Abstract
Serial sections of formalin-fixed, paraffin-embedded muscle biopsy specimens from 28 Quarter Horse, Paint, and draft-related breeds, aged 0.5-23 years, were treated with periodic acid-Schiff (PAS) stain for glycogen and were immunostained to detect ubiquitin expression. On the basis of findings in PAS-stained sections, a diagnosis of equine polysaccharide storage myopathy (EPSSM) was made in 22 horses aged 2-23 years (mean, 9.4 years); samples from 6 horses aged 0.5-15 years (mean, 7.3 years) had a normal PAS staining pattern, with no relevant lesions. Ubiquitin expression was detected in all but a 2-year-old EPSSM-affected horse and was not detected in the non-EPSSM-affected horses. Ubiquitin expression was greater than the degree of PAS-positive, amylase-resistant material, and ubiquitin was detected in aggregates of amylase-sensitive glycogen as well as in aggregates of amylase-resistant material. Results suggest that glycogen aggregates develop and are ubiquitinated prior to development of amylase-resistant inclusions. Ubiquitin immunostaining may be most useful for confirming the diagnosis of EPSSM in horses with only amylase-sensitive glycogen aggregates and in horses with early amylase-resistant inclusions. However, ubiquitin immunostaining is no more sensitive than is PAS staining for diagnosis of EPSSM.
Equine polysaccharide storage myopathy (EPSSM) is a metabolic myopathy characterized by abnormal aggregates of periodic acid–Schiff (PAS)-positive, amylase-sensitive glycogen, and PAS-positive, amylase-resistant polysaccharide within myofibers. 4, 6, 7, 16, 17, 19– 23 Affected fibers are type-2A (oxidative-glycolytic) and type-2B (glycolytic) muscle fibers. 16, 17, 20 This disorder is common in many breeds. 19 Clinical signs of muscular dysfunction include exertional rhabdomyolysis, 6, 7, 17, 18, 20, 22 generalized weakness, 20, 22 back pain, 16, 22 postanesthetic myopathy, 4, 20 muscular atrophy, 20– 22 and abnormal gait. 21, 22 There is no defect in glycolytic or glycogenolytic pathways in affected horses, 18 and the cause of this unique equine myopathy remains unknown. Muscular dysfunction may reflect abnormal muscle energy production. 3
Ubiquitin is a highly conserved protein 2, 11 that forms an integral part of the intracellular protein degradation pathway known as the ubiquitin-proteasome pathway. Covalent binding of ubiquitin marks proteins for degradation. 2, 11 Failure to degrade ubiquitinated proteins leads to cellular inclusions that can disrupt cellular homeostasis. 2, 13 Ubiquitination of abnormal cellular inclusions detected by immunohistochemical analysis performed on routinely processed tissue has been documented in various neurodegenerative and myopathic conditions in people. 5, 10, 11, 13– 15 In animals, ubiquitin expression has been described in cardiac and skeletal muscle of Holstein-Friesian cattle with hereditary myopathy of the diaphragmatic muscle 8, 9 and in degenerating axons in horses with cervical compressive myelopathy. 12 The purpose of the study reported here was to determine whether ubiquitin is expressed in muscle from horses with EPSSM and, if so, whether ubiquitin immunostaining can aid in the diagnosis of EPSSM.
Materials and Methods
Formalin-fixed biopsy specimens of semimembranosus or semitendinosus muscle from 28 Quarter Horse, Paint, and draft-related breeds were obtained by use of an open biopsy technique 22 and were submitted for evaluation for evidence of EPSSM. Specimens were strips of muscle approximately 2–3 cm long and 1 cm in diameter. To minimize sampling error, 5 to 8 sections of each specimen, to include multiple transverse sections and 1 longitudinal section, were prepared. 19, 22, 23 Sections were processed in a routine manner and treated with PAS stain for glycogen and were immunostained to detect ubiquitin. The PAS stain was optimized to recognize amylase-sensitive glycogen and amylase-resistant complex polysaccharide in formalin-fixed, paraffin-embedded equine muscle. Briefly, all solutions (0.1% α-amylase, 0.5% periodic acid, Schiff's reagent; Sigma, St. Louis, MO) were fresh at the time of use and were preheated to 37°C. Two serial sections were prepared, and sections on one slide were incubated in amylase solution for 45 minutes at 37°C. Digested and undigested sections were incubated in periodic acid solution for 15 minutes, followed by 20 minutes' incubation in Schiff's reagent. Sections were counterstained for 10 minutes with hematoxylin (Harris modified; Sigma). Serial sections were treated with avidin-biotin endogenous blocking reagent and pronase (Biomeda, Foster City, CA) for 5 minutes each, incubated overnight at 4°C with a polyclonal rabbit anti-ubiquitin antibody (Biomeda), diluted 1 ° 500 and 1 ° 1,000, and processed for routine immunohistochemical analysis using the avidin-biotin complex procedure with Nova Red chromogen (Vector Laboratories, Burlingame, CA) and Mayer's hematoxylin counterstain (Sigma). Positive-control sections were formalin-fixed and paraffin-embedded sections of human brain with corpora amylacea 2, 5, 14 (provided by Dr. Melvin Ball, Portland, OR) and formalin-fixed and paraffin-embedded sections of equine brainstem with corpora amylacea. Normal rabbit serum was used as a negative control.
Immunohistochemical preparations and PAS-stained serial sections were examined in blinded manner by 2 of the authors (BAV, THF), and were evaluated for abnormal polysaccharide storage and ubiquitin expression. Criteria for diagnosis of EPSSM on PAS-stained sections were based on previous studies:
19–
23
specifically, presence of numerous (average > 6/10x field) subsarcolemmal aggregates of amylase-sensitive glycogen, any number of central inclusions of amylase-sensitive glycogen (central bodies), and any number of amylase-resistant polysaccharide inclusions. The distribution of age within both groups (EPSSM and non-EPSSM) was Gaussian, and mean age was calculated for each group. Results were analyzed using the Student's
Results
Muscle biopsy findings and breeds of horses studied are summarized in Tables 1 and 2. Twenty-two horses were diagnosed with EPSSM on the basis of findings in PAS-stained sections. Four horses were diagnosed with EPSSM on the basis of numerous subsarcolemmal aggregates or central bodies of amylase-sensitive glycogen. Eighteen horses had amylase-resistant inclusions as well as glycogen aggregates (Table 1). The number of amylase-resistant inclusions varied; ≤5/biopsy specimen were seen in four horses, 6–15/biopsy specimen were present in four horses, and 10 horses had >15 amylase-resistant inclusions in the sections examined. Fibers with amylase-resistant inclusions often were seen in clusters, frequently at the periphery of fascicles. Fibers with central bodies most often were evident in small clusters within fascicles, and >5 central bodies were seen in sections from the two horses in which they were present.
Muscle biopsy findings in 28 Quarter Horse, Paint, and draft-related breeds of horses.∗

SS = Subsarcolemmal glycogen aggregates; CB = central bodies; AR = amylase-resistant inclusions.
Breeds of horses studied and histopathologic diagnosis.∗
QH = quarter horse.
Thirteen EPSSM-affected horses were male, and nine were female, with mean age of 9.4 years (range, 2–23 years). Six horses did not have evidence of EPSSM on the basis of findings for PAS-stained sections. Three were male and three were female, with mean age of 7.3 years (range, 0.5–16 years). Mean age of EPSSM-positive horses was not significantly different than that of EPSSM-negative horses (
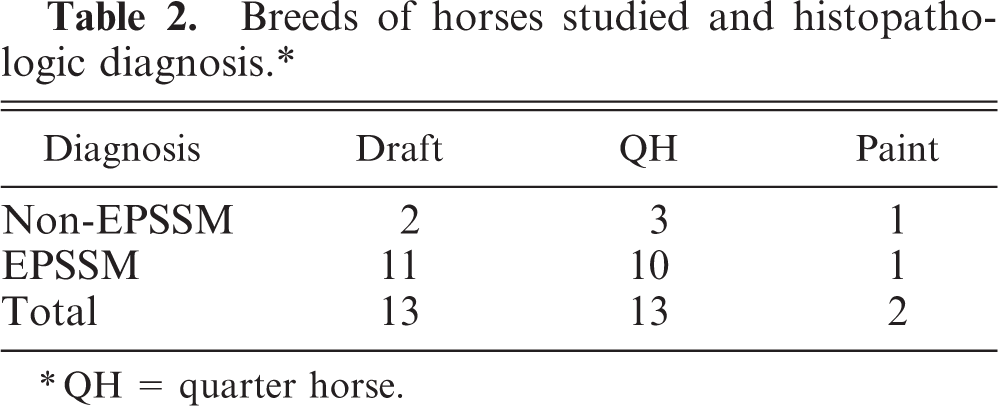
Ubiquitin expression was detected in corpora amylacea of human brain, as described, 2, 5, 14 and in corpora amylacea of equine brain. Dilutions of 1 ° 500 and 1 ° 1,000 resulted in similar intensity of ubiquitin expression, but the higher dilution resulted in less nonspecific background staining. Abnormal PAS-positive material in 21 EPSSM-affected horses also expressed ubiquitin. One horse, a 2-year-old spotted draft gelding, was diagnosed with EPSSM on the basis of numerous subsarcolemmal aggregates of amylase-sensitive glycogen, but none expressed ubiquitin. Ubiquitin expression was not detected in horses judged to be EPSSM negative on the basis of PAS staining. In all horses with amylase-resistant material, the degree of ubiquitin expression was greater than that of amylase-resistant material (Fig. 1). In addition, ubiquitin was expressed by many amylase-sensitive subsarcolemmal glycogen aggregates and by all amylase-sensitive central bodies (Fig. 2), albeit less intensely than that seen associated with amylase-resistant inclusions (Fig. 1).
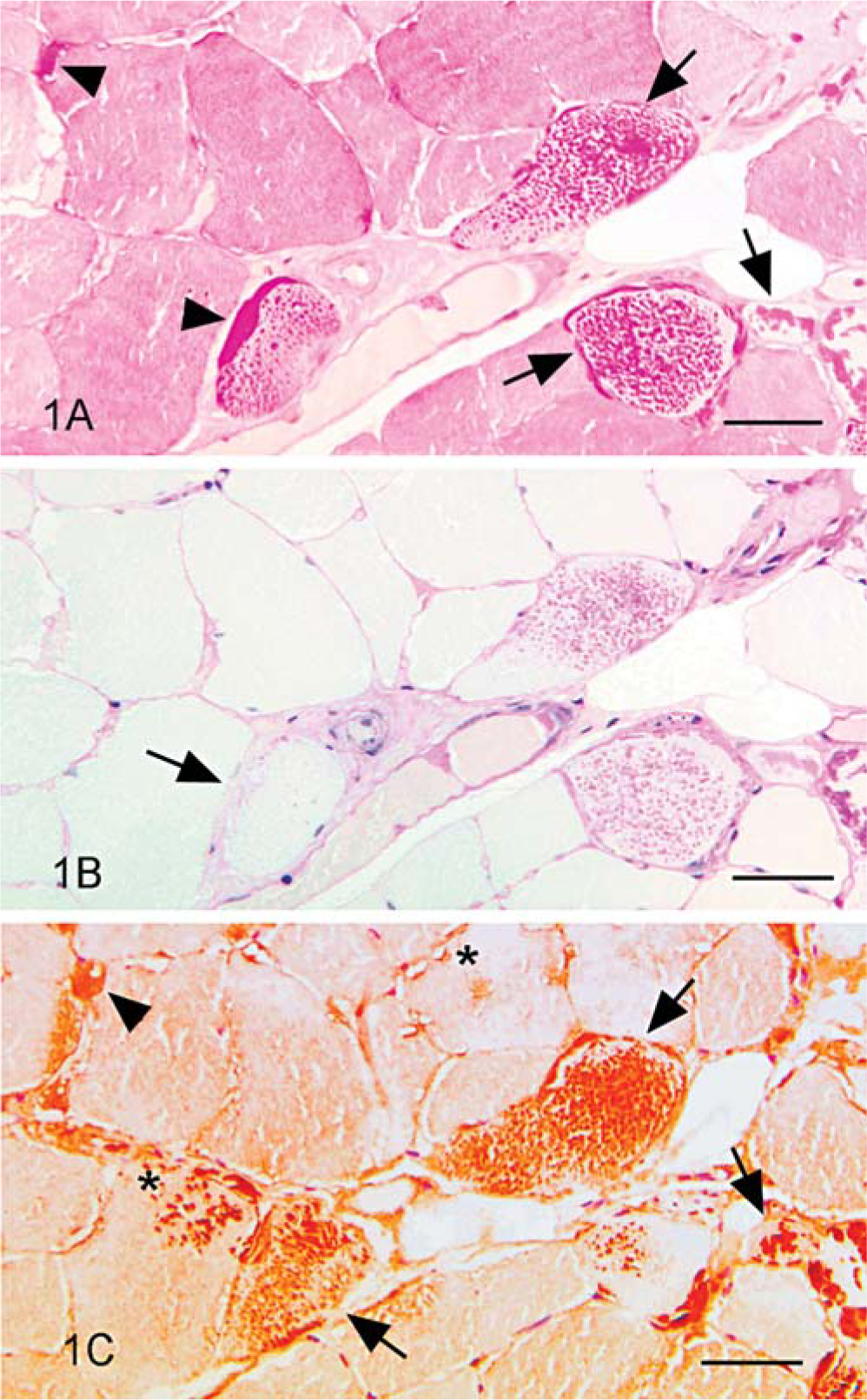
Serial transverse sections of skeletal muscle; horse with polysaccharide storage myopathy (EPSSM).
Serial transverse sections of skeletal muscle; horse with EPSSM. Multiple myofibers contain subsarcolemmal aggregates and central bodies composed of amylase-sensitive glycogen, all of which express ubiquitin. Ubiquitin expression is less intense than that of the amylase-resistant material in Fig. 1.
Amylase-resistant material was detected in horses aged 2–23 years. All horses with amylase-resistant polysaccharide also had subsarcolemmal aggregates of amylase-sensitive glycogen. The 2 horses with amylase-sensitive central bodies also had many amylase-sensitive subsarcolemmal aggregates of glycogen, and one had inclusions of amylase-resistant polysaccharide. Amylase-resistant inclusions were small and faint after amylase digestion in three horses aged 2, 3, and 5 years. Ubiquitin was more readily detected than was amylase-resistant material in these young horses. The mean age of these three horses (3.3 years) was significantly less than the overall mean age of the EPSSM-affected horses (9.4 years;
Discussion
Ultrastructural studies have characterized the skeletal muscle inclusions of EPSSM as nonlysosomal-bound intracytoplasmic aggregates of β-particles of glycogen or aggregates of glycogen admixed with filamentous material. 16, 17, 20 Ubiquitin detects abnormal proteins, 2, 11, 13– 15 and the findings of this study indicated that inclusions in muscle from horses with EPSSM contain abnormal protein that is ubiquitinated. Similar findings have been reported in polysaccharide storage myopathy in people; the protein component of the ubiquitinated polysaccharide inclusions was not identified. 10 Intracellular glycogen exists as two pools, proglycogen and macroglycogen. 1 Formation of proglycogen and macroglycogen is regulated principally by glycogenin, an autocatalytic protein that forms a part of the glycogen molecule. 1 Ubiquitin expression of glycogen-related polysaccharides in myopathic conditions may reflect abnormal folding or other alteration of the protein component of the glycogen molecule. There is evidence to suggest that there is an increased rate of glucose uptake in EPSSM-affected muscle, 7 but it is not yet known whether there is an alteration in the ratio of proglycogen to macroglycogen in affected horses. Intramyofiber inclusions in EPSSM-affected horses displace and disrupt myofibrils, 16, 17, 20 and affected fibers often exhibit reduced glycogen staining in the remainder of the cytoplasm. 17 In addition to abnormal energy production, 3 disruption of normal myofiber function due to the presence of ubiquitinated inclusions is possible.
Findings in routinely sectioned EPSSM-affected muscle can be subtle, and diagnosis has relied on evaluation of sections stained with PAS for glycogen. 4, 6, 7, 16– 23 It has been proposed that glycogen storage precedes development of amylase-resistant inclusions in EPSSM-affected horses, 23 and the findings of this study support that hypothesis. Results of this study suggest that ubiquitin expression precedes the development of amylase-resistant inclusions. It is possible that ubiquitination contributes to development of amylase resistance of glycogen aggregates. The rate of progression from glycogen aggregates to amylase-resistant inclusions is variable. In some horses (up to 13 years of age in this study) only aggregates of amylase-sensitive glycogen, either in a subsarcolemmal location or as central bodies, were detected. Some horses develop amylase-resistant inclusions at an early age (as early as 2 years in this study), whereas others experience much slower progression. Age-related progression of abnormal polysaccharide storage has also been reported in studies of EPSSM-affected foals. 6
Some investigators diagnose EPSSM only in cases with amylase-resistant material. 16, 17 Results of ubiquitin immunohistochemical analysis support the PAS stain criteria used for diagnosis of EPSSM that include the presence of amylase-sensitive subsarcolemmal aggregates of glycogen and of amylase-sensitive central bodies. 20– 23 Ubiquitin expression was greater than the degree of abnormal PAS-stained material in all but one of the 22 EPSSM-affected horses studied. Muscle from this 2-year-old horse did not express ubiquitin despite a diagnosis of EPSSM made on the basis of PAS-stained sections containing numerous subsarcolemmal aggregates of amylase-sensitive glycogen. Given the age-related progressive development of abnormal findings in muscle of EPSSM-affected horses, 6 it is possible that young EPSSM-affected horses, in which biopsy specimens contain only nonubiquitinated glycogen aggregates, might develop more severe muscle changes over time.
This study relied on archival samples submitted for evaluation for evidence of EPSSM. Therefore, true control horses were not studied. Additional studies of ubiquitin expression in muscle from clinically normal horses and from horses with other neuromuscular disorders are clearly needed.
Ubiquitin expression as a diagnostic aid was most useful in horses with only amylase-sensitive glycogen aggregates and in horses with early amylase-resistant inclusions. The results of this study suggest that, although ubiquitin expression is often greater than the degree of abnormal polysaccharide detected with PAS staining, detection of ubiquitin expression is no more sensitive for diagnosis of EPSSM than is careful evaluation of an adequate and appropriately handled muscle specimen stained with PAS for glycogen.
