Abstract
Bovine viral diarrhea virus (BVDV) belongs to the Pestivirus genus, which is further divided into subgenotypes (1a–1u and 2a–c). When persistent infection occurs, the calf will be immunotolerant to BVDV and possibly develop mucosal disease. This study describes an outbreak of BVDV-1d–induced mucosal disease lacking intestinal lesions. Eleven calves presented with anorexia, sialorrhea, lameness, recumbency, and death. Three calves were necropsied, showing ulceration of the interdigital skin and the oral and nasal mucosa; linear ulcers in the tongue, esophagus, and rumen; and rounded ulcers in the abomasum. Microscopically, mucosa and skin had superficial necrosis, with single-cell necrosis and vacuolation in epithelial cells, and severe parakeratosis. Immunohistochemistry (IHC) showed BVDV antigen in the cytoplasm of epithelial cells in skin and mucosa. All 11 dead calves were positive upon reverse transcription-polymerase chain reaction (RT-PCR) for the detection of Pestivirus along with another 11 live calves from the herd, which were positive again by RT-PCR and IHC after a 4-week interval. Sequencing of the 5′ untranslated region and N-terminal protease showed that viruses from these 22 calves were homologous and of subgenotype BVDV-1d. Cytopathic BVDV was isolated from 8 of 11 dead calves, but only noncytopathic BVDV was isolated from the 11 live animals. The findings indicate that this was an outbreak of mucosal disease caused by BVDV-1d, with high morbidity, and lesions restricted to the upper alimentary system and skin and absent from intestine. Thus, the epidemiological and pathological features in this form of mucosal disease may be similar to vesicular diseases, including foot and mouth disease.
Bovine viral diarrhea virus (BVDV) is a member of the genus Pestivirus, a single-stranded positive-sense RNA virus within the family Flaviviridae. BVDV has been divided into 2 genotypes or species: BVDV-1 and BVDV-2. 26 Furthermore, there are at least 21 subgenotypes of BVDV-1 (1a–1u) 9,30 and 3 of BVDV-2 (2a–2c). 14 BVDV can also be classified into biotypes, which are based on the presence (cp) or absence (ncp) of a visible cytopathic effect in infected cell cultures. 2 In Brazil, BVDV infection has been reported since the end of the 1960s, 6 and recent studies have verified a predominance of BVDV-1a and BVDV-2b among Brazilian cattle. 25,31
BVDV is one of the main pathogens of cattle, 17 and reproductive losses may be the most economically important consequence of the infection. The clinical signs and consequences of bovine viral diarrhea (BVD) may vary according to the genetic and antigenic properties of these viruses. Most transient infections have a mild course, 1 while clinical disease due to acute infections is rare. 18,22
Persistent infection (PI) may result from BVDV infection between 42 and 125 days of gestation, and the PI calf will be immunotolerant to this specific strain, shedding the virus to the herd. 32 Identifying and eliminating PIs should be the major focus to control and prevent BVDV. 11 The outcome of PI is a condition called mucosal disease (MD) that is associated with the appearance of a cp BVDV biotype, generally arising from mutation of the ncp BVDV already circulating in the PI animal. 4
In a herd with a rigorous BVDV control, the prevalence of PI cattle is usually 1% to 2%, but if exposure occurs in the first trimester of pregnancy, the proportion of PIs will be higher. 31 MD usually has a low morbidity rate (less than 5%) and a high case fatality rate, although outbreaks are also described affecting up to 10% and 14% of some herds. 7,19,21,29 It generally presents with erosions, ulcerations, and large areas of mucosal necrosis in segments or throughout the entire gastrointestinal system. 21,32 It is characterized by hemorrhagic, necrotic, and ulcerative lesions, primarily in Peyer’s patches, in addition to the loss of villi and intestinal gland crypts dilated by cellular debris. 4,17
Mucosal lesions overlying Peyer’s patches are considered a hallmark of mucosal disease. 4,17,21,32 In the present study, we describe an outbreak of MD by BVDV-1d, affecting calves of a beef herd cattle with a higher morbidity, in which intestinal lesions, including those associated with gut-associated lymphoid tissue, were minimal, and lesions were restricted to the upper alimentary system and skin.
Materials and Methods
Clinical, Epidemiological, and Histopathological Features
The study was performed on a beef cattle herd that suffered numerous losses of calves in a short period. Epidemiological data and clinical signs of the herd were obtained through fieldwork and with the herd owner’s veterinarian. Samples from 3 calves that died of the disease were collected during necropsy, in which small and large intestines were examined along their full length. Samples were fixed in 10% buffered formalin for 24 to 48 hours, trimmed, routinely processed for histology, embedded in paraffin, and cut at 3 μm. Histological sections were stained with hematoxylin and eosin (HE).
Immunohistochemistry
Sections of oral mucosa, skin, and small intestine were submitted for immunohistochemistry. Monoclonal BVDV antibody was used (BVD MAb 15c5; Syracuse Bioanalytical, East Syracuse, NY) at a dilution of 1:500 in phosphate-buffered saline (PBS). The antigens were retrieved through protease XIV (Sigma Chemical Co., St Louis, MO) for 15 minutes at room temperature. Nonspecific reactions were blocked with 5% skim milk for 15 minutes. The amplification signal was achieved by using biotinylated secondary antibody, followed by labeled streptavidin-biotin-alkaline phosphatase complex (LSAB + System AP; DakoCytomation, Carpinteria, CA) for 20 minutes each at room temperature. The reaction was revealed with Permanent Red solution (DakoCytomation). Brain cortex and ear notch sections were used as positive controls as previously described. 24 Primary antibody was replaced with PBS in the negative control sections.
RT-PCR and Phylogenetic Analysis
Total RNA was isolated from the serum of all animals on the property (398) and from the spleen and lymph nodes of the 3 necropsied animals. Serum from reverse transcription-polymerase chain reaction (RT-PCR)–positive live animals was collected and tested again after 4 weeks to identify PI animals. Total RNA was obtained using TRIzol LS Reagent (Life Technologies, Carlsbad, CA), according to the manufacturer’s instructions. Complementary DNA (cDNA) synthesis and PCR were performed using SuperScript III Reverse Transcriptased and Platinum Taq DNA Polymerase (Life Technologies), respectively. For partial amplification of the 5′ untranslated region (5′UTR) of pestivirus, the PanPesti primers were used. 31 For sequencing, the positive samples were also tested with the 324/326 primers to amplify a fragment of 5′UTR. To determine the genetic identity, the sequencing of the N-terminal protease (Npro) was performed with primers BD1/BD3. 30 NADL strain was used as BVDV-positive control in these procedures.
PCR amplification products were purified using a Nucleo Spin Extract II Kit (Macherey-Nagel, Düren, Germany) according to the manufacturer’s directions. Both strands were sequenced using a Big Dye Terminator cycle sequencing kit, version 3.1, on an ABI PRISM 3100 Genetic Analyzer (Applied Biosystems, Foster City, CA). Sequences that were generated in the present study were aligned with representative Pestivirus strains that were retrieved from GenBank using a Muscle algorithm contained in the Molecular Evolutionary Genetics Analysis software package, version 6 (MEGA6). 27 Evolutionary history was inferred using the maximum likelihood method with the best-fit substitution model. 12 Consequently, Kimura 2-parameter was chosen for both genomic regions (5′UTR and Npro), with invariable sites. 15 A gamma distribution was used to model the evolutionary rate differences among the sites, with 5 categories for the Npro sequences. All evolutionary analyses were conducted in MEGA6. 27
Virus Isolation
RT-PCR–positive samples (serum, spleen/lymph nodes homogenates) were tested by virus isolation using pestivirus-free Madin-Darby bovine kidney (MDBK) cells in 24-well microtiter plates with Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 5% horse serum and antibiotics. The plates were incubated at 37°C, 5% CO2 and examined daily for up to 4 days to observe the cytopathic effect. Four consecutive passages were carried out with each sample.
Additional Examinations
This outbreak was reported to the official agency of animal sanitary control due to the clinical signs resembling vesicular diseases. Swab samples were collected from ulcers in oral mucosa. These were tested by RT-PCR for foot and mouth disease virus (FMDV) and indirect sandwich–enzyme-linked immunosorbent assay (IN-ELISA) for vesicular stomatitis (VS) in a government-certified laboratory as described. 34,35
Results
Clinical and Epidemiological Features
The outbreak occurred in an Angus beef cattle-rearing herd located in Glorinha, Rio Grande do Sul state, Brazil (29° 52′ 50′′ S; 50° 46′ 59′′ W), with 500 ha of total area. The herd was kept in native pasture, and the reproductive management consisted of natural breeding during the summer. The herd was closed, so there was no introduction of animals, and the cattle did not contact animals from other farms at the borders of the property. Vaccination was performed for FMDV, rabies, and clostridiosis, but the herd was not vaccinated for BVDV. The herd was composed of 398 cattle, including 164 three to five-month-old unweaned calves. Early embryonic loss, abortion, and stillbirth rates were similar to the previous years. The outbreak lasted for 15 days between January and mid-February 2015, wherein 11 of these calves (offspring of heifers) presented clinical signs of anorexia, severe sialorrhea, weight loss, stiff gait, and lameness, progressing to recumbency and death in a clinical course of 7 days (Fig. 1). Diarrhea and tenesmus were not observed in any of these calves. All calves that developed clinical illness died.
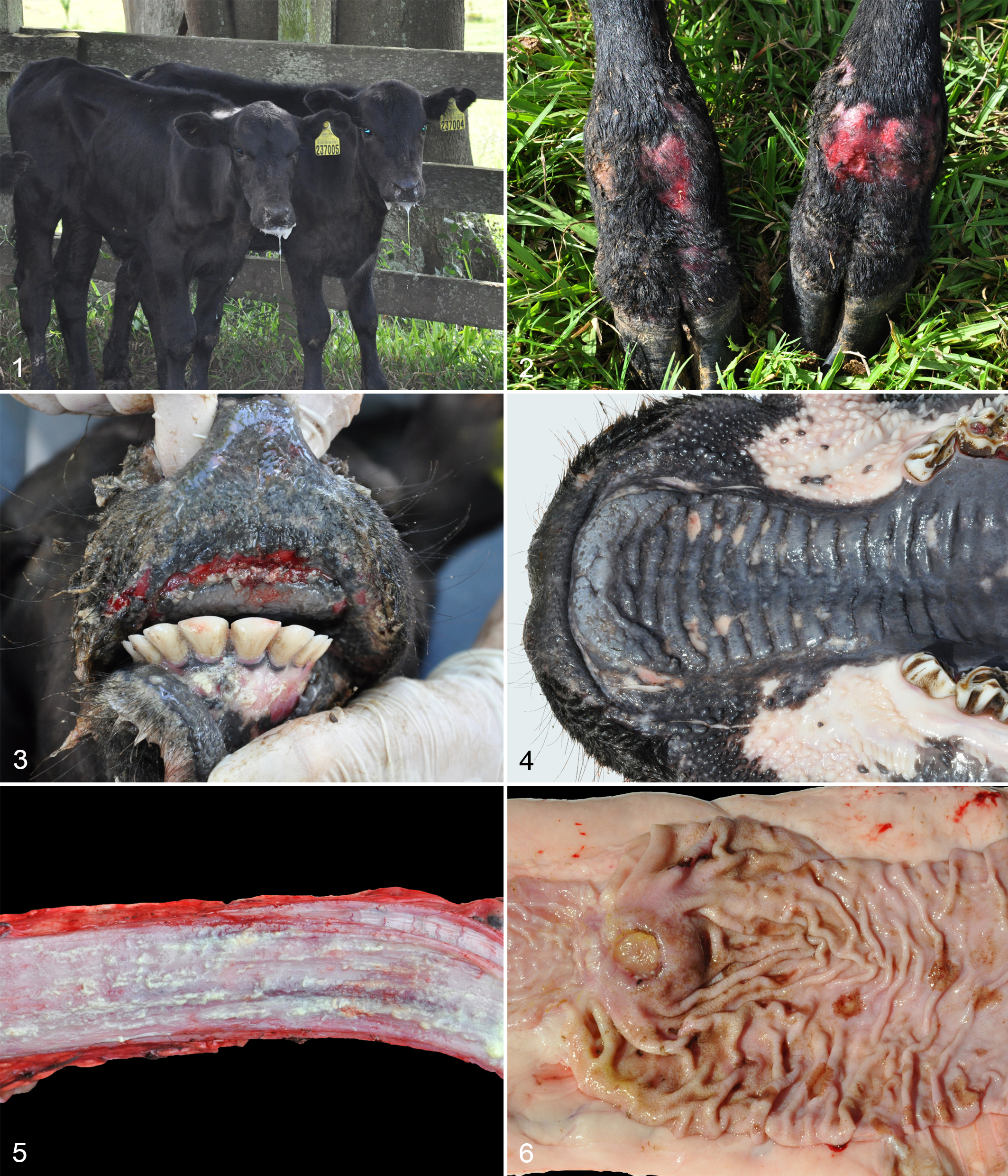
Gross Findings
During physical examinations, all 11 calves that succumbed to the disease presented extensive areas of ulceration in the oral and nasal mucosa and the interdigital region of the thoracic and pelvic limbs (Fig. 2) that varied from mild to moderate in animals that were not necropsied (8 calves). Two of these animals were euthanized (calf Nos. 1 and 2) and 1 died (calf No. 3), followed by necropsy immediately after death. The calves were in poor body condition. Oral and nasal ulcers varying from mild (calf No. 1) to severe (calf Nos. 2 and 3) (Fig. 3) often involved gingiva, hard palate (Fig. 4), soft palate, and nasal cavity (Table 1). The tongue (calf Nos. 2 and 3), esophagus (Fig. 5), rumen (calf No. 3), and abomasal (calf No. 2; Fig. 6) mucosa had severe multifocal to linear ulcers that were covered by yellowish fibrillary material. The skin of the axillary, ventral abdomen, and inguinal regions in calf No. 2 was diffusely thickened with severe crust formation and occasional ulceration. The kidneys of calf No. 2 had multifocal 2-mm-diameter rounded whitish areas on the capsular surface. No gross abnormalities were observed in small and large intestines or lymphoid tissues (lymph nodes, tonsils, and thymus).
Distribution and Severity of Lesions in Calves With Mucosal Disease Caused by Bovine Viral Diarrhea Virus–1d.

+, mild; ++, moderate; +++, severe; –, absent.
aLesions were not present in small or large intestine, Peyer’s patches, lymph nodes, or thymus.
Microscopic Findings
The ulcers in the oral mucosa, tongue, hard and soft palate, nasal mucosa, esophagus, rumen, and abomasum consisted microscopically of severe multifocal necrosis in the superficial epithelium, with numerous epithelial cells showing hypereosinophilic cytoplasm and pyknotic nuclei (single-cell death) in addition to vacuolation of epithelial cells in the stratum basale. There was a moderate inflammatory infiltrate of lymphocytes and plasma cells at the mucosa-submucosa junction. Superficially, there was severe diffuse parakeratotic hyperkeratosis intermixed with foci of ulceration of the mucosa covered by cellular debris and myriad bacterial coccobacilli (Fig. 7).
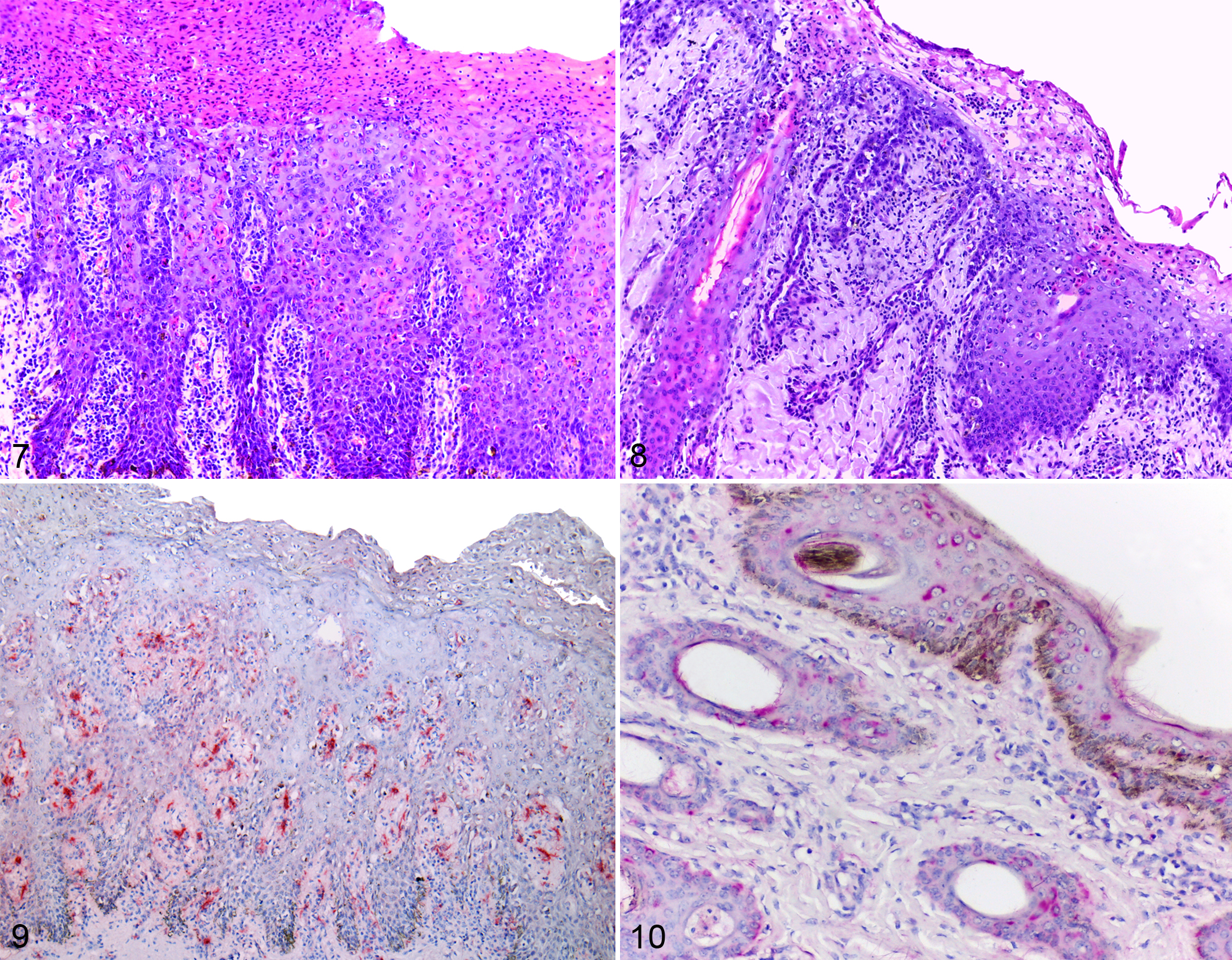
Cutaneous lesions in calf No. 2 were characterized by severe necrosis of the superficial epidermis and follicular epithelium with vacuolated cells and single-cell death. There were intracorneal pustules and severe parakeratotic hyperkeratosis (Fig. 8) and a moderate infiltrate of lymphocytes, plasma cells, and macrophages in the superficial dermis. Some sections of skin showed diffuse ulceration of the epidermis covered by fibrin.
The kidney of calf No. 2 showed mild multifocal interstitial lymphohistiocytic inflammatory infiltrate. Small and large intestines did not contain necrosis, cryptitis, or vasculitis, and lymphoid depletion was not present in gut-associated lymphoid tissue (GALT), lymph nodes, tonsils, or thymus.
Immunohistochemistry, RT-PCR, Phylogenetic Analysis, and Virus Isolation
Immunohistochemistry of the skin of the 3 calves submitted to necropsy showed marked immunostaining in the cytoplasm of epithelial cells of epidermis and hair follicles. Sections of oral mucosa showed moderate (calf No. 1) to marked (calf Nos. 2 and 3) immunostaining in the cytoplasm of epithelial cells (Fig. 9). On small intestine sections, immunostaining varied from absent (calf No. 2) to focal in Peyer’s patch (calf No. 1) and diffuse moderate in mucosa and Peyer’s patch (calf No. 3).
All 11 dead calves were positive for BVDV by RT-PCR, as were an additional 11 live calves (also the offspring of heifers) of 387 remaining cattle tested that did not show clinical signs. Virus isolation was performed on samples from the 22 infected animals, revealing that the viruses isolated belonged to the BVDV-1d subgenotype and were genetically very similar (98.8%–100% nucleotide identity in 250 nucleotides of 5′UTR). The sequence from the 5′UTR and Npro of 1 calf is shown in the phylogenetic tree (Suppl. Figs. S1, S2). Cytopathic BVDV was isolated from the samples of 8 of 11 dead animals, whereas ncp BVDV but no cytopathic virus was isolated from the samples of the 11 live calves. After a 4-week interval, the RT-PCR and immunohistochemistry were repeated on serum and ear notch biopsy samples, respectively, from these 11 live animals. All 11 live calves had immunostaining for BVDV in the cytoplasm of the epithelial cells of the epidermis and hair follicles (Fig. 10), as well as positive results in RT-PCR and virus isolation, suggesting that these animals were PI. 5 These 11 live calves were removed from the herd.
Swab samples of vesicular-like ulcers from oral mucosa were both negative by RT-PCR for FMDV and by IN-ELISA for VS.
Discussion
Despite the distinctive lack of intestinal lesions, the diagnosis of BVDV-1d–induced MD in this outbreak was obtained through gross, microscopic, and virological findings. The pathologic findings were similar to those described in acute outbreaks of BVDV-2a associated with high morbidity and mortality and extensive oral lesions and mucosal ulceration, as well as by BVDV-1b in calves with similar gross lesions predominantly at the mucous membranes of the tongue, lips, esophagus, and rumen. 13,18 However, in the present outbreak, the isolation of cp BVDV from 8 of the 11 calves that died in a short period indicates a diagnosis of MD. 33 The high morbidity observed is not common for MD, which is generally sporadic in a herd with low morbidity but high case fatality. 21,29
The lesions described in outbreaks of acute BVD overlap with those observed in MD. 18 Small intestinal crypt epithelial degeneration and necrosis and lysis of the follicular lymphoid tissues are prominent findings of typical MD. 19,29 However, the calves described here did not present with intestinal and lymphoid lesions. Based on the necropsy findings of the 3 calves, this outbreak was characterized by lesions that were restricted to the upper alimentary mucosa and skin. Similarly, the other 8 calves presented severe sialorrhea with extensive areas of ulceration in the oral and nasal mucosa but not diarrhea or tenesmus, suggesting a similar involvement of the upper rather than lower alimentary tract.
A few natural MD outbreaks have been described with absence of intestinal lesions 8,10 and a short clinical course of 7 days in 1 calf. 8 As in the calves described herein, we speculate that this short clinical course may explain the limited distribution of the lesions, whereas less fulminant infections might lead to involvement of the lower alimentary tract with development of diarrhea and tenesmus. 21
The lesions observed in the skin were characterized by a widespread dermatitis with necrosis of keratinocytes in the stratum basale and stratum spinosum, in addition to a severe parakeratosis, similar to those described in an outbreak of BVDV-1b in which 6 calves were affected. 19 However, no cp BVDV was isolated in those cases previously described, implying acute BVDV infection rather than MD. 19 A generalized dermatitis described in cattle with MD had a clinical course of 35 to 45 days, although the cases described in the present report were acute. 10 Parakeratosis in BVD is related to epithelial cell tropism of the virus, 17 and it is possible that it occurred in the skin and mucosa due to the severe necrosis of the epithelium caused by the numerous apoptotic keratinocytes.
BVDV infection results in a wide spectrum of conditions as a consequence of variation between viral strains and host factors. 19,23 Considering that the herd was closed and not vaccinated against BVD, the heifers probably lacked immunity against the virus. Thus, these animals were more susceptible to the infection, and the introduction of the virus during the pregnancy season allowed the generation of a large number of PI offspring, which explains the higher morbidity rate observed.
BVDV-1d has been detected in cattle worldwide, causing a wide range of clinical signs, 16,20,28 although this subgenotype has been detected both in healthy animals and in farms with reproductive disorders. 25 A study also suggested that subgenotype 1d is able to induce respiratory disease in immunocompetent calves, 3 although in the present cases, no pulmonary lesions or clinical signs were observed. The strain of BVDV-1d detected in the present study is genetically close to strains previously described in different regions of the country, indicating that this subgenotype of BVDV may be widespread. 20,25
Due to the high morbidity and clinical presentation, such as sialorrhea and loss of weight, associated with the gross lesions, differential diagnosis included VS, FMD, and bovine papular stomatitis. In both VS and FMD, there is a dissociation of cells in the deeper layers of the stratum spinosum and distinct spongiosis that leads to vacuole formation, 29 which were not observed in the present study. The outbreak described here affected 11 calves (3–5 months of age) that died within a short period, differing from the epidemiology of FMD, which has a very high morbidity with low mortality, with all age groups affected and a loss of productive efficiency. 29
The outbreak described here was notable because of the substantial impact of the unusual presentation of MD, with 11 dead calves in a short period, along with the PI status of 11 other animals, which remained healthy but would be expected to disseminate the virus. The source of BVDV-1d entry into this herd is unknown, as no animal was previously introduced into the herd. In conclusion, this was an outbreak of BVDV-1d–induced MD with high morbidity, short clinical course, and an unusual distribution of lesions restricted to the upper alimentary system and skin and absent from the intestines, resembling the clinical and pathological presentation of vesicular diseases.
Footnotes
Acknowledgements
We thank the technician Cíntia de Lorenzo for her assistance in the IHC performed in this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Fundação de Amparo à Pesquisa do Estado do Rio Grande do Sul (FAPERGS) and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) supported this study.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
