Abstract
An oncocytoma was diagnosed in the nasal cavity of a 12-year-old Domestic Shorthair cat who presented with periocular swelling and sneezing. Histologic examination from biopsy material revealed monomorphic sheets, anastomosing cords, tubules, and acini composed of large polygonal to oval cells that contained abundant finely granular eosinophilic cytoplasm. No vascular or lymphatic invasions were noted. Histochemical stains revealed positive staining of tumor cells with periodic acid-Schiff (PAS) (before and after diastase digestion) and phosphotungstic acid-hematoxylin. Immunohistochemical evaluation of the tumor cells demonstrated positive staining for cytokeratin and negative staining for vimentin, desmin, S-100, glial fibrillar acidic protein, and neuronal specific enolase. Ultrastructurally, the tumor cells contained large numbers of mitochondria within their cytoplasm, which confirmed a diagnosis of oncocytoma.
Oncocytomas are rare tumors that are composed of oncocytes. 2 Oncocytes are large cylindrical or polyhedral cells characterized by an abundant eosinophilic, granular cytoplasm, which is caused by the presence of large numbers of mitochondria. These cells are believed to be of epithelial origin. In humans, these tumors have been reported to occur in endocrine glands, liver, kidney, esophagus, and trachea, and virtually anywhere along the seromucous lining of the respiratory tract. 4, 14 Some information on oncocytomas in animals is available, and previous reports include an oncocytoma in the parotid region of a cat 3 and in kidney, larynx, thyroid gland, and nasal cavity of the dog. 1, 3, 5, 13 The nasal cavity is an extremely rare location for oncocytomas, with only a few human cases being reported in the literature. 7, 11, 12
Here we describe the microscopic, immunohistochemical, and ultrastructural features of a nasal oncocytoma in a cat. While nasal oncocytoma has been previously reported in the dog, the diagnosis was made on cytologic and microscopic evaluation only, and electron microscopy was not performed. 5 It has been suggested that to diagnose an oncocytoma in a new location, it is mandatory to demonstrate the presence of mitochondria through electron microscopy. 8 To the best of our knowledge the diagnosis of nasal oncocytoma based on confirmatory electron micrographs has not previously been reported in the cat or any other domestic species.
A 12-year-old female spayed Domestic Shorthair cat was presented to the veterinarian for swelling of the periocular region of the right eye and sneezing. Purulent material was released after lancing the swelling, and several courses of antibiotic therapy were given, with intermittent success. The sneezing was getting worse and eventually epistaxis, as well as intermittent mouth breathing, was noted. At presentation to the Queen Mother Hospital for Animals, Royal Veterinary College, an asymmetry of the face was seen, with swelling of the dorsomedial region of the right medial canthus. Dried blood around the right nostril and increased upper-respiratory noise were found on physical examination. Otherwise the cat was in good body condition.
Routine hematology and serum biochemistry were unremarkable. With the cat under general anesthesia, radiographs of the head and the thorax were taken, and a rhinoscopy performed. Thoracic radiographs showed no abnormality. Besides loss of several teeth, increased soft-tissue opacity, and a loss of turbinate structure in the right nasal passage were seen on nasal radiographs. Retrograde nasopharyngoscopy showed a small mass protruding from the choanae. Biopsy samples of the lesion were taken, fixed in 10% buffered formalin, and evaluated by light microscopy. Magnetic resonance imaging of the head showed a mass on the right side, extending from the nares to the olfactory bulb, filling the entire nasal cavity and frontal sinus. Surgical excision of the mass was performed; but, in the postoperative period, the cat developed ventricular tachycardia with subsequent cardiac arrest. Resuscitation was not successful. A necropsy was not permitted.
Formalin-fixed paraffin-embedded sections of the mass were stained with hematoxylin and eosin (HE), periodic acid–Schiff (PAS), PAS with diastase (both PAS stains to evaluate the presence of glycogen), Masson's trichrome, and phosphotungstic acid-hematoxylin (PTAH; to stain for mitochondria) for light microscopic examination. A panel of commercial antibodies were applied to determine whether the tumor displayed epithelial, mesenchymal, or neural differentiation. The immunohistochemical examination included pancytokeratin AE1–AE3 (Dako, cat. No. M3515, dilution 1°100), vimentin (Dako, cat. No. M0725, dil. 1°500), desmin (Dako, cat. No. M0760, dil. 1°100), glial fibrillar acidic protein (GFAP) (Dako, cat. No. Z0334, dil. 1°100), S-100 protein (Dako, Z0311, dil. 1°500), and neuronal specific enolase (NSE) (Dako, cat. No. M0873, dil. 1°100). An avidin-biotin complex method was used according to the manufacturer's specifications (DakoCytomation A/S, Glostrup, Denmark) with the various primary antibodies. Controls used normal serum instead of specific primary antibody. The remaining tissue was rinsed in 0.1 M Sorensens buffer and postfixed in 2% OsO4 in 0.1 M Sorensens buffer, and processed routinely before examination in a Philips transmission electron microscope (Philips Electron Optics, Eindhoven, The Netherlands).
Microscopic examination revealed a well-demarcated, nonencapsulated, noninfiltrative expansive neoplastic mass that was positioned in the lamina propria and extended to but did not invade the overlying respiratory epithelium. The overlying respiratory epithelium was intact. The mass was composed of large polygonal to oval cells arranged in monomorphic sheets, anastomosing cords, tubules, and acini, and was supported by a scant intervening fibrovascular stroma (Fig. 1). The lumen of the acini contained moderate amounts of eosinophilic material and cell debris. The cells had indistinct cytoplasmic margins and abundant finely granular eosinophilic cytoplasm. Nuclei were round to oval, variably positioned, and contained vesicular patterned chromatin and 1 prominent magenta nucleolus. When the tumor cells were positioned along fibrovascular stroma, the nuclei were often basally located (Fig. 2). Nuclear pallisading was also noted. Mitotic figures averaged 1 per 10 400× magnification fields. There was moderate anisocytosis and anisokaryosis. No vascular or lymphatic invasions were noted. Surrounding the mass were moderate infiltrates of inflammatory cells composed of predominantly lymphocytes and plasma cells, with occasional scattered neutrophils and macrophages.
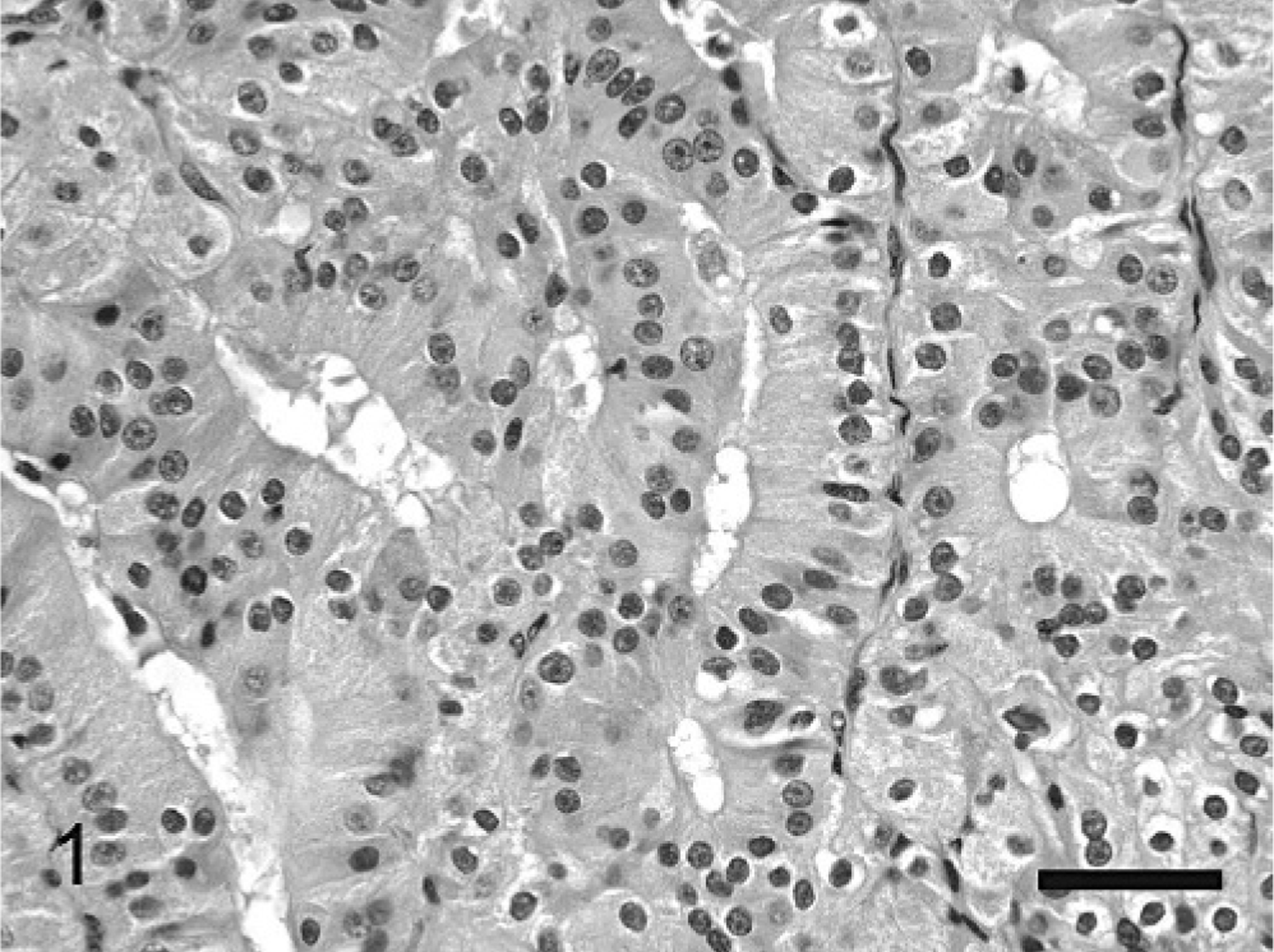
Nasal mass; cat. Note the characteristic granular cytoplasm and the round/oval nuclei. There is a tubular structure present with material present, with the nuclei of the neoplastic cells positioned along the fibrovascular stroma. HE. Bar = 40 μm.
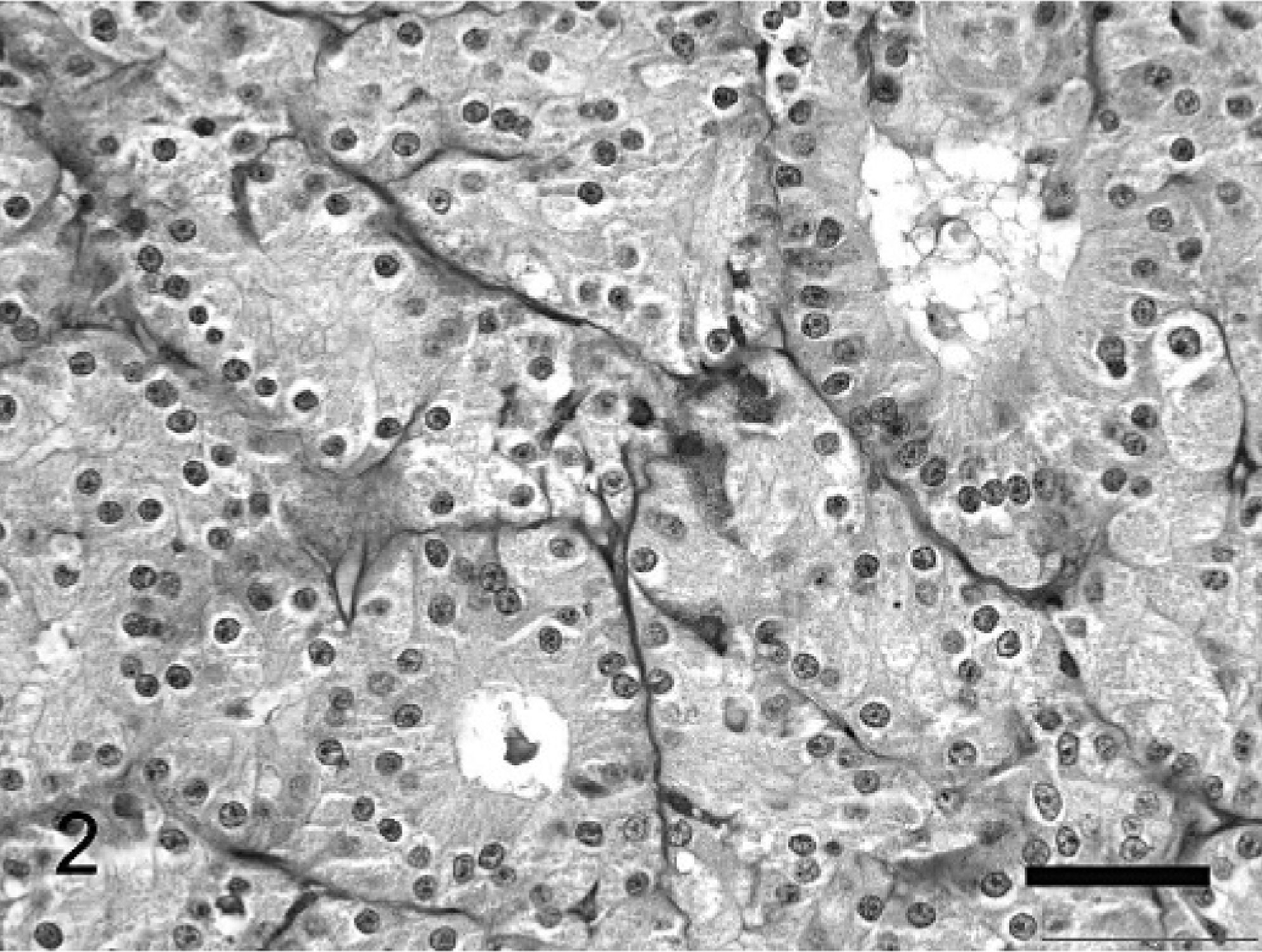
Nasal mass; cat. Note the acinar and tubularlike structures, which contain luminal material. Note the basally located nuclei lining adjacent to the fibrovascular stroma. PAS. Bar = 50 μm.
The cytoplasmic granules were moderately positive by PAS staining and were diastase sensitive. Masson's trichrome stained the cytoplasm diffusely red, and PTAH stained the cytoplasmic granules dark blue (Fig. 3). The tumor cells stained strongly positive for cytokeratin (Fig. 4) and negative for vimentin, desmin, S-100, GFAP, and NSE. Ultrastructurally, the mass was composed of oval to polygonal cells with marked numbers of mitochondria (Fig. 5). The mitochondria were of variable size, however, the cristae appeared normal. Occasional intramitochondrial glycogen inclusions were noted. Few other organelles were noted in the cytoplasm. The nuclei were large and typically contain large prominent nucleoli and homogenous karyoplasm. Occasionally, junctional complexes were noted between the neoplastic cells.
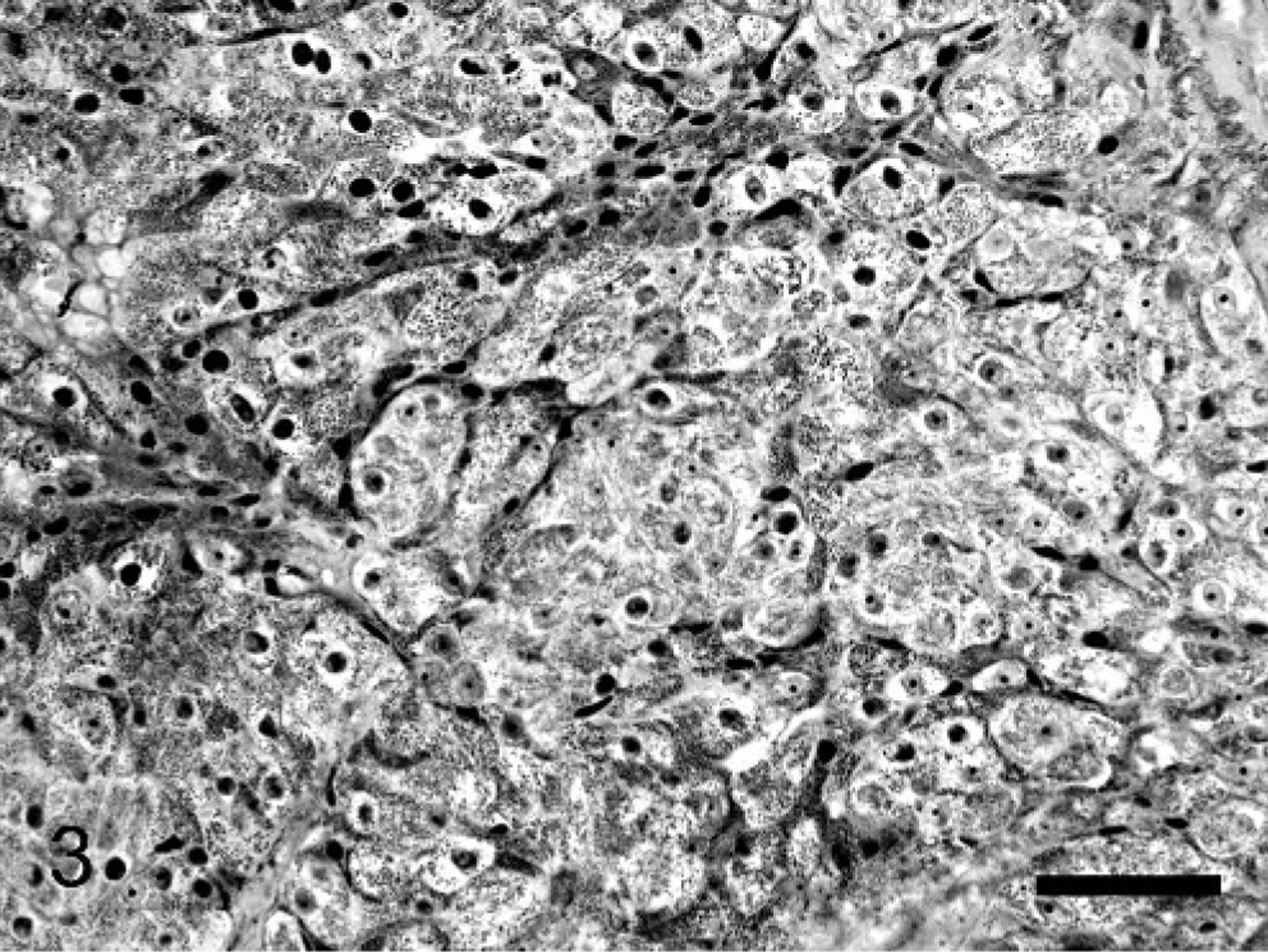
Nasal mass; cat. Note the PTAH positive granules (mitochondria) present in the cytoplasm of the tumor cells. PTAH. Bar = 50 μm.
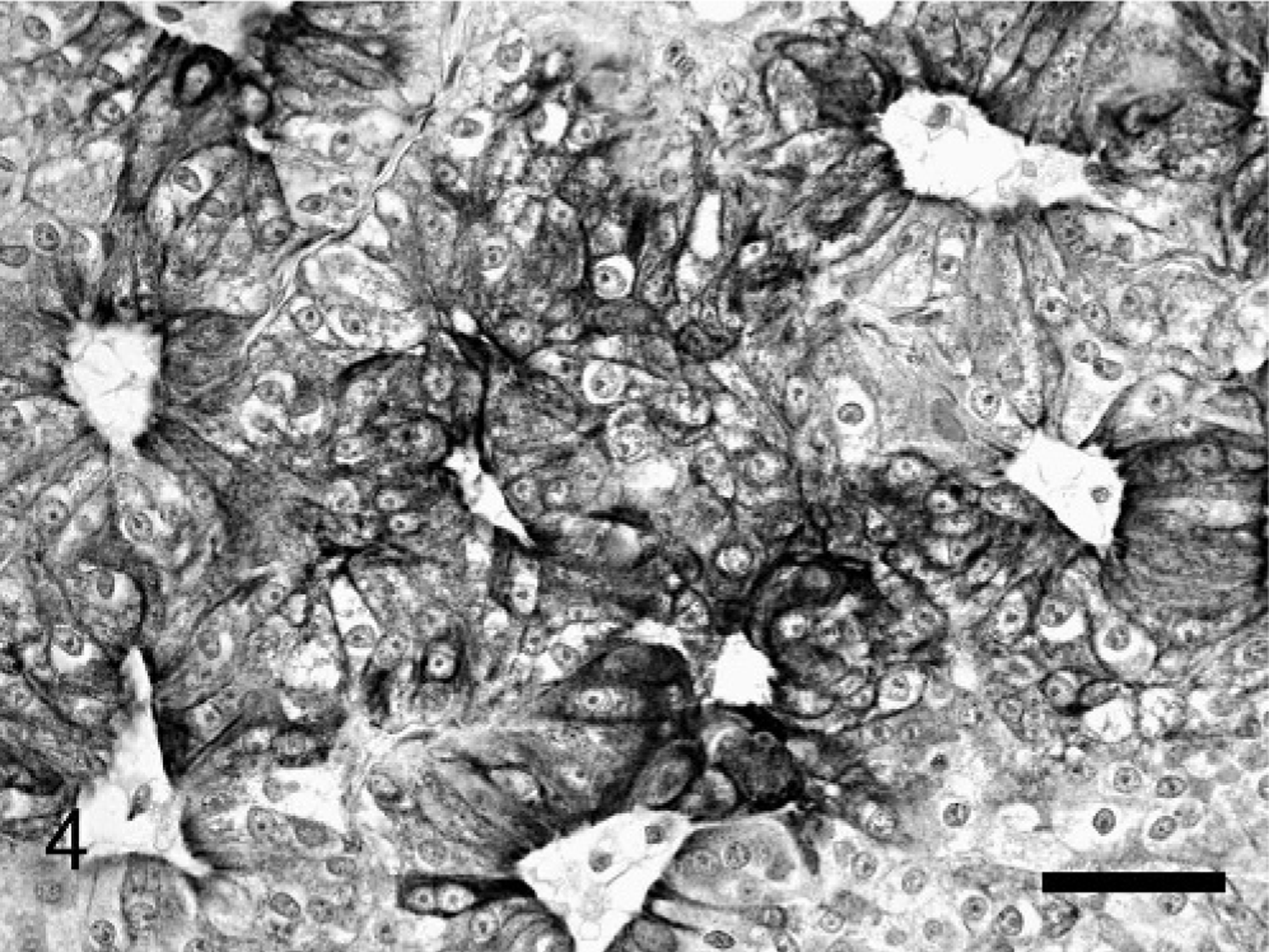
Nasal mass; cat. Representative immunohistochemical staining of the tumor cells immunoreactive for cytokeratin. Cytokeratin immunoreactivity was noted in nearly 100% of the tumor cells. Where the neoplastic cells formed acini the cytokeratin staining was typically more intense on the apical surface. Cytokeratin immunohistochemistry; DAB, hematoxylin counterstain. Bar = 50 μm.
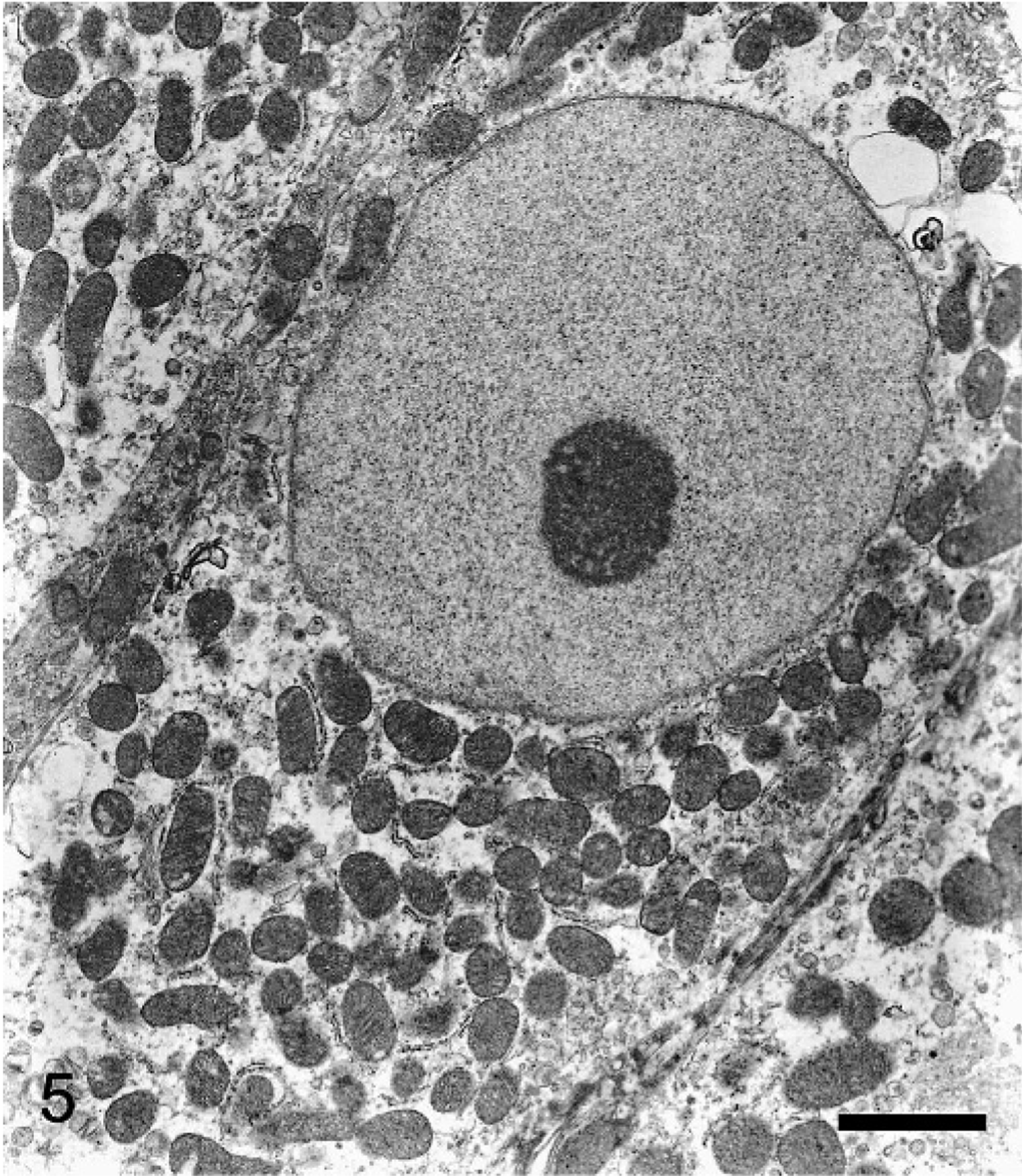
Nasal mass; cat. Transmission electron micrograph of a tumor cell with numerous mitochondria present in the cytoplasm. Note the oval nucleus with the prominent nucleolus and homogenous karyoplasm. Bar = 1 μm.
Based on the HE microscopic evaluation, the histochemical stains, immunohistochemical profile, and the ultrastructural features, the tumor was diagnosed as an oncocytoma. Because of the findings from the extensive imaging investigations undertaken in this case, the nasal cavity was thought to be the primary site from which the tumor developed.
Controversy stills exists as to the histogenesis of the oncocytoma. 14 The oncocytoma of the respiratory tract are thought to originate from the transformation of epithelial cells lining the ducts or the acini of the various seromucous glands present. In humans, oncocytes are found in many organs and are thought to represent an age-related change. 4 Histologically, these cells are large polygonal round or oval, with abundant eosinophilic granular cytoplasm. This is caused by the presence of a large number of mitochondria, which is the most characteristic feature of an oncocyte. The cause of transformation of a normal cell into an oncocyte is not fully understood, but it is thought to represent hyperplasia and hypertrophy of defective organelles because of mitochondrial dysfunction that caused defective cellular metabolism. 13 Oncocytic metaplasia is usually described in association with epithelial cells, and, in this current case, epithelial origin is put forward because of the immunohistochemical findings. However, rare cases of the same process have been reported within mesenchymal neoplasms, including rhabdomyoma, epithelioid leiomyosarcoma, and glomus tumor. 14 Whether or not oncocytomas arise from already established oncocytes or transform directly from “normal” epithelial cells is not known. It has been suggested that oncocytomas that occur in salivary glands may originate from seromucous glands, and, hence, nasal oncocytomas may arise from similar glands present in the nasal cavity or minor salivary glands adjacent to the nasal cavity. 6, 11
The diagnosis of oncocytoma solely by light microscopy is normally not sufficient, with these neoplasms having several morphologic similarities with the granular cell tumor. 9 Granular-cell tumors have been reported in head and neck regions of cats and could potentially arise within the nasal cavity or extend from the primary site into the nasal cavity. 13 Both specific immunohistochemical markers and electron microscopy are helpful in making this differentiation with granular-cell tumors. Oncocytomas specifically express cytokeratin, 1 whereas the granular-cell tumors are negative for cytokeratin and positive for vimentin, S-100, and NSE. 13 Ultrastructurally, the oncocytoma characteristically contains an abundance of mitochondria in the cytoplasm, and this feature is not noted in granular-cell tumors. Other possible differential diagnoses are the rhabdomyoma or rhabdomyosarcoma. While there is potential for such a tumor to develop in the nasal cavity, the lack of a positive desmin stain in the tumor cells and the lack of aggregates of myofilaments rule out this diagnosis. 10
In humans, most oncocytomas are benign, however, there have been reports of malignant cases in the literature. 7 Malignant forms are difficult to recognize microscopically because they may not differ much from the benign forms. Oncocytomas arising in the nasal cavity normally lack a capsule, and their malignant potential is mostly because of their local aggressive nature. 12 The biologic behavior of oncocytomas cannot be generalized and predicted by their morphologic characteristics (cytologic atypia, mitotic activity). Based on these criteria, this current case may be considered benign, however, the size and the extent of the tumor noted via medical imaging and a full examination of the nasal cavity would have been required to more appropriately classify the tumor as benign or malignant.
Footnotes
Acknowledgements
We thank John Bredl, Royal Veterinary College, University of London for his assistance with the tissue processing for the electron microscopic evaluation.
