Abstract
Unilateral testicular interstitial (Leydig) cell tumor and gynecomastia were diagnosed in an adult male rabbit. The interstitial cell tumor was a well-circumscribed, 2-mm diameter, pale tan nodule composed of a uniform population of polygonal cells. Neoplastic interstitial cells exhibited diffuse, granular cytoplasmic staining with Melan A, a marker of steroid-producing cells in humans and dogs. Multiple subcutaneous masses in the caudal abdomen were associated with enlarged nipples and consisted of hyperplastic mammary gland tissue with proliferation of ducts and alveoli, marked lobule formation, and pseudolactational hyperplasia. Many epithelial cells lining the hyperplastic ducts and alveoli exhibited intense nuclear expression of progesterone receptor antigen, whereas myoepithelial cells showed strong nuclear staining for p63 antigen. This is the first report of concurrent interstitial cell tumor and gynecomastia in a rabbit and also the first description of gynecomastia in this species.
Gynecomastia is defined as benign enlargement of the male mammary gland caused by an increased ratio of estrogens relative to androgens. 2, 20 Imbalances between estrogen and androgen may be caused by increased levels of free estrogen, decreased levels of free androgen, or androgen receptor defects. 2, 20 In men, specific causes of gynecomastia include hypogonadism associated with pseudohermaphroditism and Klinefelter's syndrome, puberty, endocrine dysfunction, malnutrition, hepatic cirrhosis, drug therapy, and, rarely, testicular neoplasms. 2, 20 In men with testicular neoplasia, gynecomastia is most commonly associated with interstitial (Leydig) cell tumors but may also occur with Sertoli cell tumor and seminoma. 2, 20
In veterinary medicine, gynecomastia is most often recognized as a component of the feminization syndrome that develops in up to 20 to 30% of dogs with Sertoli cell tumor, 8, 9 and less commonly in dogs with interstitial cell tumors. 11, 14 Gynecomastia is also a component of milking buck syndrome, a genetic disorder of various dairy goat breeds. 1 Although interstitial cell tumors are common testicular neoplasms in older domestic bulls and Fischer 344 rats, occurrence of gynecomastia or other signs of apparent hyperestrogenism have not been described in these species. 9, 13
Testicular neoplasms are considered rare in domestic rabbits, 6 with interstitial cell tumors being one of the most commonly documented types. 3, 10, 17, 18, 21 Most cases involved adult rabbits between the ages of 3 and 9 years, 3, 10, 17, 18 suggesting that, as in other species, interstitial cell tumors develop primarily in older bucks; none was associated with gynecomastia. One case, 21 however, involved bilateral interstitial cell tumors in a juvenile rabbit.
A 2.7-kg, white, adult male rabbit was presented to the Purdue University Veterinary Teaching Hospital by a local humane society for elective castration. Multiple subcutaneous masses were identified during the initial physical examination, and humane euthanasia was elected because the rabbit was deemed a poor candidate for adoption. The testes were removed immediately after euthanasia and submitted along with the rabbit for postmortem examination.
At necropsy, the size and external contour of both excised testes appeared normal. A solitary, well-demarcated, pale tan, 2-mm diameter nodule was observed in 1 testis upon longitudinal sectioning. The nodule was surrounded by grossly normal testicular parenchyma. Several raised, sparsely haired mammary masses measuring up to 1 cm in diameter were identified in the subcutis of the caudal abdomen and left flank. The masses were fluctuant on palpation and contained inspissated brown secretions. Two of the masses were associated with enlarged nipples (Fig. 1). Moreover, all nipples were readily visible because of mild enlargement and partial alopecia of the surrounding skin. Multiple firm, well-circumscribed masses up to 4 cm in greatest dimension were identified in the dermis on the right side of the thorax and on the ventral midline overlying the sternum. The dermal masses were in haired skin and were homogenously white and fibrous on cut section.
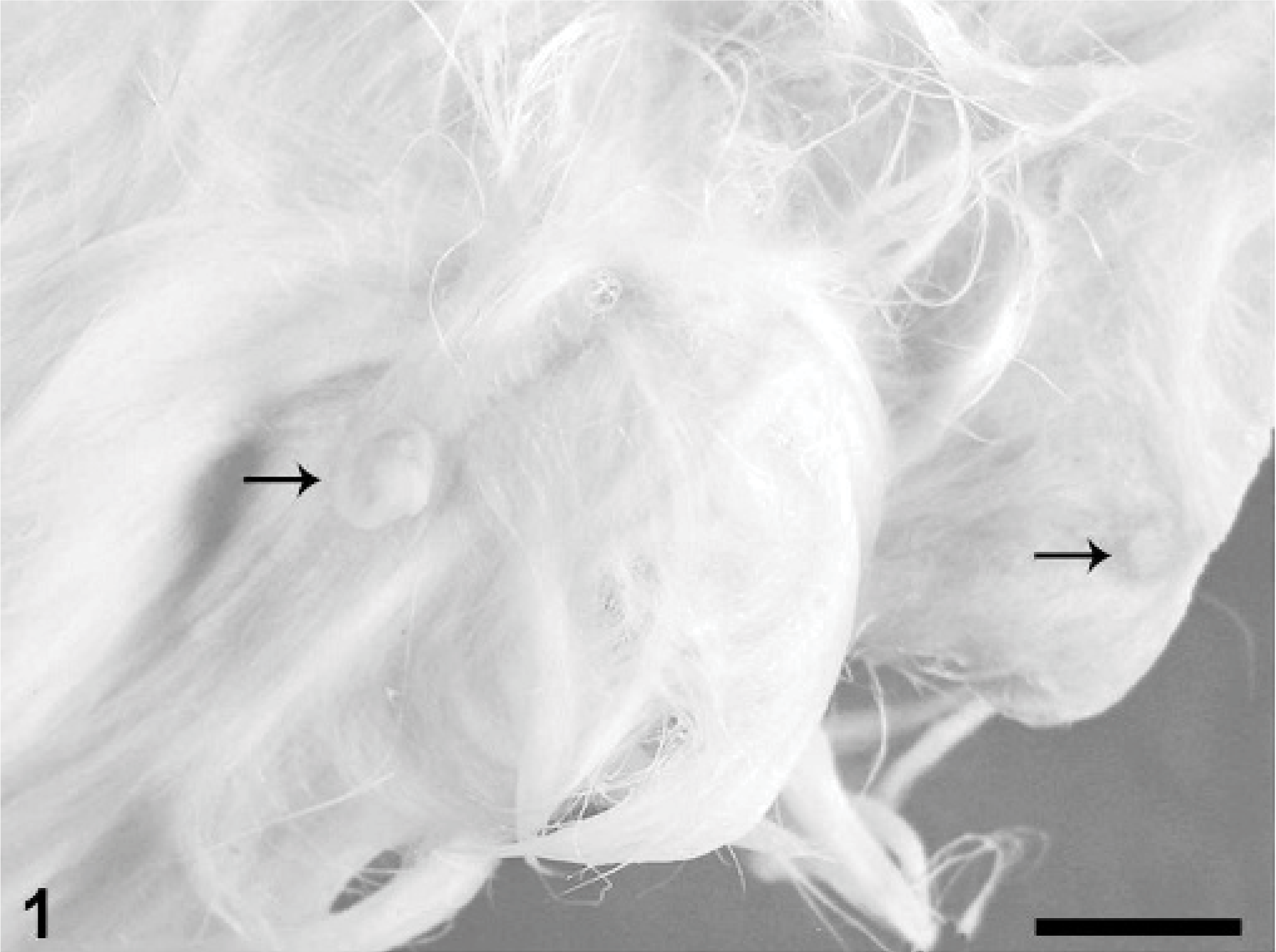
Haired skin of ventrum; rabbit. Two enlarged nipples (arrows) are associated with fluctuant subcutaneous masses. Bar = 1 cm.
Tissue specimens were fixed in 10% neutral buffered formalin, routinely processed, paraffin-embedded, sectioned, and stained with hematoxylin and eosin. Immunohistochemical tests using primary mouse monoclonal antibodies to Melan A (clone A103, 1:160, Dako Corp., Carpinteria, CA), p63 (clone 4A4, 1:200, Santa Cruz Biotechnology, Inc., Santa Cruz, CA), progesterone receptor (PR) (clone 16, 1:20, Novocastra Laboratories Ltd., Newcastle upon Tyne, UK), and estrogen receptor (ER) (clone CC4-5, 1:40, Novocastra Laboratories Ltd., Newcastle upon Tyne, UK) were performed on paraffin-embedded tissue sections. For antigen retrieval, slides were heated with a decloaker for 10 minutes in EDTA buffer (pH 9.0) for Melan A and p63 or citrate buffer (pH 6.5) for PR and ER. Detection systems were a non-avidin-biotin-peroxidase method (EnVision+, Dako Corp., Carpinteria, CA) for Melan A and p63 and a labeled streptavidin-biotin-peroxidase method (LSAB+, Dako Corp., Carpinteria, CA) for PR and ER, with diaminobenzidine (DAB) as the chromagen and Mayer's hematoxylin as the counterstain. Tissue controls were canine melanoma for Melan A, canine mammary gland for p63, and canine uterus for PR and ER.
Microscopically, the testicular mass was composed of a uniform population of polygonal cells arranged in ill-defined lobules by delicate fibrovascular stroma (Fig. 2). The polygonal cells had indistinct borders, ample finely vacuolated eosinophilic cytoplasm, and ovoid hyperchromatic nuclei with indistinct or single prominent nucleoli. The tumor was well demarcated and partially separated from surrounding parenchyma by a fine capsule, with minimal compression of adjacent tissue. Mitotic figures were not observed. Seminiferous tubules in the surrounding parenchyma and in the contralateral testis were spermatogenically active, with no apparent atrophy.
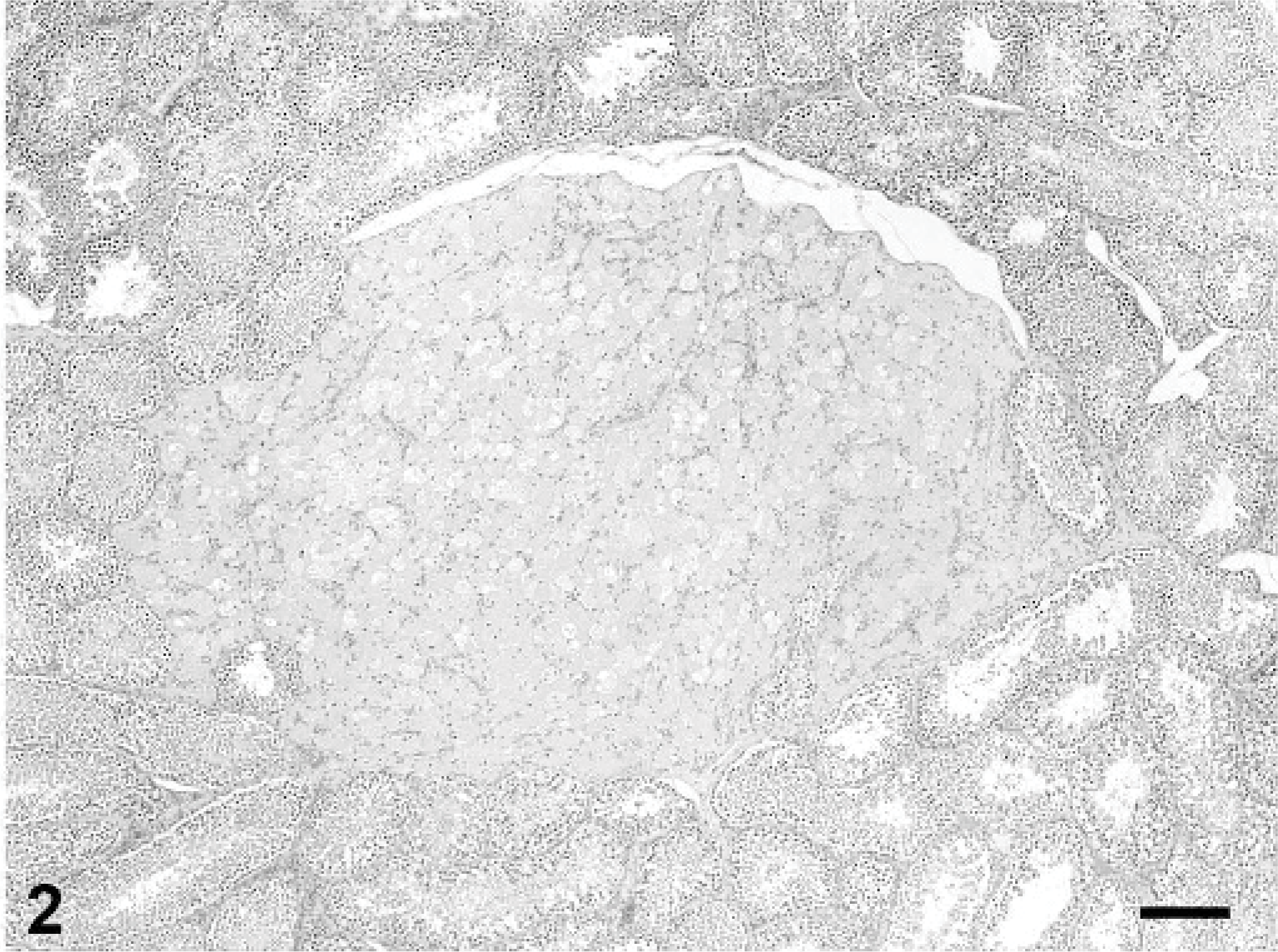
Testis; rabbit. The interstitial cell tumor is composed of large polygonal cells with abundant, finely vacuolated eosinophilic cytoplasm. HE stain, bar = 200 μm.
Immunohistochemically, neoplastic interstitial cells exhibited strong, granular cytoplasmic staining for Melan A (Fig. 3), a marker of melanocytic tumors and steroid-producing cells in humans and dogs. 16 In the rabbit, neoplastic interstitial cells lacked nuclear staining, while adjacent normal interstitial cells sometimes exhibited strong nuclear staining. This pattern of reactivity for Melan A has been previously described in some dogs with interstitial cell tumors. 16 As in dogs, normal Sertoli cells in this rabbit had diffuse cytoplasmic immunoreactivity for Melan A. 16
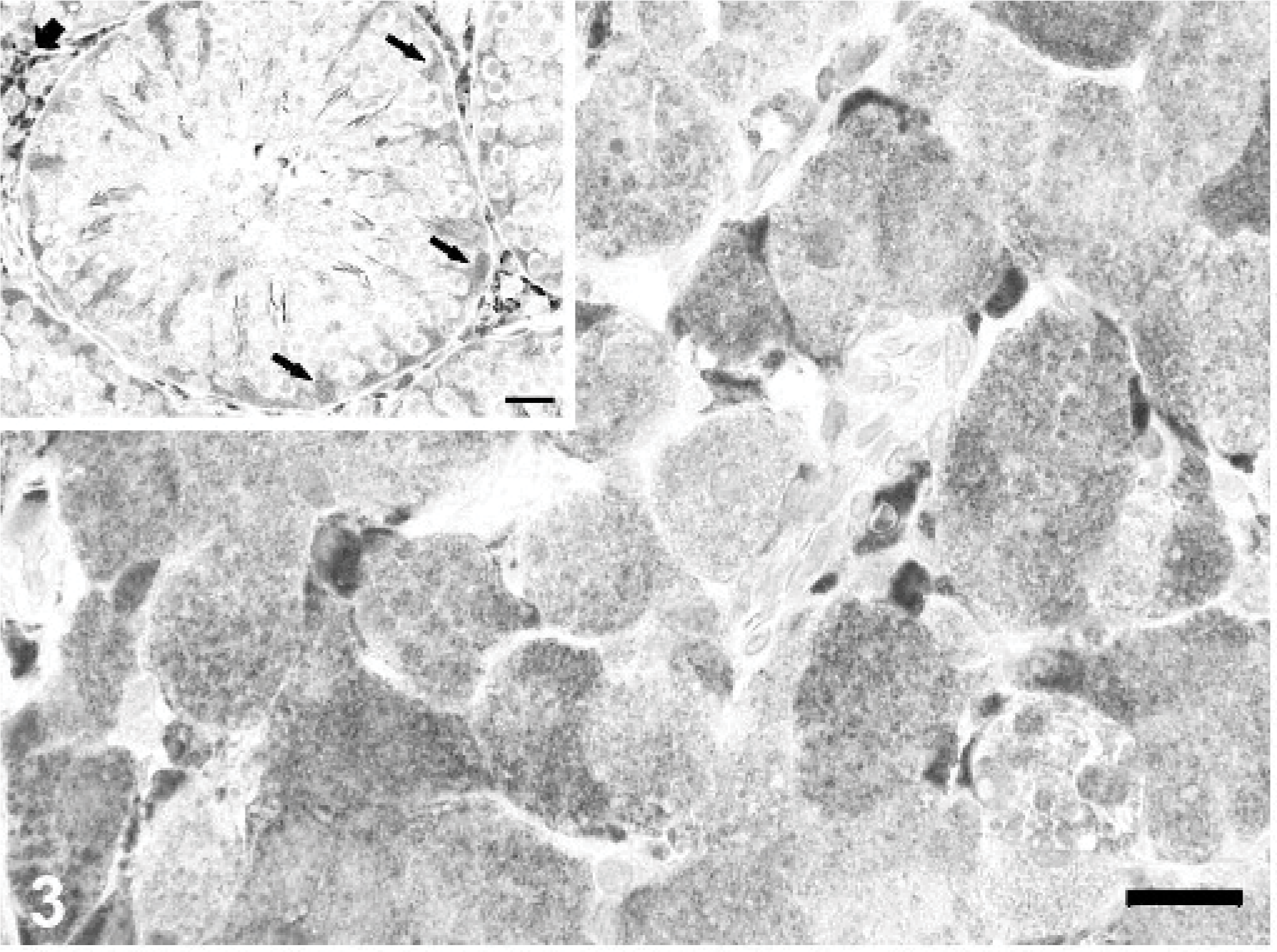
Testis; rabbit. Neoplastic interstitial cells have diffuse, granular cytoplasmic immunoreactivity for Melan A. Inset: Seminiferous tubule with cytoplasmic reactivity of Sertoli cells (black arrows) and non-neoplastic interstitial cells (short arrow). Immunoperoxidase-DAB for Melan A. Mayer's hematoxylin counterstain, bar = 20 μm; inset bar = 25 μm.
The fluctuant mammary masses from the caudal abdomen and left flank were composed of lobules of hyperplastic mammary gland tissue situated in the deep dermis and subcutis. Hyperplastic lobules were subdivided by dense fibrovascular stroma containing interlobular ducts lined by 1 to 2 layers of well-differentiated cuboidal epithelium (Fig. 4). Intralobular ducts were generally lined by a single layer of flattened or low cuboidal epithelium, with occasional branching. Many hyperplastic lobules had areas of apocrine metaplasia characterized by apical blebbing and formation of intraluminal micropapillary structures (Fig. 5) and other areas in which alveoli displayed pseudolactational hyperplasia (Fig. 6). Many ducts and alveoli were dilated and contained lakes or globules of eosinophilic secretory material.
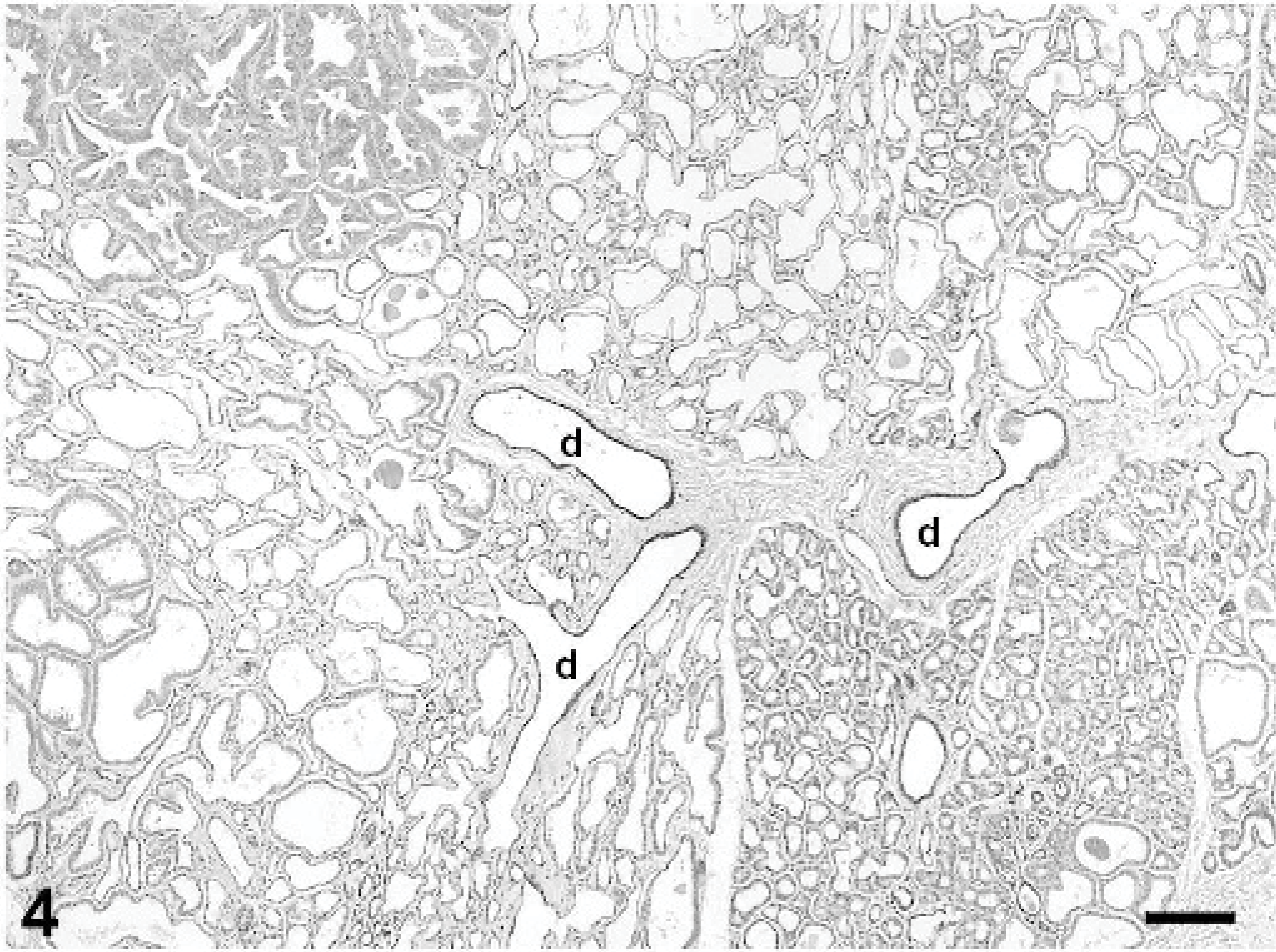
Mammary gland; rabbit. Lobules of hyperplastic mammary tissue are subdivided by collagenous stroma containing interlobular ducts (d). Alveoli and ducts are variably dilated; some contain proteinaceous secretory material. HE stain, bar = 200 μm.
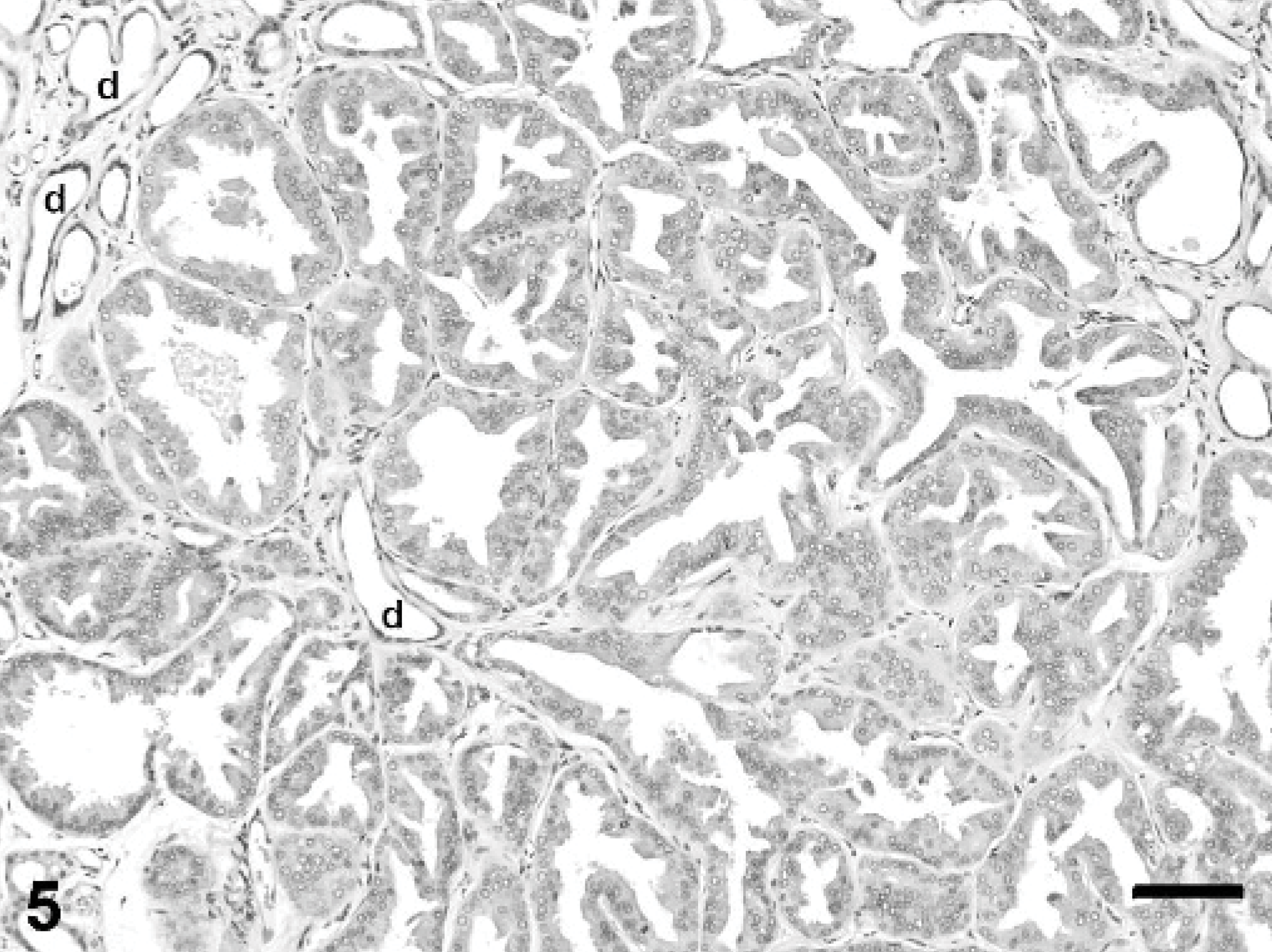
Mammary gland; rabbit. Gynecomastia with apocrine metaplasia. Hyperplastic alveolar epithelial cells have apical blebbing and are crowded with formation of intraluminal papillary projections. Several small ducts (d) are lined by 1 layer of flattened epithelium. HE stain, bar = 100 μm.
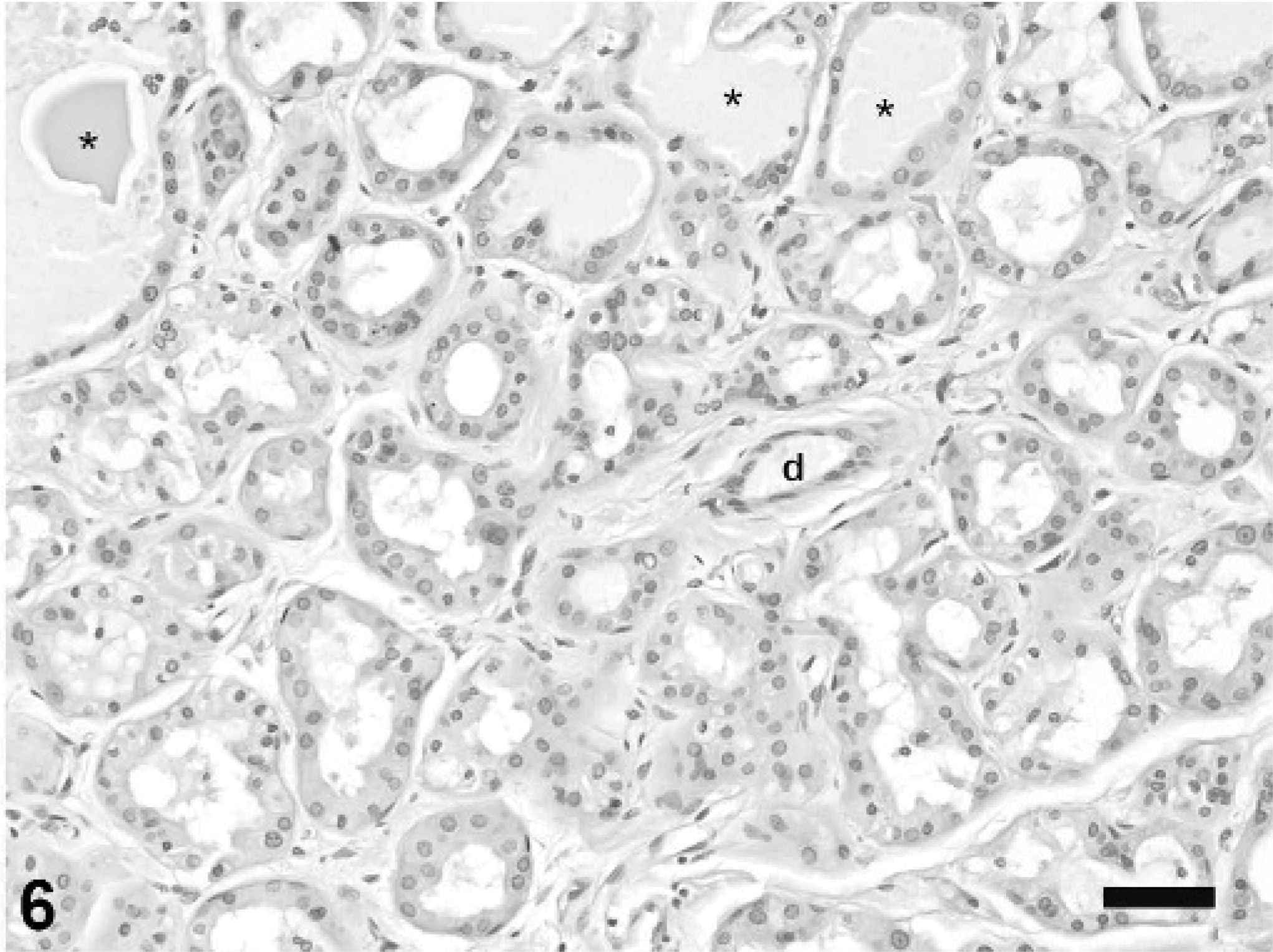
Mammary gland; rabbit. Gynecomastia with pseudolactational hyperplasia. Alveoli are lined by vacuolated epithelial cells, variably dilated, and filled with eosinophilic secretion (asterisks). An intralobular duct (d) is lined by 1 layer of low cuboidal epithelium. HE stain, bar = 50 μm.
Immunohistochemical testing for p63 antigen, a p53 homolog expressed in myoepithelial cells of human and canine mammary glands, 5 demonstrated a distinct myoepithelial cell component within the hyperplastic mammary gland lobules (Fig. 7). Similar to what has been reported in normal and hyperplastic canine mammary glands, 5 p63 staining was restricted to nuclei of myoepithelial cells. Strong staining for p63 was also observed in basal epithelial cells of the epidermis and hair follicles overlying the hyperplastic mammary tissue. Many cells lining the hyperplastic ducts and alveoli exhibited variably intense nuclear immunoreactivity for PR antigen (Fig. 8). The immunohistochemical test for ER antigen, also located in the nucleus, was inconclusive due to excessive cytoplasmic background staining.
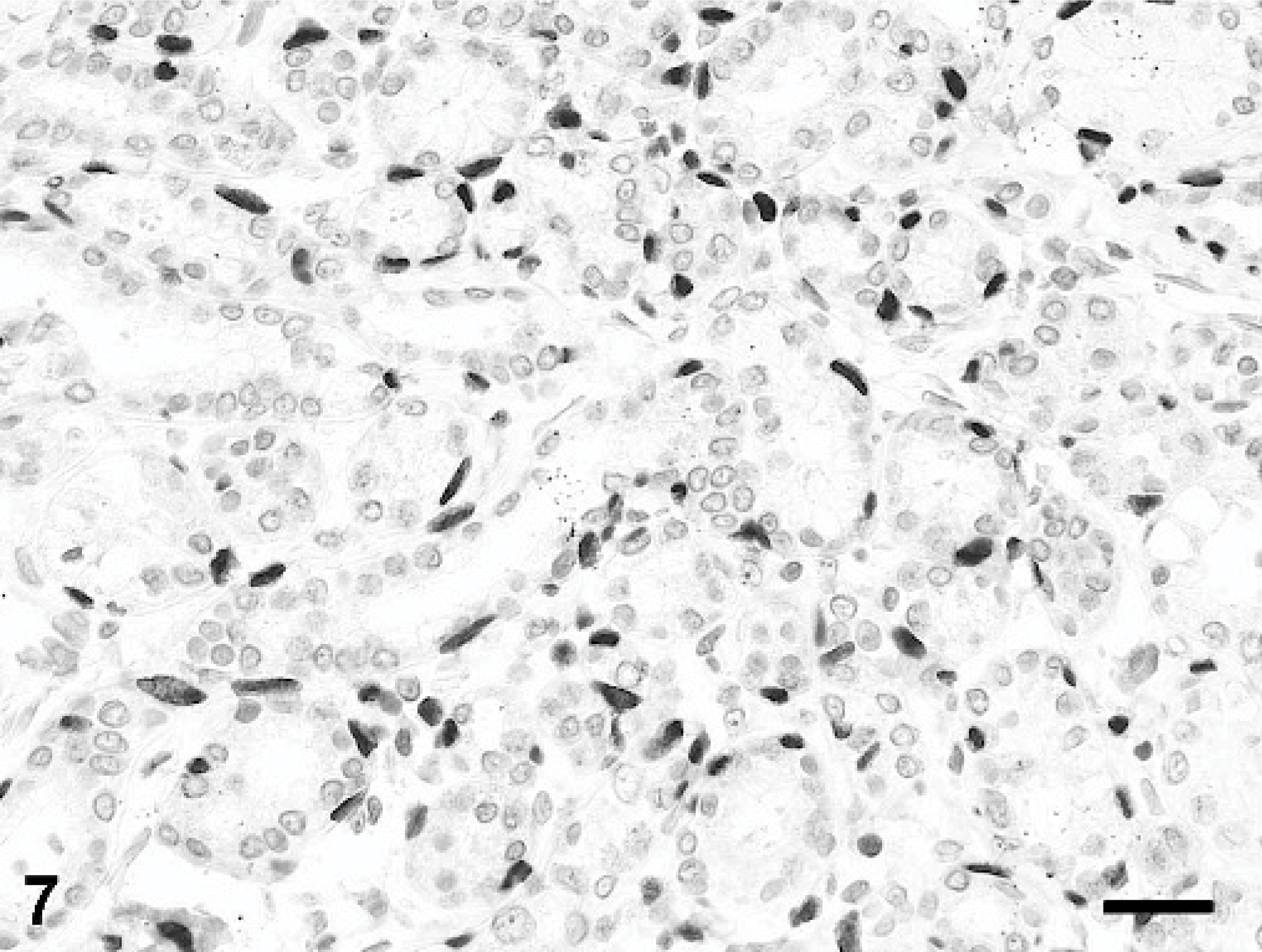
Mammary gland; rabbit. Myoepithelial cell nuclei exhibit strong nuclear reactivity for p63. Immunoperoxidase-DAB for p63. Mayer's hematoxylin counterstain, bar = 25 μm.
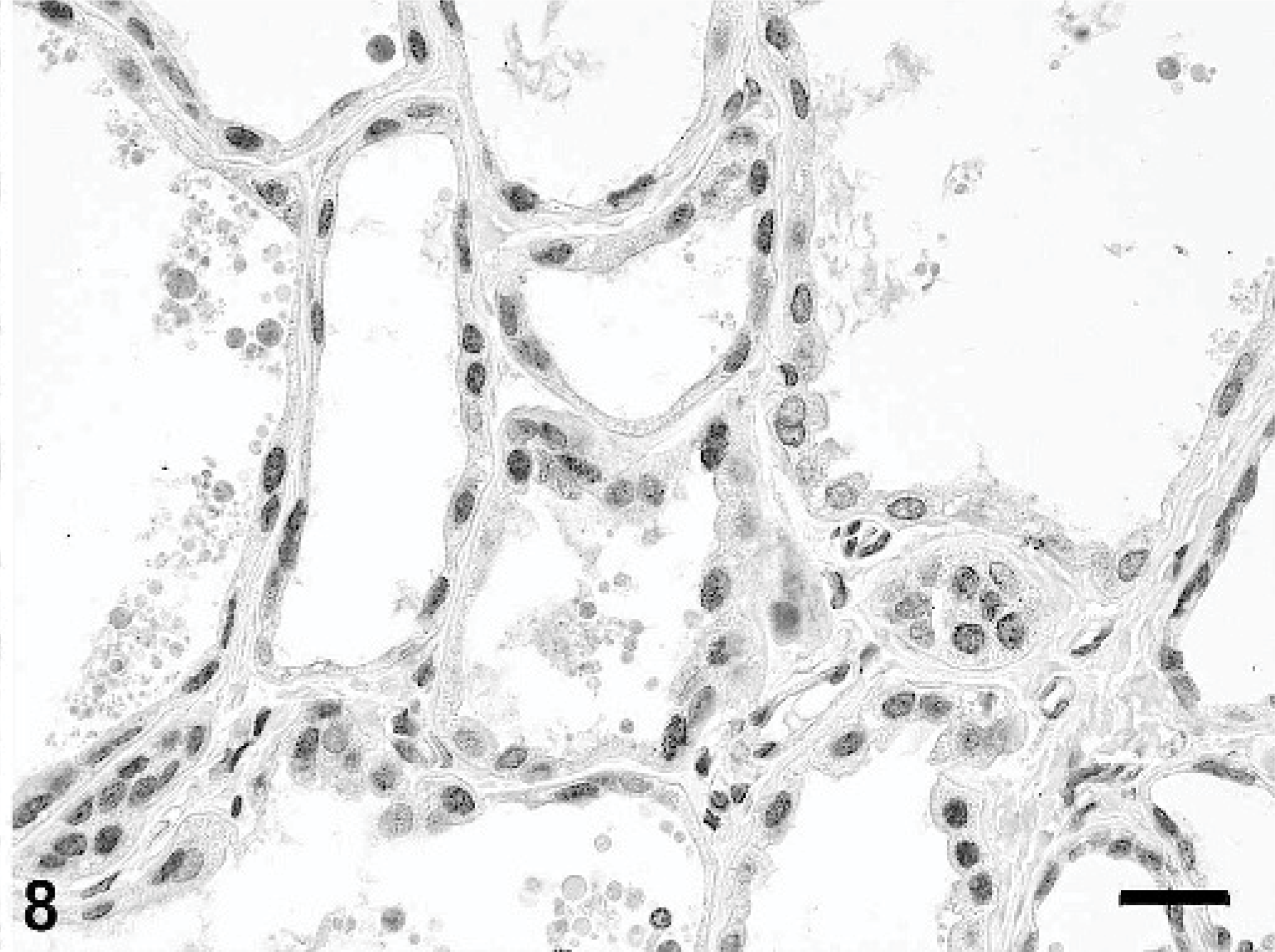
Mammary gland; rabbit. Epithelial cells lining hyperplastic alveoli have nuclear progesterone receptor (PR) expression. Immunoperoxidase-DAB for PR. Mayer's hematoxylin counterstain, bar = 25 μm.
The firm masses from the right lateral thoracic and ventral midline skin, which were not associated with mammary tissue, were diagnosed as dermal fibromas. Microscopically, the fibromas were composed of well-organized, poorly cellular, dense collagenous connective tissue that blended with surrounding dermal collagen. Additional microscopic lesions included focally extensive myocardial fibrosis at the junction of the septum and left ventricular free wall and mild, multifocal lymphocytic-plasmacytic interstitial nephritis, both of undetermined cause.
In men with functional interstitial cell tumors, gynecomastia results from elevated levels of estradiol and progesterone and reduced serum testosterone; 20 gynecomastic tissue frequently contains receptors for progesterone and estrogen. 19 Neoplastic interstitial cells synthesize and secrete increased amounts of estrogens, leading to suppression of androgen, luteinizing hormone, and testosterone secretion. 2 Estrogen inhibits the activity of the testicular cytochrome P450 enzymes required for normal testosterone biosynthesis. 2 Excessive local estrogen production, possibly due to overexpression of P450 aromatase, has been proposed in cases in which serum concentrations of estrogen and androgens are not altered. 7 Interestingly, overexpression of aromatase has also been shown to cause unilateral or bilateral testicular interstitial cell tumors in a subset of male transgenic mice expressing the human aromatase gene. 4 Regardless of the cause, human gynecomastia is characterized microscopically by florid proliferation of ducts, increased stromal and periductal connective tissue, and periductal edema, with rare alveolar development. 2, 20
In gynecomastia associated with canine testicular tumors, elongation of nipples and proliferation of ducts and stroma are common, although alveoli may also be present. 9, 12 Proposed mechanisms of gynecomastia and feminization in dogs with testicular tumors include increased production of estrogen, 14 decreased testosterone-to-estrogen ratio, 11 increased production of inhibin by tumor cells, 9 and a combination of these.
It is unknown whether the striking lobule formation and pseudolactational hyperplasia observed in this case are typical of gynecomastia in rabbits. Administration of immunosuppressive doses of cyclosporine A and methylprednisolone to female New Zealand white rabbits induced hyperprolactinemia, hyperestrogenemia, and mammary gland hyperplasia with many secretion-filled alveoli. 15 Male rabbits were not used in that study, so it is unknown whether mammary hyperplasia would develop in similarly treated bucks.
Immunohistochemical detection of Melan A or p63 has not been reported previously in rabbit tissues. Immunoreactivity of both antibodies was comparable to what has been described in canine tissues, 5, 16 suggesting that Melan A and p63 will be useful markers for steroid-producing cells and myoepithelial cells, respectively, in formalin-fixed, paraffin-embedded rabbit tissues.
Testicular interstitial cell tumor should be considered as an underlying cause in bucks that have elongated nipples, palpable mammary masses, or microscopic evidence of mammary hyperplasia. Because serum hormone levels were not assayed in this rabbit, however, the ultimate cause of gynecomastia and its potential association with the interstitial cell tumor are undetermined. Had the animal survived, it is possible that castration would have resulted in reversal of gynecomastia, as it does in dogs with benign testicular tumors. 9
