Abstract
The purpose of the study reported here was to determine the effect of three methods of fixation of skeletal muscle biopsy specimens on the histopathologic appearance of muscle sections and to determine criteria that were most consistently associated with a diagnosis of polysaccharide storage myopathy (PSSM) in horses. Surgically excised semimembranosus muscle biopsy specimens were obtained from nine horses previously diagnosed with PSSM and from 15 control horses. Portions of each specimen were fixed in formalin, frozen immediately, and chilled for 24 hours prior to freezing. Sections stained with hematoxylin and eosin (HE), periodic acid-Schiff (PAS), and amylase-PAS were scored for histopathologic criteria by three investigators blinded to the sample origin. The presence of amylase-resistant, abnormal polysaccharide was found to be the most sensitive and specific diagnostic indicator for PSSM, and was readily detected regardless of the fixation technique or investigator. Other less-specific features associated with PSSM included atrophy and cytoplasmic and subsarcolemmal vacuoles; however, their histologic scores varied among fixation technique and investigators. Scores for subsarcolemmal and cytoplasmic amylase-sensitive glycogen in horses with PSSM were similar to those for control horses and varied among fixation techniques. In conclusion, PSSM is most accurately diagnosed in muscle biopsy specimens on the basis of appearance of amylase-resistant, abnormal polysaccharide, not amylase-sensitive glycogen, regardless of fixation technique. In general, frozen sections appeared to be better suited for studying myopathies because many histopathologic features of skeletal muscle were obscured by formalin fixation.
An equine glycogen storage disorder called polysaccharide storage myopathy (PSSM) was first described in 1992 in nine Quarter Horse-related breeds that had a history of chronic exertional rhabdomyolysis. 21 The disorder was characterized by the presence of periodic acid–Schiff (PAS)-positive inclusions in the cytoplasm of muscle cells that were resistant to amylase digestion in frozen sections of skeletal muscle biopsy specimens. 21, 23 Electron microscopy indicated that the PAS-positive inclusions consisted of β-glycogen particles and an array of filamentous material. 21 In addition, skeletal muscle fibers from some of the affected horses had increased staining for normal amylase-sensitive glycogen, subsarcolemmal vacuoles, necrosis, atrophy, and regeneration. 21 Since this first account, PSSM has also been reported in a variety of other breeds including Draft Horses 5, 20, 26, 29– 31 and Warmbloods. 17 Although the clinical presentation of PSSM in these breeds of horses varies from no clinical signs of disease 26, 28 to muscle soreness, exertional rhabdomyolysis, gait abnormalities, severe weakness, and recumbency, 5, 20, 26– 31, 34 the histopathologic findings on evaluation of skeletal muscle biopsy specimens seemingly are identical. 17, 21, 29
The “gold standard” for diagnosis of PSSM in horses is generally accepted to be the presence of amylase-resistant, PAS-positive, abnormal polysaccharide inclusions in skeletal muscle fibers. 12, 14, 17, 21, 23 However, others have suggested that excess amylase-sensitive glycogen in the absence of complex polysaccharide may also be used to diagnose PSSM. 27, 28, 30– 33 This alternate diagnostic criterion has resulted in a wide spectrum of breeds diagnosed with PSSM and a high purported prevalence of the disease. 31, 33 A prevalence of 33% of all horses outside of draft and Quarter Horse bloodlines 28 and up to 80% of draft horse breeds 5, 31 has been suggested for PSSM when amylase-resistant, abnormal polysaccharide is not a required diagnostic criterion. These high estimates of the prevalence of PSSM suggest that the use of the criterion of increased amylase-sensitive glycogen to diagnose PSSM may lack specificity for PSSM, leading to misinterpretation of the breeds affected by this disorder.
Muscle biopsy is becoming a routine diagnostic technique for veterinary practitioners when evaluating horses with muscular disorders, 14 and several diagnostic laboratories are now examining muscle biopsy specimens for PSSM. Some neuromuscular laboratories receive fresh chilled muscle biopsy specimens shipped overnight to the laboratory. These specimens are frozen the next day using specialized equipment for handling, processing, and storage. 19 Other laboratories receive formalin-fixed specimens that are processed by use of routine methods. Formalin fixation is an easier method of tissue preparation, compared with the expertise required to avoid freeze artifact formation. The disadvantage of formalin fixation of a highly metabolically active tissue such as skeletal muscle is that muscle fibers may shrink by as much as 30%, enzyme activity is reduced or abolished, and storage products such as glycogen and lipids are not optimally retained. 6, 10 In addition, it is impossible to identify metabolic or contractile muscle fiber types in formalin-fixed muscle.
On the other hand, preparation of muscle biopsy specimens for freezing followed by sectioning has the disadvantage of a delay in fixation of tissue during shipping and the requirement of specialized equipment. 19 As yet and to the authors' knowledge, there is no specific information regarding whether and how accurately a diagnosis of PSSM can be established in formalin-fixed specimens, compared with specimens frozen immediately and specimens shipped on ice packs prior to freezing. Establishing specific diagnostic standards for all laboratories is important for the accurate diagnosis of this disorder, particularly because this diagnosis is used by owners in their breeding programs.
The purpose of the study reported here was to determine: 1) the effect of various methods of preparing muscle biopsy specimens (immediately frozen, chilled then frozen, and formalin fixed) on the histopathologic characteristics of PSSM; 2) the most specific diagnostic criteria for PSSM, and 3) the variation in histopathologic interpretation of skeletal muscle biopsy specimens between observers.
Materials and Methods
Horses
Twenty-four adult horses older than 2 years were studied. Of these horses, nine were Quarter Horse/Quarter Horse-cross previously diagnosed with PSSM. A diagnosis of PSSM was based on a history of clinical signs of exertional rhabdomyolysis during treadmill exercise testing, increased 4-hour postexercise serum creatine kinase (CK) activity, skeletal muscle glycogen concentration >1.5 times normal, and the presence of amylase-resistant, PAS-positive, abnormal polysaccharide inclusions found in specimens from repeated gluteus medius skeletal muscle biopsy. The control population consisted of 10 horses of Quarter Horse-type that had been obtained from a local auction for the University of Minnesota Teaching herd. These horses had no available pedigree records and were kept in tie stalls with 4 hours of turnout in a paddock daily. Five of these horses were sufficiently sound to perform a 15-minute treadmill exercise test with no clinical signs of exertional rhabdomyolysis and no increase in 4-hour postexercise serum CK activity. A disease control group consisted of five Thoroughbreds previously diagnosed with recurrent exertional rhabdomyolysis (RER), on the basis of increased 4-hour postexercise serum CK activity, abnormal muscle contracture response to caffeine and halothane, 15 and normal muscle glycogen concentration.
Muscle biopsy
An open surgical biopsy specimen from the semimembranosus muscle was obtained from all 24 horses under local anesthesia. A line block was performed using 2% lidocaine injected beneath the skin avoiding infiltration into the underlying muscle. A 3-cm vertical incision, starting 5 cm distal to the tuber ischii, was made through the skin and underlying fascia. Two parallel incisions into the muscle, 2 cm apart and 2 cm deep, were made longitudinal to the muscle fibers, taking care to handle the muscle in only one corner using forceps. Dorsal and ventral transverse incisions 5 cm apart were then made to free the muscle biopsy specimen. After excision, the end held by the forceps was trimmed with a sharp razor blade and the biopsy specimen was divided into three 1 × 2-cm cross-sections. The fascia and skin were then closed with sutures and staples, respectively.
Tissue fixation
One section of muscle (known as “frozen immediately”) was wrapped in saline-moistened gauze and taken directly to the laboratory. The cross-section of tissue was placed in optimal cutting temperature a mounting medium on thin cork squares and plunged for 20 seconds into 400 ml of isopentane previously chilled in liquid nitrogen until a 1-cm ring of solidified isopentane formed over top of the liquid isopentane. The specimen was then removed from the isopentane, placed in a sealed airtight plastic bag and kept at − 80°C until processing. The second portion of muscle (known as “chilled”) was wrapped in saline-moistened gauze in an airtight container and placed in a refrigerator at 4°C for 24 hours (to imitate overnight shipping of the specimen on ice packs to the laboratory), then was frozen in chilled isopentane and stored as the immediately frozen sample. The remaining piece of muscle (known as “formalin-fixed”) was kept in air for 3 minutes, then was aligned in cross-section in a holding cassette and placed in buffered 10% formalin for 48 hours prior to embedding in paraffin by personnel at the University of Minnesota Veterinary Diagnostic Laboratory's (UMNVDL) histopathology service. This resulted in a total of 72 skeletal muscle biopsy specimens for evaluation (24 horses × 3 methods of fixation).
Staining
Ten-micrometer-thick sections of frozen muscle were stained with hematoxylin and eosin (HE) and periodic acid–Schiff (PAS) by use of routine methods. For the amylase-PAS stain, sections were incubated in 0.35% amylase b solution for 15 minutes at 38°C and rinsed thoroughly prior to performing PAS staining with hematoxylin counterstaining. Four-micrometer-thick sections of formalin-fixed, paraffin-embedded muscle were treated with the HE, PAS, and amylase-PAS stains with methods routinely used at the UMNVDL. Sections of frozen and formalin-fixed kidney served as positive controls for the PAS and amylase-PAS stains.
Each set of 3 stained muscle specimens (HE, PAS, and PAS-amylase) was randomly assigned a number (1–72) and evaluated by one experienced investigator (SJV) blinded as to the method of preparation and identity of the horse. Two other investigators (AMF, EJA) familiar with interpreting muscle biopsy specimens also evaluated all slides to determine variability between interpreters.
The presence or absence (1 = present, 0 = absent) of the following features was determined: muscle fiber size variation (HE), muscle necrosis with macrophage infiltration (HE), cytoplasmic, rimmed vacuoles characterized by a vacuole surrounded by a basophilic ring (HE), cytoplasmic, nonrimmed vacuoles (HE), anguloid atrophy (HE), centrally located nuclei (HE), glycogen-depleted/lytic cells characterized by absence of glycogen staining or disruption of sarcolemmal membranes, or both (HE, PAS), and PAS-positive sarcoplasmic masses characterized by central PAS-positive homogenous material (PAS) evident in one focal plane. 1 The intensity of PAS staining was graded as: 1 = light pink, 2 = dark pink, and 3 = deep purple. Amylase-resistant, crystalline, abnormal polysaccharide (amylase-PAS) was scored as: 0 = not present, 1 = present in >1 fiber in one 20x random microscopic field (rmf), 2 = present in >1 fiber in 2 rmf, and 3 = present in >1 fiber in ≥3 rmf. Granular cytoplasmic glycogen (PAS), subsarcolemmal glycogen (PAS), and subsarcolemmal vacuoles (HE, PAS) also were graded. Subsarcolemmal vacuoles were scored for the HE and PAS stains because their appearance varied between the two stains. The scoring system used for these features at 20x magnification was: 0 = not present, 1 = present in >1 fiber in 1 rmf, 2 = present in >1 fiber in 2 rmf, 3 = present in >1 fiber in 3 rmf, and 4 = present in >1 fiber in ≥4 rmf.
Once all investigators completed muscle biopsy specimen evaluations, the key to the identity and previously established diagnosis was revealed. The true phenotype was based on results of the clinical, histologic and physiologic tests performed previously that identified horses as PSSM, control, or RER.
The most accurate method of PSSM diagnosis identified from the first part of this study was reassessed by a board certified veterinary pathologist (DWH) not previously experienced with evaluation of frozen or formalin muscle biopsy sections for PSSM. A random selection of muscle biopsy specimens was evaluated, and the accuracy of the diagnosis was then compared with the previously determined phenotypic diagnosis.
Statistical analysis
Contingency table analyses (chi-square tests) were used to compare the difference in the appearance of the 14 histologic features among the three fixation methods (frozen immediately versus chilled and formalin-fixed, chilled versus frozen immediately and formalin-fixed, and formalin-fixed versus frozen immediately and chilled). Contingency table analyses (chi-square tests) also were used to determine whether there was a difference in the appearance of the 14 histologic features between control horses with no signs of exertional rhabdomyolysis and control horses with RER. Since no difference in any of the features was found, the two control groups were combined to produce one control group of 15 horses for comparison with nine horses with PSSM. Contingency table analyses (chi-square tests) were then performed comparing all 15 control horses with horses with PSSM to determine whether a relationship existed between each histologic feature and diagnosis of PSSM.
A score was calculated for each muscle section (including all three fixation techniques) as the sum of the scores given to the histologic features that were determined to be associated with a diagnosis of PSSM. This was reported as the mean ± SEM score for horses with PSSM and control horses. The sensitivity of using the best-derived diagnostic criteria to diagnose PSSM was calculated for each fixation method as the number of horses that tested positive for PSSM divided by the total number of true positives; the specificity was calculated as the number of horses that tested negative for PSSM divided by the total number of true negatives. Positive predictive value (PPV) was calculated as the number of horses that actually had PSSM and tested positive divided by the total number of horses that tested positive. The negative predictive value (NPV) was determined as the number of horses that did not have PSSM and tested negative divided by the total number of horses that tested negative.
The individual interpretations of muscle biopsy specimens were compared among the three evaluators to determine whether there was significant interindividual variation using a kappa analysis. A simple kappa analysis was performed to evaluate variation among investigators for features that were determined to be present or absent. Weighted kappa analysis was used to determine the variation in agreement among evaluators when a scoring system of 0–4, 1–3, or 0–3 was used, such that a difference between a score that was one grade apart was given less emphasis than a score that was two or three grades apart. A kappa value that was closer to 1 indicated greater agreement among the investigators.
Statistical analyses were conducted with use of computer software programs.
c
,
d
,
e
For all analyses, a value of
Results
Fixation techniques
Formalin-fixed sections
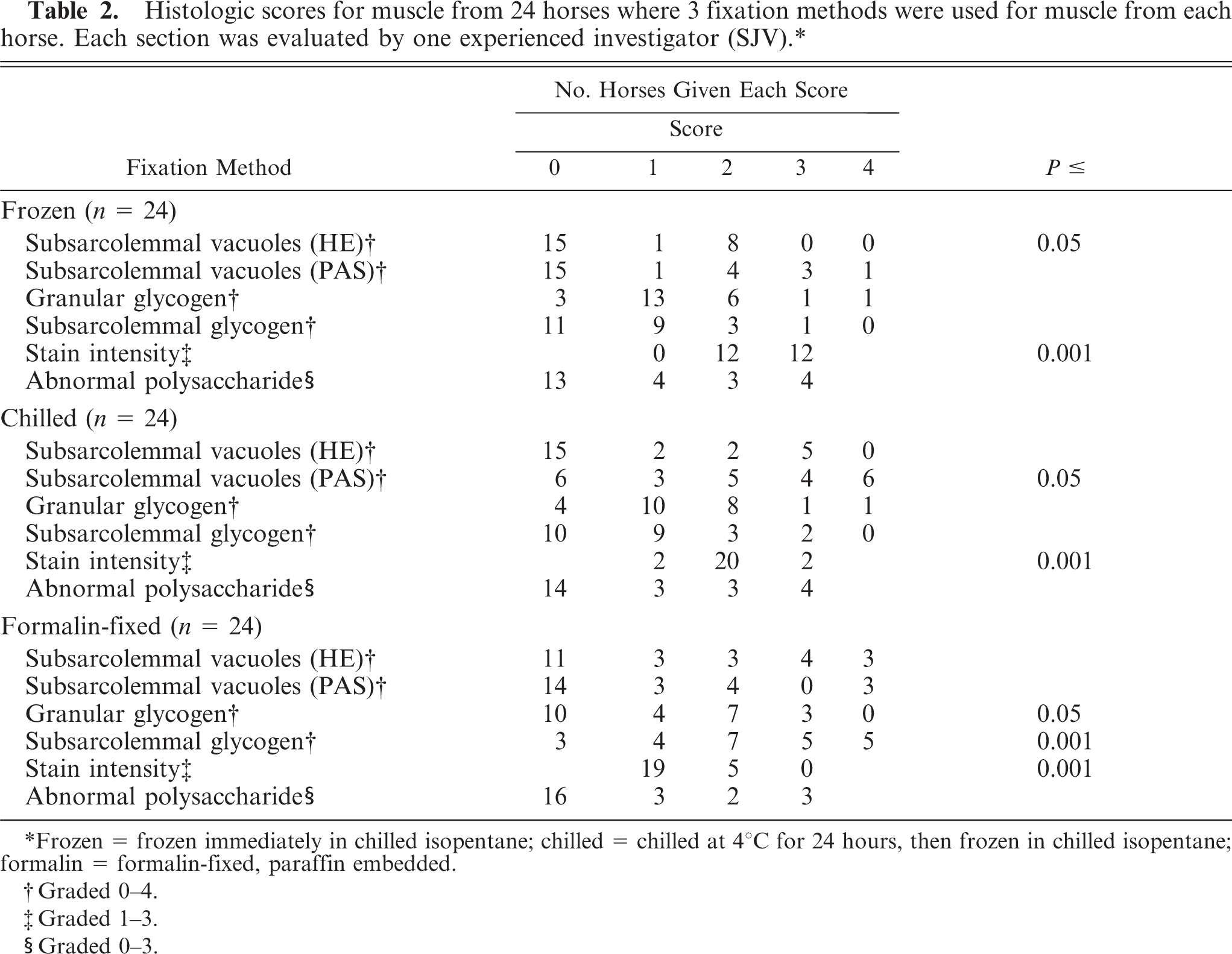
Fiber size variation, anguloid atrophy, central nuclei, and rimmed vacuoles were not as readily apparent in formalin-fixed sections of skeletal muscle, compared to sections that were frozen immediately or chilled (Table 1, Figs. 1, 2). In fact, sarcoplasmic masses and rimmed vacuoles were not observed in formalin-fixed sections, but were apparent in frozen immediately (Figs. 3, 4) and chilled sections. Formalin-fixed sections contained significantly more cytoplasmic vacuoles, more subsarcolemmal glycogen, and less granular glycogen than that in sections that were frozen immediately or chilled (Tables 1, 2). The PAS stain was lighter (Table 2) and glycogen tended to sediment to one end of each cell in some areas of the formalin-fixed PAS stain (Figs. 4–6). Lighter PAS staining was expected as thinner sections were used with formalin fixation (Figs. 5–10). There was no significant difference in the frequency of observation of amylase-resistant, abnormal polysaccharide (Figs. 5, 7, 9, 11) or necrosis or subsarcolemmal vacuoles in the HE stain between formalin-fixed and frozen sections (Tables 1, 2).
Presence of histologic features in muscle from 24 horses where three fixation techniques were used for each horse. Each section was evaluated by one experienced investigator (SJV).∗
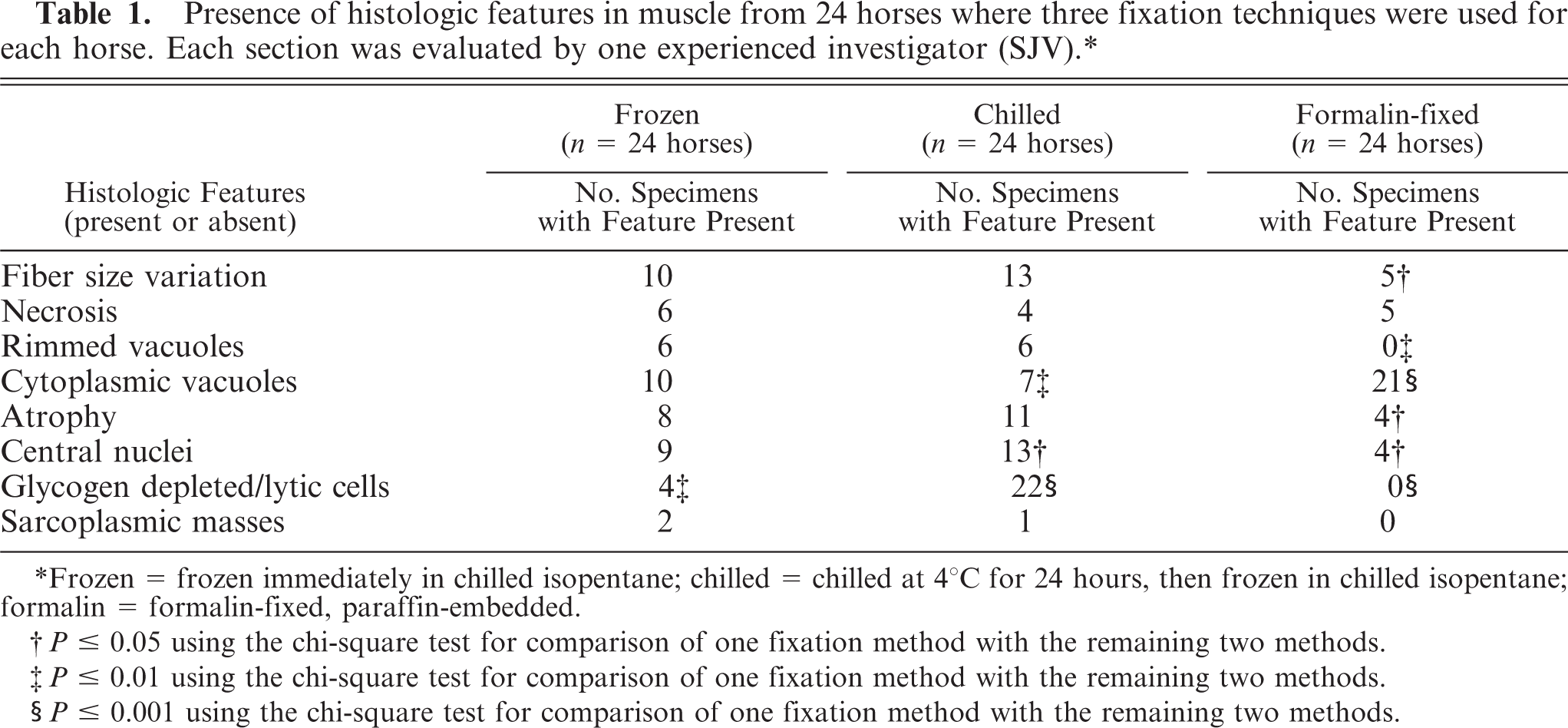
Frozen = frozen immediately in chilled isopentane; chilled = chilled at 4°C for 24 hours, then frozen in chilled isopentane; formalin = formalin-fixed, paraffin-embedded.
Histologic scores for muscle from 24 horses where 3 fixation methods were used for muscle from each horse. Each section was evaluated by one experienced investigator (SJV).∗
Frozen = frozen immediately in chilled isopentane; chilled = chilled at 4°C for 24 hours, then frozen in chilled isopentane; formalin = formalin-fixed, paraffin embedded.
Graded 0–4.
Graded 1–3.
Graded 0–3.
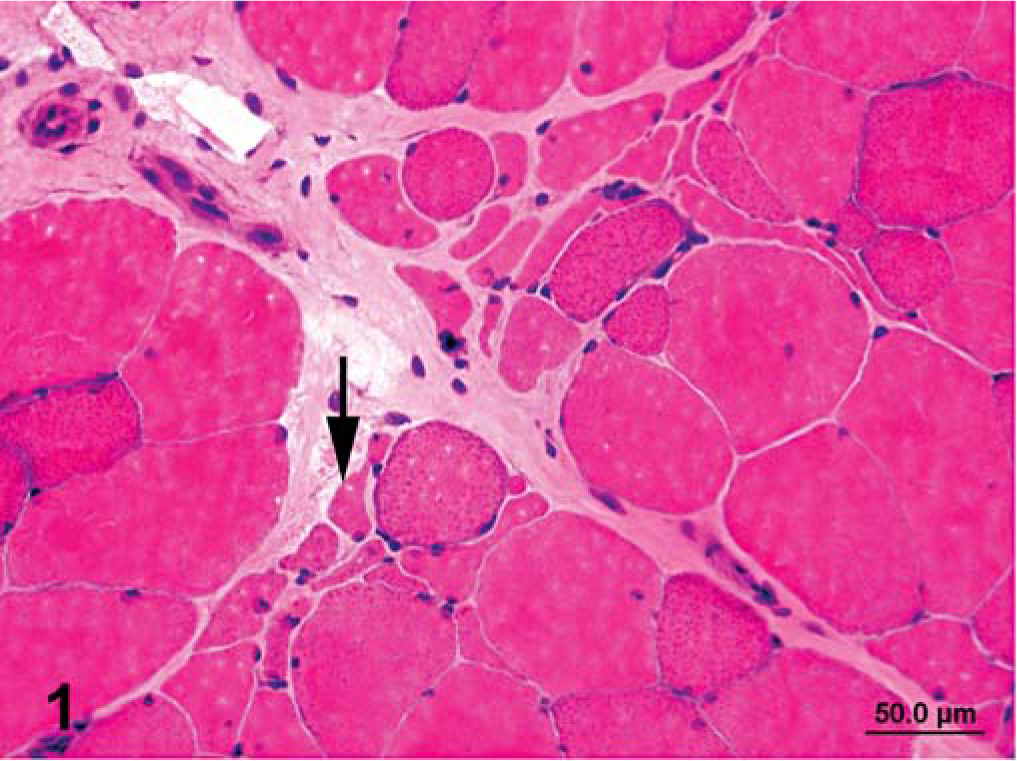
Semimembranosus muscle that was frozen immediately in chilled isopentane; horse No. 1, control Thoroughbred mare with recurrent exertional rhabdomyolosis (RER). Notice clusters of cells (arrow) with a concave appearance typical of angular atrophy. HE. Bar = 50 µm.
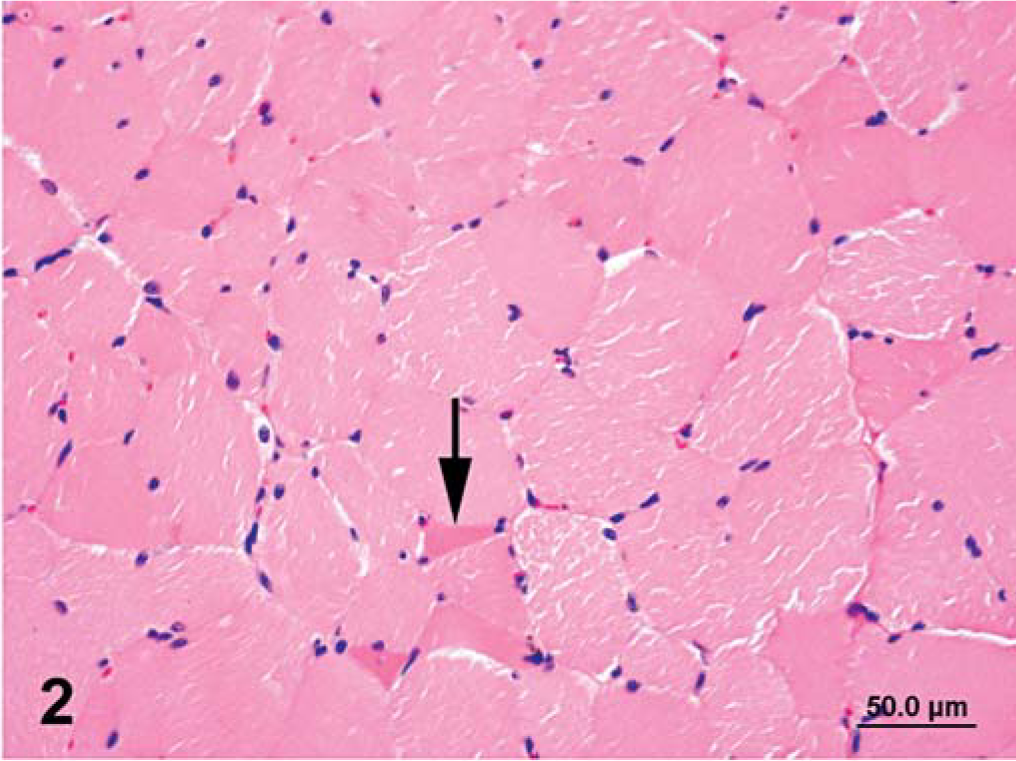
Semimembranosus muscle that was formalin fixed and paraffin embedded; horse No. 1, control Thoroughbred mare with RER. Angular atrophied fibers are less distinct (arrow) than those in a muscle specimen that was frozen immediately. Notice also widespread artifactual cracking. HE. Bar = 50 µm.
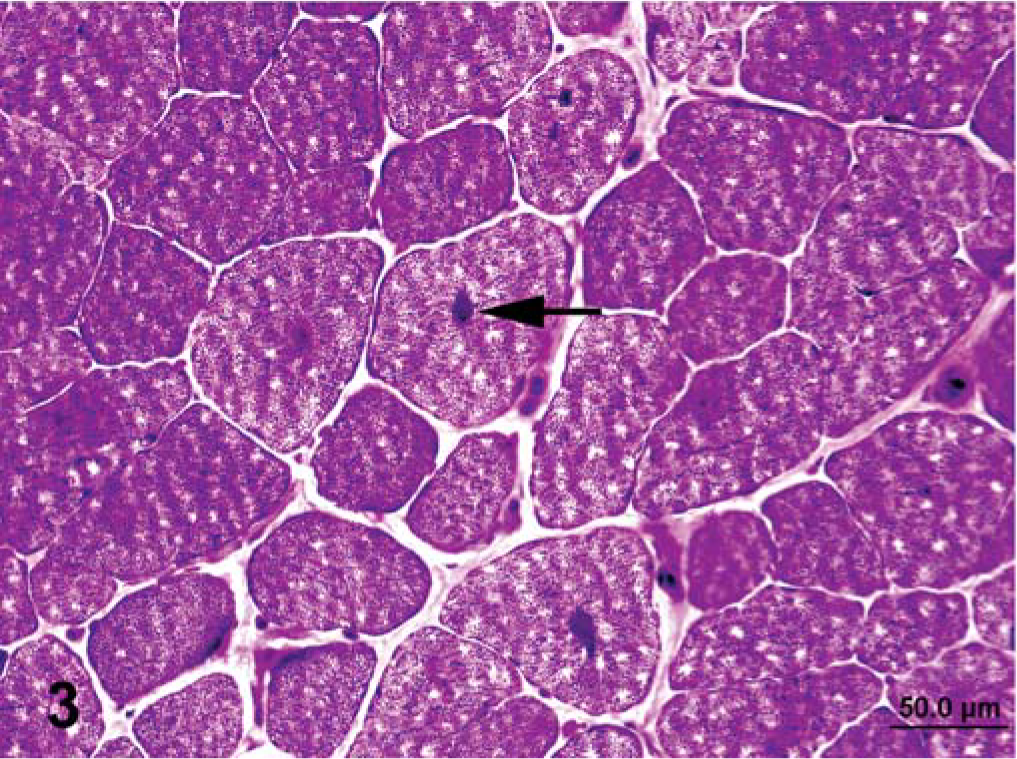
Semimembranosus muscle that was frozen immediately in chilled isopentane; horse No. 2, control Quarter Horse mare. Notice sarcoplasmic masses (arrow) and small amounts of subsarcolemmal glycogen. Notice also the widespread presence of small vacuoles resembling freeze artifacts. PAS. Bar = 50 µm.
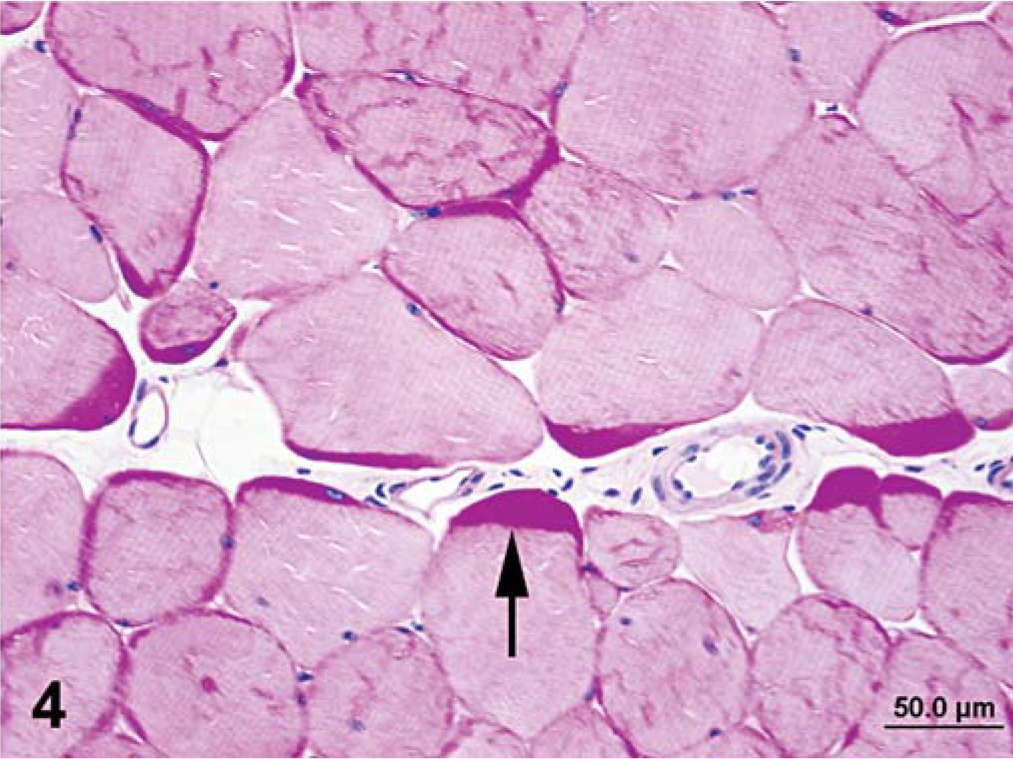
Semimembranosus muscle that was formalin fixed and paraffin embedded; horse No. 2, control Quarter Horse mare. Notice typical appearance of thick deposits of subsarcolemmal glycogen (arrow), lack of sarcoplasmic masses, and presence of artifactual cracking when this method of fixation is used. PAS. Bar = 50 µm.
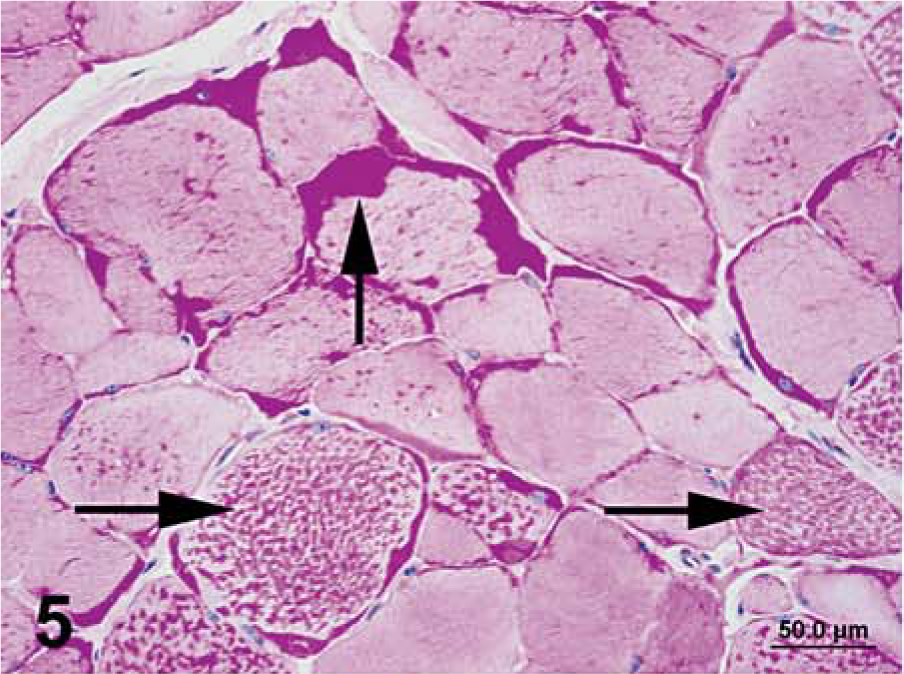
Semimembranosus muscle that was formalin fixed and paraffin embedded; horse No. 3, Quarter Horse mare with polysaccharide storage myopathy (PSSM). Notice intracytoplasmic accumulations of abnormal polysaccharide (horizontal arrows) and subsarcolemmal glycogen (vertical arrow) PAS. Bar = 50 µm.
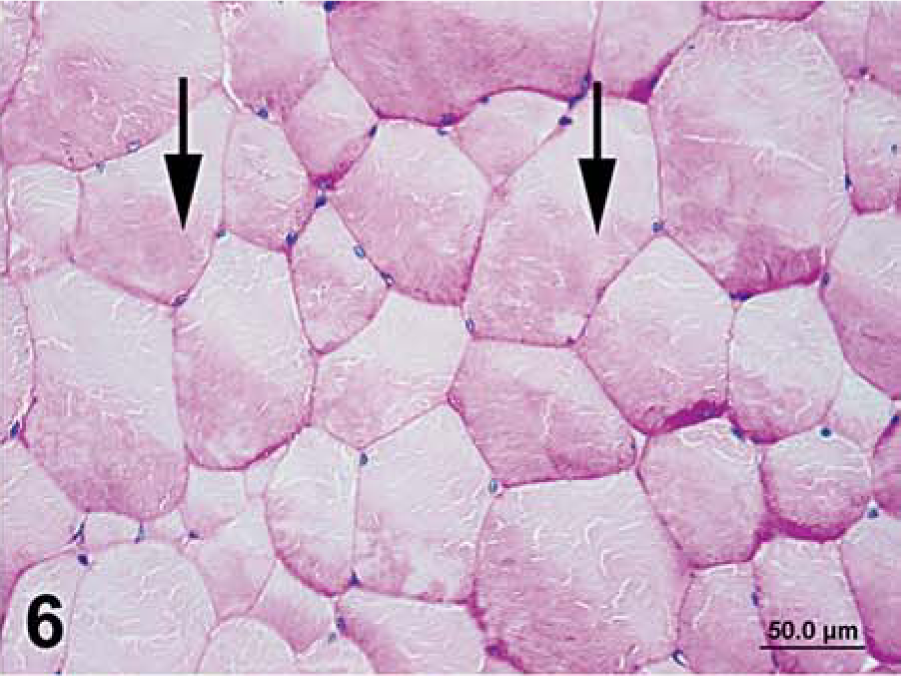
Semimembranosus muscle that was formalin-fixed and paraffin embedded; horse No. 4, control Quarter Horse mare. Notice settling of glycogen in one portion of the cell (arrows). PAS. Bar = 50 µm.
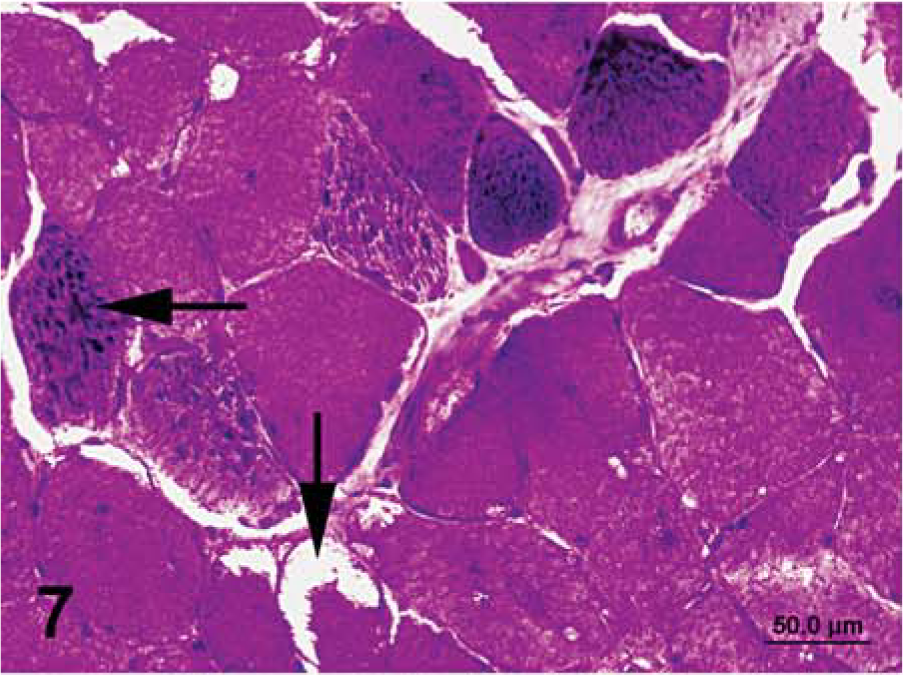
Semimembranosus muscle that was chilled for 24 hours, then frozen in chilled isopentane; horse No. 3, Quarter Horse mare with PSSM. Notice intracytoplasmic accumulations of abnormal polysaccharide (horizontal arrow), subsarcolemmal vacuoles (vertical arrow), and lighter staining intensity, compared with that of immediately frozen sections. PAS. Bar = 50 µm.
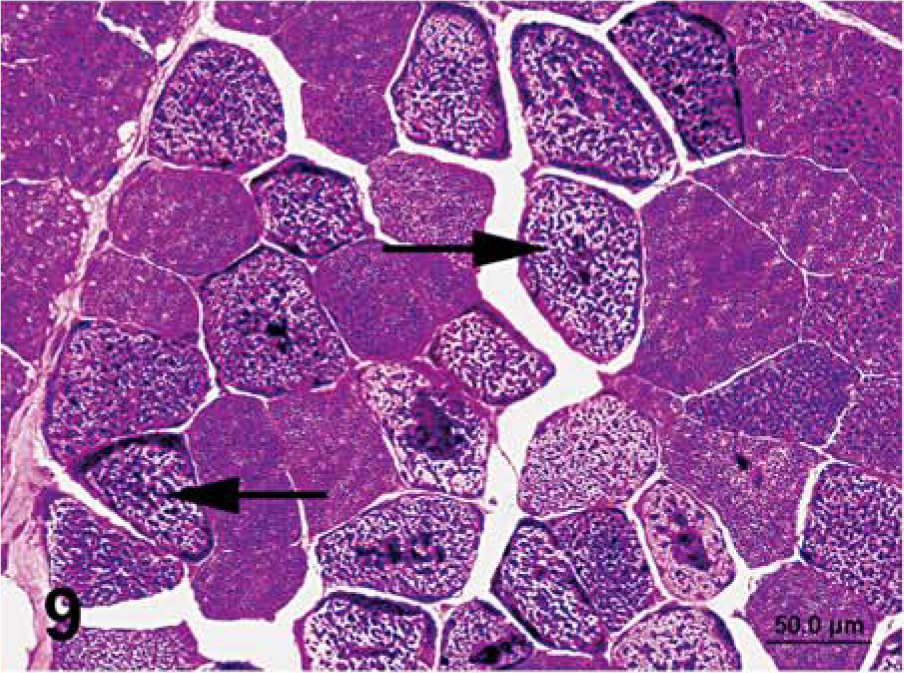
Semimembranosus muscle that was frozen immediately in chilled isopentane; horse No. 3, Quarter Horse mare with PSSM. Notice intracytoplasmic accumulations of abnormal polysaccharide (arrows). PAS. Bar = 50 µm.
Semimembranosus muscle that was frozen immediately in chilled isopentane; horse No. 4, control Quarter Horse mare. Notice absence of amylase-resistant, abnormal polysaccharide in the control horse. PAS. Bar = 50 µm.
Semimembranosus muscle that was frozen immediately in chilled isopentane; horse No. 3, Quarter Horse mare with PSSM. Notice intracytoplasmic accumulations of abnormal polysaccharide are resistant to digestion with amylase (arrows). PAS plus amylase. Bar = 50 µm.
Frozen sections
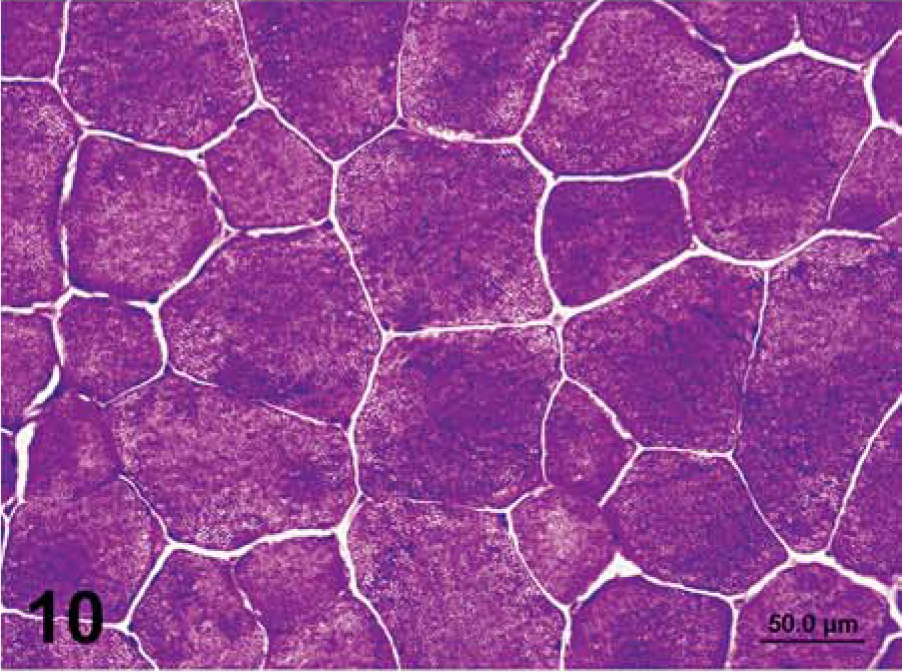
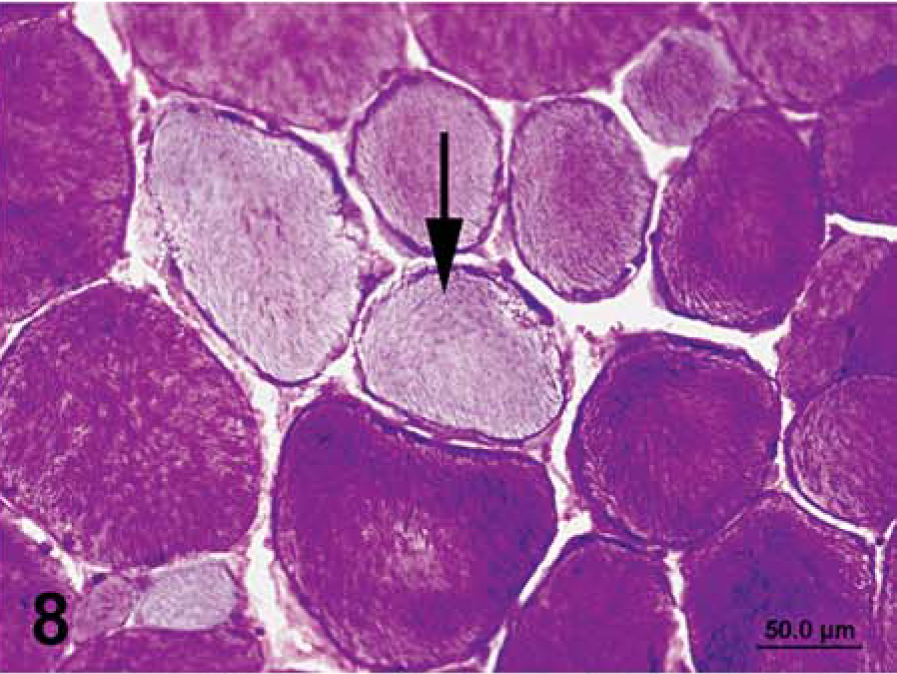
Fiber size variation, anguloid atrophy (Fig. 1), central nuclei, macrophage infiltration, rimmed vacuoles, subsarcolemmal vacuoles (Fig. 7), sarcoplasmic masses (Fig. 3), and amylase-resistant, abnormal polysaccharide (Fig. 9, 11) were readily apparent in sections that were frozen immediately (Tables 1, 2). Sections that were frozen immediately had more subsarcolemmal vacuoles visible by HE staining and darker PAS staining (Fig. 10), compared with sections generated by use of the other fixation techniques (Table 2). In some central areas of muscle sections, small cytoplasmic vacuoles were apparent that were typical of ice crystal formation developing during the freezing process (freeze artifact) (Fig. 3).
Chilled sections
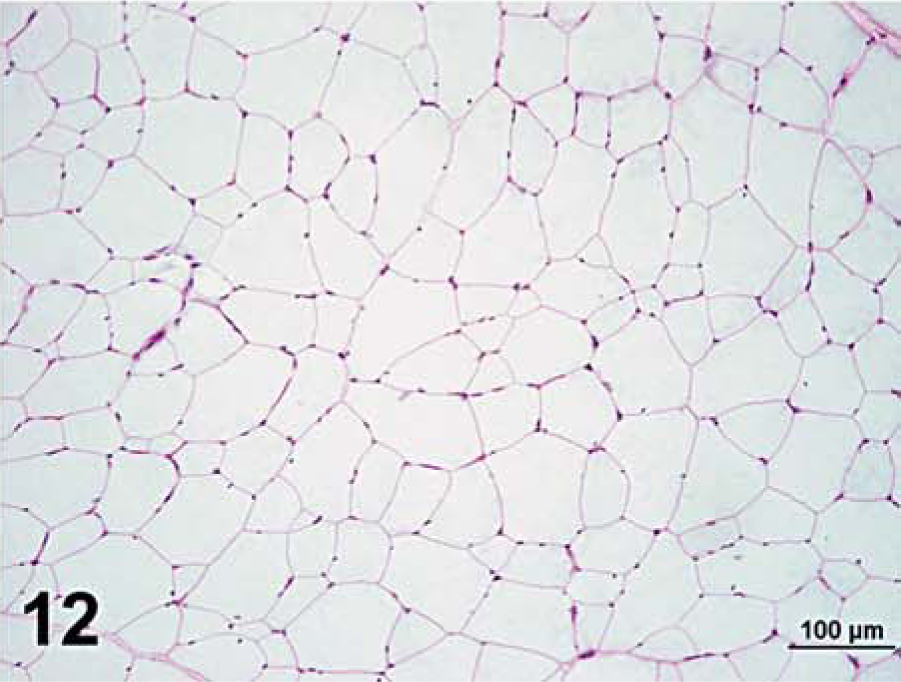
Sections that were chilled for 24 hours before freezing had a higher proportion of glycogen-depleted/lytic cells (92% of sections), compared with sections that were frozen immediately (or formalin-fixed and paraffin embedded (Table 1, Fig. 8). In addition, subsarcolemmal vacuoles were more likely to be observed in the PAS-stained specimens that were chilled before freezing, compared with the other fixation methods (Table 2, Fig. 7). Specimens chilled prior to freezing also were generally lighter in PAS staining intensity relative to specimens frozen immediately (Table 2). In some central areas of muscle sections, small cytoplasmic vacuoles typical of freeze artifact were apparent.
Semimembranosus muscle that was chilled for 24 hours, then frozen in chilled isopentane; horse No. 4, control Quarter Horse mare. Notice glycogen-depleted cells (arrow) and lighter staining intensity, compared with that of immediately frozen sections. PAS. Bar = 50 µm.
Histologic features of control horses
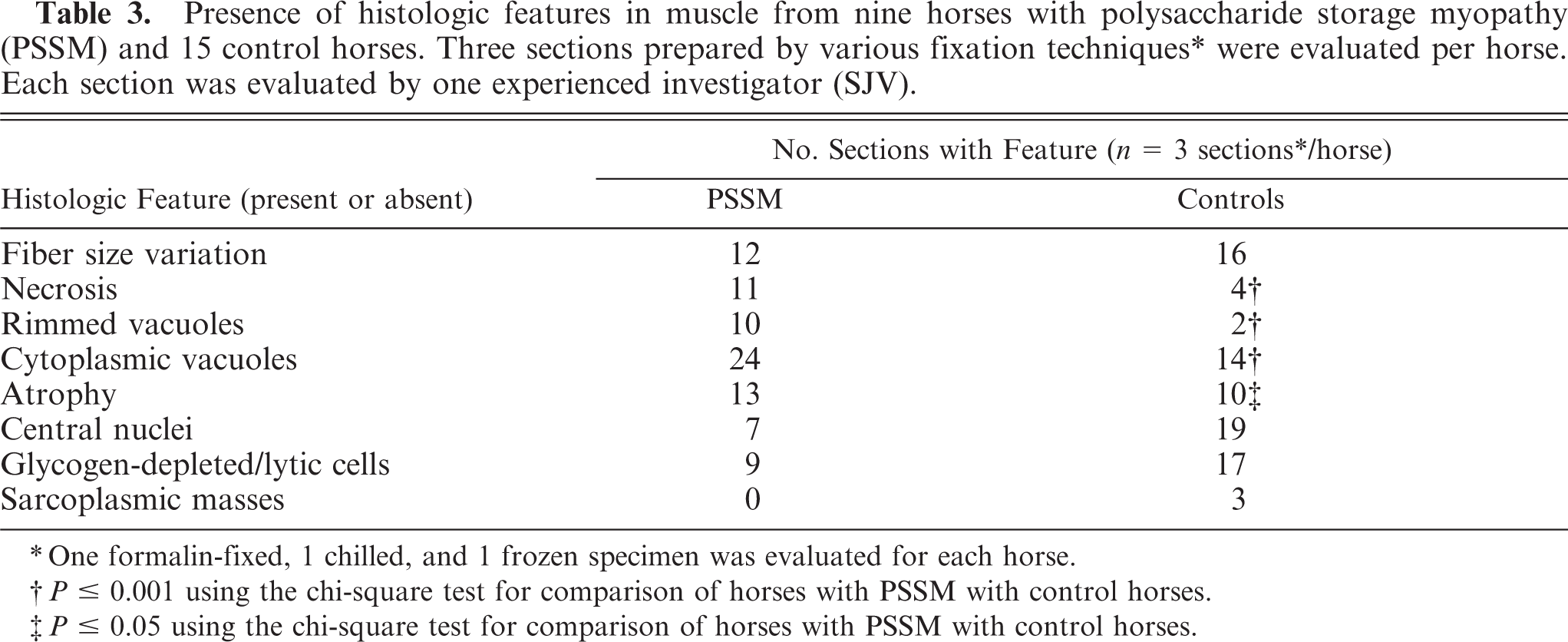
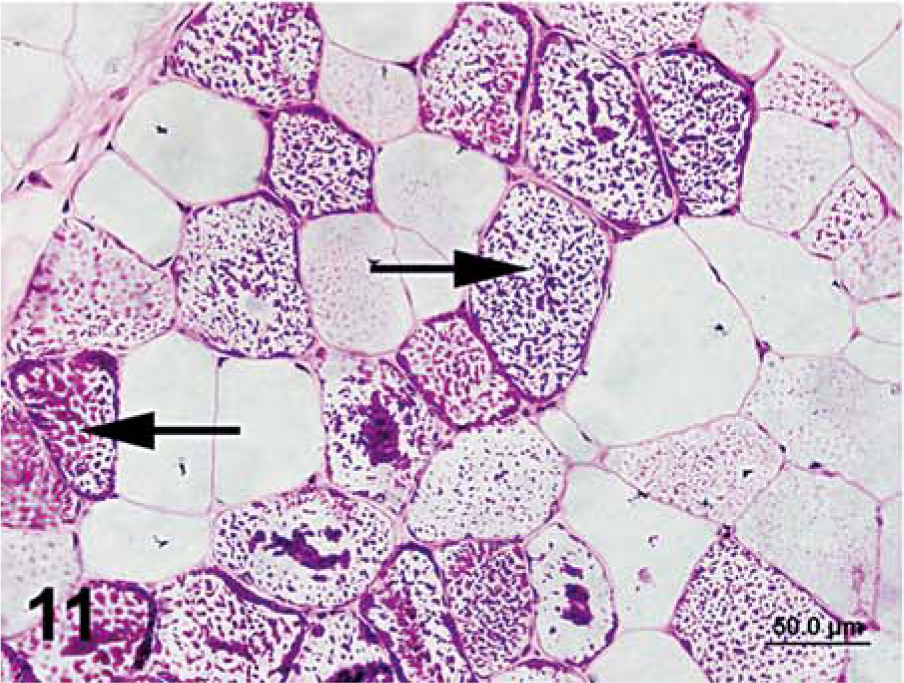
A mild degree of variation in muscle fiber size, a few anguloid atrophied fibers, and centrally displaced nuclei were found in HE-stained sections from control Quarter Horses (Fig. 13, Table 3). However, the mean histopathologic score for control horses was low (1.2 ± 0.3). A few subsarcolemmal vacuoles were observed in PAS-stained sections from some control horses, and less frequently in HE-stained sections (Table 3, Fig. 13). A few fibers undergoing macrophage infiltration were identified in one section from a control horse.
Presence of histologic features in muscle from nine horses with polysaccharide storage myopathy (PSSM) and 15 control horses. Three sections prepared by various fixation techniques* were evaluated per horse. Each section was evaluated by one experienced investigator (SJV).
One formalin-fixed, 1 chilled, and 1 frozen specimen was evaluated for each horse.
Glycogen had a granular cytoplasmic appearance in 60% of control horses (Fig. 13, Table 4) and was readily apparent in subsarcolemmal locations in 80% of control horses, principally in the formalin-fixed sections (Figs. 4, 13). One horse of the control group had evidence of amylase-resistant, abnormal polysaccharide in one microscopic field of chilled and frozen sections, but not in the formalin-fixed section. One other control horse had evidence of a few fibers with amylase-resistant, abnormal polysaccharide in one microscopic field in the immediately frozen section only.
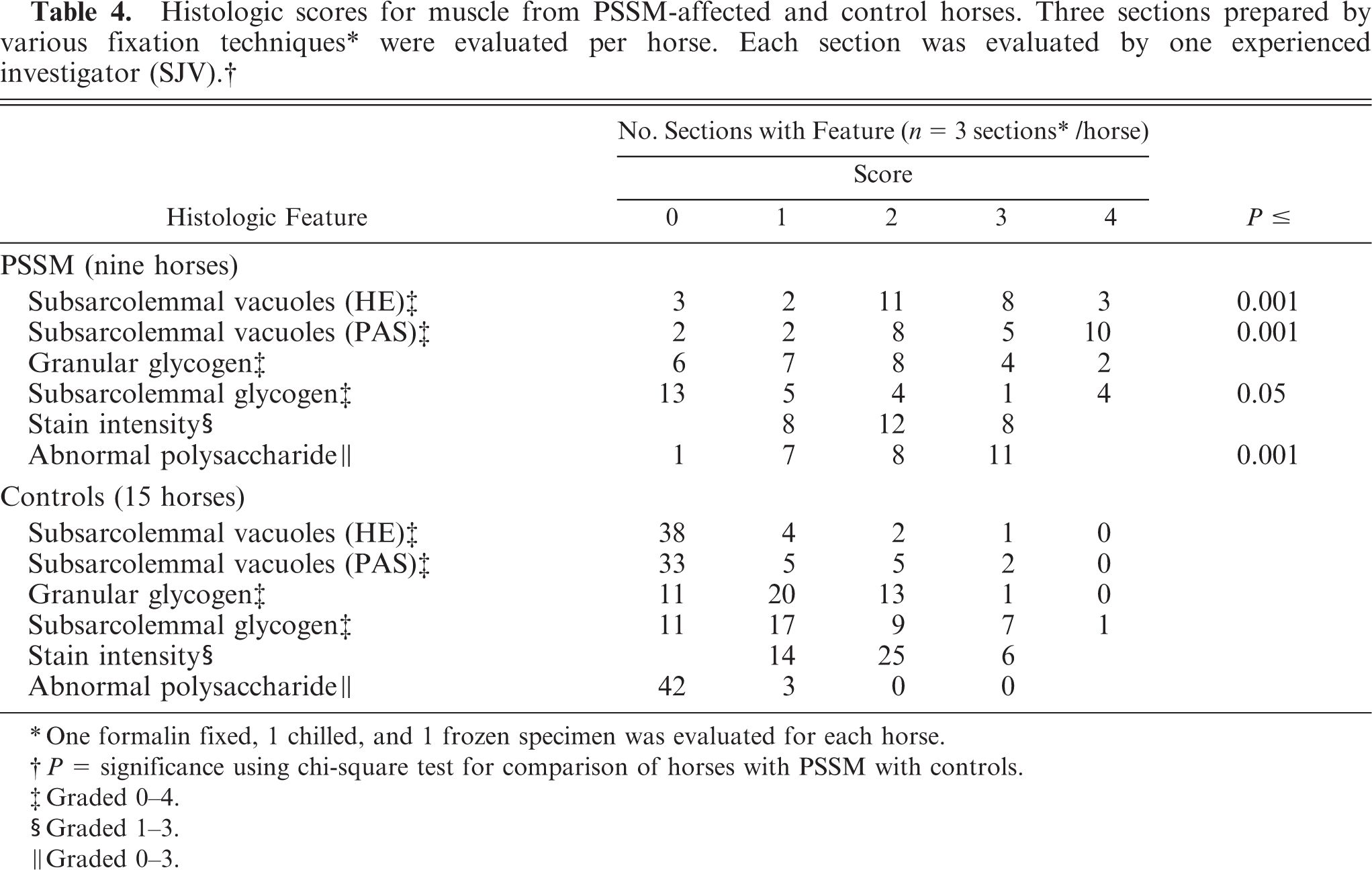
Histologic scores for muscle from PSSM-affected and control horses. Three sections prepared by various fixation techniques∗ were evaluated per horse. Each section was evaluated by one experienced investigator (SJV).†
One formalin fixed, 1 chilled, and 1 frozen specimen was evaluated for each horse.
Graded 0-4.
Graded 1-3.
Graded 0-3.
Histologic features associated with RER
Muscle necrosis with macrophage infiltration, subsarcolemmal vacuoles (HE) and amylase-resistant, abnormal polysaccharide were not observed in any sections from RER-affected horses (Fig. 13). The mean histologic score for RER horses was 1.1 ± 0.2. Since there were no significant differences in the 14 histologic features between control and RER-affected horses, the two control groups were combined to obtain 15 horses for comparison with the PSSM group (Tables 3, 4).
Histologic features associated with PSSM
Sections from horses with PSSM were more likely to show necrosis, with macrophage infiltration, rimmed vacuoles, cytoplasmic vacuoles, increased numbers of fibers with anguloid atrophy, and subsarcolemmal vacuoles (HE and PAS), compared with those from control horses (Tables 3, 4). There was no significant difference in fiber size variation, centrally located nuclei, glycogen-depleted/lytic cells, sarcoplasmic masses, granular glycogen, and PAS stain intensity between control and PSSM-affected horses (Fig. 13, Tables 3, 4). Significantly lower scores for the number of fibers with subsarcolemmal glycogen staining were given to horses with PSSM, compared with control horses (Fig. 13, Table 4). The total number of fibers with subsarcolemmal glycogen was fewer in PSSM-affected horses; however, in formalin-fixed, PAS-stained sections, there was subjectively a thicker rim of subsarcolemmal glycogen in skeletal muscle fibers from horses with PSSM, compared with control horses (Fig. 5). Amylase-resistant, abnormal polysaccharide was more likely to be present in skeletal muscle from PSSM-affected horses, compared with controls (Fig. 5, 7, 9, 11, Table 4). The mean histopathologic score for horses with PSSM ranged from 2 to 16 (combined controls, 0–6) and the mean ± SEM score for horses with PSSM was 9.5 ± 0.7 (combined controls, 1.5 ± 0.2). The 95% confidence interval for the scores given to horses with PSSM was 8.0–10.9, and that for controls was 1.1–1.95.
Variation in muscle biopsy interpretation among evaluators
Kappa values approaching 1 indicated closer agreement among investigators. Agreement among evaluators was good for the presence of amylase-resistant, abnormal polysaccharide (range, 0.80–0.86) and for stain intensity (range, 0.50–0.78). Agreement among examiners was moderate for necrosis (range, 0.30–0.68), central nuclei (range, 0.31–0.47), lytic cells (range, 0.39–0.52), subsarcolemmal vacuoles (HE stain: range, 0.35–0.50; PAS stain: range, 0.40–0.62), and subsarcolemmal glycogen (range, 0.15–0.41). Agreement among examiners was poor for features such as fiber size variation (range, −0.01 to +0.33), rimmed vacuoles (range, 0.11–0.53), cytoplasmic vacuoles (range, 0.14–0.41), anguloid atrophy (range, 0.07–0.42), sarcoplasmic masses (range, 0.03–0.22), and granular glycogen (range, 0.10 to 0.31). Interestingly, the mean ± SEM kappa value of all the features for each pair of investigators was similar (SJV vs. AMF, 0.37 ± 0.07; AMF vs. EJA, 0.31 ± 0.06; SJV vs. EJA, 0.40 ± 0.06).
Testing of diagnostic criteria
Of the 14 histologic features studied, the presence of amylase-resistant, abnormal polysaccharide in a skeletal muscle biopsy specimen provided the strongest association (
For muscle that was frozen immediately, the sensitivity, specificity, NPV, and PPV for the use of amylase-resistant, abnormal polysaccharide for a positive diagnosis of PSSM was 100.0, 86.7, 81.8, and 100.0%, respectively. For muscle that was chilled for 24 hours then frozen, the sensitivity, specificity, NPV, and PPV for the use of amylase-resistant, abnormal polysaccharide for a positive diagnosis of PSSM was 100.0, 93.3, 90.0, and 100.0%, respectively. For muscle that was formalin-fixed and paraffin embedded, the sensitivity, specificity, NPV, and PPV for the use of amylase-resistant, abnormal polysaccharide for a positive diagnosis of PSSM was 88.9, 100.0, 100.0, and 93.8%, respectively. The combined sensitivity, specificity, NPV, and PPV for the use of amylase-resistant, abnormal polysaccharide as a diagnostic criterion across the three fixation techniques was 96.3, 93.3, 89.7, and 97.7%, respectively.
Assessment of the new diagnostic criteria
Blinded reassessment by a board-certified veterinary pathologist (DWH) of randomly selected muscle biopsy specimens from seven PSSM-affected and nine control horses resulted in 100% true positives, 89% true negatives, 11% false positives, and 0% false negatives.
Discussion
In human medicine, frozen fixation and cryostat sectioning is the preferred method of muscle biopsy specimen preparation because of the need to preserve muscle architecture and use of a greater variety of staining techniques available for frozen sections. 6 Immediately frozen biopsy specimens were ideal for evaluating muscle tissue for the histopathologic criteria used in the study reported here, and provided a high degree of sensitivity for diagnosing PSSM. The difficulty in appropriately freezing skeletal muscle requires that specimens obtained in the field be shipped fresh on ice packs to laboratories where they are frozen within 24 hours. The results of this study suggest that this fixation method did not significantly impact most of the histopathologic features evaluated. However, the autolytic process that took place prior to freezing appeared to increase the number of glycogen-depleted/lytic cells (Fig. 8) and the number of subsarcolemmal vacuoles in PAS-stained sections (Fig. 7). Thus, acute necrosis may be difficult to distinguish from glycogen-depleted/lytic cells in shipped fresh, skeletal muscle specimens. In addition, a generalized decrease in PAS staining intensity in chilled, relative to immediately frozen sections likely was a result of ongoing glycogenolysis prior to freezing. Freeze artifacts formed in some sections likely because muscle has a high water content, and if it is not rapidly frozen at low temperatures, ice crystals form inside the cells and create small artifactual vacuoles. Rechilling of isopentane may have been inadequate between freezing of the large number of skeletal muscle specimens obtained for this study.
Formalin fixation and paraffin embedding are the most commonly used methods for processing biopsy specimens and are easy methods to use for shipping such specimens to a laboratory. In the study reported here, fiber size variation, anguloid atrophy, and central nuclei were observed less frequently in HE-stained sections that were formalin fixed, compared with specimens that were frozen. This appeared to be due to the relative difficulty in visualizing the cell membranes in formalin-fixed, HE-stained specimens, compared with specimens that were frozen (Fig. 2 compared with Fig. 1). Sarcoplasmic masses, as represented by central PAS-positive stellate material (Fig. 3), 1 and rimmed vacuoles also were not readily observed in formalin-fixed, compared with frozen sections. In addition, abnormal cytoplasmic vacuoles such as rimmed vacuoles were difficult to distinguish from artifactual “cracking,” which occurs commonly in formalin-fixed skeletal muscle (Fig. 2). 10 Thus, in general, formalin fixation of skeletal muscle was not ideal for characterizing myopathies. The appearance of glycogen differed between formalin and frozen sections. The PAS-stained sections were lighter in formalin-fixed specimens, probably due to the thinner sections used and the displacement of glycogen from the muscle cell due to its solubility in the dehydrating and clearing media used during fixation. 10 Furthermore, glycogen appeared to settle in one end of muscle fibers in some sections, creating an artifactual gradient of PAS-positive staining within fibers (Fig. 6) and the appearance of focal subsarcolemmal glycogen accumulation (Fig. 4). Therefore, frozen sections appear to be better suited for studying myopathies because many histopathologic features of skeletal muscle were obscured by formalin fixation, and a wider variety of metabolic stains could potentially be used to identify specific features of the disorder.
The ability to detect amylase-resistant, abnormal polysaccharide, however, was not significantly affected by freezing or chilling of samples or formalin fixation; therefore, this diagnostic criterion for PSSM could be applied to formalin-fixed sections. All PSSM-affected horses were accurately phenotyped, with a high sensitivity and PPV (90–100%), by investigators using this diagnostic criterion even when investigators were blinded to the horse's identity. There was only one formalin-fixed, PAS amylase-stained section of 27 sections from PSSM-affected horses, in which amylase-resistant, abnormal polysaccharide was not identified. Other features that were significantly associated with PSSM but not always present in biopsy specimens included myofiber necrosis, rimmed vacuoles, cytoplasmic vacuoles, anguloid atrophy, and subsarcolemmal vacuoles. The common presence of these features in horses with PSSM resulted in higher mean scores, compared with those for controls.
A scoring system that includes these features may be useful in conjunction with amylase-resistant, abnormal polysaccharide to characterize muscle biopsy specimens from horses with exertional myopathies. However the criteria should not be used in the absence of the amylase-resistant, abnormal polysaccharide, as they are present in other myopathies, 4 and are not consistently present in all PSSM-affected horses. The high degree of variation among examiners and among fixation techniques when evaluating these histopathologic features weakens their usefulness as diagnostic criteria. A high score, however, may indicate that it is prudent to evaluate several PAS-stained sections to definitively identify or rule out the presence of amylase-resistant, abnormal polysaccharide. 14 The only feature that had excellent agreement among all individuals and was not affected by fixation technique was the presence of amylase-resistant, abnormal polysaccharide.
It is important to note that the accumulation of amylase-resistant, abnormal polysaccharide may not be evident in muscle biopsy specimens until horse are at least 18 months old, whereas rhabdomyolysis may develop as early as 1 month of age. 9 Thus, when examining muscle specimens from young horses, a diagnosis of PSSM cannot confidently be excluded using amylase-resistant, abnormal polysaccharide until horses are older than 18 months. Unfortunately, until further genetic or biochemical studies identify the gene or protein that is responsible for PSSM, the diagnosis of PSSM will have to be based on clinical signs of disease and examination of a skeletal muscle biopsy specimen.
Polysaccharide storage myopathy is one of the most common inherited muscle disorders in horses. 7, 12, 23 Accurate diagnostic criteria, therefore, become important for clinical recognition of the disease, for selection of breeding animals, and for establishing correct phenotypes for horses used in research. It has been suggested that PSSM can accurately be diagnosed on the basis of increased PAS staining, granular glycogen deposits or increased subsarcolemmal glycogen, or both, in the absence of amylase-resistant, abnormal polysaccharide. 26– 28, 30– 32, 34 However, granular cytoplasmic glycogen, subsarcolemmal glycogen, or PAS stain intensity were not significantly associated with a previous diagnosis of PSSM in the horses of this study. In fact, granular cytoplasmic glycogen was present in the specimens from both control groups (Quarter Horses and RER), and those from the PSSM-affected horses had less subsarcolemmal glycogen, particularly in frozen sections. The results of this study suggest that the previously reported high prevalence of PSSM (up to 80%) 5 in a variety of breeds (Norwegian Fjord, Standardbreds, Thoroughbred crosses, American Saddlebreds, and American Miniature Horses), 27, 28, 31– 34 may be a result of nonspecific diagnostic criteria.
Semimembranosus muscle that was frozen immediately in chilled isopentane; horse No. 4, control Quarter Horse mare. Notice absence of amylase-resistant, abnormal polysaccharide in the control horse. PAS. Bar = 100 µm (lower magnification to show the overall lack of amylase-resistant, abnormal polysaccharide).
Amylase-resistant, abnormal-polysaccharide was identified in small numbers of muscle fibers from 2 of 15 control horses. Thus, the specificity and NPV of amylase-resistant, abnormal polysaccharide as a diagnostic criterion across all three fixation techniques was 96 and 98%, respectively. The presence of amylase-resistant, abnormal polysaccharide in these control horses either indicates that there was some PAS-positive artifact in sections that was interpreted as abnormal polysaccharide or that some horses of the control group had PSSM. An ideal control group would have been healthy, competitive Quarter Horses that were known to not have clinical signs of PSSM. However, we were unable to find owners willing to allow us to take a sizable surgical biopsy specimen from such horses. Most horses of the control group were lame, which likely is why they were sent to auction, and they could not be consistently exercised to rule out PSSM on a clinical basis. Thus, we were not able to determine whether the finding of amylase-resistant, abnormal polysaccharide in our control group was consistent with a clinical diagnosis of PSSM.
To the authors' knowledge, the actual prevalence of PSSM has not been established in Quarter Horses. Using the diagnostic criteria of amylase-resistant, abnormal polysaccharide, the prevalence of PSSM in Belgian Draft horses is as high as 34%. 11 It would be an unexpected finding to have such a high prevalence in Quarter Horses, as the degree of inbreeding in them is less than that in Belgian Draft horses. 3, 22 Obviously a prospective study using the criterion of amylase-resistant, abnormal polysaccharide needs to be performed in healthy Quarter Horses to determine the prevalence of this disorder in this breed.
In conclusion, controlled research studies have indicated that, when PSSM is diagnosed by the presence of amylase-resistant, abnormal polysaccharide, 21 Quarter Horses have consistent features of increased postexercise serum CK activity, 2, 13, 23– 25 enhanced insulin sensitivity, 2, 8 and a familial incidence of disease. 9, 22 Thus, there appears to be a high degree of specificity to amylase-resistant, abnormal polysaccharide associated with an inherited clinical syndrome. To accurately perform studies of PSSM, the diagnostic criteria should include the presence of amylase-resistant, abnormal-polysaccharide in tissues that are frozen or fixed in formalin. Substitution of other characteristics of amylase-sensitive, normal glycogen substantially decreases specificity of the diagnosis of PSSM and likely results in the false inclusion of breeds and individual horses that are normal or have other myopathies under the diagnosis of PSSM. In situations where muscle biopsy specimens are used to evaluate individual clinical cases, there is a small chance of a false-negative diagnosis using amylase-resistant, abnormal polysaccharide as the “gold standard” for diagnosis. The impact of a false-negative diagnosis on an individual animal can be largely negated if horses with exertional myopathies are provided with recommendations for a generic low-starch, high-fat diet and regular exercise. A good clinical response to these regimens is reported for PSSM and other exertional myopathies. 12, 16, 18, 34
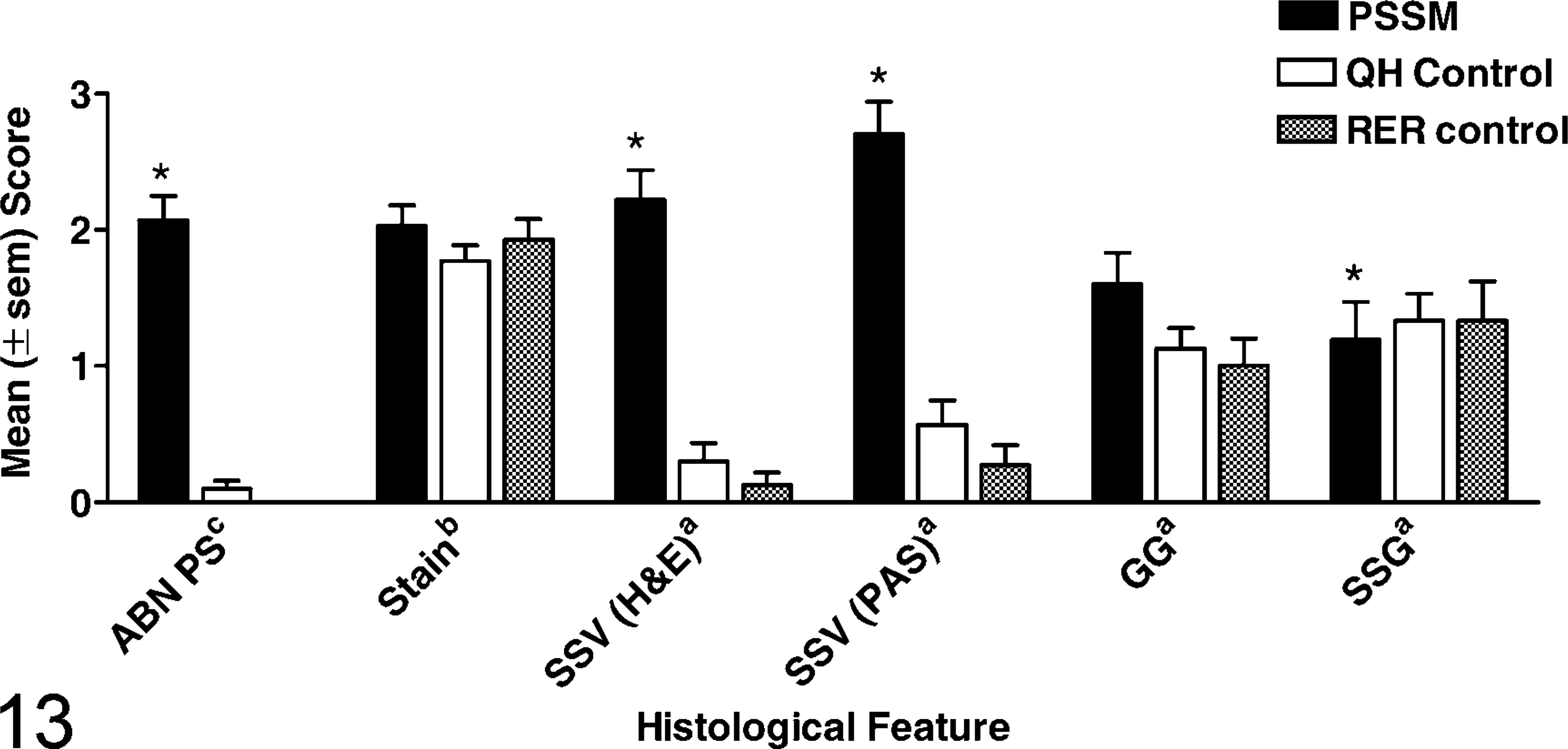
Bar graph showing mean ± SEM score given to 6 histologic features of skeletal muscle from nine horses with PSSM, 10 control Quarter Horses, and five control horses with RER. One formalin-fixed, one chilled, and one frozen specimen was evaluated for each horse. ∗ = a significant difference from control horses using a chi-squared test at P < 0.05. ABN PS = amylase-resistant PAS-positive abnormal-polysacchride, Stain = stain intensity, SSV = subsarco lemmal vacuoles, GG = granular glycogen, SSG = subsarco lemmal glycogen. a graded 0 to 4, b graded 1 to 3, c graded 0 to 3.
Footnotes
O.C.T. Compound. Embedding medium for frozen tissue specimens. Miles Inc., Elkhart, IN.
Alpha amylase from hog pancreas. Sigma-Aldrich, Atlanta GA)
GraphPad Prism. Version 4.00, San Diego, CA 92130
Statistix, version 8 (Analytical Software). Tallahassee, FL
SAS, SAS Institute Inc., Cary, NC
