Abstract
Equine polysaccharide storage myopathy (EPSSM) is a metabolic myopathy of unclear origin that results in excessive accumulation of polysaccharides in the skeletal muscle fibers. This muscle disorder has been reported in many horse and pony breeds, 4, 10 with the highest incidence recorded in draft horses. 5 Percheron, Belgian, Clydesdale, and Shire draft horse–related breeds have been previously reported affected. 6 Cob Normand breed is an average-sized horse named after the English Cob and developed as a distinct breed at the beginning of the 20th century from crossings of native horses with Thoroughbred. In 1995, there were 803 brood mares in activity. The sudden death of 2 full-sister mares with histologic features resembling EPSSM elicited a broader study on incidence, histologic, morphometric, and ultrastructural characteristics of the disorder in this breed.
Biopsies from the
Intracytoplasmic presence of amylase-resistant material, used as diagnosis criteria for EPSSM, was observed in biopsies of 20 out of 53 horses, either subsarcolemal or centrally located, displacing the myofibrils (Fig. 1). Aggregates of this material appeared gray with HES, purple with PAS and Best carmine, brown with Lugol's iodine and IKI-glycerol, blue with Alcian blue, and light blue with Hale following or not incubation with hyaluronidase. These were unstainable with methenamine silver, toluidine blue, Red Congo, Oil Red O, and acid phosphatase. PAS and Best carmine stains were resistant to α-amylase and diastase short digestion but sensitive to long digestion in diastase (4 hours and more). Type II fibers, showing a positive ATPase histoenzymatic reaction (pH = 10.4), were preferentially affected, although type I fibers could also be affected in severe cases. With PAS, aggregate-containing fibers showed a pale cytoplasm in comparison with adjacent aggregate-free fibers displaying purple to dark blue cytoplasm. This suggests an extensive intracytoplasmic accumulation of glycogen. Biopsies of 24 horses exhibited numerous subsarcolemmal vacuoles with granular basophilic material with HES. Ten plus or minus eight percent of the muscular fibers and up to 10% of the affected fiber area was involved. PAS staining was irregular and, where present, was α-amylase sensitive; other staining procedures were negative. No oxidative or ATPase activities were observed. In the majority of cases (n = 18 out of 24), both vacuoles and amylase-resistant material were present, but the numbers of vacuoles were not correlated with the numbers of material aggregates. In the other 6 cases, subsarcolemmal vacuoles were the sole myocytic lesion, and the corresponding horses were considered negative for EPSSM. Vacuoles were either rarely observed (less than 1% of fibers, n = 5) or numerous (7.3% of fibers, n =1). The last case corresponded to the biopsy of a 1-year-old horse, the myocytic vacuoles being associated with mild interstitial infiltration by macrophages and lymphocytes. Ultrastructural features in the 5 horses examined included large masses of nonmembrane-bound granular material with heterogeneous electron density that were unevenly distributed throughout the cytoplasm of the fibers. In some cases, filamentous material (2 to 4 μm wide, up to 15 μm long) was admixed. Myofibers also showed mild myofibrillar and mitochondrial loss.
Diameters of muscle fibers were measured using minimal Ferret. Type I fibers were too rarely observed to yield statistically relevant results. Diameters of type IIA and type IIB fibers were measured according to the absence of myocytic lesion, the presence of vacuoles, or the presence of aggregates (respectively 60.6 ± 21.3; 77.3 ± 18.4; 69.0 ± 22.3 μm for type IIA fibers; 53.1 ± 16.9; 72.1 ± 17.4; 66.0 ± 20.4 μm for type IIB fibers). Standard deviations were not significantly different between control fibers and vacuolated fibers, on the one hand, and between control fibers and aggregate-containing fibers, on the other hand. The minimal Ferret mean value for fibers containing vacuoles or aggregates was higher compared with the mean value for adjacent normal fibers (
Quantitative evaluation of the myocytic changes was performed on the biopsies of affected horses (Fig. 2). The percentages of affected muscle fibers were distributed bimodally, and 2 groups of biopsies can be distinguished: 1) 1 group composed of biopsies of 14 horses showing less than 3% (0.98 ± 0.92%) of type II fibers with aggregates occurring in the center of a single or small clusters of perifascicular fibers and 2) a second group of biopsies of 6 horses showing 9 to 30% (18.1 ± 5.7%) of fibers of both types, with aggregates evenly scattered through the sample. Segmental necrosis was also associated. There were no significant differences (
The necropsied horse belonged to the second group (18.9% of affected fibers). PAS-positive and amylase-resistant aggregates were observed in all sampled striated muscles, except in lingual muscle and in the masseter,
This report is the first description of the disease in the Cob Normand breed. In this breed, EPSSM has many features in common with the disease as described previously in other breeds, i.e., preferential involvement of type II fibers, material staining characteristics consistent with abnormal polysaccharide, and diffuse accumulation of excessive intramuscular glycogen. 4, 6 In this study, diameter of fibers with aggregates is shown to be larger compared with unaffected fibers of the same fiber type. This could be caused by compensatory hypertrophy or by the accumulation of abnormal material.
Definitive diagnosis of EPSSM relies on the pathologic evaluation of a muscle biopsy sample, although the criteria for histologic diagnosis are somewhat controversial. This report restrict diagnosis to the finding of PAS-positive and amylase-resistant material within muscle fibers, 4 but others extend diagnosis to include horses with only PAS-positive and amylase-sensitive vacuoles. 5, 9 As others draft horses, Cob Normand horses are prone to develop higher numbers of aggregates than others breeds. 9 The abundance of aggregates also increases with age, 2, 6 with the youngest affected animals of our study (3 years old, n = 3) typically exhibiting very few aggregates. Subsarcolemmal vacuoles are sometimes reported to be the first detectable finding in young affected horses, prior to the development of amylase-resistant aggregates. 2 In our study, this feature was observed in a 1-year-old horse for which a further biopsy to confirm diagnosis was refused by the owners.
PAS and Best carmine stains were consistent with the aggregates having a polysaccharide nature. Resistance to α-amylase digestion distinguishes this polysaccharide from glycogen. Moreover, the brown staining with iodine and the sensitivity to long diastase digestion distinguish the polysaccharide from amylopectin. 3 This partial resistance to glycogenolytic enzymes and the uneven electron density of the granular material may be related to an abnormal structure of the polysaccharide molecule. The blue Hale staining and the presence of large arrays of ultrastructural filamentous material can be attributed to the partial incorporation of another kind of molecule such as an acidic mucopolysaccharide, which could also explain the glycogenolytic resistance. Taken together, these features strongly suggest a polysaccharide mixture. Ubiquitine expression has been recently detected in muscle fibers of horses with EPSSM. This could reflect abnormal folding or other alteration of the protein component of the glycogen molecule. 8
Previous studies on muscular aggregates distribution focused on a few skeletal muscles including limb and epaxial muscles. 4, 6, 7, 9 Extensive numbers of striated muscles were evaluated here for the first time in 1 horse. The majority of striated muscles showed severe lesions, including myocardial and diaphragmatic muscles, as already reported in other draft horses. 9 Aggregates were also found in mononuclear cells of the deep inguinal lymph node. Polysaccharides from necrotic affected fibers can be phagocyted by muscular interstitial macrophages, 6 which could explain the presence of these polysaccharides in macrophages in lymph nodes draining severely affected muscles. Presence of aggregates in smooth muscle is reported here for the first time in a polysaccharide storage disease. In this aging animal, polyglucosan Lafora-type bodies and lipofuscin were ruled out, as the former appears black with methenamine silver and blue with Lugol's iodine 1 and the latter appears red with Oil Red O and has acid phosphatase activity. Further study of these rarely affected tissues is clearly needed to identify reasons and mechanisms involved in broad amylase-resistant material deposition.
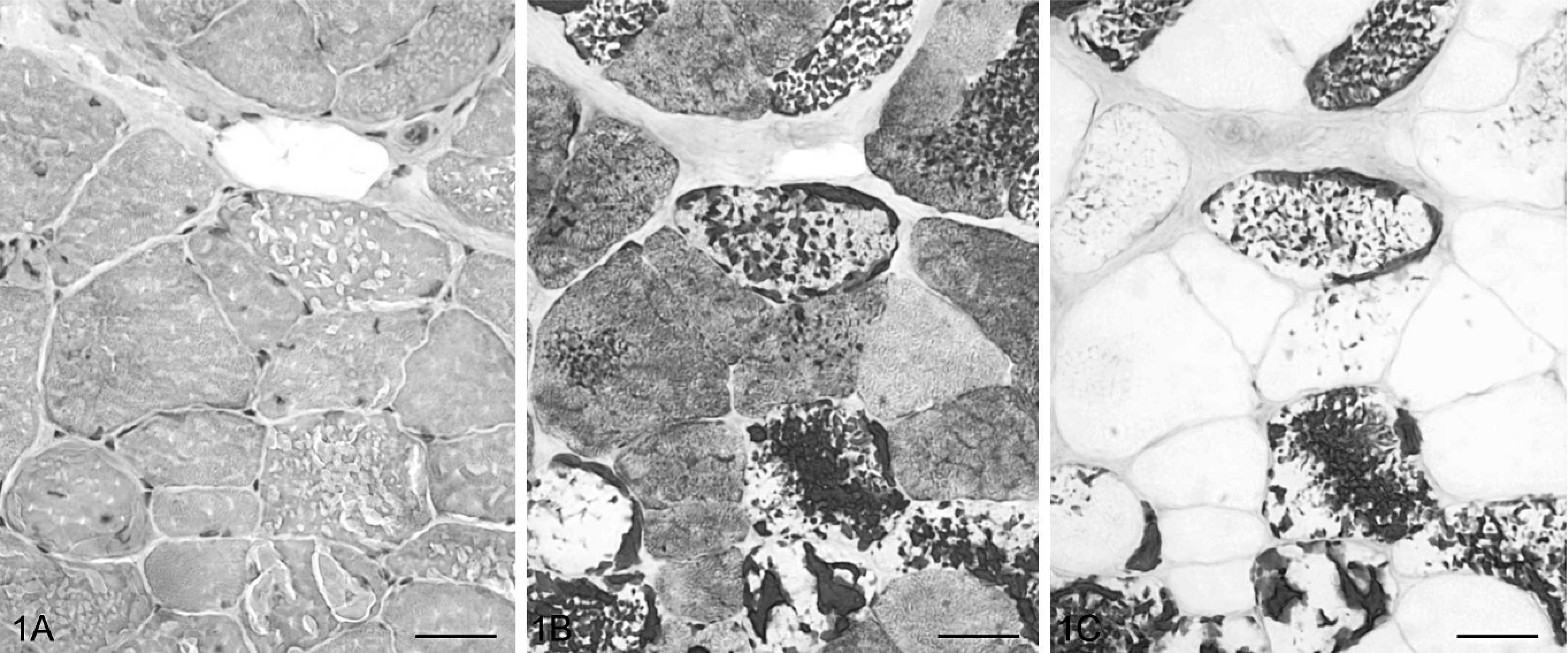
Serial transverse sections of gluteal muscle; Cob Normand horse with polysaccharide storage myopathy (EPSSM). Numerous fibers contained large aggregates, either peripheral or intermyofibrillar, stained with PAS and showing α-amylase resistance. HES (1A), PAS (1B), PAS following α-amylase digestion (1C). Bar = 20 μm.
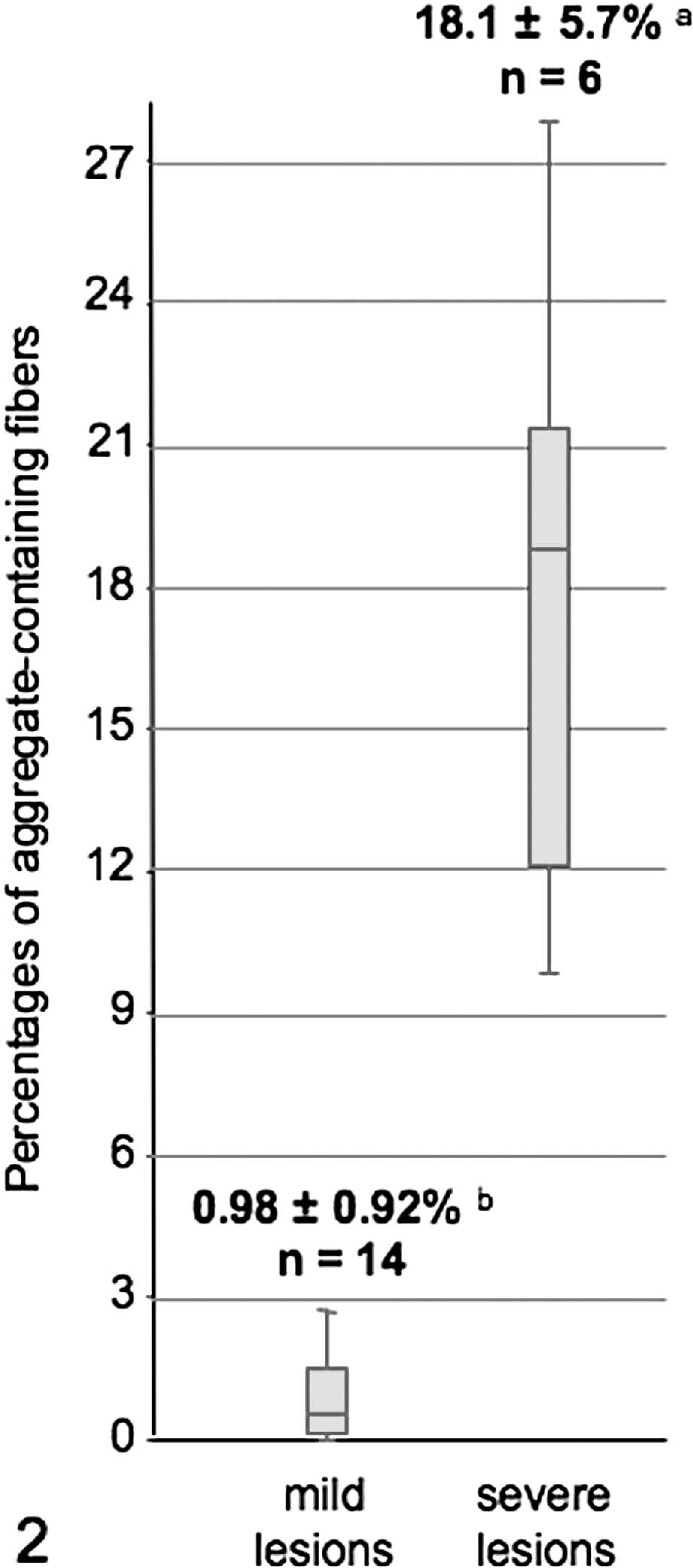
Mean percentages of aggregate-containing fibers from 2 muscle biopsy groups with distinct histological presentation of EPSSM. a and b were significantly different. n = number of horses.
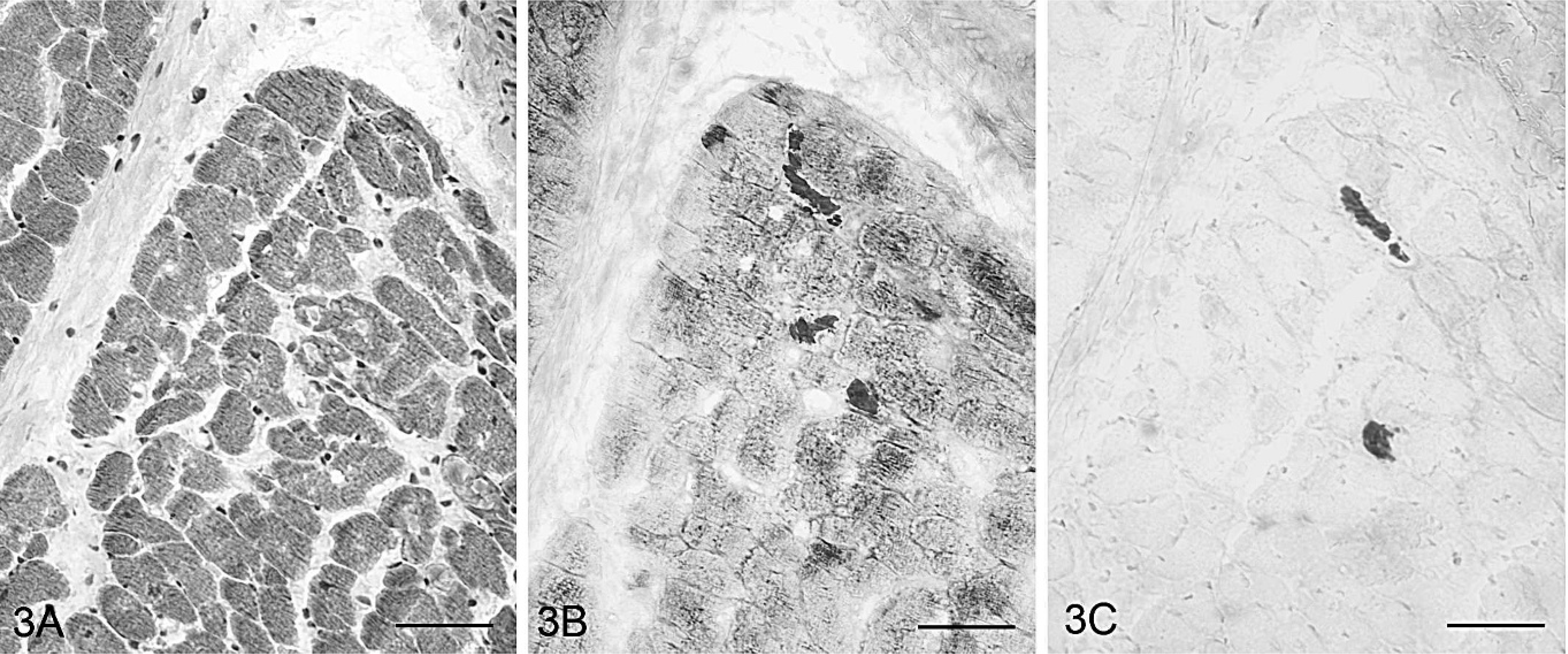
Serial sections of heart; Cob Normand horse with EPSSM. Some cardiac muscle fibers contained aggregates stained with PAS and showing α-amylase resistance. HES (3A), PAS (3B), PAS following α-amylase digestion (3C). Bar = 20 μm.
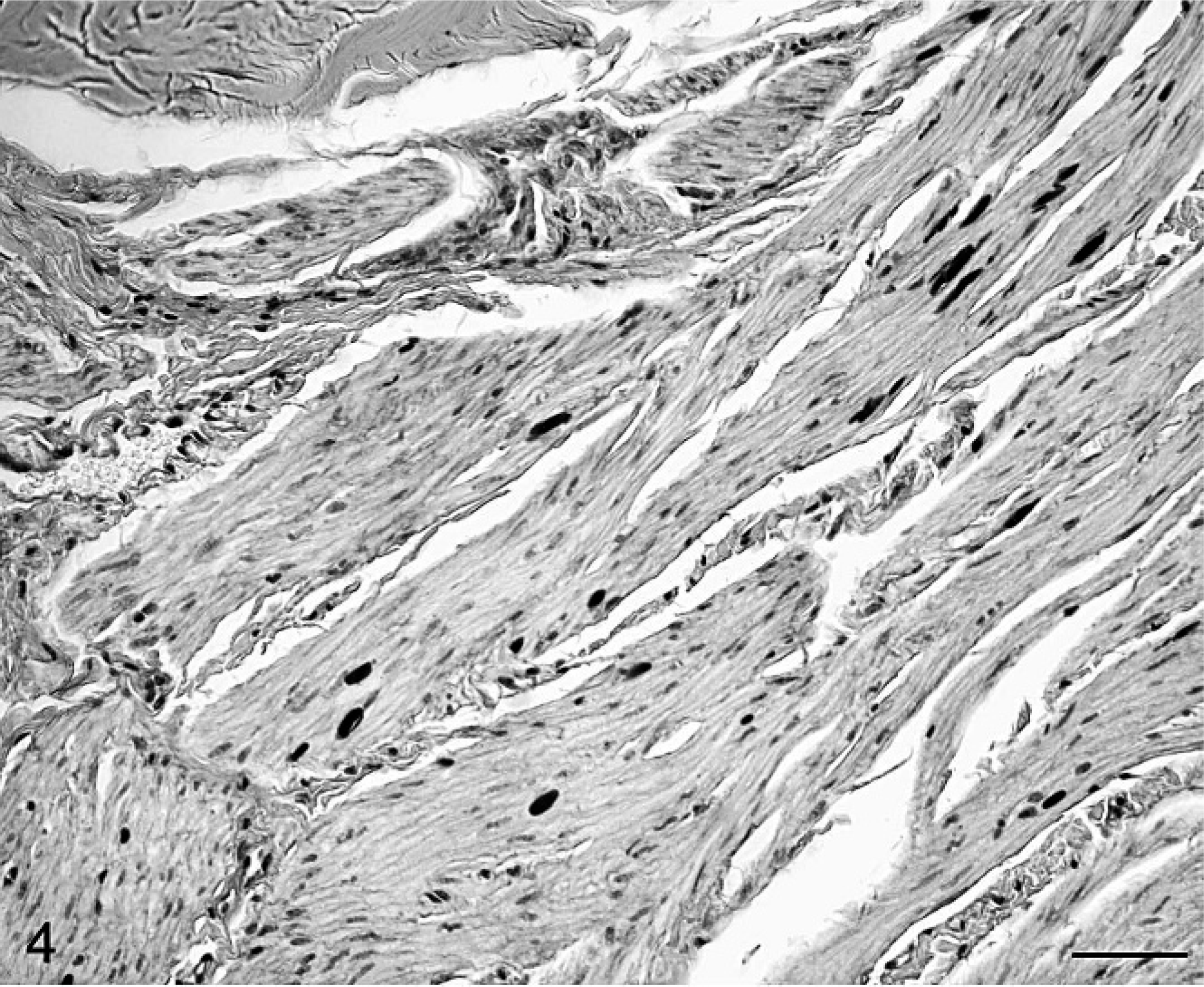
Bladder; Cob Normand horse with EPSSM. PAS-positive and amylase-resistant aggregates were observed in cavernous muscle fibers. PAS following α-amylase digestion. Bar = 20 μm.
Footnotes
Acknowledgement
This project was supported by the Haras Nationaux.
