Abstract
A 14-month-old heifer with a 17-day history of unresponsive bloody diarrhea was necropsied. There were focal, pink-red erosions of the nares and hard palate; ulcers and fissures of the tongue; and multiple ulcerative lesions of the alimentary canal. Interdigital skin of both rear limbs was ulcerated and bleeding; and the margins of the vulva contained punctiform red ulcers. The gross lesions were consistent with mucosal disease. Histopathology and laboratory testing ruled out rinderpest, foot-and-mouth disease, and vesicular stomatitis, and identified bovine virus diarrhea virus to be the cause of this disease. Lesions of the vulva similar to those seen in some stages of infectious pustular vulvovaginitis were negative for bovine herpesvirus-1 and tested positive for bovine viral diarrhea virus antigen by immunohistochemistry.
Bovine virus diarrhea virus (BVDV) is a positive-sense RNA virus classified as a member of the Pestivirus genus, within the Flaviviridae family. BVDV has been subdivided into 2 major genotypes, 1 and 2, and each genotype is further subdivided into subgenotypes. 3 There are 11 subgenotypes within genotype 1, and 2 subgenotypes within genotype 2. Both genotypes contain cytopathic and noncytopathic (NCP) biotypes, based on their effects on tissue culture cells. Both the cytopathic and noncytopathic BVDV cause “virus diarrhea of cattle,” a disease recognized earlier as an “apparently new transmissible disease of cattle” 12, 13 that occurs postnatally between 8 and 24 months of age. BVDV infection of cows during early pregnancy commonly results in embryonic death and resorption. Infection of a fetus in utero with noncytopathogenic BVDV before the onset of immunocompetency (80–125 days of gestation) can cause severe congenital defects that result in fetal death, low weight at birth, or “weak calf syndrome.” A proportion of calves that survive the infection at this stage become persistently infected (PI) and immunotolerant to the NCP virus. 4 Within the first 2 years of life the NCP BVDV in live-born PI animals often undergoes recombination, resulting in mutation to CP biotype, and the animal succumbs to a severe form of disease called mucosal disease (MD). BVDV infection is immunosuppressive, and a variety of opportunistic infections, distorting its gross appearance and presentation, have been reported in cases of mucosal disease. 14 The lesions and presentation of this heifer had the appearance of severe mucosal disease. However, similarity to the lesions of rinderpest and genital bovine herpesvirus (BHV) infection made it necessary to conduct virologic, serologic, and immunohistochemical tests.
The case discussed here was a 14-month-old, red-and-white Hereford and Maine Anjou heifer weighing 300 kilograms. It was brought to the Connecticut Veterinary Medical Diagnostic Laboratory at the beginning of July. This animal came from a 50-acre beef production farm and was one of 14 animals housed at the same location. The herd had a history of intermittent diarrhea since the spring. All animals on the farm, with the exception of this one, were vaccinated in the spring with a commercial polyvalent killed virus vaccine (bovine rhinotracheitis, virus diarrhea, parainfluenza 3, respiratory syncytial virus, with Leptospira canicola-grippotyphosa-hardjo-icterohemorrhagiae-pomona and Hemophilus somnus bacterins) (Fort Dodge, IA) and separately with a rabies vaccine. This animal had been skipped during spring vaccination because it had been vaccinated as above in November and December, at 6 and 7 months of age.
The history indicated that this heifer had been sick for approximately 2 weeks. Clinical signs included diarrhea, that had been bloody at times, anorexia, and dehydration. The animal's condition deteriorated despite treatment by an attending veterinarian. Seventeen days after the onset of the illness, the animal was euthanized, and necropsy followed immediately.
On gross examination, the animal had sunken eyes, and pale-pink conjunctiva. There were several small erosions of the mucosa of the nares and lips (Fig. 1) and numerous small ulcers and erosions in the buccal mucosa. The hard palate contained 18–20 pink–red erosions, each 2–3 mm in size (Fig. 2). These were depressed and stellate or irregular in shape. The dorsal surface of the tongue had 20–40 punched-out ulcers and fissures of 2–8 mm size (Fig. 3). Internally, the mucosa of the esophagus had numerous dark-red to brown, depressed, linear ulcers, varying in size from 2 × 3 to 2 × 10 mm (Fig. 4). The nonpapillary portion of the rumen and the abomasum contained numerous 1–2 mm red ulcers with white borders (Fig. 5, 6). The latter occurred on the mucosal folds, as well as in the pyloric antrum (Fig. 6). The distal half of the small intestine had reddened Peyer's patches (Fig. 7). The mucosa of the jejunum contained numerous pinpoint to pinhead-sized red foci of necrosis, often along the ridges of the transverse folds. Some of these red foci were hemorrhages, others were ulcers.
The mucosae of the ileum, spiral colon, and cecum were thickened. Segmentally, the mucosa of the cecum and colon was dark red to brown to black and necrotic (Fig. 8). Mucosa was raised or covered by pseudomembranes (Figs. 8, 9), which were negative by culture for Salmonella. The wall of these affected parts was thickened with edema, and the mesentery was edematous as well, with enlarged, reddened regional lymph nodes. The lesions extended throughout the distal colon and rectum.
There were bleeding ulcerative lesions of the interdigital skin of both rear limbs (Fig. 10). The rectum was prolapsed 3 cm, and the exposed mucosa was dark red–brown or black and covered with dry necrotic mucosa. The margins of the vulva contained 10–12 small, punched-out, red ulcers, each 2–3 mm in diameter (Fig. 11), and there was reddening and superficial ulceration of perineal skin.
At necropsy, samples of serum and tongue were submitted for further testing at the Foreign Animal Disease Diagnostic Laboratory (FADDL) (Greenport, NY), and the spleen was sent to the Animal Health Diagnostic Center at Cornell University (Ithaca, NY).
The heifer's serum submitted to FADDL tested negative for antibodies against foot-and mouth-disease virus infection–associated antigen (VIAA) and vesicular stomatitis (serotypes New Jersey and Indiana 1) by complement fixation and ELISA, and rinderpest virus by virus neutralization. Lamb kidney cells, which had been inoculated with lingual tissue, showed Cytopathic Effect after 1 week, and electron microscopy revealed a 100 nm herpesvirus. Virus neutralization ruled out bovine herpesvirus-1 (BHV-1), the causative agent of infectious bovine rhinotracheitis. The presence of BHV (BHV-1 and BHV-5) viral DNA was further assessed by PCR 8 in our laboratory. Total DNA was extracted from 6 pooled 20 μ samples of paraffin-embedded tissues, including vulva, using a DNeasy Blood and Tissue Kit (Qiagen, Valencia, CA) according to the manufacturer's instructions. Primers targeting BHV gC glycoprotein named B1, specific for BHV-1 5′-CAA CCG AGA CGG AAA GCT CC-′3 (nt. 185–204); B5, specific for BHV-5 5′-CGG ACG AGACGC CCT TGG-′3 (nt. 322–339); and Bcon, a common reverse primer 5′-AGT GCA CGT ACA GCG GCT CG-′3 encompassing nt. 519–538 of BHV-1 and nt. 461–480 of BHV-5, were used to amplify a 354 bp product for BHV-1 and 159 bp product for BHV-5. PCR was carried out in a 50 μl reaction containing 200 μM dNTPs, 20 pmoles of each primer, 5 μl 10× Advantage-2 buffer (Clontech, Mountain View, CA), 1 μl 50× Advantage-2 polymerase (Clontech), and 5 μl total DNA. Cycling conditions consisted of 1 step hold of 3 minutes at 94°C, followed by 40 cycles of 1 minute at 94°C, 1 minute at 56°C, 1 minute at 68°C, and a final elongation step of 3 minutes at 68°C. Amplified products were visualized in 2% (w/v) agarose gel stained with ethidium bromide. All samples tested negative for BHV-1 and BHV-5. The spleen submitted to the Cornell University Animal Health Diagnostic Center (Ithaca, NY) was shown to contain BVDV genotype 1b. This virus was cytopathic on bovine testicular cells. Spleen did not yield herpesvirus.
Microscopically, there were focal ulcerative lesions of the nares and vulva and necrotic dermatitis of the interdigital skin of the hind limbs. There was multifocal, ulcerative stomatitis (Fig. 12), esophagitis (Fig. 13), rumenitis, and abomasitis (Fig. 14) in the alimentary canal. The ileum and colon had diffuse, necrotizing, pseudomembranous enterocolitis. The regional lymph nodes had diffuse lymphoid depletion, with sinus histiocytosis, edema, and occasional neutrophils. There was lymphoid depletion in the spleen. The adrenal glands had multiple cortical hemorrhages. Immunohistochemical (IHC) staining specific for BVD was applied to sections of the abomasum, jejunum, and colon. There were numerous immunoreactive cells within the lamina propria of different segments of the intestinal tract (Fig. 15). The morphology and locations of the positive cells were consistent with macrophages, epithelial cells lining the crypts, and endothelial cells lining capillaries. Immunohistochemical staining specific for BHV-1 was applied to sections of intradigital skin, and nasal and vulvar mucosae. There was no positive staining for BHV-1 observed in any of these tissues. The ulcers of the vulva (Fig. 16), which resembled infectious pustular vulvovaginitis, were positive for BVD viral antigen (Fig. 17) and negative for BHV-1 antigen by IHC.
The microscopic lesions and laboratory tests established a diagnosis of mucosal disease in this heifer. It is likely that this heifer was persistently infected (PI) and immunotolerant to a NCP BVDV strain as a result of maternal–fetal infection in utero. Transplacental infection with NCP biotypes occurs much more frequently than with cytopathic isolates. Some time after birth, the persistent virus may have undergone homologous or heterologous recombination, 3, 19 resulting in conversion into a cytopathic biotype, which in turn caused severe mucosal disease (MD). The BVDV isolated from the spleen of this heifer was a CP biotype of genotype 1B. NCP BVDV strains are more prevalent in nature, and it is from these that CP BVDV strains arise. 3 A NCP and a CP BVDV pair may be isolated from MD cases, and these can be distinguished by the appearance of nonstructural protein 3 (NS3) in the CP virus. 10, 19 Nevertheless, NCP viruses by themselves are capable of causing severe postnatal disease in cattle. Outbreaks of severe mucosal disease caused by NCP genotype 2 BVDV have been reported before. 7 NCP genotype 2 has also been associated with severe acute and sometimes hemorrhagic BVD, which was distinguishable from mucosal disease. 18 Another possibility is that the PI animal was superinfected, most likely with a homologous antigenically related CP BVDV strain 5, 6, 20 or even a heterologous strain. 2, 17 There is no possibility that the superinfecting virus could have been vaccine-derived, 1, 16 because the only vaccine used in this herd was inactivated.
Although ear-notch immunohistochemistry, repeat virus isolation, or RT-PCR were not performed to reveal persistent infection, 9, 11 the age of the animal, severity and distribution of the lesions, and the isolation of a CP biotype of BVDV from the spleen suggest that there was persistent BVDV infection that progressed into severe mucosal disease. The reason for the herpesvirus isolated from tongue in this case is unknown. The ulcerative lesions in the vulva observed in this heifer could be confused with some stages of infectious pustular vulvovaginitis caused by BHV-1. In early BHV-1 infection, intranuclear inclusion bodies occur. In this instance, laboratory confirmation was employed to differentiate BVDV from BHV-1 infection. At an earlier time “necrotic vulvitis of the labia” was reported in some animals with mucosal disease. 15 However, it is not clear if these authors were reporting lesions similar to those shown here or some other less well-defined necrotic change. Ours may be the first report of BVDV-induced punctiform ulcerations of the vulva in mucosal disease.
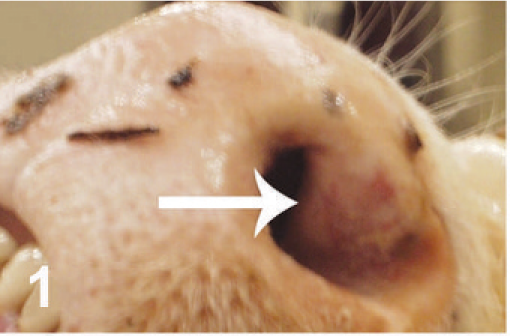
Nares; cow. Reddened erosions of the external nares.
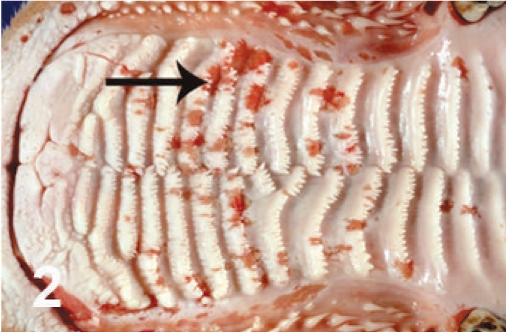
Hard palate; cow. Multiple red erosions along the ridges.
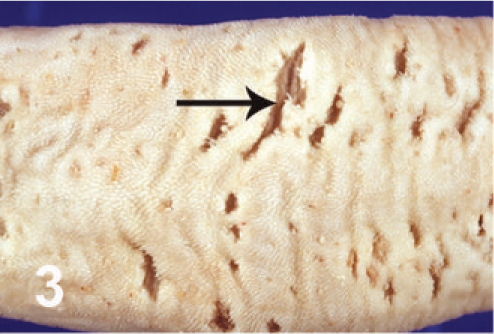
Tongue; cow. Ulcers and fissures of the dorsal surface.
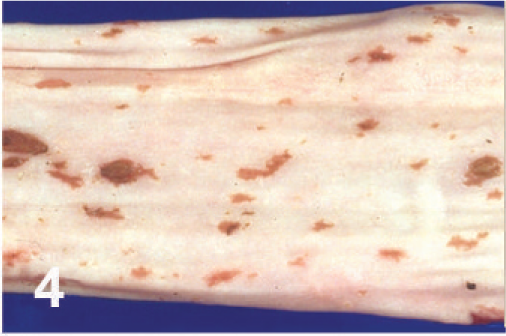
Esophagus; cow. Linear erosions.
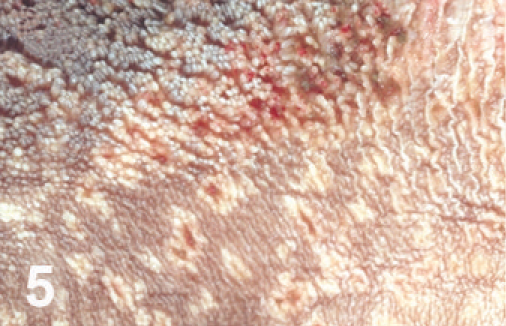
Rumen; cow. Punctate ulcers.
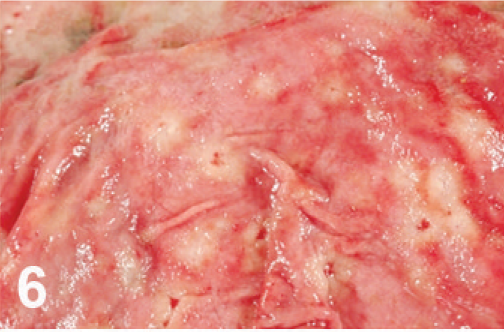
Abomasum; cow. Punctate ulcers.
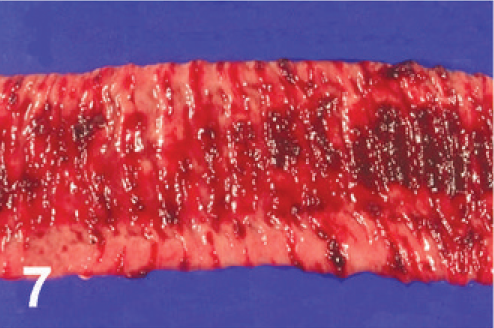
Jejunum; cow. Multiple punctate and stellate ulcerations and hemorrhage with ulceration of a Peyer's patch.
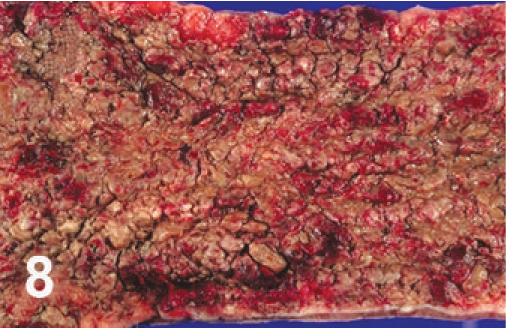
Cecum; cow. Necrotic typhlitis.
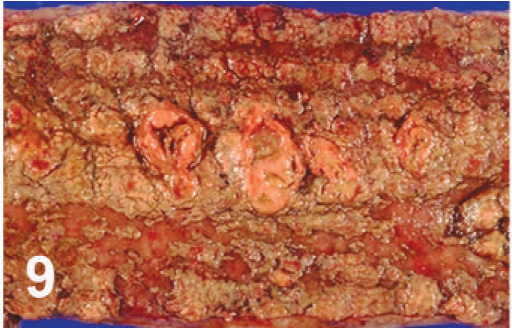
Colon; cow. Pseudomembranous necrotic colitis.
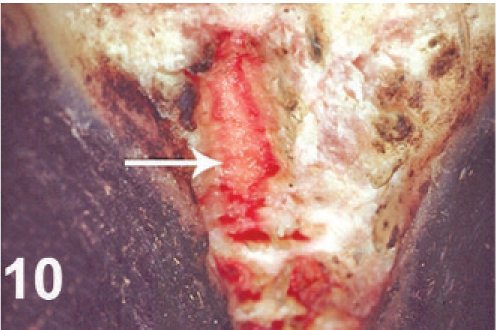
Rear limb; cow. Ulcerative interdigital pododermatitis.
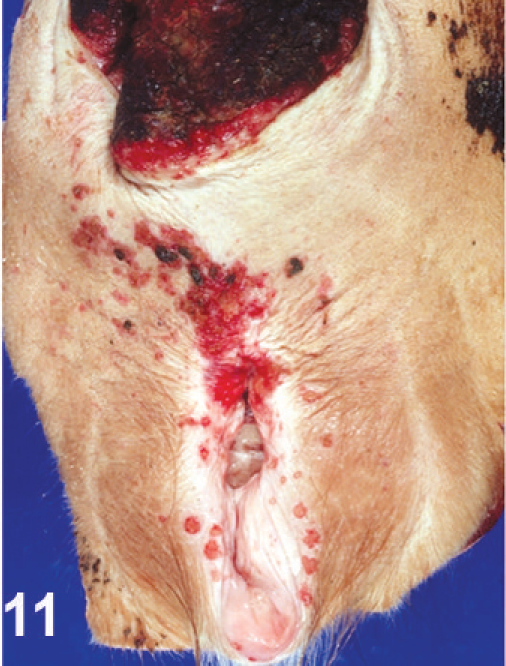
Vulva; cow. Red ulcers of the vulvar mucosa, and reddening and superficial ulceration of the perineal skin. Prolapsed rectum above.
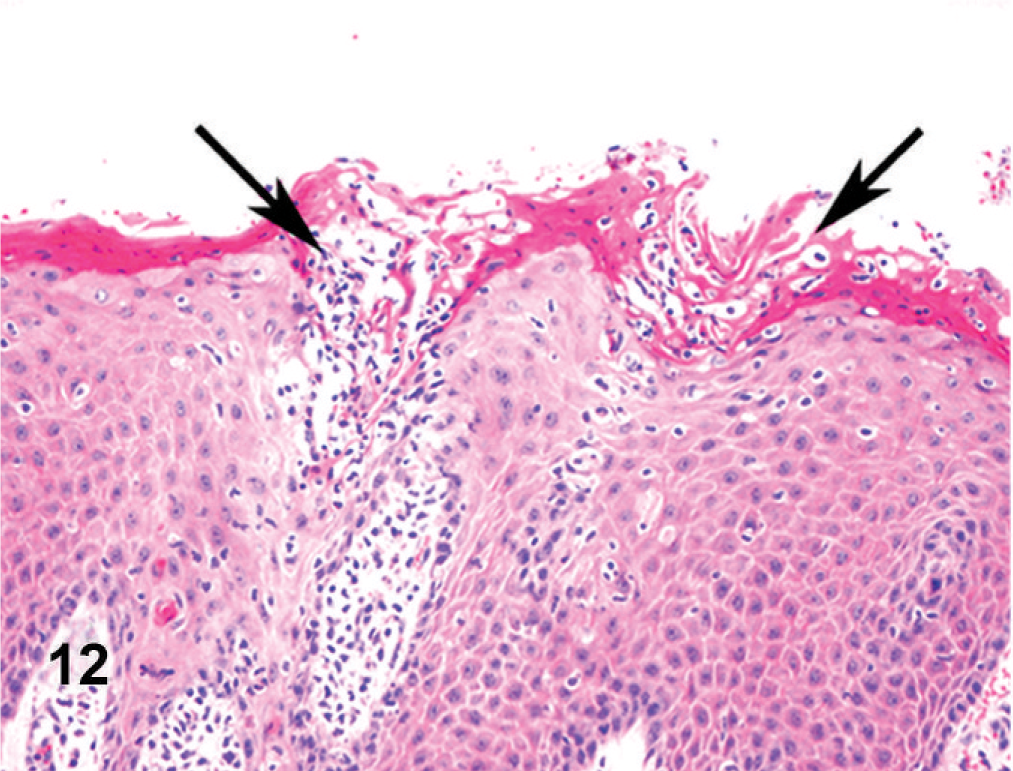
Oral mucosa; cow. Mucosal erosions and intracorneal pustules (arrows).
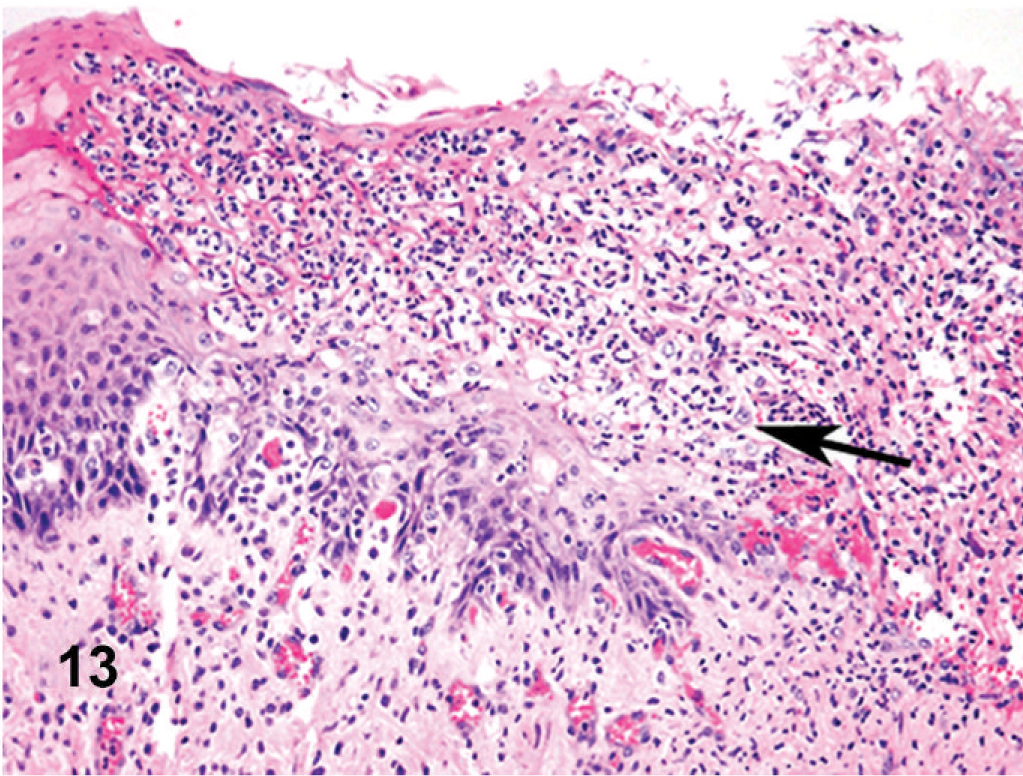
Esophagus; cow. Surface ulceration coated over by fibrinopurulent exudate (arrow).
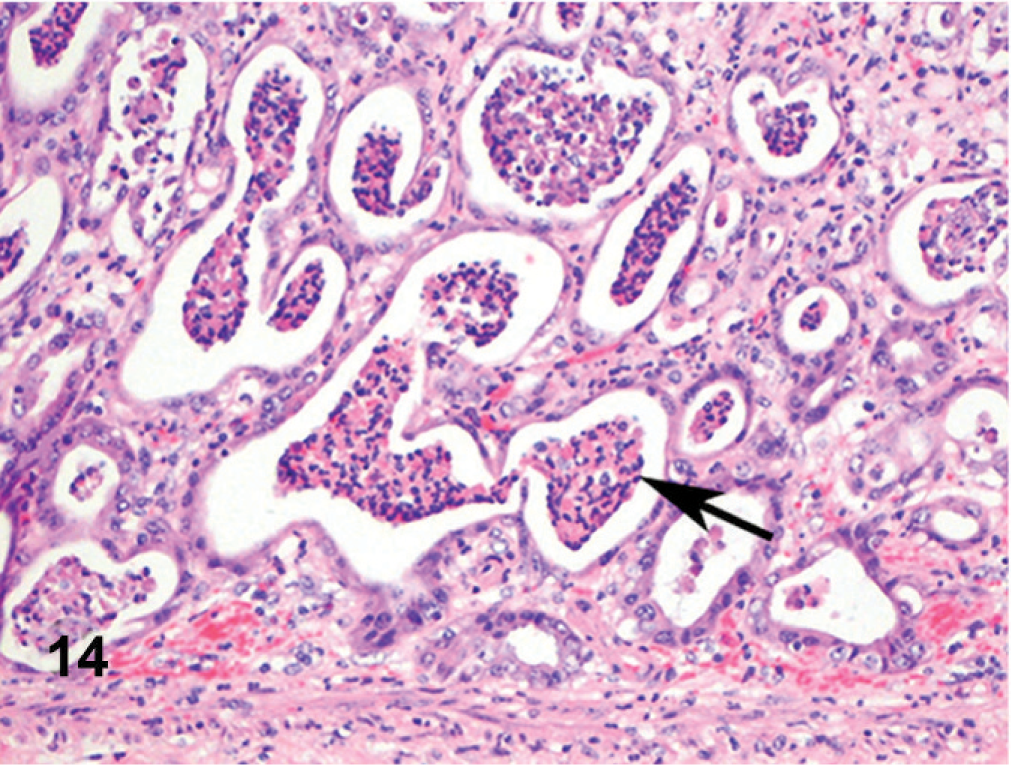
Abomasum; cow. Gastric glands are dilated and contain aggregates of neutrophils and sloughed epithelial cells.
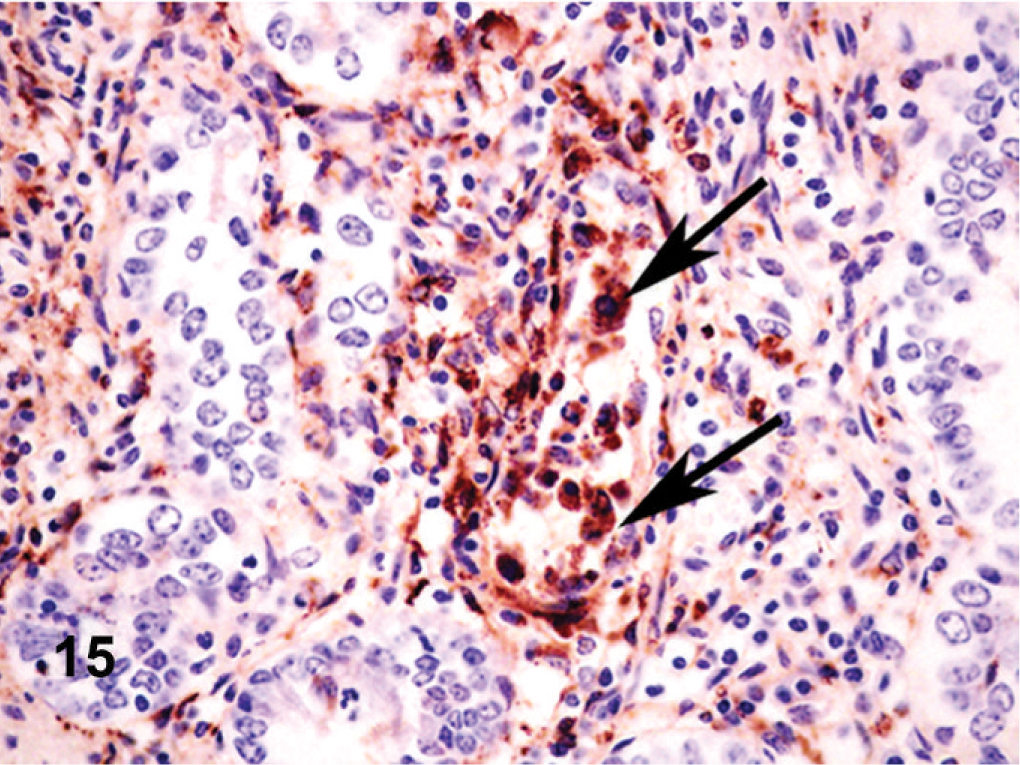
Small intestine; cow. Crypt epithelial cells (arrows) and endothelium lining mucosal capillaries show immunoreactivity for BVD antigen.
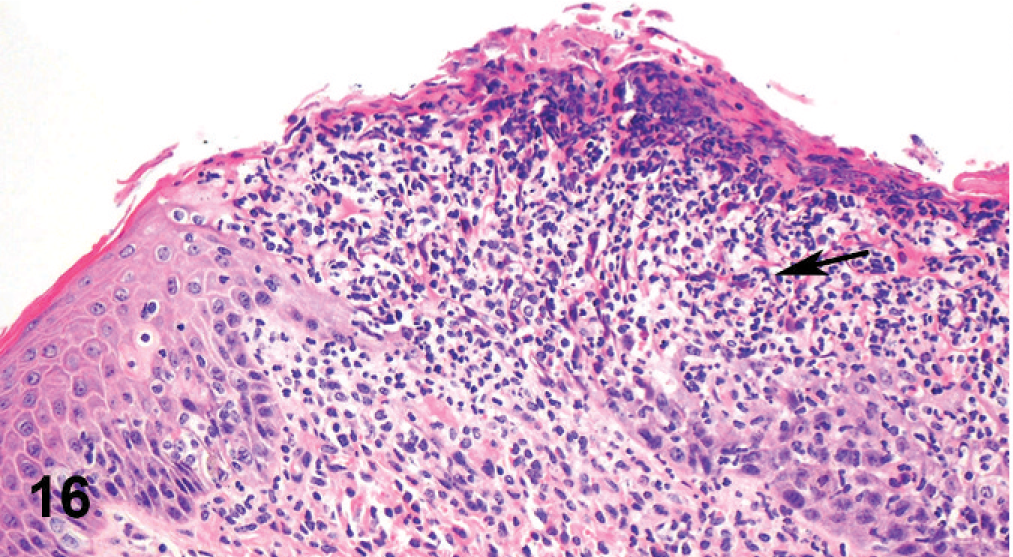
Vulva; cow. Ulceration of the mucosa coated by fibrinopurulent response.
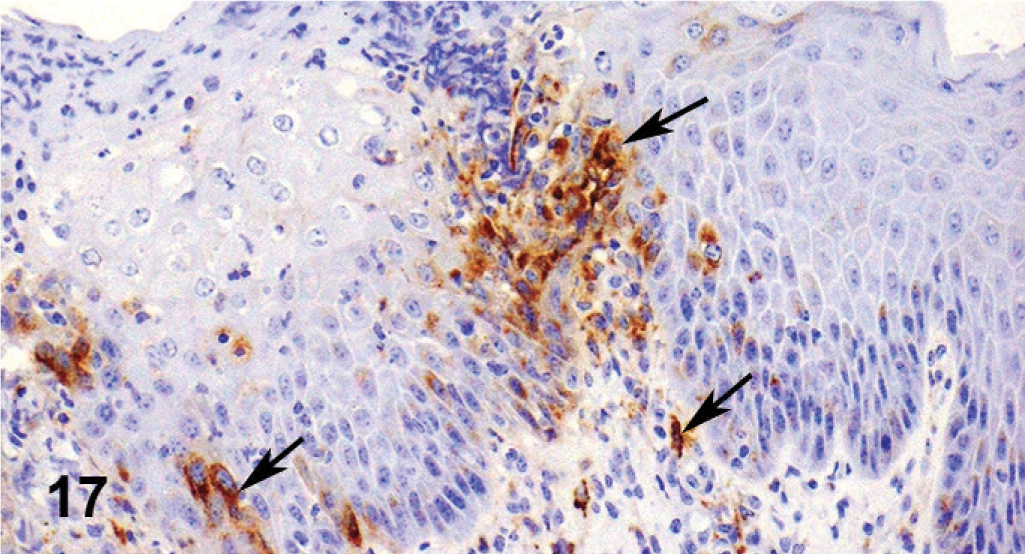
Vulva; cow. Immunoreactivity for BVD antigen in stratified squamous epithelial cells (arrows).
