Abstract
Polysaccharide storage myopathy is an equine neuromuscular disorder characterized by accumulation of glycogen-related polysaccharide inclusions within skeletal muscle fibers. The pathologic criteria for diagnosis of this disorder are somewhat controversial; however, periodic acid-Schiff-positive, amylase-resistant inclusions are considered pathognomonic. Although these inclusions are most often found in affected horses related to the Quarter Horse, draft horse, and Warmblood breeds, this report describes these characteristic inclusions in muscle of five horses from nonrelated breeds (two Morgans, one Arabian, one Arabian X Thoroughbred, and one Standardbred) and two Welsh cross ponies. Affected horses had histories of recurrent exertional rhabdomyolysis, and one developed progressive weakness leading to increased recumbency. The affected ponies were part of an unrelated research project and had no apparent clinical signs.
Equine polysaccharide storage myopathy (EPSSM) is an emerging neuromuscular disorder of horses. Although previously recognized in a small number of horses with various signs of neuromuscular dysfunction, 2 , 7 the first report of this disorder in a larger group of horses was in Quarter Horse-related breeds with recurrent exertional rhabdomyolysis. 8 It is also recognized to occur in draft horse-related breeds 1 , 12–14 and in Warmbloods. 10 Exertional rhabdomyolysis is a common finding in affected horses, 8 , 14 , 16 although generalized muscle atrophy and weakness, abnormal pelvic limb gait, pelvic limb muscle atrophy, postanesthetic myopathy, and spontaneous recumbency with inability to rise also occur. 1 , 12 , 14 , 16 The disorder is thought to be inherited 9 , 12 , 13 but may be clinically inapparent for many years. 10 , 12–14, 16
The criteria for histopathologic diagnosis of EPSSM and the breeds affected are somewhat controversial. All investigators agree that the finding of periodic acid–Schiff (PAS)-positive, amylase-resistant inclusions of complex polysaccharide within muscle fibers is pathognomonic for this disorder. 10 , 15 Some investigators believe that the diagnosis of EPSSM can also be made based on the presence of intramyofiber accumulations of glycogen aggregates. 15 One investigator reported that the PAS-positive, amylase-resistant inclusions occur only in Quarter Horse-, drafthorse-, and Warmblood-related breeds and that these are the only breeds in which EPSSM occurs. 10 This disorder has been described in other breeds; however, biopsy samples of muscle from these cases contained aggregates of glycogen but no complex polysaccharide. 16 Here, we describe PAS-positive, amylase-resistant inclusions within skeletal muscle fibers of seven horses and ponies not of Quarter Horse, drafthorse, or Warmblood breeding.
The signalment and histories are summarized in Table 1. Muscle samples of semimembranosus and sacrocaudalis dorsalis medialis muscle from horse No. 1 were collected at necropsy following euthanasia, which was performed because of progressive weakness. Samples of semimembranosus or semitendinosus muscle were obtained by biopsy from horse Nos. 2–5. Pony Nos. 6 and 7 were noninfected controls in a Lyme disease study, and muscles collected at necropsy were labeled proximal thoracic limb and proximal pelvic limb muscle. Frozen-section muscle histochemistry was performed on horse Nos. 2–4. Portions of the muscle samples from these horses were snap-frozen in isopentane cooled in liquid nitrogen, and 8–10-µm transverse sections were cut at −25 C and stained with hematoxylin and eosin (HE), modified Gomori's trichrome, NADH, PAS, and myofibrillar ATPase at pH 10.0 and 4.35. Muscle samples from all animals were fixed in formalin and embedded in paraffin, and 4–6-µm transverse and longitudinal sections were stained with HE, PAS for glycogen, and PAS following amylase digestion. A section of liver containing glycogen served as a control for the PAS for glycogen stain. The samples of sacrocaudalis dorsalis medialis collected from horse No. 1 were fixed in formalin and embedded in paraffin, and sections were stained with HE and Masson's trichrome stains.
Signalment and history of seven horses and ponies with polysaccharide storage myopathy.
∗ F = female; Mc = male, castrated.
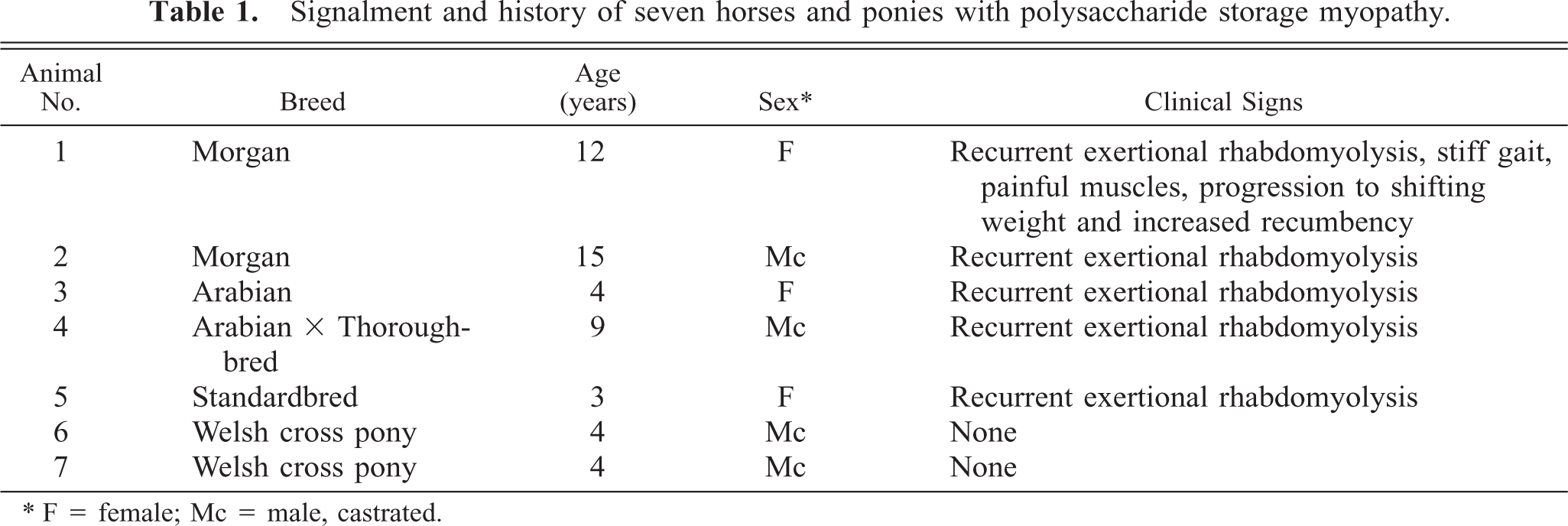
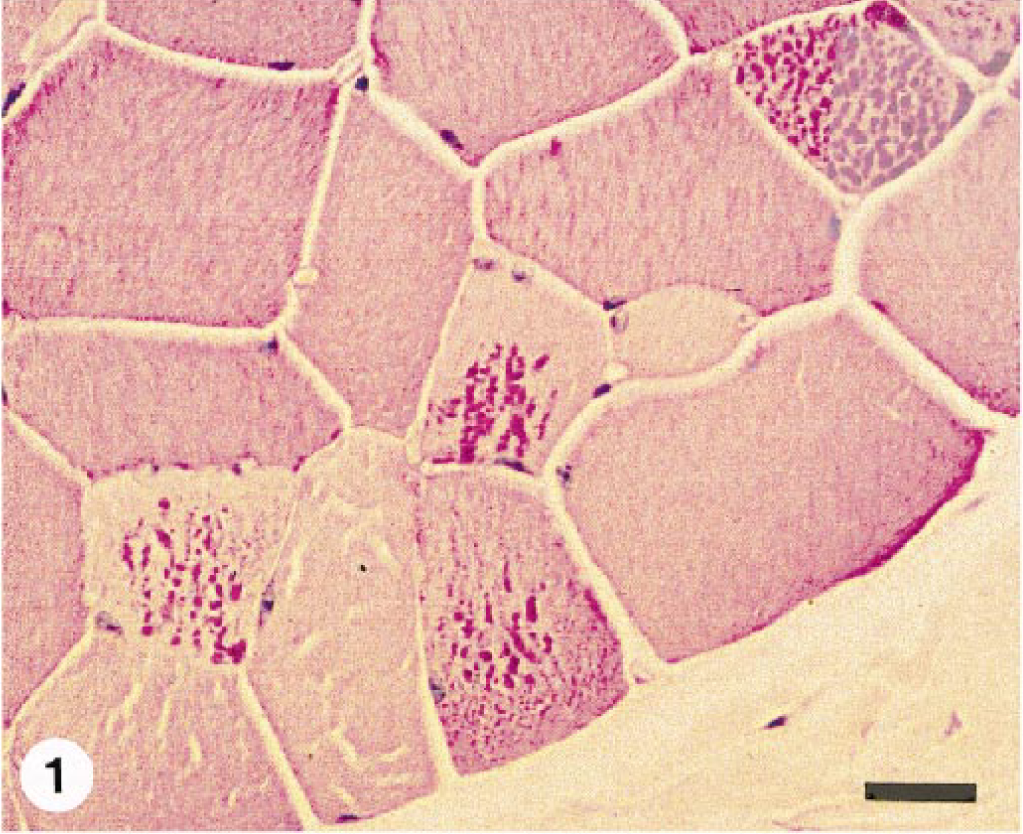
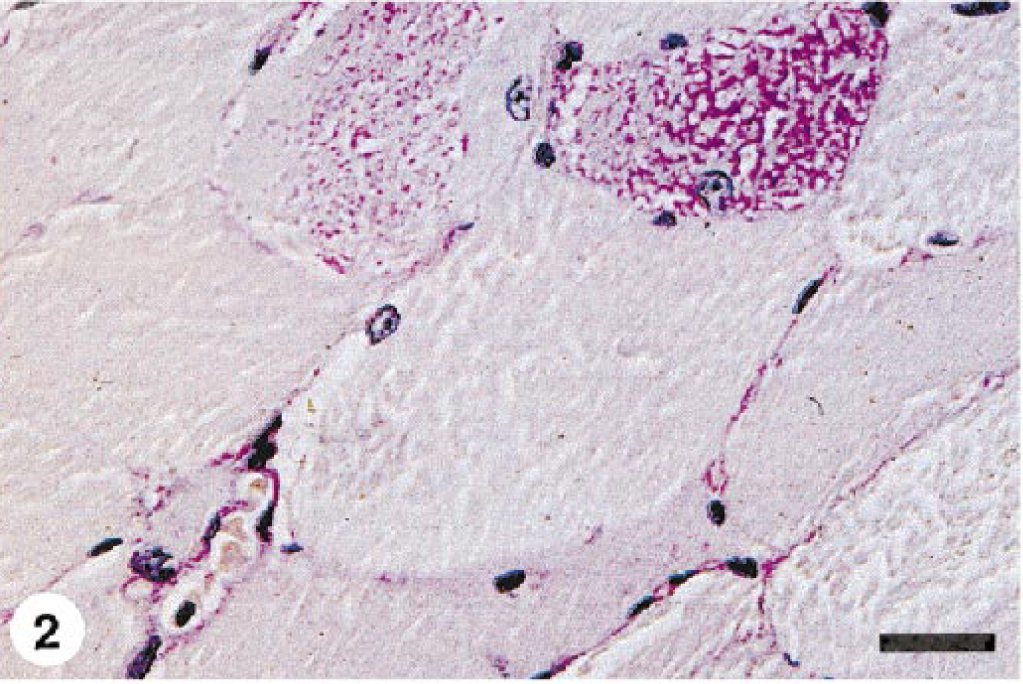
Excessive fiber size variation and increased numbers of internal nuclei were found in all semimembranosus and semitendinosus samples examined. The sacrocaudalis dorsalis medialis muscle from horse No. 1 was normal. Angular atrophied fibers or large group atrophy were not seen in any case. Scattered fibers in formalin-fixed, HE-stained sections contained subsarcolemmal zones that were either clear or contained pale pink hyaline material. In the frozen sections from horse Nos. 2–4, these zones occurred within type 2A and 2B fibers and stained pale pink with HE and pale blue-grey with modified Gomori's trichrome stain. These areas did not stain with NADH or myofibrillar ATPase but stained intensely with PAS and were amylase sensitive, consistent with glycogen. In all cases, a small number of fibers in formalin-fixed samples contained either numerous small “lakes” or larger irregular, confluent aggregates of material that stained pale blue-grey with HE and intensely magenta pink with PAS (Figs. 1–4) and resisted amylase digestion (Figs. 2–4), consistent with complex polysaccharide. Affected fibers most often occurred singly or in small clusters and were not uniformly distributed within the muscle sections examined. Amylase-resistant complex polysaccharide was found in the proximal thoracic limb muscle sample of pony No. 6 and in both proximal thoracic and proximal pelvic limb muscle samples from pony No. 7.
Semitendinosus muscle; horse No. 3. Scattered fibers contain multiple small intracytoplasmic “lakes” of PAS-positive material. There is overall increased intensity and irregularity of PAS staining. PAS for glycogen, formalin-fixed, paraffin-embedded tissue. Bar = 50 µm.
Semitendinosus muscle; horse No. 2. Two fibers contain multiple small intracytoplasmic “lakes” of PAS-positive, amylase-resistant material. PAS with amylase digestion, formalin-fixed, paraffin-embedded tissue. Bar = 50 µm.
Semimembranosus muscle; horse No. 5. A single fiber contains multiple small intracytoplasmic “lakes” of PAS-positive, amylase-resistant material. PAS with amylase digestion, formalin-fixed, paraffin-embedded tissue. Bar = 50 µm.
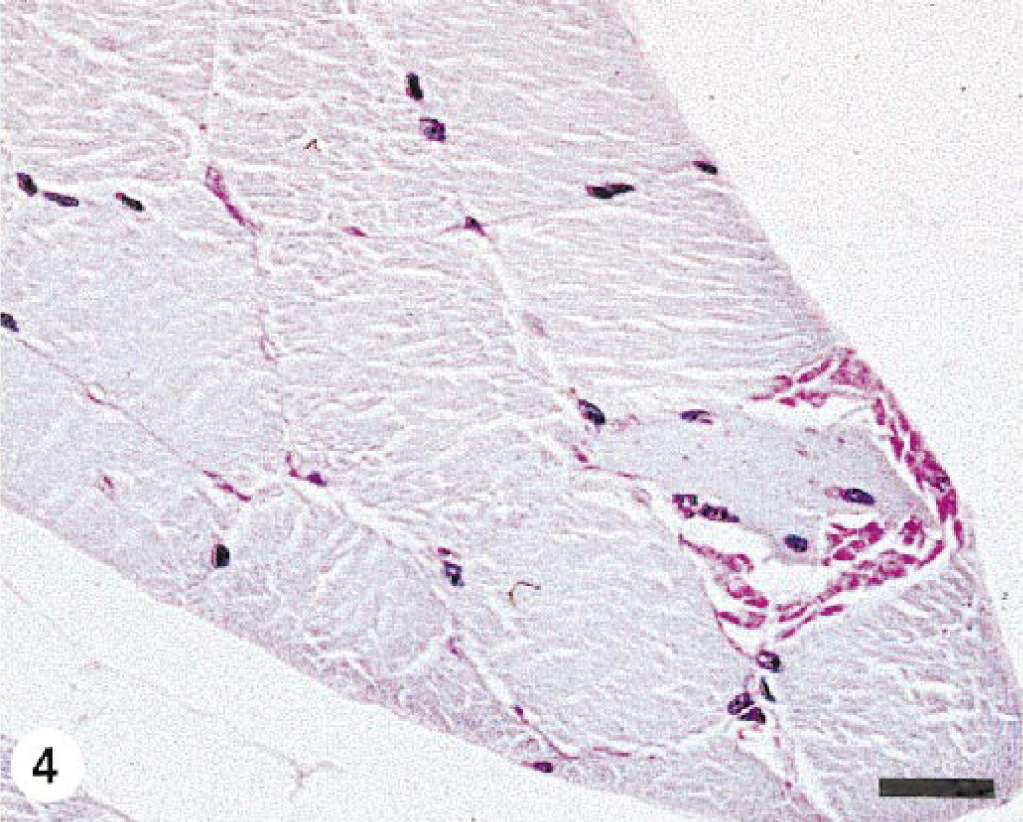
Proximal thoracic limb muscle; pony No. 7. A large, irregularly shaped PAS-positive, amylase-resistant inclusion replaces approximately 50% of the diameter of the affected fiber. This fiber also contains multiple internal nuclei. PAS with amylase digestion, formalin-fixed, paraffin-embedded tissue. Bar = 50 µm.
Clearly, the severity and nature of the changes in skeletal muscle of horses with EPSSM is somewhat variable. The findings reported here were less severe than those often seen in affected Quarter Horses 8 , 10 and draft horses, 13 suggesting that different breeds may have a different form of this disorder. Myopathic changes and aggregates of glycogen and complex polysaccharide are not, however, found in normal equine skeletal muscle stained with PAS for glycogen (Fig. 5). In particular, the morphology of the PAS-positive, amylase-resistant inclusions in muscle from horses with EPSSM is variable. The most common finding is multiple intracytoplasmic “lakes” of material (Figs. 1–3). Larger, confluent aggregates of material replacing part or all of the affected fiber segment were seen in pony Nos. 6 and 7 (Fig. 4). Because these distinctive inclusions were an unexpected finding at necropsy of clinically normal ponies, they may be nonspecific and not indicative of underlying myopathy. Similar inclusions occur in draft horses with clinical signs of neuromuscular dysfunction due to EPSSM (Fig. 6), however, and are considered part of the spectrum of changes possible in this disorder. The lack of clinically apparent disease in these ponies is probably due to the fact that they were maintained as a college-owned research herd and were never put into training. Although EPSSM is thought to be an inherited disorder, onset of clinical signs often occurs in affected Quarter Horses when they are put into training at 2–4 years of age, 10 and in drafthorses obvious clinical signs may not occur until 21 years of age. 12 The clinical histories in horse Nos. 1–5 are typical of those in EPSSM horses being ridden or driven and are indicative of neuromuscular dysfunction consistent with underlying EPSSM. The progressive weakness and increased recumbency in horse No. 1 would also be characteristic of equine motor neuron disease. 5 , 15 Although spinal cord was not examined in this horse, the lack of evidence in the sacrocaudalis dorsalis medialis muscle of angular atrophy or large group atrophy indicative of denervation makes this diagnosis unlikely; 6 , 15 progressive wasting and weakness also occur in animals with EPSSM. 12–14 The failure to identify complex polysaccharide inclusions in a single transverse frozen section of muscle from horse Nos. 2–4 is probably due to sampling error because the number of affected myofibers in all the animals was relatively small and affected fibers were not uniformly distributed. Multiple transverse and longitudinal sections from different areas of the formalin-fixed samples were embedded and examined, thus increasing the likelihood of finding single or small clusters of affected fibers.
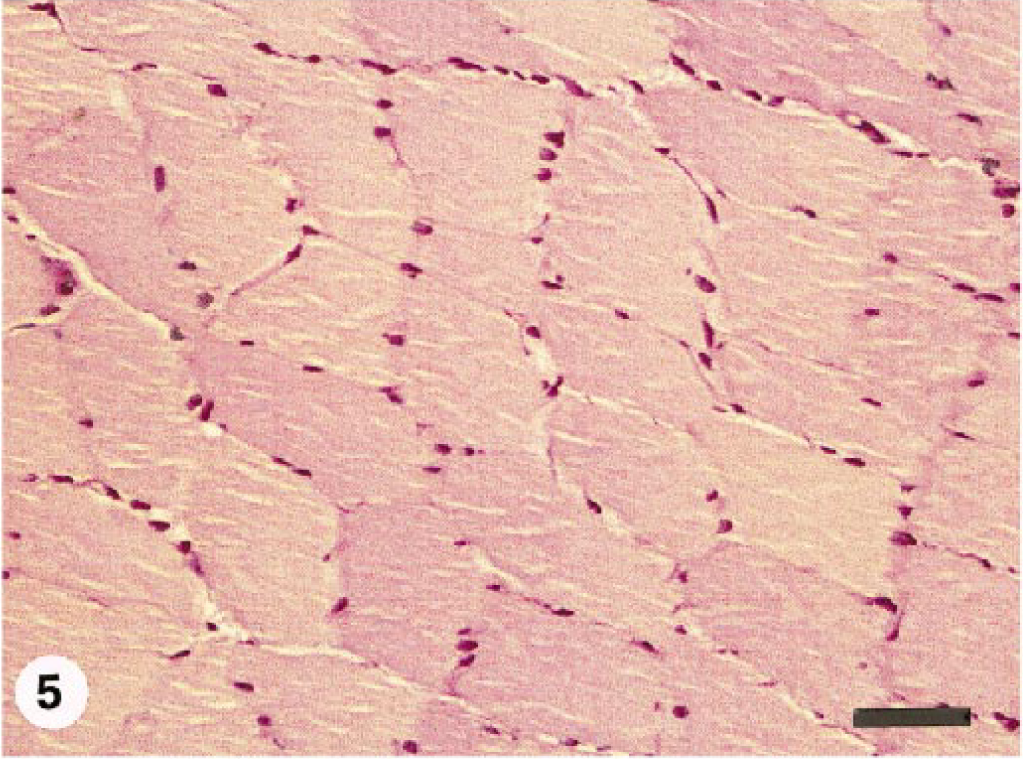
Semitendinosus muscle; normal horse. Relatively uniform fiber diameter with no internal nuclei. Glycogen staining intensity varies somewhat based on fiber type, but intracytoplasmic glycogen staining is finely granular and even distributed. PAS for glycogen, formalin-fixed, paraffin-embedded tissue. Bar = 100 µm.
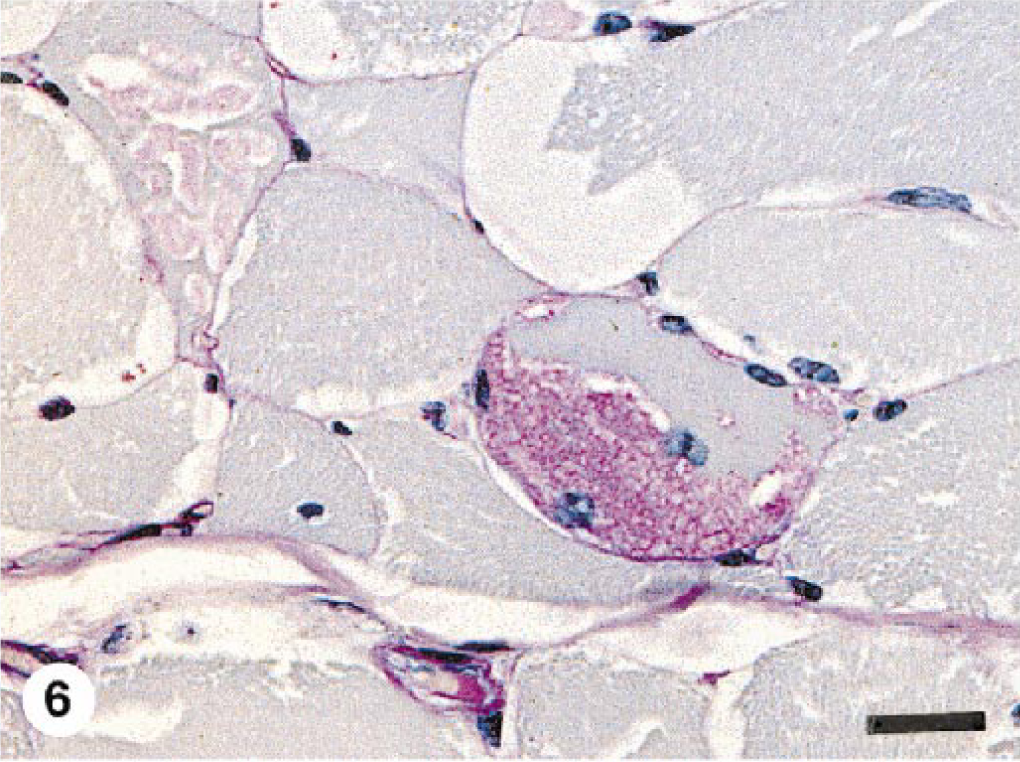
Semitendinosus muscle; 9-year-old Percheron × Thoroughbred mare with recurrent exertional rhabdomyolysis due to EPSSM. There is excessive fiber size variation. One fiber contains a large confluent PAS-positive, amylase-resistant inclusion (similar to that in pony No. 7) that replaces approximately 50% of the fiber diameter. Internal nuclei are present in this and in an adjacent fiber. Another fiber contains multiple small intracytoplasmic “lakes” of PAS-positive, amylase-resistant material similar to that illustrated in horse Nos. 2, 3, and 5. The prominent, clear round-to-oval vacuoles contain PAS-positive, amylase-sensitive glycogen. PAS with amylase digestion, formalin-fixed, paraffin-embedded tissue. Bar = 50 µm.
Some evidence suggests that EPSSM is inherited as an autosomal recessive trait. 9 The dam of horse No. 3 was retired from racing because of recurrent exertional rhabdomyolysis, but a muscle biopsy was not performed. No information was available regarding the sire. Pony Nos. 6 and 7 were sired by the same Welsh pony stallion, and their dams were mixed-breed ponies that were daughters of this stallion. Clinical signs of neuromuscular dysfunction were not apparent in the sire or in either of the dams, although none were being ridden or driven. No pedigree information was available for horse Nos. 1, 2, 4, and 5.
Polysaccharide storage myopathy in horses is clinically and pathologically similar to myopathies due to carbohydrate metabolic defects in humans, 3 although extensive studies of affected horses have failed to identify a defect in glycolytic or glycogenolytic pathways. 11 , 17 Storage of glycogen and complex polysaccharide occurs only within skeletal muscle, 13 and increased skeletal muscle uptake of blood glucose in affected horses has been proposed. 4 Inclusions of PAS-positive, amylase-resistant material in skeletal muscle of EPSSM horses are always accompanied by aggregates of glycogen, 10 , 13 , 15 and glycogen storage probably precedes formation of complex polysaccharide.
These seven cases document that EPSSM occurs in a wide range of breeds and indicate that examination of muscle samples for myopathy with storage of glycogen and complex polysaccharide should be performed in any horse with a history of exertional rhabdomyolysis or other clinical signs of neuromuscular disease.
Footnotes
Acknowledgements
We thank Drs. John Cowley, Rick Henninger, and Gary Kubala for referral of cases and Alexis Wenski-Roberts and Bob Demyan for preparation of photomicrographs. This study was supported in part by a grant from the Harry M. Zweig Memorial Fund for Equine Research.
