Abstract
A granular cell tumor involving the pituitary gland, optic chiasm and ventral pyriform lobes was discovered in a 12-year-old Labrador Retriever. Clinical signs included acute blindness, seizures, ataxia, weakness, and behavioral changes. The diagnosis was established by histopathologic and ultrastructural examination of neoplastic tissues collected at necropsy. Granular cell tumors involving the central nervous system are well documented in humans but rarely have been described in dogs. The location of the neoplasm and the clinical symptoms seen in this dog closely parallel those of a rare syndrome in humans commonly described as symptomatic parasellar or pituitary granular cell tumors. The cell of origin for these tumors is still highly debated, and attempts to characterize human granular cell tumors through immunohistochemistry have produced conflicting results. An immunohistochemical profile of this neoplasm revealed focal positive staining for vimentin with a lack of staining for neuron-specific enolase, glial fibrillary acidic protein, S-100, and synaptophysin. All neoplastic cells were strongly positive with the periodic acid-Schiff reaction.
Keywords
Granular cell tumors (GCTs) are rare in domestic animals. They have been reported most commonly in dogs and horses, and there are several reports describing GCTs in cats and a bird. 4,8,11 Primary GCTs occur at several sites in the body, and in dogs these tumors are most commonly described in the oral cavity. Additionally, canine GCTs have been reported occasionally at other sites such as heart, lymph node, brain, and skin. 3,8 GCTs involving the central nervous system of any species are rare. Most have been described in the meninges, although several reports of cerebral GCTs exist. 10,13 Regardless of location, these neoplasms exhibit uniform morphologic features. Most are benign and rarely metastasize. Despite their characteristic morphologic features, tumor histogenesis is still disputed, and many believe that GCTs may arise from several tissue types. 10,13,16 The majority of peripheral GCTs involving soft tissue are believed to arise from Schwann cells. 4,11 Here, we describe the features of a GCT located in the parasellar and intrasellar regions and involving the pituitary gland.
A 12-year-old spayed female Labrador Retriever was presented to the Texas A&M University Veterinary Medical Teaching Hospital with a 1-month history of mild seizure-like activity, occasional erratic behavior, disorientation, and progressive loss of vision. Neurologic evaluation revealed bilaterally fixed and dilated pupils that lacked a menace response. Fundic examination indicated edema of both optic nerves. All other aspects of the neurologic exam were within normal limits. Differential diagnoses considered after initial examination included intracranial neoplasia, cerebral atrophy/cognitive dysfunction, retinal atrophy, and sudden acquired retinal degeneration syndrome. A hemogram, blood chemistry profile, and urinalysis were within normal limits. Thoracic radiographs showed mild changes in the bronchial walls consistent with aging, but there was no evidence of metastatic disease. A normal electroretinogram indicated that the blindness was secondary to a disease process involving the optic nerves or brain. The results of an ACTH stimulation test were equivocal for hyperadrenocorticism. During a routine venipuncture, the dog experienced a mild seizure that consisted of forelimb extension, neck rigidity, and loss of bowel and urination control. The dog's clinical condition steadily deteriorated, and because of the poor prognosis, the owners elected to euthanatize the dog.
Necropsy revealed a diffusely tan, roughened to nodular, poorly circumscribed mass centered on the optic chiasm and hypophysis and encompassing both optic nerves. The central portion of the mass was approximately 0.5 × 0.8 × 1 cm, and it contained irregular projections that extended onto the ventral surface of both pyriform lobes and the adjacent cranium. Prominent bony roughening was noted in areas of the cranial vault associated with the neoplasm. Aside from the entrapment and destruction of the optic nerves, no other ocular abnormalities were detected. Impression smears were made for cytologic evaluation. The sample was highly cellular and displayed a monomorphic population of large round to oval cells with single, small, eccentrically placed nuclei. The cells contained abundant basophilic, coarsely granular cytoplasm. Aside from moderate anisocytosis and an occasional binucleate cell, few criteria of malignancy were identified (Fig. 1).
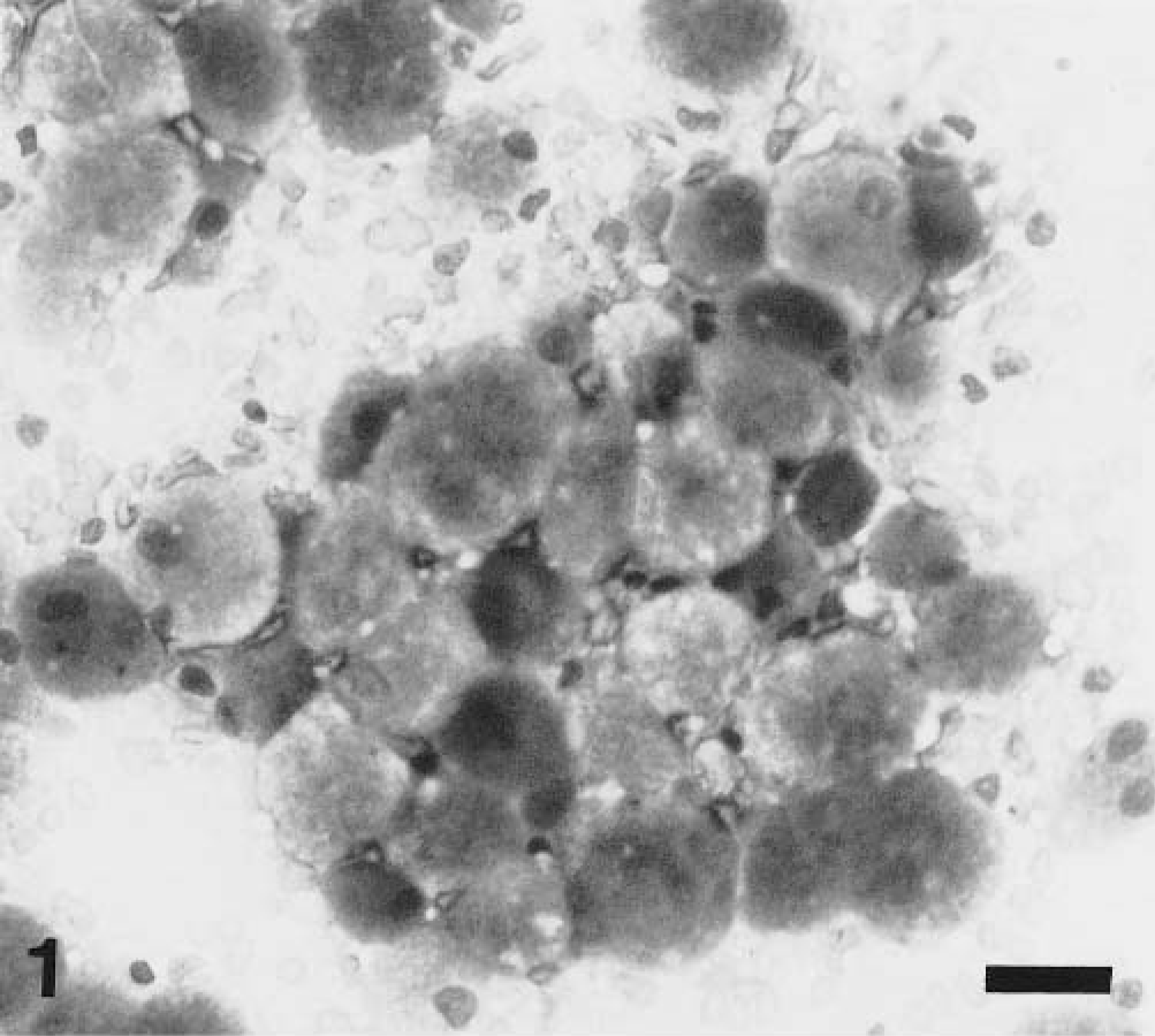
Impression smear. Granular cell tumor; dog. Large cells with abundant granular cytoplasm and a single, small, eccentrically placed nucleus. Wright-Giemsa. Bar = 25 µm.
Multiple sections involving the brain and cranium were fixed in buffered formalin for routine light microscopy. Histologically, the neoplastic cells had a uniform morphologic appearance but were poorly circumscribed. Multiple sheets and clusters of large polygonal cells were located on the surface of the pyriform lobes and within areas of bony proliferation on the inner surface of the cranium. Additional clusters were located in the adenohypophysis (Fig. 2). The neoplastic cells had distinct cell borders and contained abundant eosinophilic granular cytoplasm. Nuclei were small, round to oval, eccentric, and occasionally multiple. Necrosis and mitotic figures were absent (Fig. 3). A marked perivascular response consisting predominantly of lymphocytes was detected around many of the vessels in both the pyriform lobes and other areas of the cerebral cortex. Based on morphology, a list of differential diagnoses included GCT, oncocytoma, and rhabdomyoma. Additional sections were stained with periodic acid–Schiff (PAS) and Sudan black. All of the neoplastic cells were lipid negative but demonstrated diffuse, punctate cytoplasmic positivity for PAS (Fig. 4).
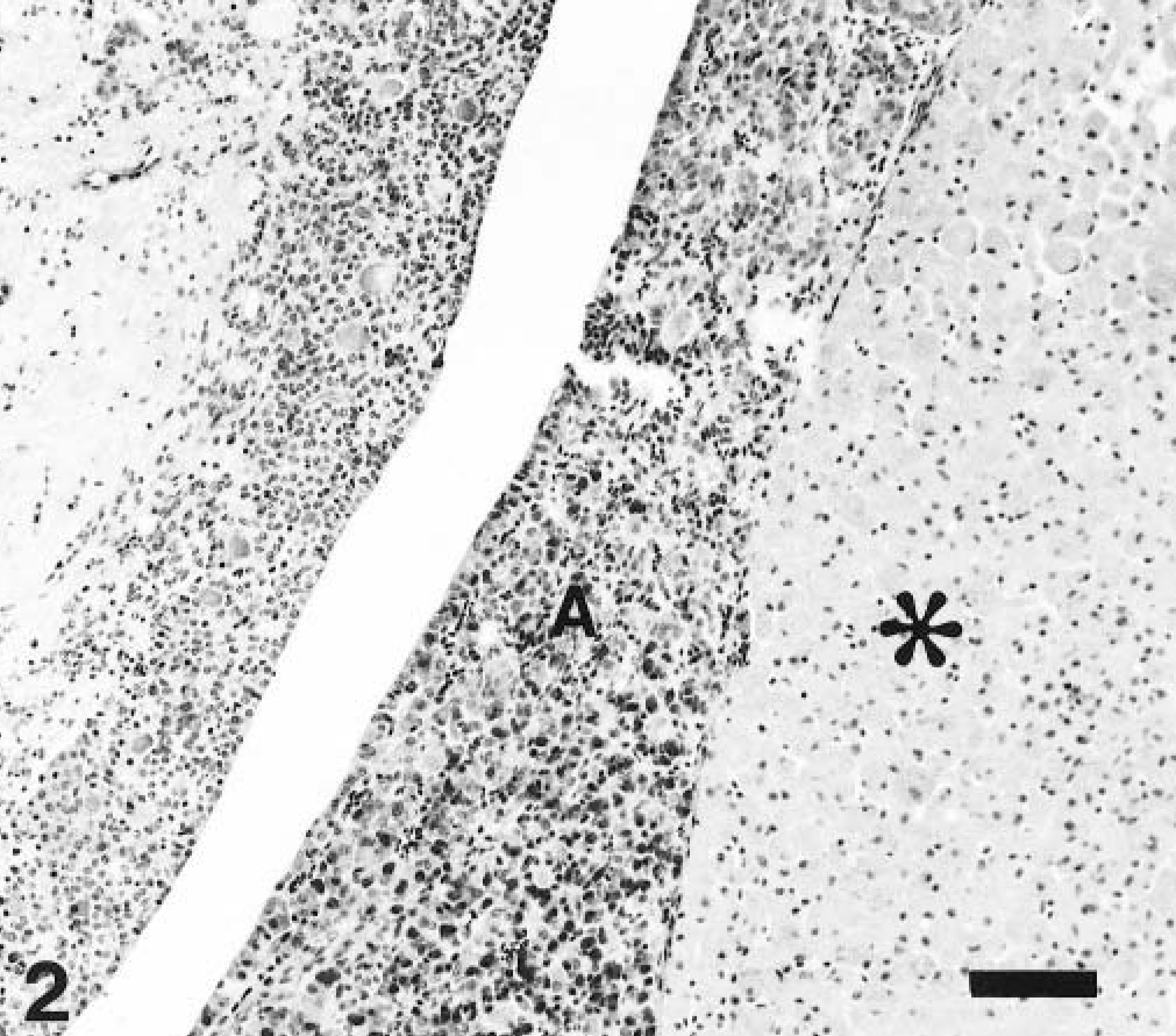
Pituitary gland; dog. Granular cell tumor (asterisk) located in the adenohypophysis (A). HE. Bar = 100 µm.
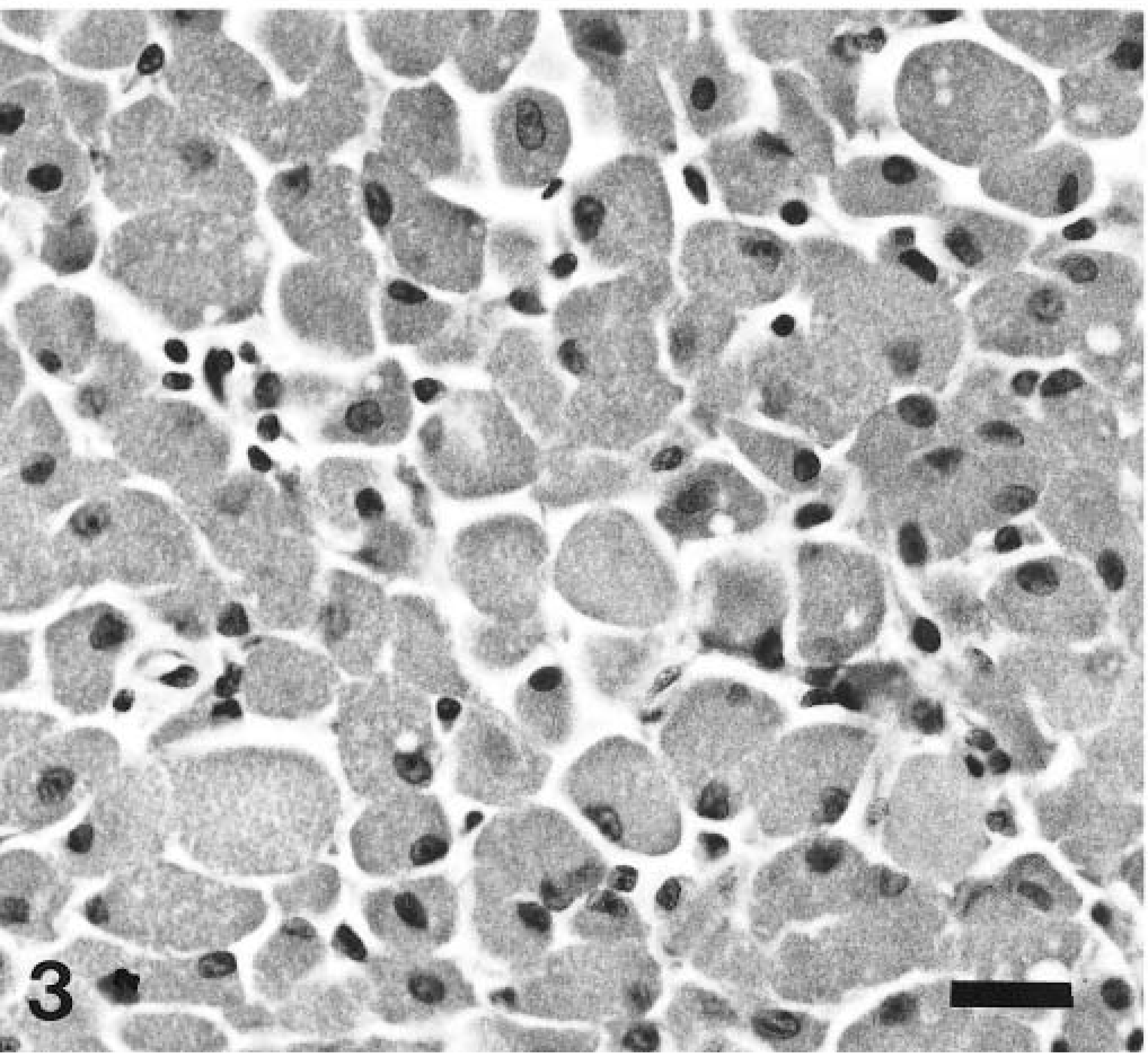
Granular cell tumor; dog. A monomorphic population of cells containing abundant granular, eosinophilic cytoplasm. HE. Bar = 25 µm.

Granular cell tumor; dog. Neoplastic cells exhibiting diffuse, granular cytoplasmic staining. PAS. Bar = 50 µm.
Immunohistochemistry for pancytokeratin, vimentin, S-100, glial fibrillary acidic protein (GFAP), neuron-specific enolase (NSE) and synaptophysin was performed using a diaminobenzidine–horseradish peroxidase method. Cytoplasmic staining for vimentin was detected focally in approximately 30–40% of the neoplastic cells. Staining for cytokeratin, GFAP, S-100, and synaptophysin was uniformly negative.
Electron microscopy was performed on tissue collected in buffered formalin. Ultrastructurally, the cells demonstrated a paucity of normal cytoplasmic organelles. The cytoplasm contained numerous membrane-bound vacuolar structures that displaced the nucleus to the periphery. Many of the vacuolar structures contained irregular electron-dense material and were interpreted as lysosomes (Fig. 5). Abundant cytoplasmic lysosomes that displace and obscure normal cytoplasmic structures are the hallmark feature of GCTs. In contrast, the cytoplasmic granularity of both oncocytomas and rhabdomyomas is a result of abundant mitochondria. The histologic and ultrastructural features of the neoplasm confirmed a diagnosis of GCT. Although the neoplasm was confined to the cranial vault, it was locally invasive. There was complete destruction of the optic chiasm and entrapment of the optic nerves, which accounted for the acute blindness. The mild seizure-like activity and behavioral changes likely resulted from involvement of the pyriform lobes and the hypothalamic region. Many of the morphologic and clinical features closely paralleled those of an entity in human medicine often described as parasellar or pituitary symptomatic GCTs. 1,14
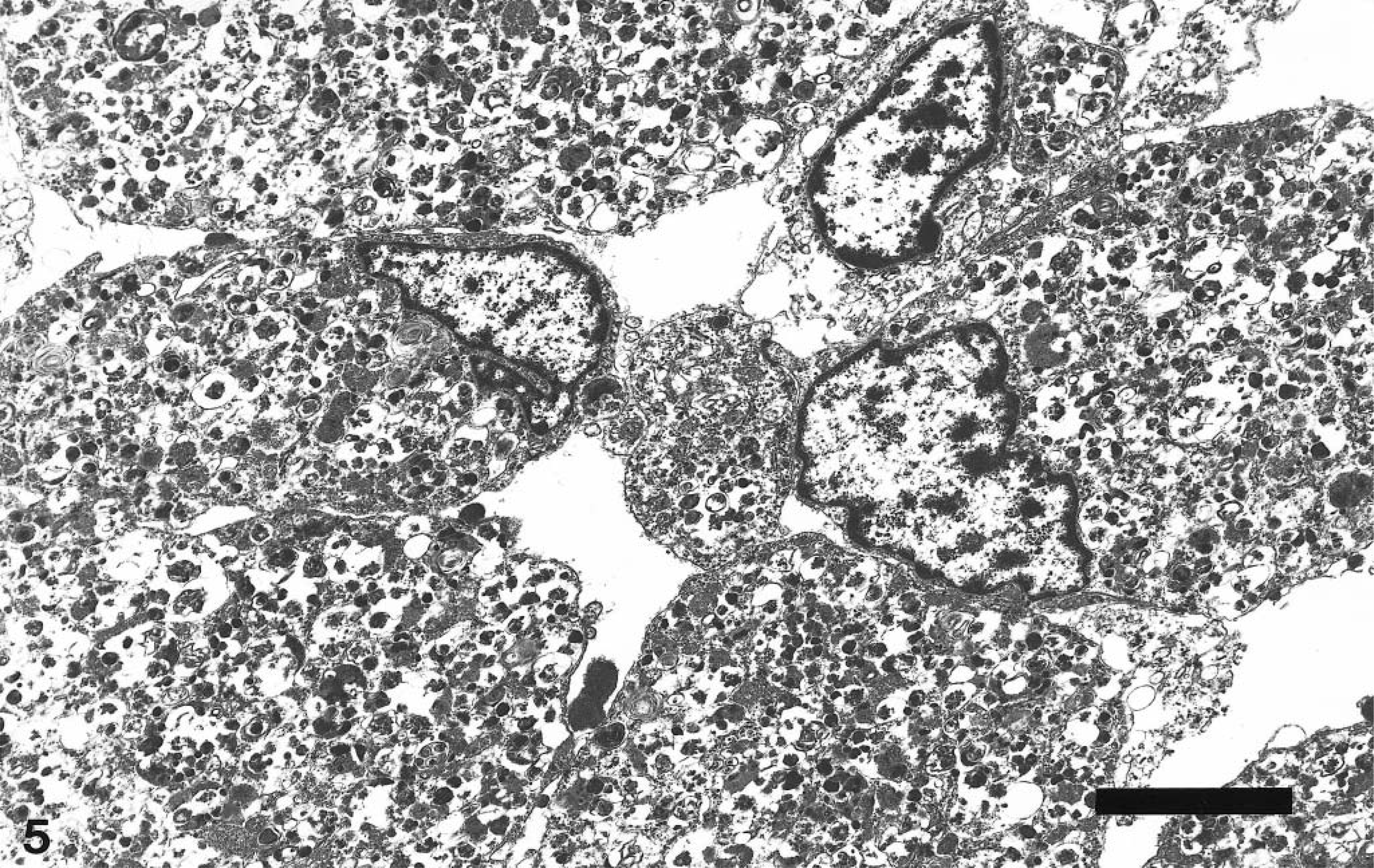
Transmission electron micrograph. Granular cell tumor; dog. Neoplastic cells containing numerous membrane-bound cytoplasmic vacuoles that obscure normal organelles and cause margination of the nucleus. Many of the vacuoles contain electron-dense material. Bar = 10 µm.
Small nests of granular cells are commonly identified in the posterior pituitary gland and/or pituitary stalk during routine postmortem examinations of human patients. These nests are occasionally referred to as choristoma, tumorlet, granular myoblastoma, and granular pituicytoma, but the preferred term is GCT. 5 The majority of these GCTs are incidental findings and are not associated with clinical symptoms. 10,14,15 Greater numbers of granular cells have been identified in the pituitary gland of older persons, and some researchers have claimed that parasellar GCTs are an age-related metabolic disorder of lysosomes. 15 Currently, there are approximately 40 reports of clinically symptomatic GCTs involving the pituitary gland. Initially, these tumors are benign and slow growing, but once they become symptomatic they seem to progress rapidly and are associated with an unfavorable outcome. 5 They frequently compress and compromise function of the pituitary gland, optic chiasm, and hypothalamus. Ophthalmologic deficits, including blindness and optic atrophy, are reported in approximately 90% of the cases. Other frequently reported symptoms include disorientation, dementia, and hormonal insufficiency or excess. 1,6,14 Although, the pituicyte has been proposed as the cell of origin for symptomatic parasellar GCTs, this proposal is still widely debated. 1,10,16
The morphologic features of GCTs involving the pituitary gland are very similar to those of GCTs found in other locations. 6,14 Histologically, GCTs are described as multiple clusters of large round to polygonal eosinophilic cells containing abundant granular cytoplasm and a small eccentrically placed nucleus. The granularity of the cytoplasm is due to the presence of numerous lysosomal storage vacuoles. Mitotic figures are uncommon. Characteristic morphology and the presence of numerous PAS-positive cytoplasmic granules are the most consistent criteria for diagnosing GCTs. 4,11 One interesting feature of the neoplasm in this report is the perivascular cuffing of lymphocytes around many of the cortical vessels. Although infectious causes of nonsuppurative encephalitis were considered, perivascular cuffing has also been described in association with several human pituitary GCTs. 14
Many researchers have attempted to describe the immunophenotype of GCTs in the central nervous system and soft tissue sites in both human and veterinary cases. The majority of the reports involving animal species indicate frequent positive staining for vimentin in GCTs of the meninges and the oral cavity. Positive staining for NSE, S-100, and cytokeratin is sporadic and inconsistent. 4,8,11 There were no reports of positive immunostaining for GFAP in domestic animal species. Two immunohistochemical studies of cerebral GCTs in laboratory rats demonstrated negative to occasional positive staining for GFAP. 8,12
Few consistent immunohistochemical staining features have been identified in human GCTs. Although peripheral soft tissue GCTs are often reported to be S-100 positive, immunostaining for S-100 in cerebral nonhypophyseal human GCTs has been inconsistent. 13,16 In one study involving nonhypophyseal neoplasms of the central nervous system, researchers detected a high level of NSE and alpha-1-antichymotrypsin (ACT) in a majority of tumors. S-100, GFAP, and vimentin were occasionally positive. 13 Alternatively, additional studies produced only occasional positive staining for ACT and GFAP and negative staining for vimentin and NSE. 2,16 ACT is often regarded as a histiocytic marker, but it is not found exclusively in histiocytic cells. ACT staining may reflect the lysosomal activity that is characteristic of GCTs. 13
The limited number of immunohistochemical studies on hypophyseal GCTs also produced variable staining patterns. 7,9,10,14 Many researchers failed to demonstrate positive staining for S-100; however, in one study three of six tumors had convincing reactivity. 10,14 Two of the six tumors had strong positivity for ACT, whereas the remaining four tumors had either no positive cells or only a few positive cells. Although most studies show negative staining for vimentin, there is a single report of weak vimentin staining. 7 Few studies produced immunoreactivity for GFAP. 7,9,10,14 This finding is surprising given the demonstration of GFAP intermediate filaments in some ultrastructural analyses and does not support the prevailing hypothesis that the pituicyte is the cell of origin. Some researchers have speculated that the dense accumulation of lysosomal storage vacuoles surrounding the intermediate filaments affects the ability of the markers for GFAP to adhere to the appropriate receptors. 10
The lack of S-100, GFAP, pancytokeratin, and NSE immunopositivity in neoplastic cells in this dog correlates with the features that generally have been described for human pituitary GCTs. This neoplasm also expressed focal positivity for vimentin, which has typically been associated with nonhypophyseal tumors and is a rarely reported feature of human pituitary GCTs. 7 Because of the small number of cases evaluated to date and inconsistency in the results of various studies, it is difficult to define stringent criteria for the immunophenotype of pituitary GCTs. Conflicting reports regarding immunostaining of GCTs from any site are common. Because the designation of GCT refers to the morphologic appearance, these differences may result from differing histogenesis. Studies testing for a broader range of antigens and using both archived and new cases will need to be implemented for a more complete understanding and characterization of GCTs.
