Abstract
Detection of the microsporidian Encephalitozoon cuniculi in tissue samples is considered difficult. The aim of the current study was to determine whether immunohistochemistry (IHC) and in situ hybridization (ISH) represent reliable methods for the detection of E. cuniculi in postmortem tissue samples of rabbits. Paraffin-embedded tissue sections of brain and kidneys of 48 naturally infected pet rabbits, 10 negative controls, and the eyes of 3 further rabbits were used for all investigations. By IHC in 19 animals (37.3%), spores could be clearly detected and were all equally stained. By ISH using a digoxigenin-labeled oligonucleotide probe, only 6 animals (11.8%) proved undoubtedly positive. In these cases, many parasite-like objects revealed strong typical purple-black positive signals. However, several of the examined samples showed only partial staining of the pathogen or unclear results. Thus, in order to find an explanation for these inconsistent ISH results and to take a more detailed look at the different developmental stages of the organism, electron microscopy was applied. Empty spores, which had already discharged their polar filaments, prevailed in total number. Taken together, both techniques are rather insensitive, but under the condition that sufficient numbers of microsporidia are present, IHC can be recommended for specific identification of E. cuniculi in tissue samples. In contrast, ISH failed to detect some developmental stages of the organism, and, as such, ISH is therefore considered an inappropriate diagnostic method.
The microsporidian eukaryote Encephalitozoon cuniculi is an important pathogen of rabbits and other mammals.7,10,14 Phylogenetic analysis proved the close relationship of this organism, which was traditionally considered as protozoa, with fungi. 17 The unicellular ellipsoidal spores exhibit a size of approximately 2 µm in length11,14 and a thick spore wall, which is composed of 3 layers: an outer proteinaceous exospore, a chitin-rich endospore, and an inner plasma membrane.11,14
After an animal is infected with the pathogen and the pathogen has spread through the bloodstream, the brain and kidney are final predilection sites.3,6 Each step of the parasitic development takes place inside a parasitophorous vacuole presumably of host origin, where proliferative forms are closely associated with the membrane of the vacuole, and advanced stages are arranged in the inner part. 11 Latent chronic infections prevail in rabbits. 4 The typical pathohistological findings in rabbits consist of chronic interstitial nephritis and granulomatous meningoencephalitis.4,6
In such cases, the detection of E. cuniculi is considered difficult due to the small quantity of organisms in the affected organs and the tiny size of single spores, both of which contribute to the long and intense searches required for identification in microscopic slides. The aim of the current study was to determine whether immunohistochemistry (IHC) and in situ hybridization (ISH) represent reliable diagnostic methods that could facilitate the detection of E. cuniculi in postmortem tissue samples of rabbits.
Tissue samples from 48 pet rabbits a with microscopic lesions suspicious for encephalitozoonosis (granulomatous encephalitis, interstitial nephritis) were chosen. Ten additional clinically healthy rabbits with no pathological alterations served as negative controls. From all animals, paraffin-embedded tissue samples of kidney and brain (mainly sectioned transversally at the level of thalamus) were examined with IHC and ISH. The majority of the animals had already been tested for E. cuniculi by conventional and nested polymerase chain reaction (PCR) and with the help of special stains (Ziehl–Neelsen and acid-fast trichrome).4,5 Additionally, paraffin-embedded eyes of 3 further patients were used for the study.
Immunohistochemistry was performed using the peroxidase-conjugated avidin–biotin complex method according to an established protocol.b,15 Briefly, antigen retrieval was performed by heating the slides in protease c (1 mg/ml; brain, kidney) or citrate buffer (pH 6.0; eye). The primary polyclonal antibody d was used at a dilution of 1:1,000. Previous specificity tests of this antibody revealed no cross-reactivity with tissues containing the protozoa Toxoplasma gondii, Neospora caninum, Plasmodium elongatum, Leishmania spp., Tritrichomonas foetus, Giardia duodenalis and the fungi Aspergillus spp., Candida spp., and Pneumocystis carinii. There was a slight cross-reactivity with spores of Cryptococcus spp. AEC (3-amino-9-ethylcarbazole) e served as chromogenic substrate, which delivered a red reaction product at the location of the pathogen. In each run, a positive control (brain section of a naturally infected rabbit containing numerous E. cuniculi–bearing vacuoles) and a negative control (see previous) were used.
In situ hybridization was performed based on a previously published protocol. 2 The pretreatment was conducted with a combination of proteinase Kf (30 ng/µl) and chitinase g (50 ng/µl). An E. cuniculi–specific, commercially synthetized, digoxigenin-labeled oligonucleotide probe h (20 ng/µl) complementary to a portion of the 18S ribosomal RNA (rRNA) was used, with the following probe sequence: 5’-AAGCAGCTTCGTCAGCCGCTATTACTAGGA-3’. The digoxigenin-labeled hybrids were detected by incubating the slides with anti–digoxigenin-AP (alkaline phosphatase) Fab fragments i (dilution 1:200). The positive and negative controls were the same as those used in the IHC. For transmission electron microscopy, cubes of 1 mm3in size from formalin-fixed kidney tissue of a strongly positive rabbit were processed according to standard techniques 18 and examined with a transmission electron microscope. j
Out of 51 suspicious animals, 19 cases (13 brain, 4 brain and kidney, and 2 eye; 37.3%) tested positive by IHC (Table 1). In these slides, the spores were clearly detected by their typical red color reaction. All spores were entirely and uniformly labeled. In the brain, the spores were located either singly and scattered within granulomas (Fig. 1A) and/or densely packed within vacuoles, without showing any surrounding inflammatory reaction (Fig. 1B). Organisms lying singly in the tissue were infrequently seen. In many cases, numerous granulomas without any detectable spores could be found in the brain samples. In positive kidneys, labeled spores were discernible within tubular epithelium cells and sporadically free in tubular lumina (Fig. 1C). Usually, E. cuniculi spores were present without any accompanying tissue lesions. Spores were not identified in the kidneys with chronic interstitial nephritis. With a few exceptions, the number of identified spores was low in the examined cases. The eyes contained very sparse round to ellipsoidal spores in the lens. A positively reacting fine granular material within the lens could also be observed. None of the negative controls showed labeled spores or other parasite-like objects.
Tissue samples of pet rabbits with clinical signs and/or microscopic lesions suspicious for encephalitozoonosis that were examined by immunohistochemistry (IHC) and in situ hybridization (ISH).

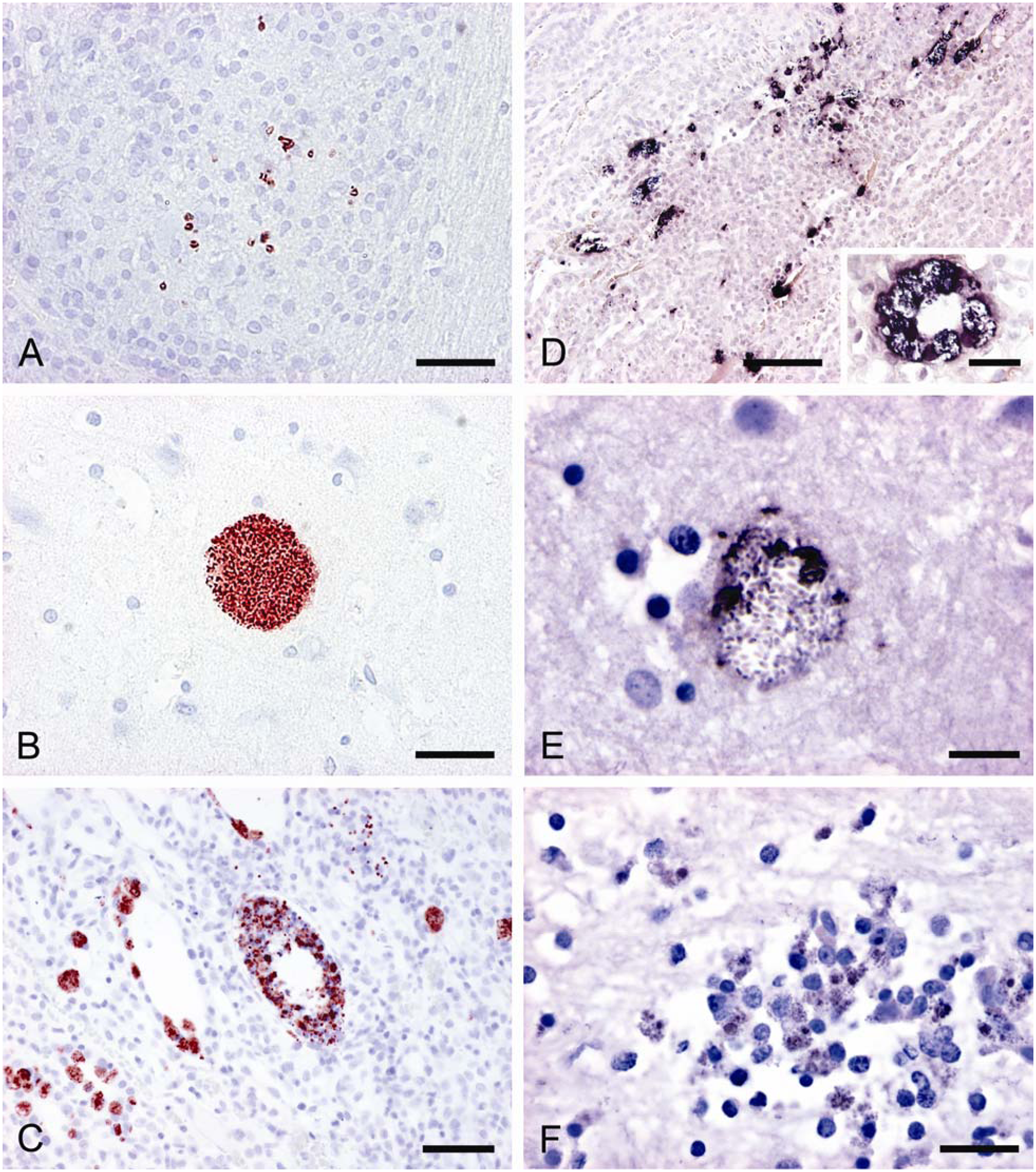
Results of immunohistochemistry (IHC) and in situ hybridization (ISH).
In ISH, the parasite-like objects revealed strong typical purple-black positive signals. The microsporidians were situated within granulomas or parasitophorous vacuoles, but vacuoles definitely predominated. As with the previous detection method, many granulomas contained no specific signals, and similarly, staining occurred in lesion-free adjacent tissue. In some cases, particularly in the kidney (Fig. 1D), the localization of the detected pathogen exactly corresponded with the signals obtained by IHC. However, several of the examined samples showed only partial staining of the pathogen or unclear results. In such cases, spores with distinct signals were situated together with very weakly or unevenly stained spores inside the vacuoles or granulomas (Fig. 1E). Similar to the IHC finding in the lens, in some brain samples, a positive reaction of granular material could be observed within the cytoplasm of macrophages, which were involved in granuloma formation (Fig. 1F). Nonetheless, only clear results were positively assessed. Therefore, only 6 IHC-positive cases (4 brain and kidney and 2 brain only; 11.8%) showed unequivocally positive signals by ISH (Table 1). None of the IHC-negative samples revealed positive signals by ISH. In order to find an explanation for the difficulties in spore labeling by ISH, ultrastructural examinations were initiated. With this method, different stages of spore development were clearly discernible within parasitophorous vacuoles of kidney tissue. According to their ultrastructural features, meronts, sporonts, sporoblasts, and spores could be distinguished (Fig. 2A). Among the spores, partly empty forms, which had discharged their polar tubes, prevailed in total number (Fig. 2B). In these spore types, an irregular layer of dark, granular material lining the inner spore wall was often retained.
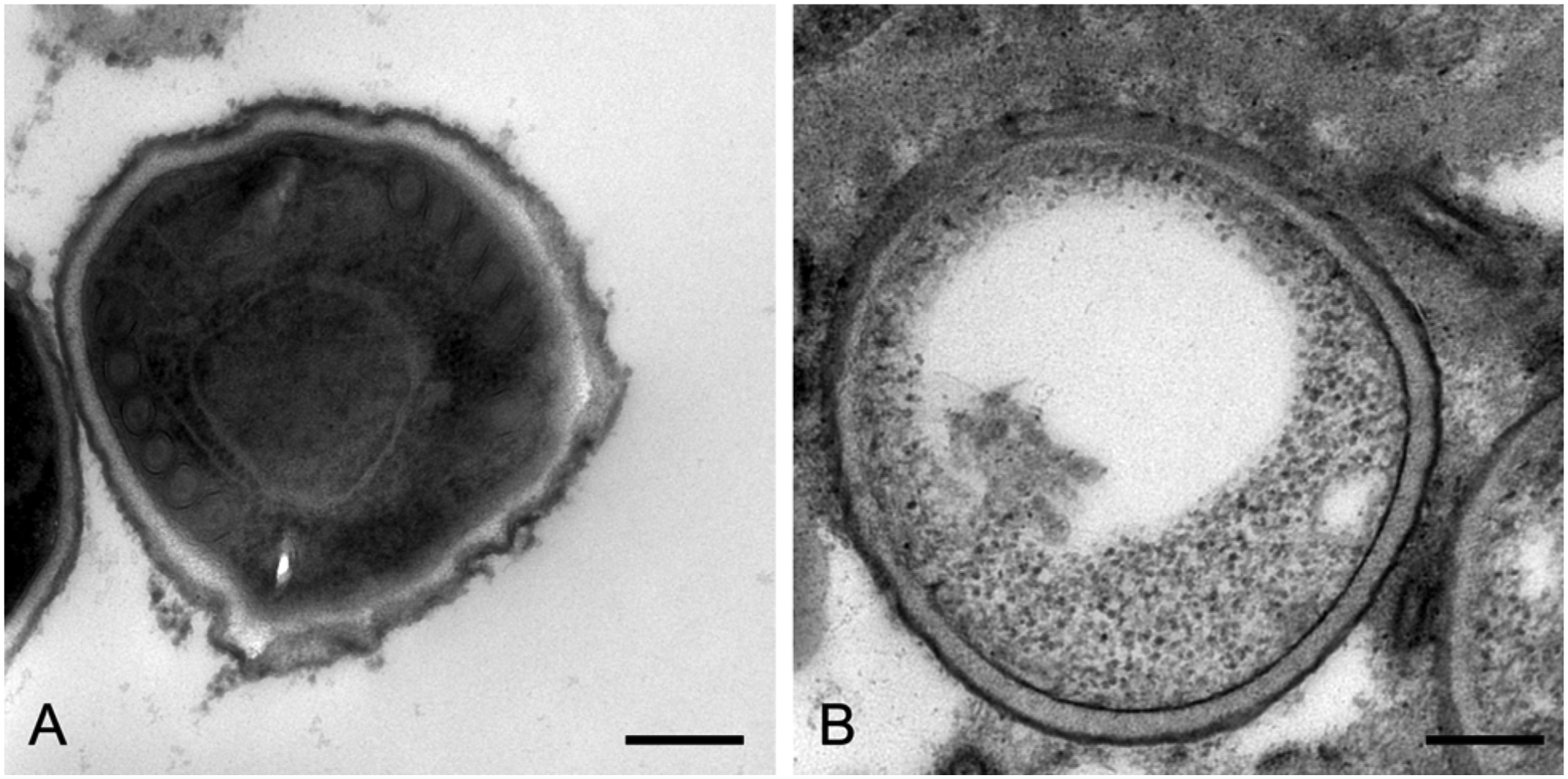
Electron microscopy of Encephalitozoon cuniculi spores.
Earlier studies have attempted to find a simple and robust spore detection method for E. cuniculi infection. They predominantly tried to microscopically visualize spores in tissue samples by certain labeling methods, like acid fast trichrome or Ziehl–Neelsen stains. 4 Moreover, E. cuniculi was successfully identified using IHC in former studies,1,8,9,12 which seemed to be a useful tool for specific demonstration of the pathogen and its localization within tissues.
In the present work, with a few exceptions, only sparse spores were identified in the tissues with both tested techniques, IHC and ISH, and many granulomas contained no detectable spores at all. This may be explained by the chronicity of most infections as the number of organisms decreases as the lesions become older. 7 The resulting small quantity of spores, their unpredictable dissemination in tissues, and the tiny size of single spores contribute to a high probability to overlook the pathogen in tissue sections. Nevertheless, the low sensitivity of IHC correlated well with the sensitivity of conventional PCR, which had been assessed in a preceding study. 5 In general, assessments of sensitivity and specificity of diagnostic procedures for E. cuniculi infections suffer from the lack of a defined gold standard indicative of a proven infection. It seems that standard histological examination of predilection sites, like brain, kidney, and lens, identifies typical lesions in many cases in which the causative organism is not even detectable by the most sensitive procedure described to date (i.e., nested PCR). 5 However, due to its extreme sensitivity, nested PCR is highly susceptible to laboratory contamination, and generation of false-positive results may occur. So despite the increasing use of sophisticated molecular methods for pathogen discovery, conventional histology still plays a central role in diagnosing E. cuniculi infections in rabbits. The problems associated with all methods aimed at detecting the microsporidian spores are not related to the specific techniques per se but rather reflect the very sparse or even absent organisms in these chronic infections. Nonetheless, there was a significant difference in the staining properties of the 2 techniques. In IHC, the label was most distinct at the spore wall, and there were no weakly or unevenly stained spores. The polyclonal antibody used most likely detects proteins of the spore wall, which seem present in all developmental stages and are easily accessible for the reagents. Based on the present findings, a positive IHC result can be considered as proof for the presence of the pathogen in the host tissue. However, a negative result does not rule out E. cuniculi as causative agent of characteristic histological lesions.
For ISH, with regard to the composition of the E. cuniculi spore wall and the necessity of the probe to react with intracellular structures, a combination of proteinase and chitinase was chosen for pretreatment. Numerous trials were conducted, including the variation of different proteinase and chitinase concentrations, exposure times, hybridization temperatures, and color substrate reaction times. The ISH method described herein represents the procedure that produced the best possible labeling reaction. This staining protocol and the properties of the probe generally allow specific labeling of E. cuniculi, which was demonstrated by the clear signal of many spores. In addition, a fine granular material showing positive signals of varying intensity was found within macrophages in cerebral granulomas. These round structures were considered to be early developmental forms of E. cuniculi. This finding is comparable to a previous report, 16 where similar immature structures are described in light microscopic examinations. However, ISH potentially failed to detect all developmental stages of the organism. Several of the examined samples showed only partial staining of the pathogen or ambiguous results. Spores with distinct signals and others with weak or no detectable signals were situated together inside the same vacuoles. Potential reasons for these disappointing results were that the rRNA, which is visualized by ISH, could not be accessed by the probe, has been degraded in degenerate spores, or has been lost in spores with already discharged polar tubes including the infectious sporoplasm.
To test some of these hypotheses, kidney tissue containing parasitophorous vacuoles with unevenly labeled spores by ISH was investigated using electron microscopy. This enabled a detailed look at the different developmental stages of the organisms and their typical position within vacuoles. Particularly, various empty spores, which had already discharged their polar filaments and thereby released the infectious sporoplasm including the nucleus, were found. This could be traced back to the fact that E. cuniculi spores undergo a rapid life cycle, 11 and thus, the empty spores could serve as an explanation for weakly stained spores in the samples. In many instances, an irregular layer of dark, granular material lining the inner spore wall was often retained in empty spores. This feature could also be observed in past investigations by electron microscopy 11 and be the reason for only marginally labeled spores in the ISH results, as the remaining sporoplasm may be insufficient to stain the entire spore. Usually, microsporidians contain high rRNA contents (approximately 60%). 17 It is possible that, despite the use of chitinase, the applied pretreatment was not able to perforate the thick wall of mature spores. Thus, only spores that were cut during preparation of histological sections and had therefore exposed their inner structures would deliver detectable signals.
In situ hybridization seems to be a useful tool for detection of fully intact spores with comparably thin walls in acute infections. 13 However, in the context of E. cuniculi infections of rabbits with sparsely occurring organisms of unknown morphological characteristics, ISH is a very insensitive method that is clearly inferior to IHC. For certain research applications, however, it may prove useful. To conclude, it became apparent that IHC is the preferable tool for specifically localizing E. cuniculi spores in tissue sections. However, the examiner should make note of the problem of mainly latent chronic infections resulting in low numbers of detectable organisms, which sometimes makes etiological diagnosis impossible.
Footnotes
Acknowledgements
The authors would like to thank Klaus Bittermann for his professional help with the digital artwork and the team of the Institute of Pathology for excellent technical support and input.
a.
Tissue bank of the Institute of Pathology and Forensic Veterinary Medicine, Vienna, Austria.
b.
Vectastain® ABC Kit, Vector Laboratories Inc., Burlingame, CA.
c.
Protease, Sigma-Aldrich, St. Louis, MO.
d.
Guinea pig antiserum against E. cuniculi, Medicago AB, Uppsala, Sweden.
e.
AEC (3-amino-9-ethylcarbazole), substrate-chromogen Ready-to-Use, Dako Denmark A/S, Glostrup, Denmark.
f.
Proteinase K, Roche, Basel, Switzerland.
g.
Chitinase, Sigma-Aldrich, St. Louis, MO.
h.
Digoxigenin-labeled oligonucleotide probe, Eurofins MWG Operon, Ebersberg, Germany.
i.
Anti-digoxigenin-AP Fab fragments, Roche, Basel, Switzerland.
j.
Zeiss EM 900 transmission electron microscope, Carl Zeiss MicroImaging GmbH, Göttingen, Germany.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
