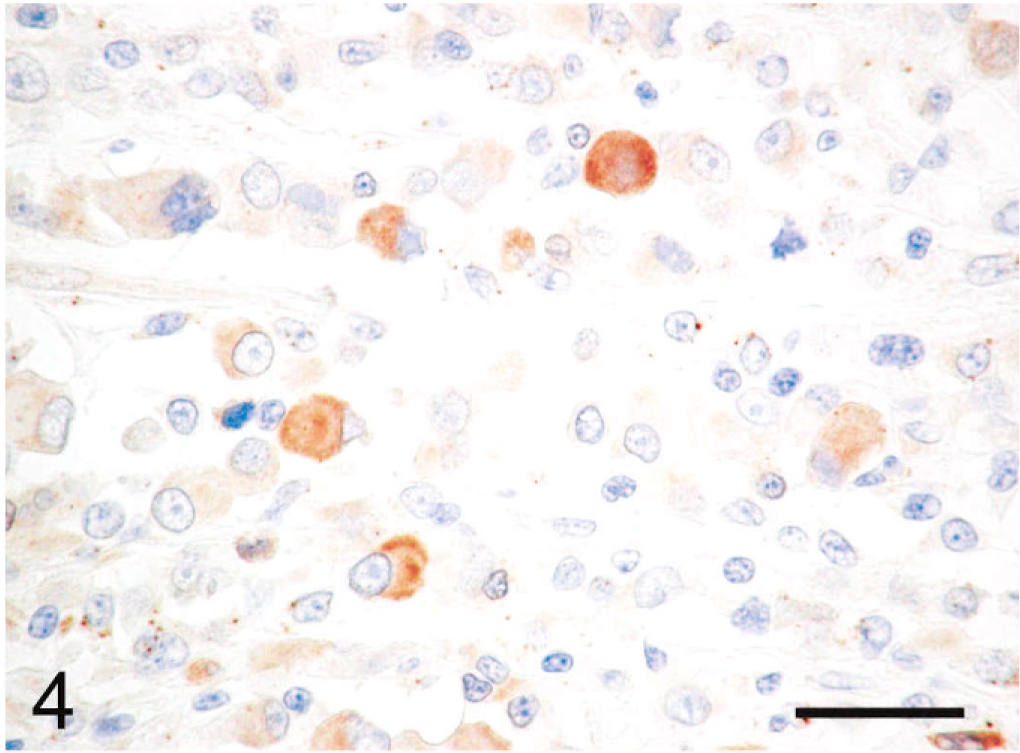
Subcutaneous rhabdoid tumor; cat. The cytoplasm of relatively large neoplastic cells with cytoplasmic inclusions is diffusely positive for NSE. Immunoperoxidase method for NSE, hematoxylin counterstain. Bar = 30 µm.
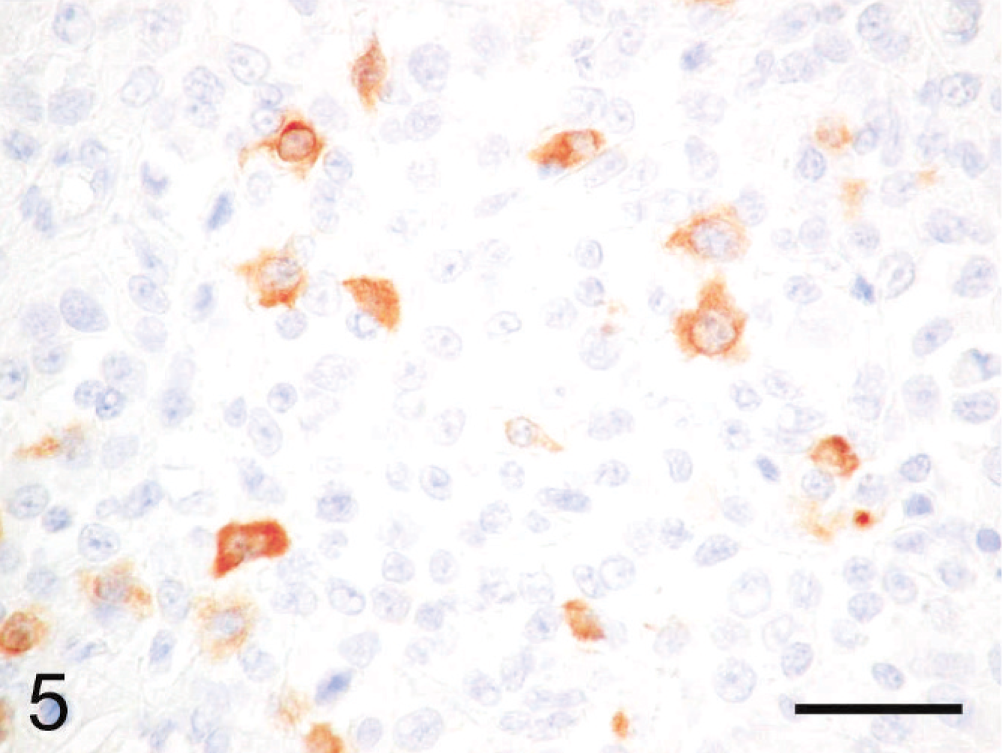
Subcutaneous rhabdoid tumor; cat. The cytoplasm of some neoplastic cells without cytoplasmic inclusions is diffusely positive for neurofilament. The neurofilament-positive cells are angular; some have cytoplasmic processes. Immunoperoxidase method for neurofilament, hematoxylin counterstain. Bar = 30 µm.
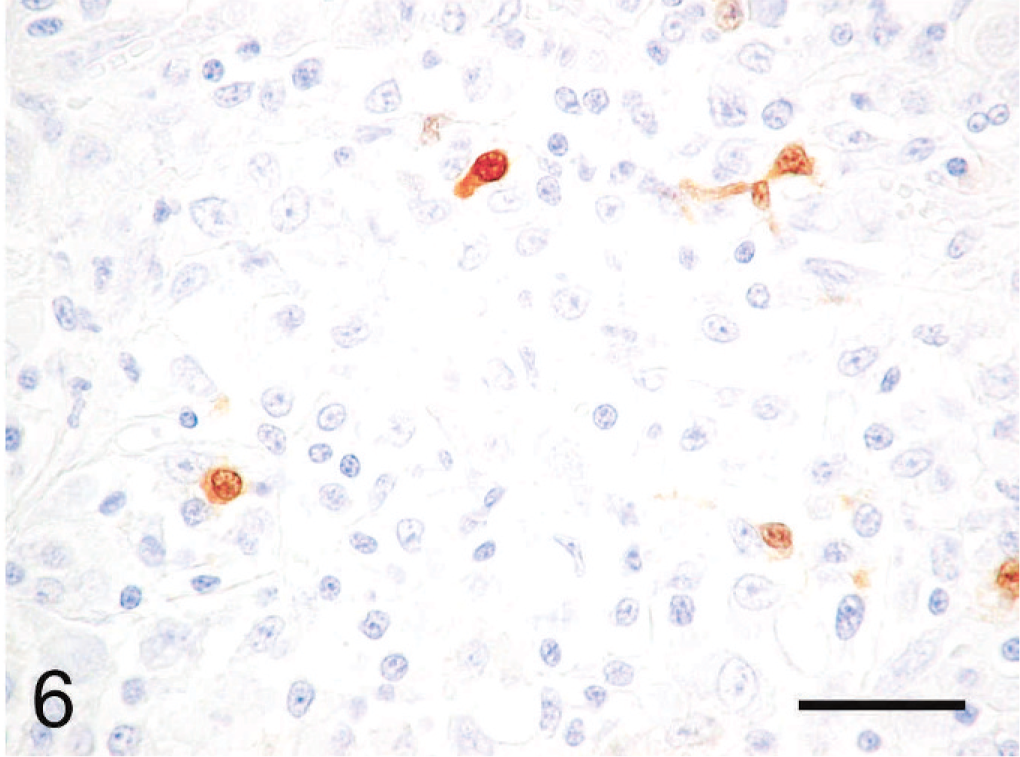
Subcutaneous rhabdoid tumor; cat. The cytoplasm and nucleus of some small neoplastic cells are positive for S-100 protein. Immunoperoxidase method for S-100 protein, hematoxylin counterstain. Bar = 30 µm.
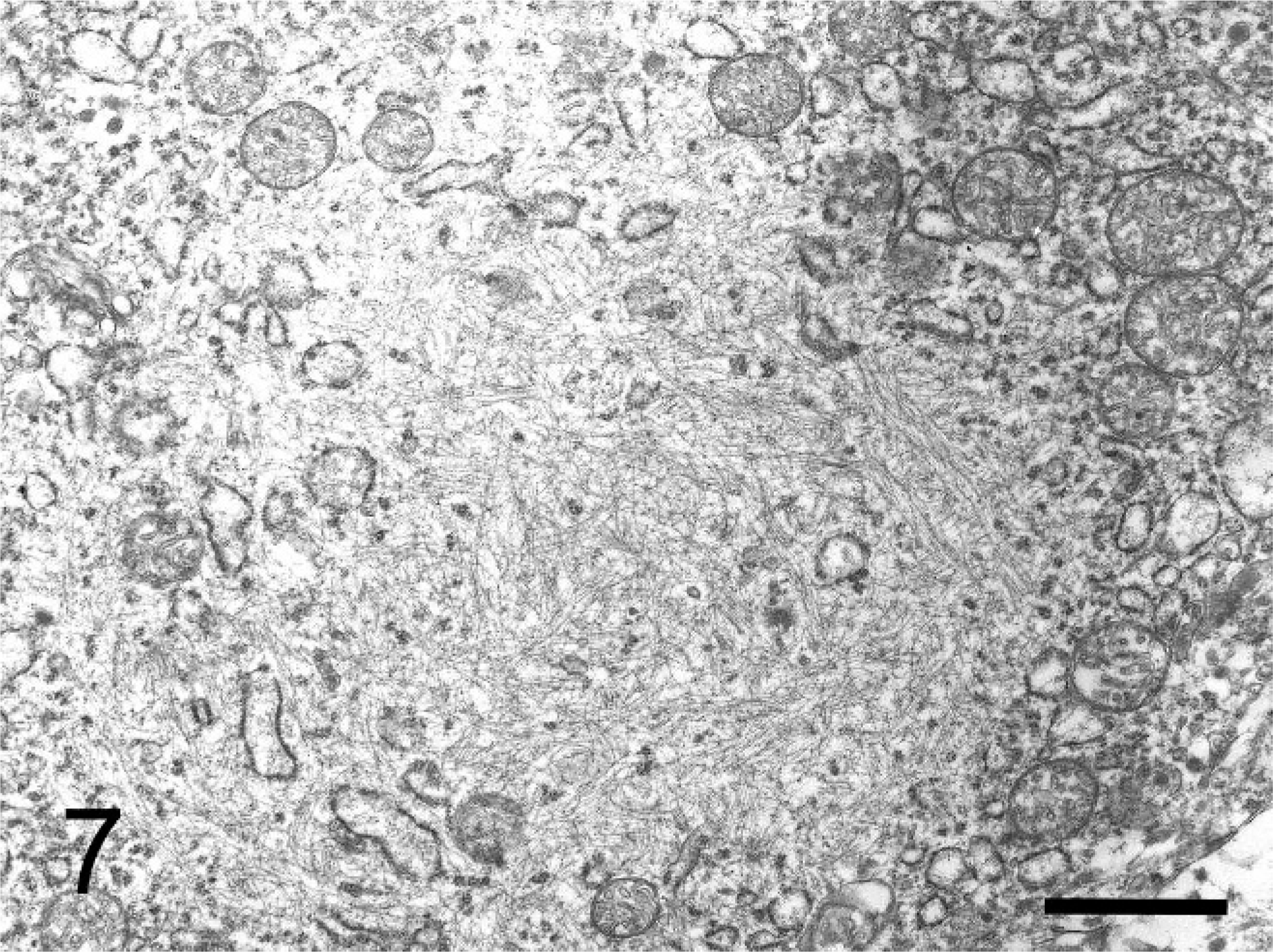
Transmission electron micrograph. Subcutaneous rhabdoid tumor; cat. The cytoplasmic inclusions are composed of aggregates of intermediate filaments that entrap mitochondria and rough endoplasmic reticulum. Bar = 2 µm.
The most striking feature of this case is the presence of rhabdoid cells that were histologically and ultrastructurally quite similar to those of human RTs. Regardless of tumor location, the neoplastic cells in human RTs are consistently positive for vimentin and often coexpress vimentin and epithelial markers (cytokeratin and EMA). 5, 8, 14, 15 However, some human RTs are negative for cytokeratin, and cytokeratin was not detected immunohistochemically in the previously reported canine RT 10 and was only focally expressed in a hepatic metastatic lesion of the RT in an orangutan. 9 In addition to vimentin and epithelial markers, immunoreactivity for NSE or S-100 protein has been found in some renal 14 and extrarenal RTs, 5 and neurofilament expression has been reported in renal 14 and extrarenal RTs 11 and central nervous system ATRTs. 8 Our results demonstrate that the histologic, ultrastructural, and immunohistochemical features of this feline tumor are quite similar to those of human RTs and the few RTs reported in animals.
In humans, tumor cells with rhabdoid morphology are focally present in a variety of malignant tumors, especially in malignant soft tissue tumors (so-called composite rhabdoid tumors). 5 Therefore, it is necessary to determine whether such tumors are “pure” rhabdoid tumors or a different type of tumor with a focal rhabdoid phenotype. Although the definition of RTs is still controversial, it is generally thought that the term “rhabdoid tumor” should be restricted to those tumors with predominant rhabdoid features in which no other clear line of differentiation can be found. In the present case, the immunopositivity for vimentin, NSE, neurofilament, and S-100 protein suggests a neuroectodermal origin for the tumor. Malignant melanoma is uncommon in cats and found predominantly in the skin. 2 Almost all feline melanomas are positive for vimentin, NSE, and S-100 protein. 7, 12 About two-thirds are positive for melan-A, although most amelanotic melanomas are negative for this marker. 7 In humans, the presence of rhabdoid cells has been reported in malignant melanoma as an unusual variant. 4 In our case, however, the tumor was localized to the subcutis, in which melanocytes are normally absent, and had no connection to the epidermis. Additionally, our case was negative for melan-A, and no apparent melanosomes were found by electron microscopy. Although the possibility of malignant melanoma cannot be completely ruled out, we could find no indication of melanocytic differentiation in this tumor.
This tumor affected an old cat (13 years). In humans, renal RTs usually occur in infants or children, 14, 15 whereas soft tissue RTs affect patients over a wider age range. 5, 15 Furthermore, the orangutan with an RT in the gastric wall was aged. 9 It is likely that, as in humans, the age range for soft tissue RTs is wider in animals. Human RTs are characterized by highly aggressive behavior with frequent local recurrence, distant metastasis, and low survival rate despite therapy. 5, 8, 14, 15 A survival clinical course has also been reported in animal RTs. The gastric RT in the orangutan metastasized to abdominal organs, 9 the brain tumor of the dog extended from the optic chiasm to the pons, 10 and the orbital RT in the horse metastasized to lymph nodes of the head and neck. 3 In contrast, in this feline case, the tumor was localized to the subcutis with evidence of survival 11 months after surgical excision.
In conclusion, this is the first known report of RT in the cat. The neoplasm in this cat shared several distinctive histologic, ultrastructural, and immunohistochemical features with human RTs and the few RTs reported in animals. However, the apparently benign behavior of this case is quite different from that reported in human and veterinary RTs. Examination of additional cases may provide further insight into this rare and poorly defined tumor.
Footnotes
Acknowledgements
This work was supported by Grant-in-Aid for Scientific Research from Japan Society for the Promotion of Science (JSPS; nos. 20580341 and 19580359).
