Abstract
An adult pet rabbit (
Keywords
Testicular tumors are uncommonly reported in rabbits. Zwicker and colleagues postulated that the apparent low incidence of cases of testicular tumors in laboratory rabbits may be due to infrequent microscopic evaluation of rabbit testes and because most laboratory rabbits do not live long enough to develop these neoplasms.
34
Testicular tumors reported to occur in rabbits include Leydig cell tumor,
9,11,17,18,34
seminoma,
3,4,24
teratoma,
21
adenocarcinoma,
14
and mixed cell–type tumors.
28,32
Leydig cell tumor, alone or in combination with another testicular tumor, appears to be the most frequently encountered histologic type.
9,11,17,18,28,32,34
Granular cell tumor (GCT) occurs in many animal species but to date has not been reported in rabbits.
15,19,23,26
In postnatal animals and humans, GCT may occur in the genital tract, including the vulva, clitoris, vagina, cervix, and/or uterus of cats, rodents, and/or humans, and in the skin of the penis and scrotum of humans.
5,7,16,19
Although GCT has not been reported in the testes of animals or postnatal humans, it occurred in the testes of a 29-week-old human fetus with GCT in multiple tissues.
25
A unique characteristic of GCT is the presence of numerous secondary lysosomes when examined by transmission electron microscopy (TEM).
19
The cell of origin of GCT has been the subject of much debate; however, current evidence indicates that this tumor may arise in any tissue. Many cases of GCT, but not all, are neuroectodermal in origin.
12
Neuroectoderm gives rise to Schwann cells, neuroendocrine cells, neurons, neuroglia, and pigment-producing cells. Tumors of nonectodermal origin also exhibit GCT-like changes.
12,26
These data indicate that GCT may be a phenotypic change that occurs in neoplastic cells irrespective of their embryonal origin or anatomical location. The present case report describes the exfoliative cytologic, histologic, electron microscopic, and limited immunohistochemical findings of GCT in the left testis of an adult pet rabbit (
A male, sexually intact, 2.5-kg, mixed-breed, adult pet rabbit 8 years of age presented for progressive enlargement of the left testicle of 2 months' duration. The enlarged testicle was approximately 9 cm in length and firm. There were no lesions in the scrotal skin, the testes moved freely in the scrotal sac, and there were no physical examination findings indicative of gynecomastia. Preanesthetic blood work, including serum biochemistry and hematocrit, were unremarkable. Because neoplasia was suspected, bilateral orchiectomy was performed and the rabbit was sent home after an uneventful recovery. Impression smears and scrapings of the cut surface of the left testicle were prepared and stained with an automated modified Wright's stain (Advia S60, Bayer Health Care LLC, Diagnostic Division, Tarrytown, NY) and evaluated by light microscopy. The left testicle was fixed in 10% buffered neutral formalin and processed routinely for histopathology. The cut surface of the formalin-fixed enlarged testicle was firm and very light brown. The scrotum and the contralateral testis were not submitted for microscopic evaluation. Small fragments of the left testicle were also fixed in modified Karnovsky's fixative, post fixed in osmium tetroxide, processed routinely to epoxy-embedded tissue sections, and evaluated with a transmission electron microscope (FEI/Phillips 100-CM, Hillsboro, OR). Paraffin-embedded tissue sections were immunolabeled for cytokeratins (mouse monoclonal antibody, clone AE1/AE3, M3515; Dako Corp., Carpinteria, CA), broad-spectrum cytokeratins (mouse monoclonal antibody, clone MNF116; M0821, Dako Corp.), vimentin (mouse monoclonal antibody, clone Vim 3B4; M0720, Dako Corp.), Melan-A (mouse monoclonal antibody, clone A103, M7196; Dako Corp.), neuron-specific enolase (mouse monoclonal antibody, clone BBS/NC/VI-H14, M0873; Dako Corp.), muscle actin (mouse monoclonal antibody, clone HHF35, M0635; Dako Corp.), desmin (mouse monoclonal antibody, clone D33, M0760, Dako Corp.), Ki-67 (mouse monoclonal antibody, clone 7B-11, 18-0192; Zymed, Carlsbad, CA), synaptophysin (mouse monoclonal antibody, clone SY776, M0776; Dako Corp.), CD68 (mouse monoclonal antibody, clone EBB11, M0718; Dako Corp.), and inhibin alpha (mouse monoclonal antibody, clone R1, MCA951ST; AbD Serotec, Raleigh, NC) as previously described. 22 Atrophied testicular tissue adjacent to the tumor, tissue sections from other animals, and normal rabbit testis were used as control tissues. Other markers evaluated in GCT and testicular tumors were not examined because available antibodies were rabbit polyclonal antibodies and because of limited tissue availability.
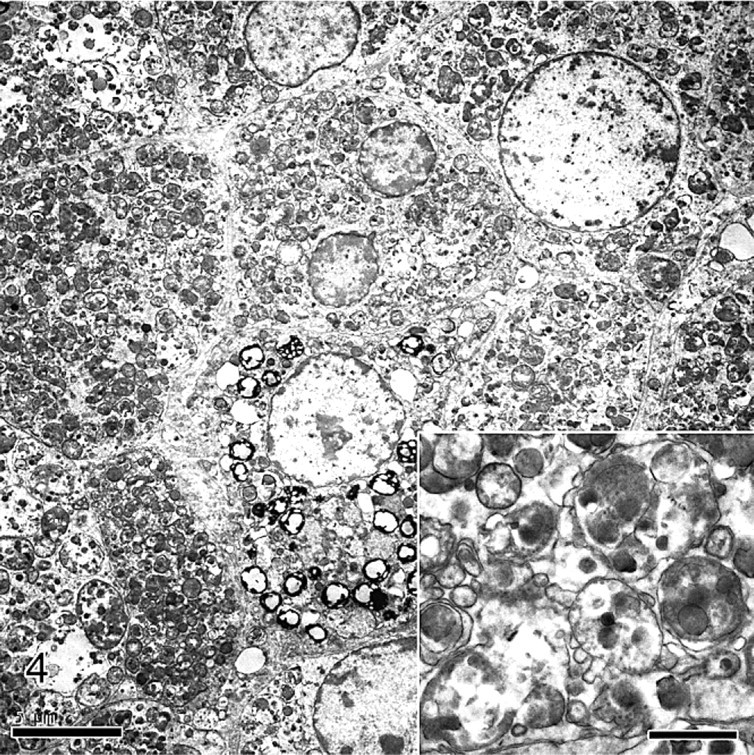
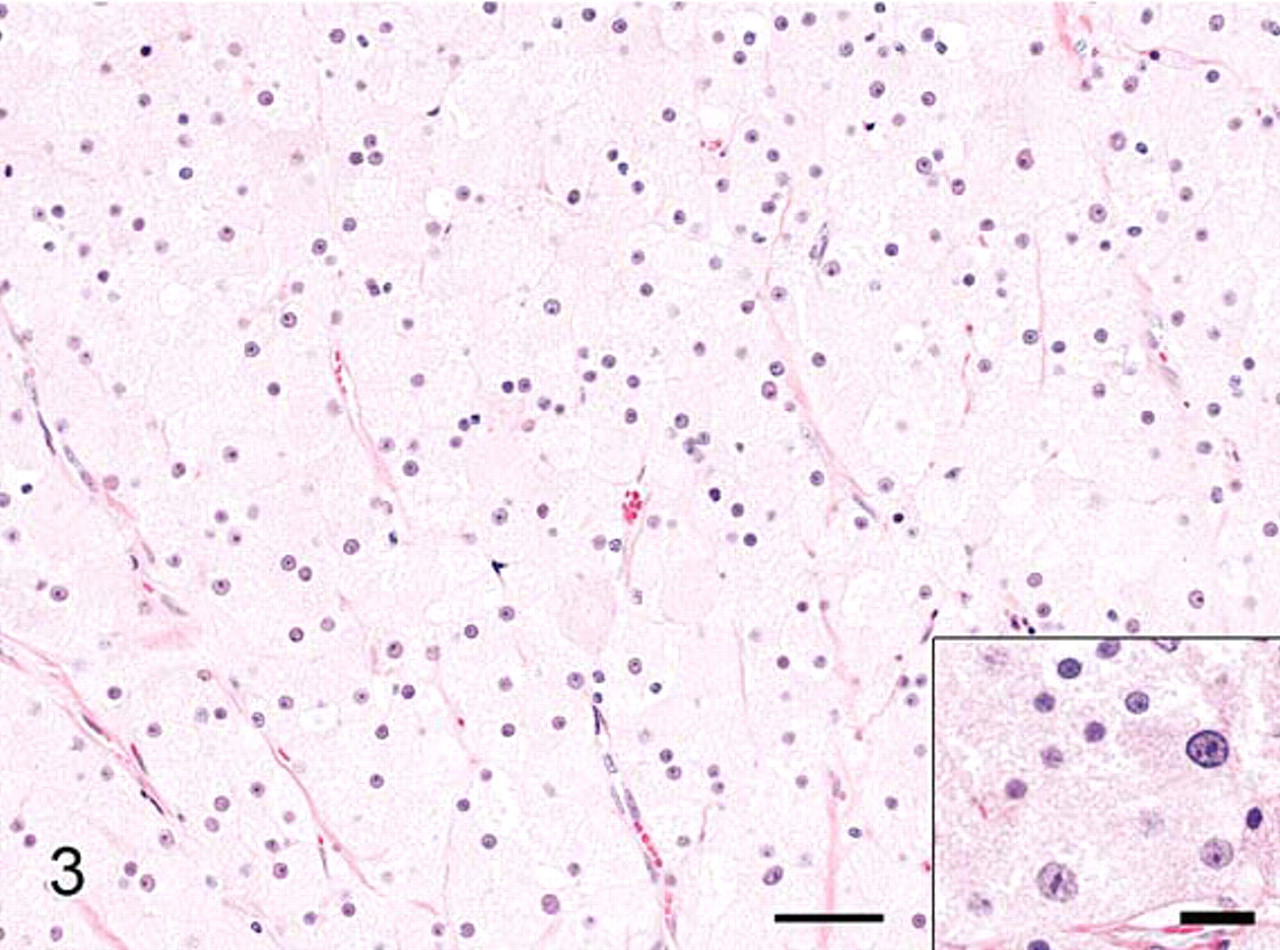
The impression smears were markedly cellular and consisted of a monotonous population of individualized polygonal cells and dense cellular aggregates in an abundant pink to magenta granular background (Fig. 1). The cells had indistinct cell borders and abundant bluish cytoplasm filled with abundant pink to magenta granular material that often obscured the nucleus. Nuclei were round to oval with finely stippled chromatin and single small nucleoli. Histologically, the testicle was replaced by a large, encapsulated, multilobular, densely cellular mass that compressed remnants of atrophied testicular tissue (Fig. 2). The mass was composed of sheets and aggregates of large polygonal neoplastic cells separated by fine fibrovascular stroma. The neoplastic cells had abundant granular eosinophilic cytoplasm, round to oval nuclei with finely clumped chromatin, and single prominent nucleoli (Fig. 3). Few cells contained small clear round cytoplasmic vacuoles that were interpreted as lipid. The degree of anisokaryosis and anisocytosis was moderate. The number of cells in mitosis was 0–1 per 40× lens objective field (26 mm in diameter). The cytoplasm of the neoplastic cells stained light to moderately pink with periodic acid Schiff stain (data not shown). By electron microscopy the cytoplasm of the tumor cells was full of dense aggregates of membrane-bound vesicles, consistent with secondary lysosomes, containing amorphous electron-dense debris, fragments of other organelles, and few membranous arrays (myelin bodies), findings diagnostic of GCT (Fig. 4). A few cells contained cytoplasmic lipid vacuoles. Immunohistochemistry (IHC) results are shown in Table 1. Melan A and vimentin were the only markers detected in the majority of the neoplastic cells. Neuron-specific enolase-positive cells occurred multifocally.
Immunohistochemical results.
∗Cytoplasmic granular staining occurred in multifocal clusters of cells throughout the tumor.
†NA = not applicable.
‡Leydig cells were negative in control rabbit testis but positive in control dog testis.
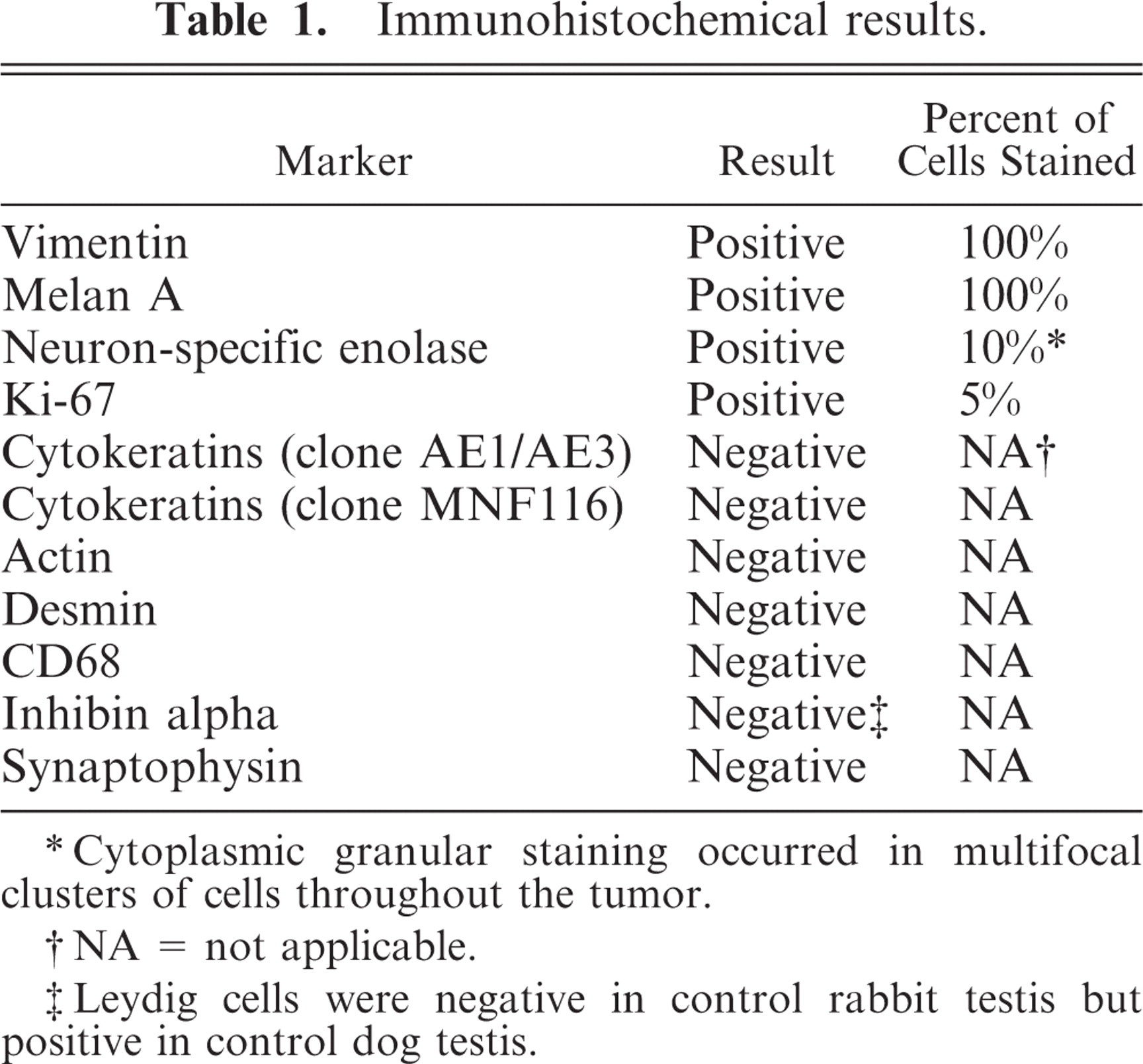
Granular cell tumor, testis; rabbit. Ultrastructurally, the cytoplasm of all neoplastic cells was full of secondary lysosomes containing amorphous debris, membranous debris, and fragmented organelles. A few cells contained cytoplasmic lipid vacuoles. Uranyl acetate and lead citrate. Bar = 5 μm. Inset, higher magnification of the cytoplasm of the tumor cells. Bar = 1 μm.
Granular cell tumor, testis; rabbit. Aggregates and lobules of polygonal neoplastic cells with abundant eosinophilic granular cytoplasm. HE. Bar = 100 μm. Inset Bar 20 μm.
Granular cell tumor, testis; rabbit. Encapsulated and lobulated expansile mass compresses adjacent atrophied testicular tissue. Few seminiferous tubules remain (arrows). HE. Bar = 200 μm.
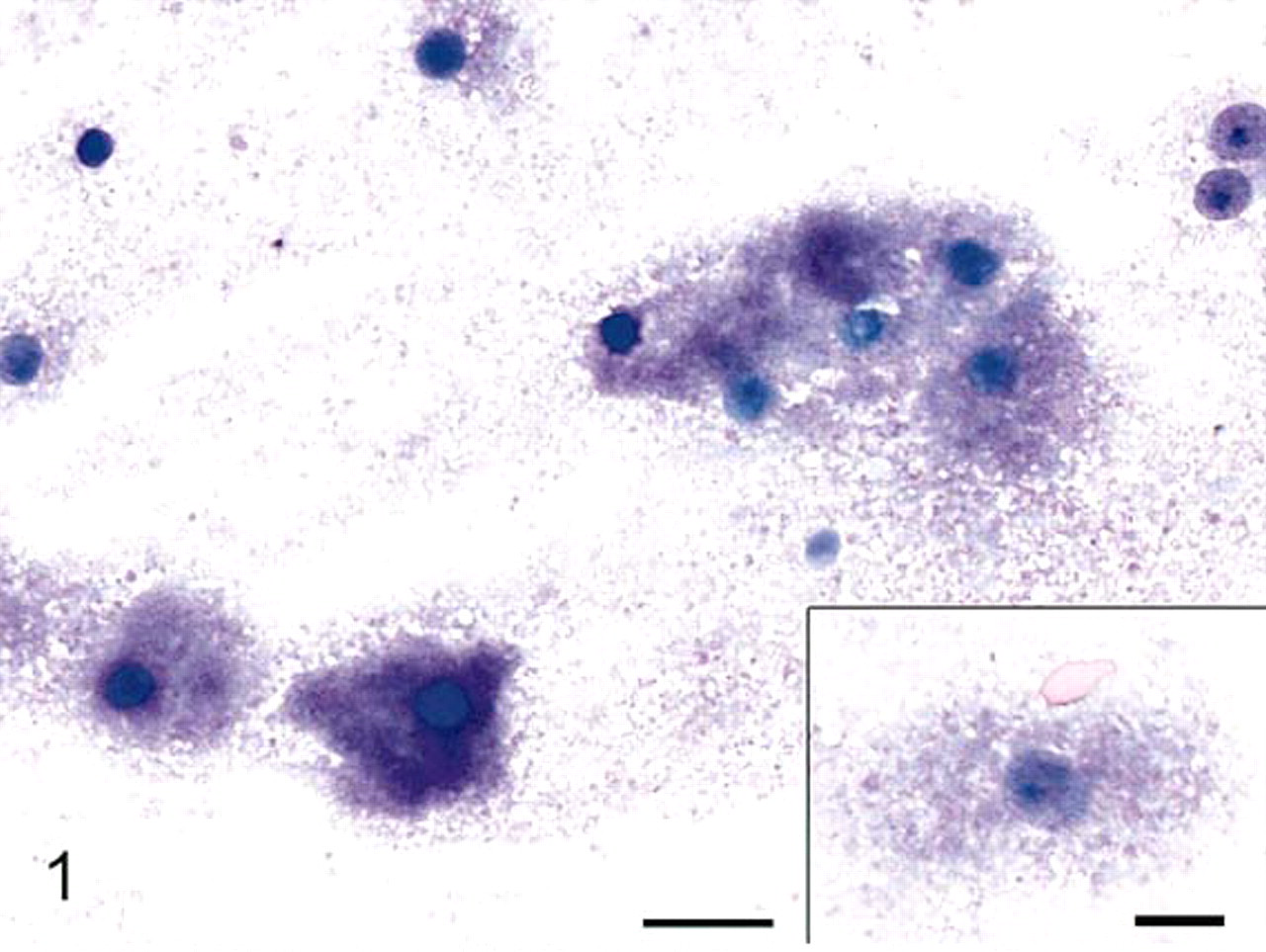
Granular cell tumor, testis; rabbit. Individualized cells and cellular aggregates in a pink to magenta granular background. The cells are filled with granular pink to magenta granular material. Modified Wright's stain. Bar = 20 μm. Inset, higher magnification of the cells. Inset bar = 10 μm.
The exfoliative cytologic features of GCT are unique and include a monotonous population of cells with abundant amounts of pink to purple/magenta cytoplasmic granular material. 1,13,31 In the present report, the exfoliative cytologic findings of the tumor were diagnostic of GCT and not of other primary testicular tumors, such as Leydig cell tumor. 8,20 Despite the confidence in the cytologic diagnosis, the tumor presented a diagnostic challenge histologically because of the anatomic location and shared histologic characteristics between GCT and Leydig cell tumor. Leydig cell tumor was the preferred differential diagnosis when hematoxylin and eosin (HE)-stained histologic sections of the rabbit tumor were evaluated blindly, that is, without knowledge of the cytologic or TEM findings, by multiple board-certified veterinary pathologists. Histologic characteristics shared by both Leydig cell tumor and GCT include cells with abundant granular eosinophilic cytoplasm and the formation of sheets and aggregates of neoplastic cells in fibrovascular stroma. 6 In contrast to the eosinophilic granular cytoplasm of GCT, which is due to great numbers of secondary lysosomes, the eosinophilic granular cytoplasm of Leydig cell tumors in HE tissue sections is due to cytoplasmic organelles, such as mitochondria with tubular cristae and abundant smooth endoplasmic reticulum. TEM findings in this rabbit were diagnostic for GCT in other animal species and were not consistent with TEM findings of Leydig cell tumor or non-neoplastic Leydig cells. 10,19,29,30,33 Unfortunately, we could not adequately compare our TEM findings with those of Leydig cell tumor in rabbits because of the limited availability of TEM evaluation in previous reports. Only one report of Leydig cell tumor in a rabbit had TEM findings, but the results were difficult to interpret because of inadequate fixation and the lack of published TEM images. 34 Similar to the current report, IHC is often utilized in cases of GCT to characterize antigen expression and cell lineage, but it is not necessary to obtain a definitive diagnosis. IHC is not frequently utilized in the diagnosis of Leydig cell tumor in animals, likely because the diagnosis is straightforward with exfoliative cytology and histology. 8,20 The IHC results of the present case were generally consistent with IHC results of GCT and Leydig cell tumor or non-neoplastic Leydig cells of other animal species and/or humans. 2,13,15,18,19,22,26,27 Although speculative, the tumor in the rabbit may have been derived from Leydig cells, as supported by Melan A immunoreactivity and anatomic location. Melan A immunoreactivity occurs in melanocytes (a neuroectoderm derivative) and in steroid-producing sex-cord-stromal cells and tumors, such as Leydig cell tumors, of domestic animals, including the rabbit, and man but has not previously been evaluated in GCT. 17,22,27 IHC for steroidogenic enzymes may have been of additional assistance in determining if this tumor was derived from Leydig cells. A full immunohistochemical evaluation of normal rabbit testes and the tumor would have been ideal but was not performed. The rabbit has remained free of local recurrence and metastatic disease for 2 years.
Footnotes
Acknowledgements
We greatly appreciate the expertise and work of the Special Microscopy (S. White, J. Baker) and Histology (B. Solomon, M. Hyten) laboratories at Lilly Research Laboratories and the Animal Disease Diagnostic Laboratory at Purdue University (D. DuSold).
