Abstract
Scrapie is a naturally occurring fatal neurodegenerative disease of sheep and goats. This study documents incubation periods, pathologic findings, and distribution of abnormal prion proteins (PrPSc) by immunohistochemistry in tissues of genetically susceptible sheep inoculated with US sheep scrapie agent. Four-month-old Suffolk lambs (QQ at codon 171) were inoculated by 1 of 3 different routes (nasal, peritoneal, and conjunctival) with an inoculum (No. 13–7) consisting of a pool of scrapie-affected sheep brains. Except for 3 sheep, all inoculated animals were euthanized when advanced clinical signs of scrapie were observed between 19 and 46 months postinoculation (MPI). Spongiform lesions in the brains and labeling of PrPSc in central nervous system and lymphoid tissues were present in these sheep. One intranasally inoculated sheep euthanized at 12 MPI had presence of PrPSc that was confined to the pharyngeal tonsil. These results indicate that the upper respiratory tract, specifically the pharyngeal tonsil, may serve as a portal of entry for prion protein in scrapie-infected environments.
Scrapie belongs to a group of diseases known as transmissible spongiform encephalopathies (TSEs), which affect man and animals. It is a naturally occurring, fatal neurodegenerative disease of sheep and goats. Susceptibility to scrapie is partially dependent on genetic variation of the host, 1,3 and in sheep, polymorphisms of the prion protein (PrP) gene at codons 136 (valine or alanine), 5 154 (histidine or arginine), and 171 (glutamine, arginine, or histidine) are the dominant factors. 1,10 In an experimental study in the USA of orally inoculated Suffolks, 61% developed scrapie. All were homozygous for glutamine (QQ) at the 171 allele. 10 Infection by the causative agent, considered to be the post-translationally modified form of a host-encoded membrane glycoprotein (PrPc), leads to spongiform encephalopathy and accumulation of an abnormal form of prion protein (PrPSc) in tissues of the nervous and lymphoid systems, as well as in placenta of infected pregnant ewes. 3
The most likely portal of entry in natural scrapie has been suggested to be the alimentary tract, with other potential routes such as dermal scarification and the conjunctiva thought to be effective experimentally. 3 There is a paucity of information on experimental studies with scrapie in Suffolks, the predominant sheep breed in USA, compared with other breeds. In particular, nonoral or nonintracerebral routes of experimental infections of US scrapie agent have not been documented previously. This study partially fills this void by documenting disease and pathology of scrapie after intranasal, intraperitoneal, and intraconjunctival, inoculations in Suffolks that were QQ at codon 171 of prion protein (PRNP) gene and, therefore, were susceptible to scrapie. 1,10
Material and Methods
Seven 4-month-old Suffolk lambs (5 castrated males and 2 females) were obtained for this study. All were AA/RR/QQ at codons 136, 154, and 171, respectively, and were from a closed scrapie-free flock at the National Animal Disease Center (NADC) in Ames, Iowa. The animals were divided into 3 groups (A, B, and C; Table 1), and each group had the inoculum administered by a different route. Group A was inoculated by the conjunctival (n = 2), group B by the peritoneal (n = 2), and group C by the nasal (n = 3) route. All animals were inoculated with inoculum No. 13-7, which has previously been demonstrated to be infectious by intracerebral and oral routes. 5
Findings in scrapie-susceptible Suffolk sheep experimentally inoculated with the US scrapie agent (No. 13-7) by 3 different routes.
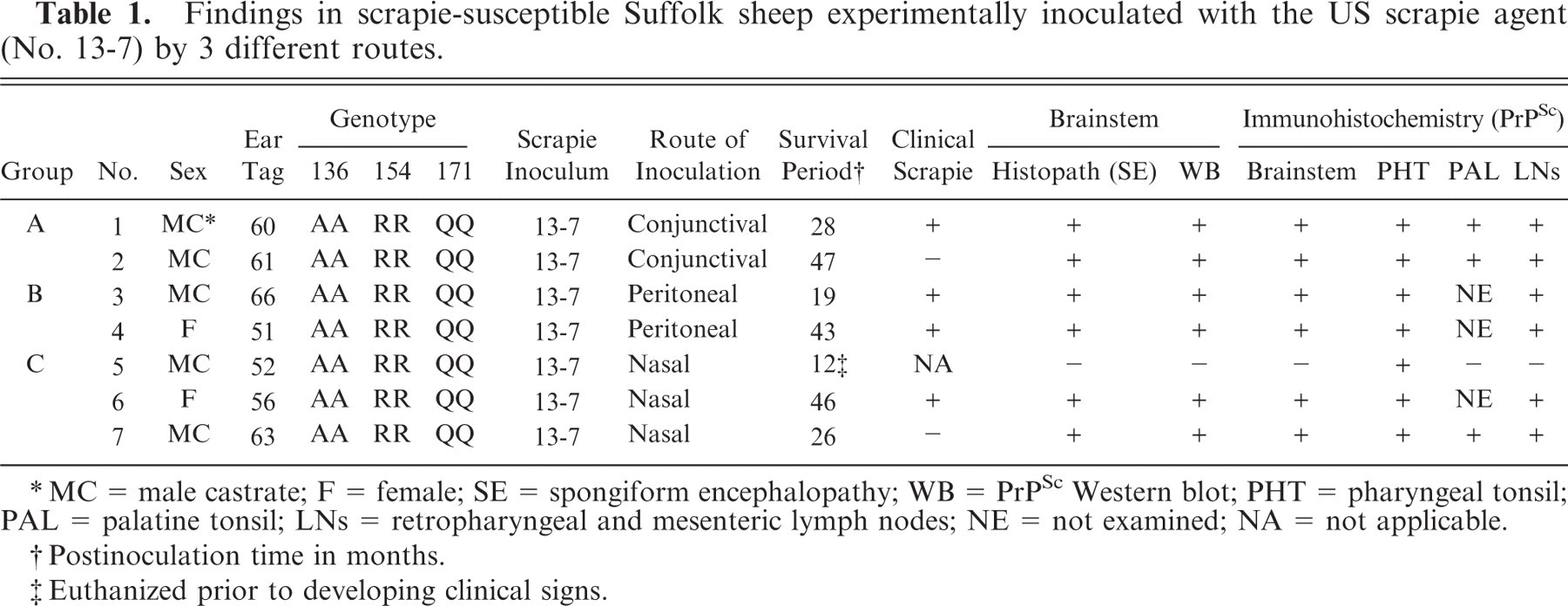
∗MC = male castrate; F = female; SE = spongiform encephalopathy; WB = PrPSc Western blot; PHT = pharyngeal tonsil; PAL = palatine tonsil; LNs = retropharyngeal and mesenteric lymph nodes; NE = not examined; NA = not applicable.
† Postinoculation time in months.
‡ Euthanized prior to developing clinical signs.
The inoculum was prepared from a pool of 13 scrapie-affected sheep brains (all were positive by immunohistochemistry, IHC) from 7 source flocks. 5 The homogenate (10% wt/vol) of infected brains was prepared in phosphate-buffered saline containing gentamicin using a mechanical grinder. All animals were inoculated with 1 ml of the inoculum (Table 1). For the intraperitoneal route, the inoculum was injected into the peritoneal cavity (right ventral abdomen) while the lamb was restrained by its hind legs. The dose for the conjunctival route was divided in 2. One half of the dose was injected under the palpebral conjunctiva, and the other half of the dose was applied topically on the conjunctiva. For the intranasal route, the inoculum was drawn into a syringe, and, without the aid of a needle, it was squirted into 1 nostril (right side).
Inoculated animals were initially housed in a biosafety level-2 containment facility for 12 months postinoculation (MPI) and later were moved to outside pens at the NADC. The sheep were fed pelleted growth and maintenance rations that contained no ruminant protein, and clean water was available ad lib.
With the exception of 3 sheep, all others were euthanized when they developed terminal clinical signs of scrapie. They were examined at necropsy, and 2 sets of tissue samples were collected. One set of tissues included representative sections of liver, kidney, spleen, skin, striated muscles (heart, tongue, diaphragm, masseter), tonsils (pharyngeal, palatine), thyroid gland, turbinates, lung, tonsils, intestines (ileum), adrenal gland, lymph nodes (retropharyngeal, mesenteric), pituitary gland, Gasserian ganglion, brain (hemisections of cerebral cortex, cerebellum, superior colliculi, and brainstem, including obex), and eye (retina). These tissues were fixed in 10% buffered formalin, embedded in paraffin wax, sectioned at 5 μm, and stained with hematoxylin and eosin for light microscopy. The second set of tissues was frozen.
All paraffin-embedded tissues were also stained by an automated IHC method for detection of PrPSc as described previously. 5 Briefly, after deparaffinization and rehydration, tissue sections were autoclaved for 30 minutes in an antigen retrieval solution (DAKO Target Retrieval Solution, DAKO Corp., Carpinteria, CA) and stained with an indirect avidin biotin system (Basic Alkaline Phosphatase Red Detection Kit, Ventana Medical Systems, Inc., Tucson, AZ) designed for an automated immunostainer (NexES IHC module, Ventana Medical Systems, Inc.).
The primary antibody was a cocktail of 2 monoclonal antibodies, F89/160.1.5 9 and F99/97.6.1, 8 each used at a concentration of 5 μg/ml, and incubation was carried out at 37°C for 32 minutes. The secondary antibody was biotinylated anti-mouse (Biotinylated anti-mouse IgG [made in horse], Vector Laboratories, Burlingame, CA) diluted 1:200 and incubated for 8 minutes at 37°C.
One half of the brain (cut longitudinally) was frozen for Western blot as described previously, 5 and monoclonal antibody P4 was utilized (R-Biopharm, Inc., Marshall, MI) for this procedure.
Results
Except for 3 sheep (Nos. 52, 61, and 63; Table 1), all other inoculated animals showed clinical signs of scrapie prior to euthanasia. Initial clinical signs of the disease consisted of a progressive decrease in appetite and associated weight loss. Later, the affected sheep developed fine head tremors, listlessness, progressive problems with locomotion, and, in terminal stages, sternal recumbency. None of the affected sheep exhibited obvious pruritus or loss of wool from their fleece.
Among the nonclinical scrapie-infected animals, sheep No. 52 was euthanized at 12 MPI (Table 1). This animal had sustained some physical trauma when caught between gates at the time it was transferred to an outside pen and had difficulty in getting up. Because its prognosis appeared to be poor, it was euthanized. Significant lesions were not observed during necropsy or on microscopic examination of its tissues. Sheep No. 61 was found dead at 47 MPI. Only the head of this sheep was available for examination, and no gross lesions were observed in the examined tissues (Table 1).
The postinoculation survival periods for the individual animals are shown in Table 1. Because the animal numbers in each of the groups are rather small, detailed statistical analysis was of no value. As a group, the intraperitoneally inoculated sheep (group B) had the shortest disease onset PI time (31 MPI). This was followed by group C (intranasal, 36 MPI) and group A (intraconjunctival, 37.5 MPI) (Table 1). However, because the number of animals per group was small, a definitive conclusion cannot be made.
The distribution of histologic lesions in the brain and the results of IHC and Western blot are given in Table 1. Microscopic spongiform lesions characteristic of prion disease were present in all sheep with clinical signs of scrapie, and characteristic PrPSc deposition was present in the central nervous system and lymphoid tissues of these animals (Figs. 1 and 2, Table 1). IHC analysis of sheep No. 52, euthanized at 12 MPI, revealed a presence of PrPSc that was confined to the pharyngeal tonsil (Fig. 3, Table 1). Pharyngeal tonsil obtained from a scrapie-free sheep did not reveal PrPSc on IHC.
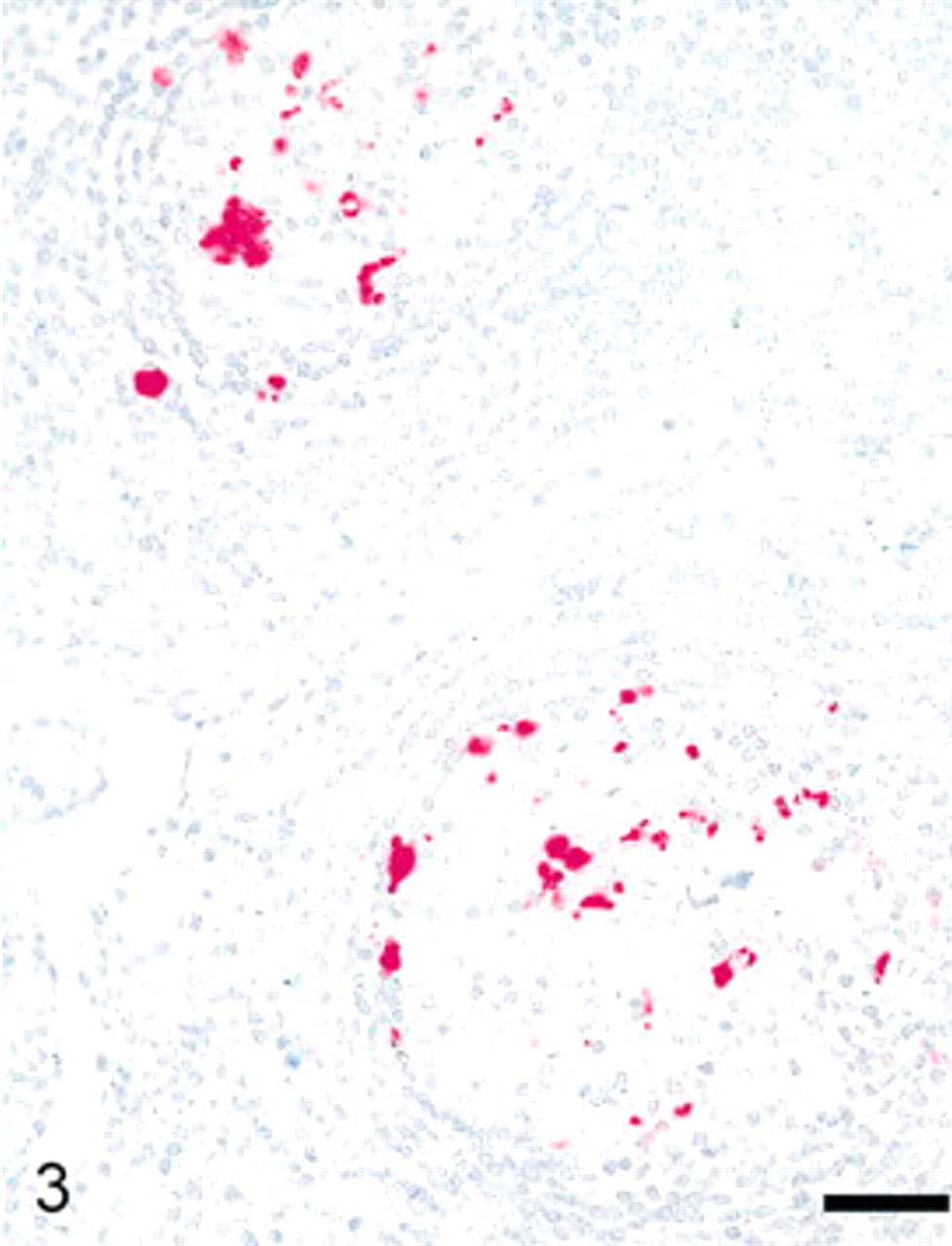
Pharyngeal tonsil of sheep No. 52 euthanized at 12 months postinoculation. The PrPSc labeling is present in the germinal centers of 2 follicles. Stained for PrPres labeling (red) by immunohistochemistry (immunoalkaline phosphatase) and counterstained with hematoxylin. Bar = 30 μm.
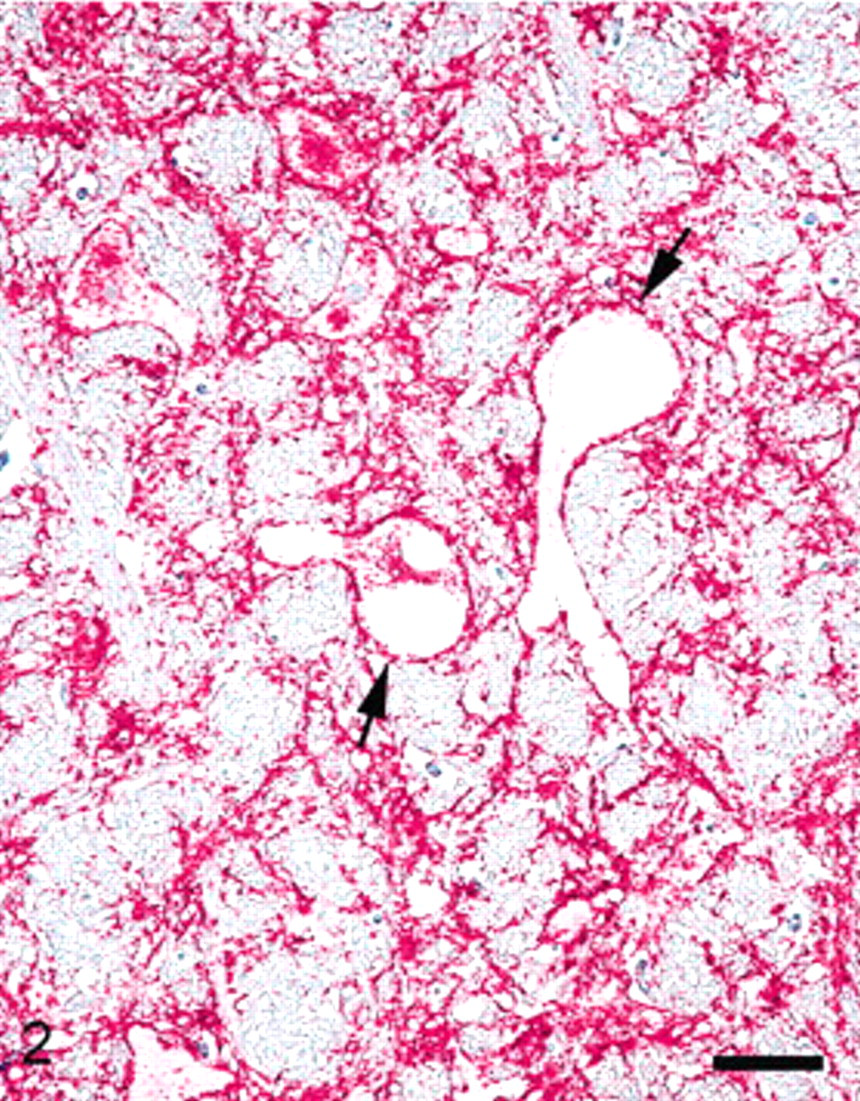
Medulla oblongata at the level of obex of sheep No. 56 with clinical scrapie. There is extensive diffuse PrPSc labeling in the neuropil. Also, there is some PrPSc labeling within neurons as well as in the 2 vacuolated neurons (arrows). Stained for PrPSc labeling (red) by immunohistochemistry (immunoalkaline phosphatase) and counterstained with hematoxylin. Bar = 60 μm.
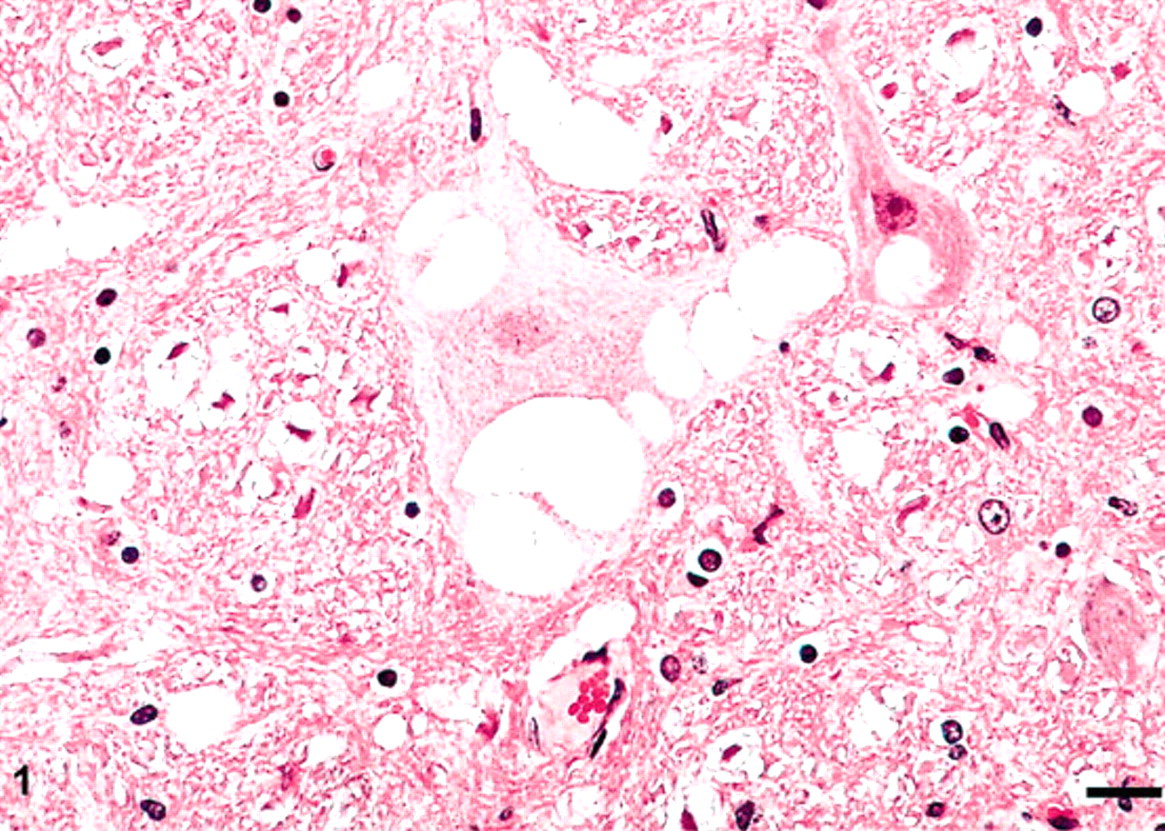
Medulla oblongata at the level of obex of sheep No. 56 with clinical scrapie. There is extensive vacuolation of the neuropil and presence of single or multiple vacuoles in neuronal perykaria. HE. Bar = 20 μm.
In addition to the IHC data presented in Table 1, lymphoid aggregates were detected in only 1 of the third eyelid samples, which was negative for PrPSc labeling by IHC.
IHC-stained tissue sections of non–central nervous system and nonlymphoid tissues, including striated muscles (heart, diaphragm, and masseter muscle), did not reveal PrPSc labeling.
Western blot analyses of brainstem samples from these animals revealed that all sheep (except No. 52 euthanized at 12 MPI) inoculated with the scrapie agent via different routes (Table 1) were positive for the presence of PrPSc, as detected by monoclonal antibody P4 (Fig. 4). Animal Nos. 61 and 63, which did not exhibit clinical TSE disease (Table 1), were both positive for PrPSc; however, No. 61 was weakly positive (Fig. 4). The PrPSc isoforms from all scrapie-positive brainstems had a similar molecular weight (Fig. 4), independent of their route of inoculation. Brainstem material for animal Nos. 52 and 61 was enriched up to a concentration of 20-mg brain equivalent (eq). Brain material of animal No. 61 showed distinct PrPSc profiles starting at 1-mg brain eq, whereas animal No. 52 and control uninfected sheep brainstem material was negative at 20-mg brain eq (data not shown).
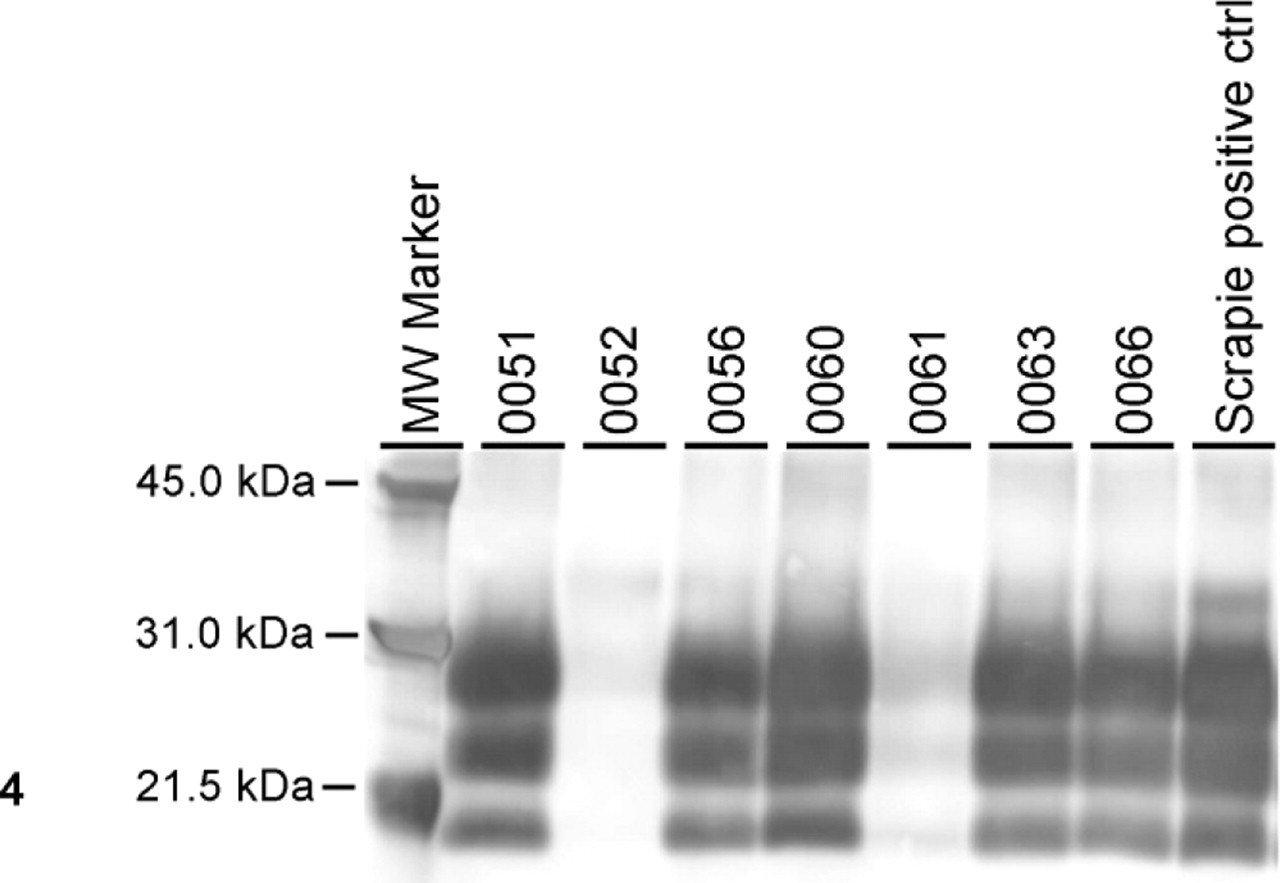
Western blot analysis with monoclonal antibody P4 showing PrPSc in the brainstem of all sheep except sheep No. 52.
Discussion
In general, the intracerebral (IC) route of inoculation of TSE agents is considered to be the fastest means of inducing disease with these agents in susceptible hosts. In sheep using the same scrapie inoculum (No. 13-7), IC inoculations revealed average incubation times of 18.8 MPI, whereas oral inoculation averaged 32 MPI. 5 In the present study, the fastest method was the intraperitoneal (mean = 31 MPI) followed by the intranasal (36 MPI) and intraconjunctival (37.5 MPI) routes. Although these 3 routes of inoculation had generally similar incubation times as to the previously reported oral inoculation (32 MPI), it must be pointed out that in the oral study a total dose of 30 ml of 10% inoculum was used. Also, despite this large oral dose, only 5 of 9 orally inoculated sheep developed clinical scrapie. 5
Although the IC route of inoculation gives the shortest incubation time, it involves the use of light anesthesia and is an invasive surgical procedure. The IC procedure carries minimal risks to the recipient animals, but complications associated with anesthesia as well as postsurgical sequelae, such as intracranial hematoma and encephalitis, have been observed. To avoid these complications, we have evaluated various less invasive routes of inoculation. Interestingly, the intralingual route results in a short incubation time that is comparable to the intracerebral inoculation. 4 The current exploration of alternate routes of experimentally induced scrapie may lead to a safer and more efficient method of challenge of TSEs to recipient animals.
All scrapie-inoculated sheep except No. 52 were positive for the presence of PrPSc in the central nervous system tissues. Animal Nos. 61 and 63, which did not exhibit clinical signs of scrapie, revealed the presence of spongiform encephalopathy and PrPSc in the brainstem, indicating that in addition to PrPSc detection by Western blot and IHC, histopathologic changes can be observed in preclinical stages. Both intraconjunctival and intranasal application of scrapie to sheep results in clinical disease, and the brainstem seems to be one of the early target areas in the central nervous system, confirming that brainstem is an appropriate tissue for diagnostic purposes in animals naturally exposed to classical strains of scrapie. In a previous study, it was speculated that an exposure route for an atypical cattle prion agent (bovine amyloidotic spongiform encephalopathy) other than the alimentary tract could result in less severe involvement of the brainstem. 2
Results of this study have shown that none of the 3 routes of inoculation produced clinical scrapie as fast as the IC route of inoculation. 5 However, it is possible for sheep to develop scrapie using peritoneal, nasal, and conjunctival inoculation routes. One intranasally inoculated sheep (No. 52; Table 1; euthanized at 12 MPI) showed presence of PrPSc labeling only in the pharyngeal tonsil; therefore, it is suggested that the tonsil was the portal of entry for PrPSc into this animal. It could be argued that inoculation into the nasal passage of sheep may also have found some inoculum in the posterior oral cavity, thereby resulting in both nasal and oral inoculation. Although this is certainly a possibility, it must be realized that in this study only a small volume of inoculum (1 ml of 10% suspension) was used, whereas in a previous study a large volume of the same inoculum (30 ml of 10% suspension) was required to induce scrapie in sheep. 5 Because a relatively small volume of inoculum was needed for the intranasal route compared with oral inoculation, 5 it is speculated that, in the present study, the upper respiratory tract (most likely the pharyngeal tonsil) served as a natural portal of entry for the scrapie agent. In a recent study using a transmissible mink encephalopathy strain (HY) in hamsters, it was also concluded that the nasal cavity was an effective portal of entry for the transmissible mink encephalopathy agent. 7 Infection via the intranasal route could also be possible in scrapie-infected environments, such as in scrapie-infected flocks. However, because in this experiment the number of animals per individual groups was rather small, this question should be addressed in a future study with larger numbers of susceptible sheep.
Footnotes
Acknowledgements
We thank Drs. L. Nusz and J. Laufer for clinical assistance, Dr. Katherine O'Rourke for prion antibodies for immunohistochemistry, and James Fosse for the photomicrographs. Expert technical assistance was provided by Martha Church, Dennis Orcutt, Deb Clouser, Kevin Hassall, Sharla Van Roekel, Ginny Montgomery, and the transmissible spongiform encephalopathy animal caretakers. This study was carried out under the guidelines of the institutional ACUC committee at the National Animal Disease Center. Mention of trade names or commercial products in this article is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture.
