Abstract
The purpose of this study was to characterize the patterns of PrPSc immunoreactivity in the retinae of scrapie-affected sheep and to determine the extent of retinal pathology as indicated by glial fibrillary acidic protein immunoreactivity (GFAP-IR) of Müller glia. Sections from the retina of 13 experimentally inoculated scrapie-affected and 2 negative control sheep were examined with immunohistochemical staining for PrPSc, GFAP, and PrPSc/GFAP double staining. GFAP-IR of Müller glia is suggestive of retinal pathology in the absence of morphologic abnormality detected by light microscopy. Sheep with the least amount of PrPSc in the retina have multifocal punctate aggregates of prion staining in the outer half of the inner plexiform layer and rarely in the outer plexiform layer. In these retinae, GFAP-IR is not localized with prion accumulation, but rather is present in moderate numbers of Müller glia throughout the sections of retina examined. The majority of sheep with retinal accumulation of PrPSc have intense, diffuse PrPSc staining in both plexiform layers, with immunoreactivity in the cytoplasm of multiple ganglion cells and lesser amounts in the optic fiber layer and between nuclei in nuclear layers. This intense PrPSc immunoreactivity is associated with diffuse, intense GFAP-IR that extends from the inner limiting membrane to the outer limiting membrane. This is the first report of a prion disease in a natural host that describes the accumulation of PrPSc in retina associated with retinal pathology in the absence of overt morphologic changes indicative of retinal degeneration.
Keywords
The transmissible spongiform encephalopathies (TSEs, or prion diseases) are a family of related neurodegenerative diseases associated with the accumulation of an abnormal form of prion protein (PrPSc). Previous studies have demonstrated that abnormal prion protein accumulates in synaptic regions of the retina in animals 12– 15, 22, 31 and humans 16, 17 affected by TSEs. The function of the normal prion protein in the healthy nervous system is not known, although there is evidence demonstrating that it is localized presynaptically and may have a synaptic function. 18, 26 Prion protein also has been localized to synaptic regions in other parts of the central nervous system. 1 Further, there is evidence that the accumulation of abnormal prion protein alters the normal localization of known synaptic proteins. 29 In some instances, measurable effects on vision of animals with TSE infections have been documented. Primates inoculated intracerebrally with brain or buffy coat derived from another primate infected with bovine spongiform encephalopathy (BSE) became symptomatic between 16 and 18 months after inoculation, with visual system abnormalities that included loss of pupillary light responses and unresponsiveness to visual stimuli when compared to control animals. 3
PrPSc accumulation, infectivity, and/or retinal degeneration have been described in numerous animal models or cross-species transmission studies. In Syrian hamsters, retinal degeneration and clinical signs progress rapidly after maximal scrapie infectivity titers plateau, 19 and PrPSc immunoreactivity is prominent in the inner and outer segments of rod photoreceptors, the sites of early pathologic changes in scrapie-infected hamsters. 5, 20 The distribution of abnormal prion protein in the retina also has been described in mouse models of scrapie. 9, 23 Transgenic mice expressing hamster PrP from the PrP promoter (tg7) developed clinical disease, retinal degeneration, and extensive apoptosis in the outer nuclear layer after intraocular inoculation of hamster scrapie. 23 Very little PrPSc immunolabeling is observed in the layers of the retina in some mouse models, despite high levels of infectivity and demonstration of scrapie-induced retinopathy. 9 In natural host species, accumulation of PrPSc in the inner and outer plexiform layers of the retina has been described in sheep 8, 12, 15 and goats 32 affected with scrapie, cattle with BSE, 4 mule deer with chronic wasting disease (CWD), 31 and humans with Creutzfeldt-Jacob Disease (CJD). 16, 17 With the exception of one report describing retinopathy in 2 scrapie-affected sheep, 2 histologic lesions affecting the visual system have not been described in sheep, a natural host of scrapie.
Upregulation of glial fibrillary acidic protein (GFAP) is an indicator of retinal stress 33 and occurs concomitant with hypertrophy of Müller cells as a consequence of retinal injury. 24 Müller cells span the retina from the outer limiting membrane, which is composed of a plane of junctions between Müller cells and photoreceptors at the base of the apical microvilli to the vitreal surface of the retina at the Müller cell endfeet (inner limiting membrane). In the normal retina, GFAP and vimentin expression is restricted to the endfeet, with occasional filaments extending between the ganglion cell layer and inner plexiform layer. 24
The purpose of this work was to describe the progression of PrPSc accumulation in the retina and to evaluate GFAP immunoreactivity (GFAP-IR) in Müller glia. Our results demonstrate retinal pathology prior to morphologic change observable by light microscopy.
Materials and Methods
Retinae described here were collected as part of a sheep scrapie pathogenesis study. 12 Suffolk sheep were inoculated by oral or intracranial routes at 4 months of age. Tissues including entire globes were collected at necropsy at 4, 9, 15, and 21 months after inoculation or when animals had terminal signs of disease. Sections examined for this study included the retina from all sheep from the previous study with immunoreactivity to PrPSc in brainstem for which the retina was available (n = 13). Entire globes with a segment of ocular nerve of approximately 1–2 cm were extracted at necropsy. Connective tissue and muscle were removed from the area surrounding the optic nerve, and a scalpel was used to make a small slit through the connective tissue tunics at the junction of the cornea and sclera before immersion into 10% neutral buffered formalin. Tissues were allowed to fix for at least 3 weeks, at which time a 5-mm-thick vertical section from the caudal aspect of the globe containing retina and optic nerve was processed by routine histologic methods and embedded into paraffin blocks. Serial 4-μm sections from the retina were cut for staining by hematoxylin and eosin (HE), as well as PrPSc, GFAP, and PrPSc/GFAP double-labeled immunohistochemistries. Sections immunostained to detect PrPSc were processed as previously described 13 using primary antisera containing monoclonal antibodies F89/160.5 27 and F99/97.6.1, 30 each at a concentration of 5 μm/ml. With each batch of slides stained for PrPSc, serial sections of brainstem from a known positive sheep also were stained for PrPSc to assess variability between runs and processed with omission of primary antibody to control for nonspecific staining. Immunostaining for GFAP was performed as described below. After being deparaffinized and rehydrated, sections were stained using a commercial kit (Dako EnVision Doublestain System, DakoCytomation, Carpinteria, CA) containing all reagents except primary antibody. Sections were treated for endogenous peroxidase for 5 minutes with a 0.03% hydrogen peroxide buffer provided. The slides were then incubated with GFAP antibody (Fibrillary Acidic Protein Antibody [rabbit anti-cow], DakoCytomation) at 1 ° 10,000 dilution overnight at 4°C. Secondary antibody was applied for 30 minutes at room temperature, followed by 3,3′-diaminobenzidine tetrahydrochloride (DAB) substrate stain for 10 minutes at room temperature. All sections assessed for GFAP-IR were processed together with a section of cerebrum serving as a positive control and a serial section of cerebrum with omission of primary antibody serving as a control for nonspecific staining. Double labeling for PrPSc/GFAP was done by following this GFAP immunostaining protocol with the PrPSc protocol. Immediately following completion of the DAB reaction of the GFAP protocol, slides were autoclaved for 30 minutes at 121°C in a target retrieval solution (DakoCytomation Target Retrieval Solution, DakoCytomation) and were stained using monoclonal antibodies as described above.
Results
Sections from all available retinae from sheep with PrPSc immunoreactivity in at least one tissue in a previous sheep pathogenesis study 12 were used to further describe PrPSc distribution, GFAP-IR, and the association between PrPSc and GFAP-IR in double-stained retinal sections. No lesions were noted on examination of HE-stained sections made from formalin-fixed tissues. Assessment of PrPSc accumulation (Figs. 1–4) in retina was done on all sheep with at least 1 PrPSc-positive tissue (Table 1). Of particular interest was the observation of PrPSc accumulation in the retina of an orally infected animal (No. 13) that was not yet displaying clinical signs of disease. Immunoreactivity in retinae with the least PrPSc accumulation was observed in the outer part of the inner plexiform layer (Figs. 1 and 2, arrows) and less frequently within the outer plexiform layer (Fig. 1, arrowhead). More PrPSc accumulation was observed in animals with clinical signs. In these animals, PrPSc immunoreactivity was observed throughout both the inner and outer plexiform layers (synaptic layers) and in multiple retinal ganglion cells (Fig. 3). In cases with the greatest amount of PrPSc accumulation, PrPSc immunoreactivity was observed in the plexiform layers in many ganglion cells and photoreceptor inner segments (Fig. 4).
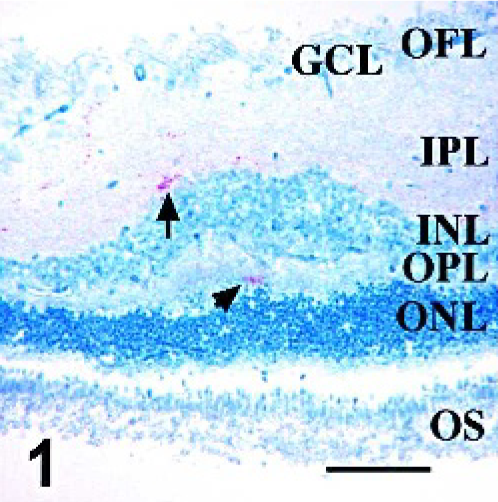
Retina; Sheep 13. Distribution of PrPSc in scrapie-infected sheep retina. PrPSc accumulation is first noted in the outer part of the inner plexiform layer (arrow) and rarely in the outer plexiform layer (arrowhead). PrPSc immunohistochemistry with monoclonal antibodies F89/160.1.5 and F99/97.6.1 and hematoxylin counterstain. OS, photoreceptor outer segments; ONL, outer nuclear layer; OPL, outer plexiform layer; INL, inner nuclear layer; IPL, inner plexiform layer; GCL, ganglion cell layer; OFL, optic fiber layer. Bar = 60 μm.
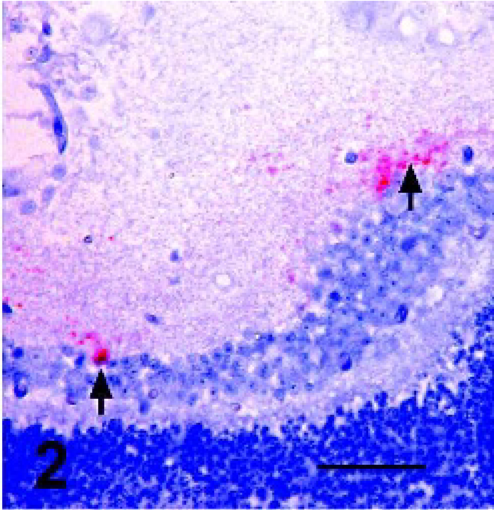
Retina; Sheep 13. Higher magnification view of punctate and globular areas of immunoreactivity in the outer margins of the inner plexiform layer (arrows). PrPSc immunohistochemistry with monoclonal antibodies F89/160.1.5 and F99/97.6.1 and hematoxylin counterstain. Bar = 20 μm.
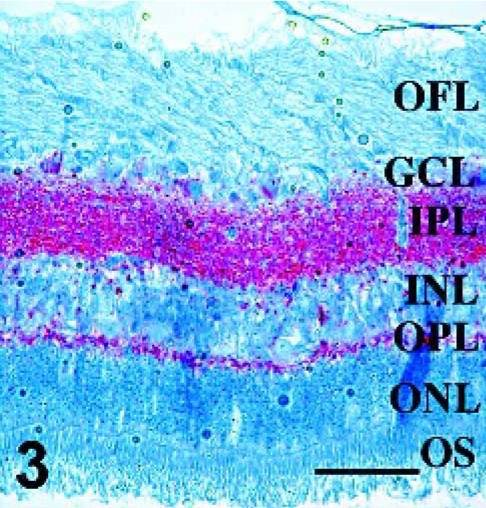
Retina; Sheep 7. Intense prion immunoreactivity is present throughout inner and outer plexiform layers, indicating moderate PrPSc accumulation. PrPSc immunohistochemistry with monoclonal antibodies F89/160.1.5 and F99/97.6.1 and hematoxylin counterstain. Bar = 60 μm.
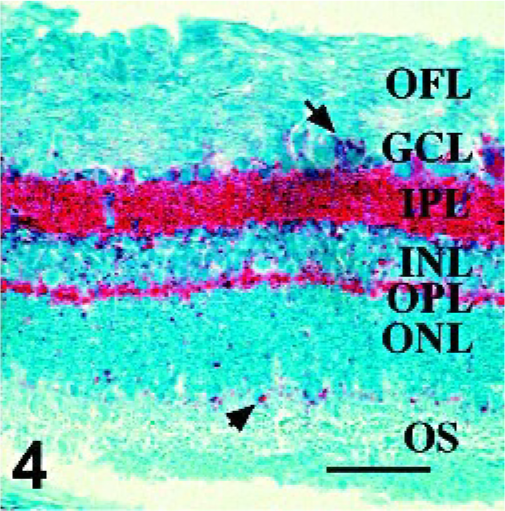
Retina; Sheep 1. In retinae with the most PrPSc accumulation, immunoreactivity is present in multiple ganglion cells (arrow), at the level of photoreceptor outer segments (arrowhead), and in lesser amounts in the optic fiber layer. PrPSc immunohistochemistry with monoclonal antibodies F89/160.1.5 and F99/97.6.1 and hematoxylin counterstain. Bar = 60 μm.
Retinal layers with PrPSc accumulation in scrapie-affected sheep after intracerebral and oral inoculation. PrPSc immunohistochemistry results for retina of selected sheep.
OS, photoreceptor outer segments; ONL, outer nuclear layer; OPL, outer plexiform layer; INL, inner nuclear layer; IPL, inner plexiform layer; GCL, ganglion cell layer; OFL, optic fiber layer; PI, postinoculation time; IC, intracerebral; +, GFAP positive; -, GFAP negative.
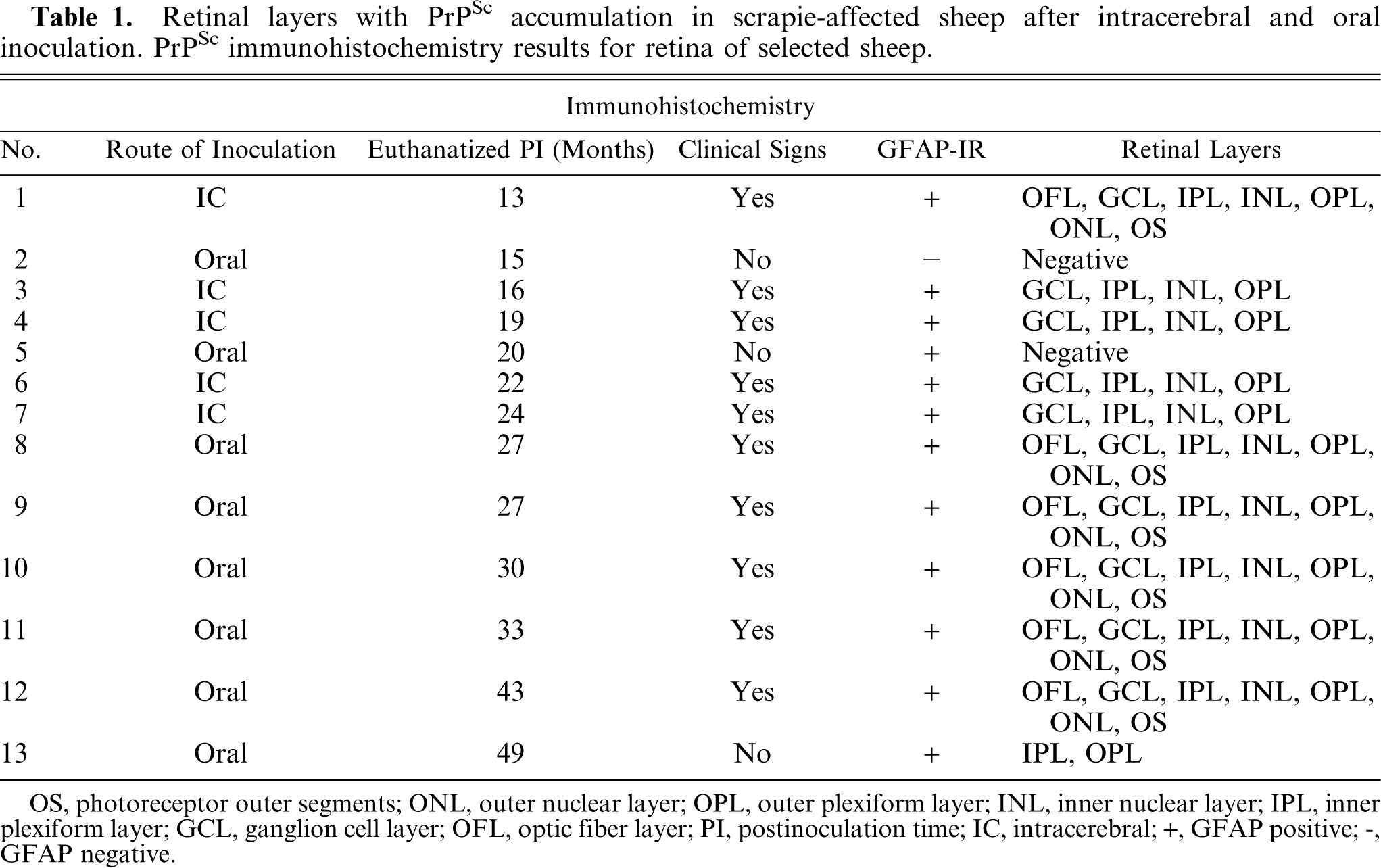
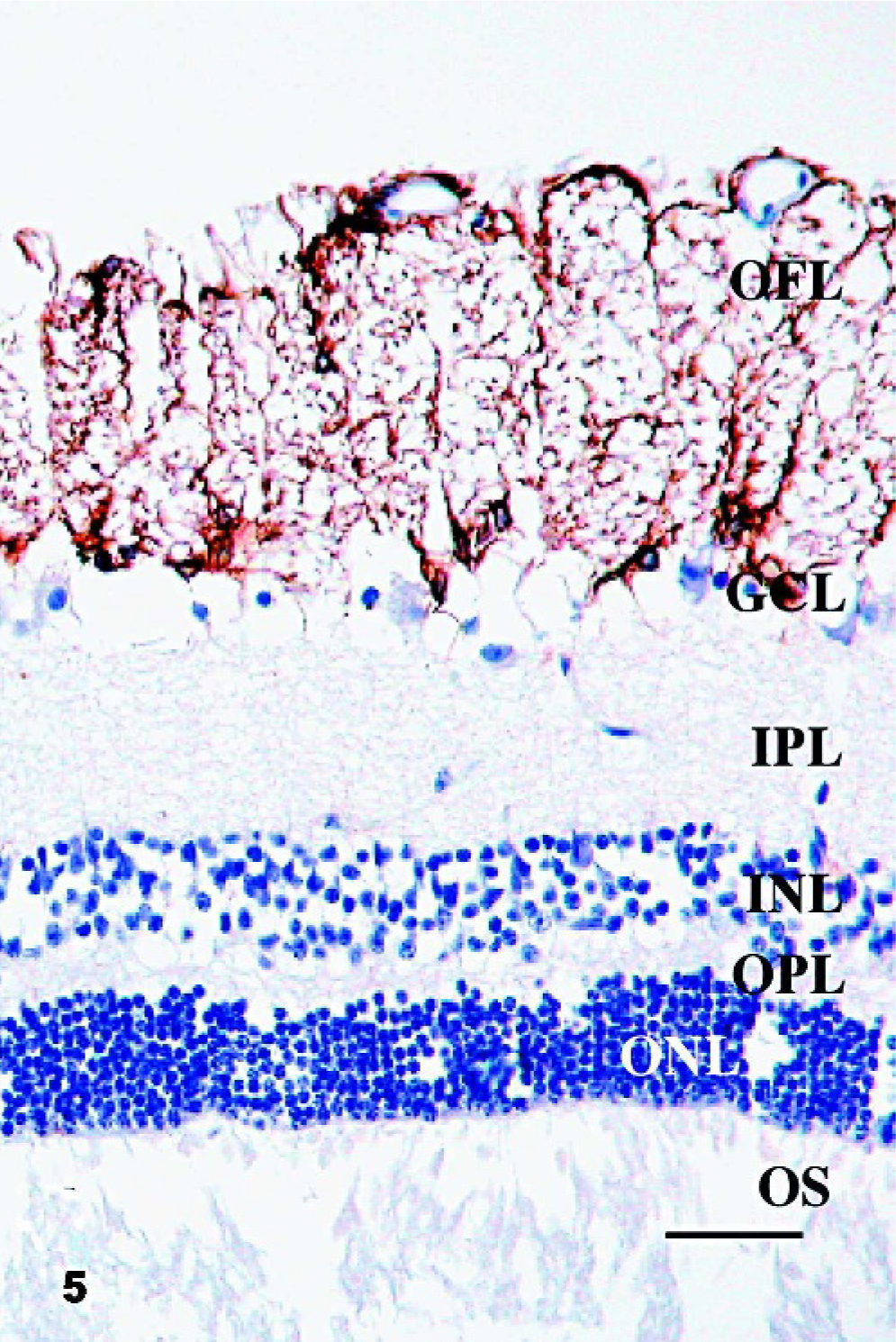
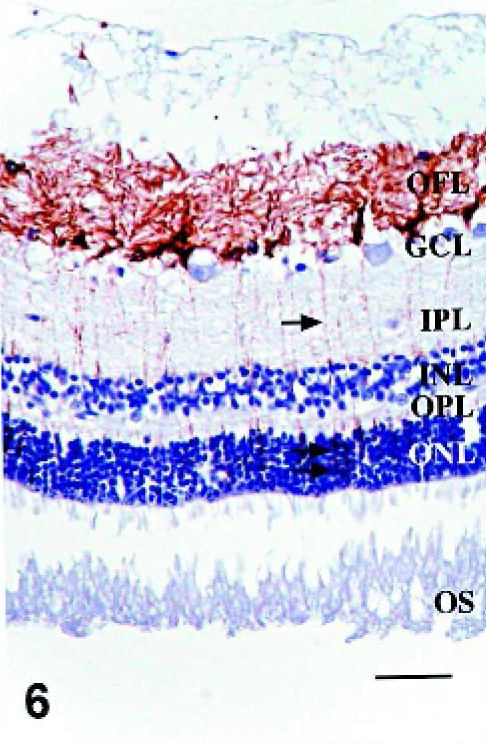
Since morphologic changes consistent with retinopathy or cell death were not demonstrated by HE-stained sections, GFAP immunostaining was performed to evaluate the extent of retinal pathology. Control specimens had scant to rare GFAP-IR, which was found predominantly in astrocytes of the optic fiber layer, with rare fibrillar extension of immunoreactivity through the inner plexiform layer (Fig. 5). In retinae with scant, multifocal areas of PrPSc accumulation, GFAP-IR was moderately increased. Cells with GFAP-IR were found not only in areas adjacent to PrPSc accumulation, but also throughout the retina (Figs. 6, 7). In retinae with intense PrPSc staining, GFAP-IR was intense throughout all layers of the retina, including areas surrounding ganglion cells, fibrillar extension through both inner and outer plexiform layers, within inner and outer nuclear layers, and diffusely at the Müller cell termination point at the inner segments of the photoreceptor layer (Figs. 8–10). GFAP-IR was increased in all retinae with PrPSc accumulation. All retinae with PrPSc accumulation had more GFAP-IR than control sheep. In addition, one sheep (No. 5) without PrPSc accumulation detectable by our methods had increased GFAP-IR in the retina.
Retina; Control sheep. GFAP-IR in control specimens was found predominantly at the inner limiting membrane and in astrocytes of the OFL with rare fibrillar extension of immunoreactivity through the inner plexiform layer. OS, photoreceptor outer segments; ONL, outer nuclear layer; OPL, outer plexiform layer; INL, inner nuclear layer; IPL, inner plexiform layer; GCL, ganglion cell layer; OFL, optic fiber layer. GFAP immunohistochemistry with hematoxylin counterstain. Bar=60 μm.
Retina; Sheep 13. In retinae with scant, multifocal areas of PrPSc accumulation, GFAP-IR was moderately increased. Fibrillar areas of GFAP-IR are present, extending through all layers of the retina between the ganglion cells and the photoreceptor outer segments (arrows). OS, photoreceptor outer segments; ONL, outer nuclear layer; OPL, outer plexiform layer; INL, inner nuclear layer; IPL, inner plexiform layer; GCL, ganglion cell layer; OFL, optic fiber layer. GFAP immunohistochemistry with hematoxylin counterstain. Bar = 60 μm.
Retina; Sheep 13. Cells with GFAP-IR were found adjacent to areas of PrPSc accumulation (arrow), and throughout the retina. GFAP/PrPSc double immunohistochemistry with hematoxylin counterstain. Bar = 60 μm.
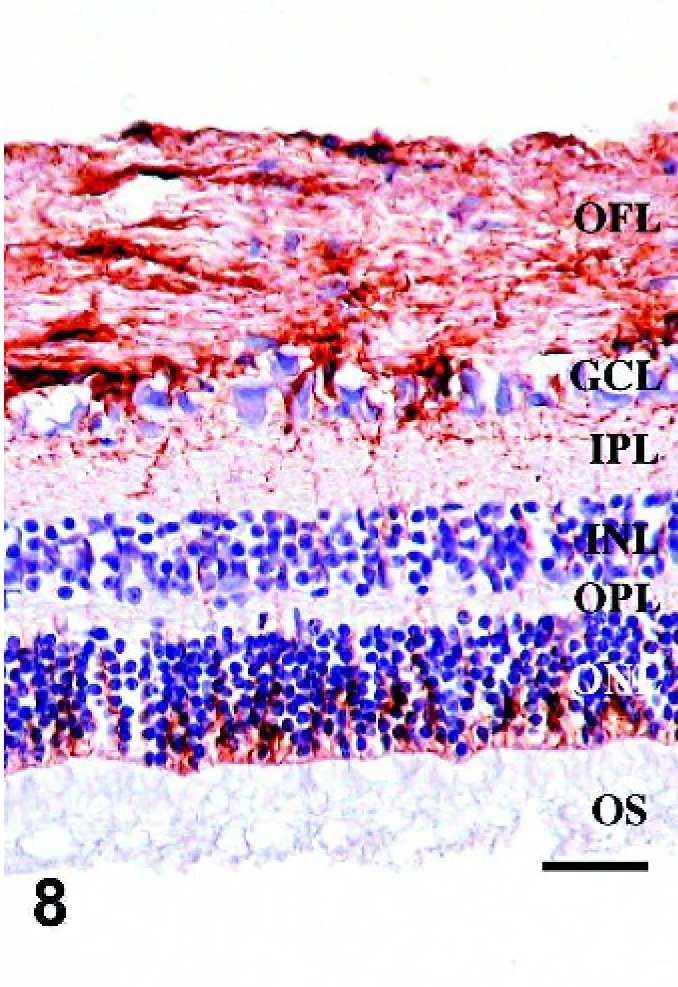
Retina; Sheep 1. GFAP-IR is increased in sheep with marked accumulation of PrPSc compared with control sheep (Fig. 5) or sheep with less PrPSc accumulation (Fig. 6). OS, photoreceptor outer segments; ONL, outer nuclear layer; OPL, outer plexiform layer; INL, inner nuclear layer; IPL, inner plexiform layer; GCL, ganglion cell layer; OFL, optic fiber layer. GFAP immunohistochemistry with hematoxylin counterstain. Bar = 60 μm.
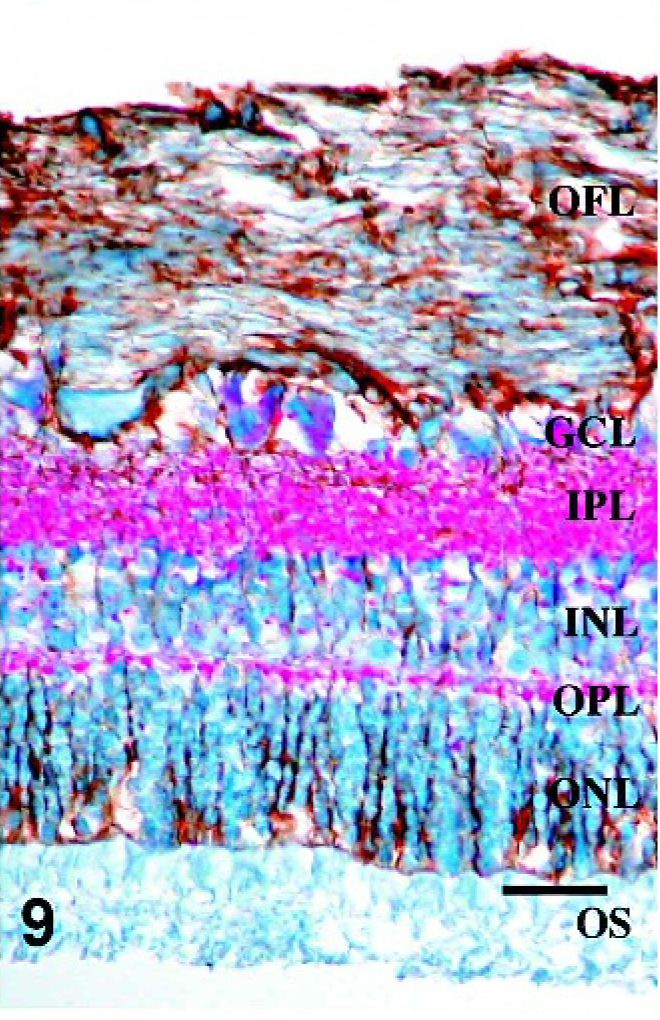
Retina; Sheep 1. Intense PrPSc staining is associated with GFAP-IR throughout all layers of the retina, including areas surrounding ganglion cells, fibrillar extensions through both inner and outer plexiform layers, within inner and outer nuclear layers, and diffusely at the Müller cell termination point at the inner segments of the photoreceptor layer. GFAP/PrPSc double immunohistochemistry with hematoxylin counterstain. Bar = 60 μm.
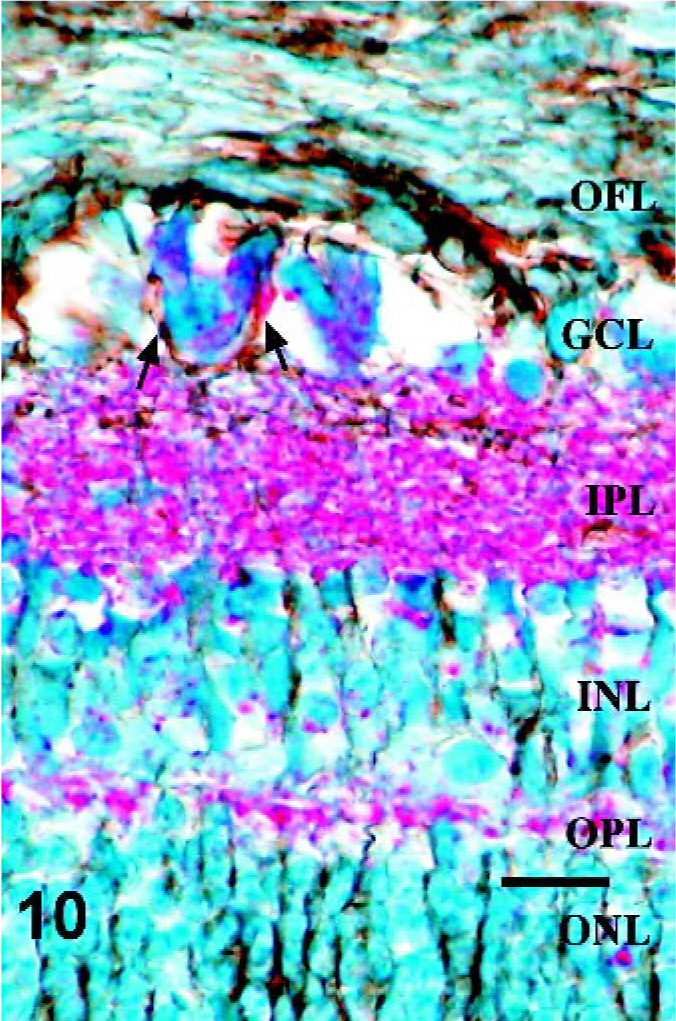
Retina; Sheep 1. Higher magnification of the retina in Fig. 8. Note GFAP-IR surrounding multiple ganglion cells with cytoplasmic PrPSc immunoreactivity (arrows). GFAP/PrPSc double immunohistochemistry with hematoxylin counterstain. Bar = 20 μm.
Discussion
This is the first report of prion disease in a natural host that describes the accumulation of PrPSc in retina associated with retinal pathology in the absence of overt morphologic changes indicative of retinal degeneration. In the retina of sheep with the fewest foci immunoreactive for PrPSc, immunoreactivity is present predominantly in the outer portion of the inner plexiform layer, with rare foci of immunoreactivity in the outer plexiform layer. As PrPSc immunoreactivity increases, the inner and outer plexiform layers are diffusely affected with lesser staining throughout all layers, from the inner limiting membrane to the outer limiting membrane. Prion immunoreactivity in outer and inner plexiform layers suggests accumulation of PrPSc in association with synapses. It is not known whether abnormal prion accumulation is associated with functional impairment, but evidence suggests that normal prion protein may be involved in regulation of normal synaptic function. 18 Further, PrPSc replication has been shown to alter the distribution of synaptic machinery, suggesting that it may alter synaptic function. 29
Expression of GFAP in normal retina occurs in astrocytes of the optic fiber layer and ganglion cell layer, with some expression in Müller cells 33 at the inner limiting membrane. Increased GFAP expression in Müller glia is considered an indicator of retinal degeneration. 21 In this study, patterns of GFAP-IR were compared between control sheep and scrapie-infected sheep with low and high levels of PrPSc accumulation in retina.
Intraocular inoculation of some scrapie strains in mice causes retinal ganglion cell degeneration. 28 While another study shows microglial cell recruitment into the ganglion cell layer and inner plexiform layer after intraocular injection, 25 neither microglia nor morphologic changes consistent with cellular degeneration or loss were noted in any of the retinae examined in this study. It should be noted that all tissue sections examined were derived from formalin-fixed tissues. Formalin fixation of the globe can be associated with lesser tissue integrity and more artifacts that complicate assessment of tissue morphology than other fixatives, such as Bouin's solution with picric acid. Accumulation of abnormal prion protein in our infected sheep was not associated with loss of outer rod segments, inner segments, or photoreceptor nuclei, as has been reported in the hamster 6, 7, 19 and mouse 10, 11, 23 models.
In the present study, there was increased GFAP-IR in all retinae with PrPSc accumulation. Retinae with PrPSc immunoreactivity had increased GFAP expression throughout the retina, rather than only in areas of PrPSc accumulation. Diffusely throughout the sections examined, retinae with large amounts PrPSc staining had intense GFAP-IR throughout the length of Müller cells. Interestingly, the retina of one sheep without detectable PrPSc accumulation had increased GFAP-IR compared with control tissues. The significance of this finding is not yet known. Despite the absence of histologic lesions, concurrent GFAP and PrPSc immunoreactivity suggests retinal pathology as a result of PrPSc accumulation.
The retina may be an interesting tissue to study, as PrPSc accumulation was observed prior to clinical signs in one animal. In this study, with the exception of 2 sheep, the retinae of all animals were positive for PrPSc by immunohistochemistry when determined to be positive in at least one other tissue. Retina may be a tissue of choice for studying the pathobiology of PrPSc accumulation because of its ordered structure and the fact that PrPSc readily accumulates in the retinae of scrapie-affected sheep.
Footnotes
Acknowledgements
We thank Dr. Robert Kunkle for his advice on PrPSc immunohistochemical staining and Dennis Orcutt, Joe Lesan, and Martha Church for their technical assistance with this project. Mention of trade names or commercial products in this article is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture.
