Abstract
Scrapie is a fatal neurodegenerative disease of sheep resulting from the accumulation of a misfolded form of the prion protein (PrPSc). Polymorphisms in the host prion protein gene (PRNP) can affect susceptibility to the scrapie agent. Lysine (K) at codon 171 of PRNP is an inadequately characterized, naturally occurring polymorphism in sheep. We inoculated Barbado sheep with PRNP genotypes QQ171, QK171, or KK171 by either the intracranial (IC, n = 2–7 per genotype) or oronasal (ON, n = 5 per genotype) routes with a scrapie isolate to investigate the effect of lysine at codon 171 on susceptibility. When neurologic signs were observed or at the end of the experiment (70 months postinoculation [MPI]), sheep were necropsied and tissue collected for histopathologic, immunohistochemical, enzyme immunoassay and Western blot examination for PrPSc. All genotypes of sheep developed scrapie after IC inoculation. After ON inoculation, sheep with the QK171 genotype had prolonged incubation periods compared to the QQ genotype. During the experiment, 2 of 5 of the ON-inoculated QK genotype sheep developed neurologic signs and had PrPSc in the brain. The other 3 of 5 sheep were asymptomatic at 70 MPI but had detectable PrPSc in peripheral tissues. None of the ON-inoculated sheep of the KK171 genotype developed signs or had detectable PrPSc. Our experiments demonstrate that sheep with the KK171 genotype are resistant to scrapie via oronasal exposure and that sheep with the QK171 genotype have prolonged incubation relative to QQ171 sheep. The K171 prion protein allele may be useful to enhance scrapie resistance in certain breeds of sheep.
Keywords
Transmissible spongiform encephalopathies (TSEs) are fatal neurodegenerative diseases that affect multiple species. Scrapie is the naturally occurring TSE of sheep and goats. 18,24,29 The causative agent is a misfolded form of the prion protein (PrPSc), which is self-replicating and infectious. 7,28 Sheep are susceptible via natural exposure to PrPSc both vertically from mother to offspring and horizontally between unrelated animals in a flock. 8,30
The normal cellular prion protein (PrPC) is encoded by an endogenous gene, PRNP. 27 Allelic variation in PRNP codons 136, 154, and 171 produces amino acid polymorphisms in PrPC that correlate with varying degrees of scrapie susceptibility in sheep. 5,9,10 Alanine (A) 136 and arginine (R) 171 lend relative resistance while valine (V) 136 and glutamine (Q) 171 correlate with an increased risk for developing scrapie. Arginine 154 has a minor association with scrapie susceptibility. Ultimately, disease susceptibility is influenced primarily by the 136/171 haplotype with a distinct influence imparted by codon 171. 4,5,11,21 The A136R154R171/ARR and ARQ/ARR haplotypes are associated with relative resistance while ARQ/ARQ is linked to scrapie susceptibility. 4,15 Homozygosity for Q171 increases susceptibility, whereas a survival advantage exists with heterozygosity (eg, QR171). 21,34
The effect of common amino acid polymorphisms at codon 171 (Q171, R171) on scrapie susceptibility has been investigated. 14,21,32 However, little is known about the contribution of lysine (K) other than the presence of naturally occurring scrapie affecting heterozygous K171 sheep in Greek and Italian sheep. 1,6 K171 has a relatively higher incidence in some Mediterranean breeds and also occurs in other breeds seen in the United States, including Barbado, Barbado/St Croix, Dorper, and Suffolk crosses. 2,17 Nonrandom sampling of flocks in the United States has also identified the polymorphism in American Blackbelly, Black Hawaiian, Painted Desert, and Texas Dall sheep (D. Sutton and D. Norden, USDA APHIS, personal communication, unpublished data, 2017). For regulatory purposes, the current scrapie eradication program in the United States considers sheep with Q, H, and K at position 171 equally susceptible to scrapie. 3 Therefore, determining the contribution of K171 to scrapie susceptibility could affect breeding programs by allowing retention of otherwise desirable rams.
We previously reported a prolonged incubation period in heterozygous QK171 Barbado sheep intracranially (IC) inoculated with the scrapie agent. 16 The purpose of the current study was to compare the relative susceptibility of sheep with PRNP encoding QQ171, QK171, or KK171 after either IC or oronasal (ON) inoculation with a US-derived scrapie isolate.
Materials and Methods
Animal Procedures
Animals for this experiment were sourced from a known scrapie-free flock housed at the US Department of Agriculture (USDA) National Animal Disease Center (NADC) in Ames, Iowa. Approval from the Institutional Animal Care and Use Committee was obtained prior to conducting this experiment (protocol number 3893). PRNP genotypes were sequenced by polymerase chain reaction (PCR) using previously described methods. 15 Amino acid sequences of the prion protein were predicted from the genotypes; they were determined to be homozygous at other potentially polymorphic sites M112, G127, A136, M137, S138, L141, R151, R154, M157, N176, H180, Q189, T195, T196, R211, Q220, and R223. For this study, genotype polymorphisms were documented at codon 171: QQ171, QK171, or KK171. The experimental groups comprised sheep with QK and KK genotypes while the QQ genotype served as a positive control. Sheep in the IC experiment were inoculated at 11 months old except the 2 positive controls (QQ) that were retired breeding ewes (24 and 59 months of age). All lambs in the ON experiment were inoculated at approximately 3 months of age.
Scrapie Isolates and Animal Procedures
A classical scrapie isolate from the continental United States (No. 13-7) was serially passaged 4 times through ARQ/ARQ Suffolk sheep prior to use as an inoculum in the current experiment. 20 Brain tissue was mechanically ground, 100 μg/ml of gentamicin was added, and the final homogenate was prepared as a 10% (w/v) solution with phosphate-buffered saline. Lambs from each genotype group were experimentally infected as described below.
The procedure for intracranial inoculation has been described. 16,19 Briefly, the sheep were sedated with xylazine and the dorsal frontal region was surgically prepared prior to making a 1-cm midline incision caudal to the junction of the parietal and frontal bones. A 1-mm hole was drilled through the underlying bone. A 22-guage spinal needle was inserted through the hole to the ventral aspect of the calvarium where 1 ml of inoculum was injected slowly as the needle was withdrawn. The skin incision was closed with tissue glue. Oronasal inoculation was performed as previously described. 25 Briefly, the head was elevated slightly, and 1 ml of inoculum was deposited into the right nostril via a needleless syringe.
All inoculated sheep were housed in biosafety level 2 facilities for 2 weeks following exposure to scrapie. After this period, the sheep were moved to outside pens at the NADC. Animals were fed a pelleted growth and alfalfa hay ration.
Sheep were monitored daily for clinical signs consistent with scrapie. Necropsy was performed when unequivocal clinical signs occurred or at the predetermined end of the study (70 months postinfection).
Sample Collection and Processing
At necropsy, duplicate tissue samples were collected and stored in 10% buffered neutral formalin (globes in Bouin’s fixative) or frozen. Specifically, tissues were collected from brain, spinal cord, pituitary, trigeminal ganglia, eyes, sciatic nerve, third eyelid, palatine tonsil, pharyngeal tonsil, lymph nodes (mesenteric, retropharyngeal, prescapular, and popliteal), spleen, esophagus, forestomaches, intestines, rectal mucosa, thymus, liver, kidney, urinary bladder, pancreas, salivary gland, thyroid gland, adrenal gland, trachea, lung, turbinate, nasal planum, heart, tongue, masseter, diaphragm, triceps brachii, biceps femoris, and psoas major.
Microscopic and Immunohistochemical Evaluation
To study the accumulation of PrPSc in tissue, bright-field microscopy was used in combination with immunohistochemistry (IHC). Paraffin-embedded tissues were sectioned at their optimum thickness (brain, 4 μm; lymphoid, 3 μm; and other, 5 μm). The presence of PrPSc and the pathologic phenotype (immunolabeling pattern and distribution) were evaluated by application of a cocktail containing 2 monoclonal antibodies (F89/160.1.5 and F99/97.6.1) each used at a concentration of 5 μg/ml using an automated processor. 25 Descriptions of the PrPSc immunolabeling patterns have been published previously. 12,31 Hematoxylin and eosin (HE)–stained sections of brain were evaluated for evidence of spongiform change in each animal.
Western Blot
To evaluate the molecular characteristics of PrPSc, Western blot detection was performed on the frozen sections of brainstem at the level of the obex from representative samples of each genotype and inoculation group. The assay was performed as previously described 16 using monoclonal antibody P4 (0.1 μg/ml; R-Biopharm, Marshall, MI), a biotinylated anti-mouse secondary antibody (0.1 μg/ml; biotinylated anti-mouse IgG, Amersham Biosciences, Waltham, MA), and streptavidin horseradish peroxidase conjugate (dilution 1:10 000; streptavidin horseradish-peroxidase conjugate, Amersham Biosciences). 16 A chemiluminescent detection system was used to acquire images.
Enzyme Immunoassay
A commercially available enzyme immunoassay (EIA) (HerdChek; IDEXX Laboratories, Westbrook, ME) was used to detect the presence of PrPSc in the brainstem at the level of the obex, retropharyngeal lymph node, and palatine tonsil. Assays were conducted according to kit instructions except that the samples were prepared as a 20% (w/v) tissue homogenate. Cutoff numbers were determined with a negative control per kit instructions; values greater than the mean optical density of negative controls + 0.180 were considered positive.
Definitions
To analyze experimental data, the following definitions were used. The attack rate was defined as the percentage of sheep from an experimental group that had detectable PrPSc in any tissue. Incubation period was defined as the amount of time that elapsed between inoculation and euthanasia at the onset of clinical signs.
Results
Attack Rate and Incubation Period
All of the IC-inoculated sheep developed clinical disease and had detectable PrPSc in the brainstem based on IHC (100% attack rate). Differences in the incubation period were associated with route of inoculation and genotype at codon 171. Among the IC-inoculated sheep, the mean incubation periods for QQ, QK, and KK genotypes were 8.8, 26.6, and 49.4 months postinoculation (MPI), respectively (Table 1; Fig. 1).
Summary of Results by Genotype of Barbado Sheep Inoculated With US Scrapie Isolate 13-7.
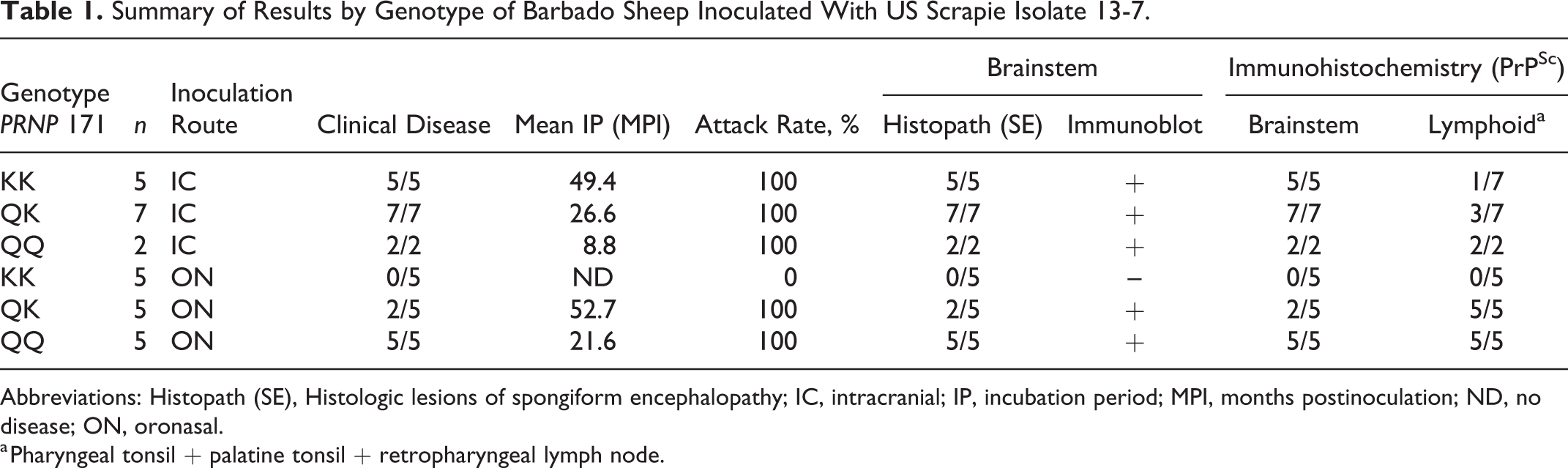
Abbreviations: Histopath (SE), Histologic lesions of spongiform encephalopathy; IC, intracranial; IP, incubation period; MPI, months postinoculation; ND, no disease; ON, oronasal.
a Pharyngeal tonsil + palatine tonsil + retropharyngeal lymph node.
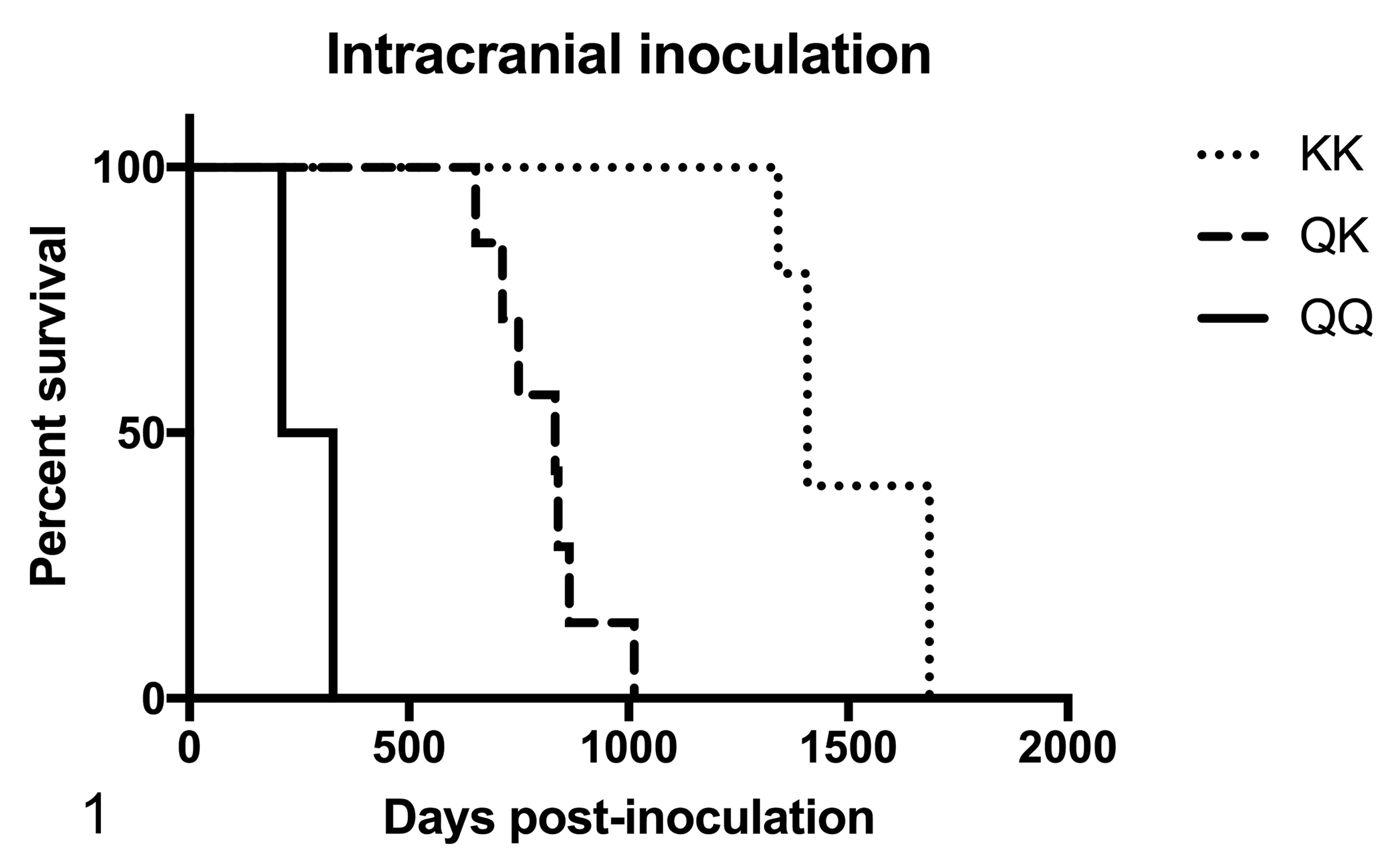
Survival plots of Barbado sheep intracranially (Fig. 1) and oronasally (Fig. 2) inoculated with the No. 13-7 scrapie agent. The survival curves for the KK, QK, and QQ genotypes are illustrated by the short dash, long dash, and solid lines, respectively.
The attack rates for sheep with QQ, QK, and KK genotypes inoculated via the ON route were 100%, 100%, and 0%, respectively. The QQ and QK genotypes of ON-inoculated sheep with neurologic signs had mean incubation periods of 21.6 and 52.7 months, respectively (Table 1; Fig. 2). At the experimental end point (70 MPI), none of the sheep with the KK genotype had developed clinical signs.
PrPSc Accumulation in Intracranially Inoculated Sheep
To investigate the microscopic changes in the brain, HE-stained sections of brain were evaluated for spongiform change. Spongiform change occurred in the brainstem of all IC-inoculated sheep. Immunolabeled sections of brain, spinal cord, and peripheral tissues were examined for PrPSc accumulation. PrPSc accumulation occurred throughout the central nervous system in all IC-inoculated sheep. Differences were observed in the PrPSc accumulation patterns between genotypes and were most prominent in the frontal cortex (Figs. 3–8) and cerebellum.
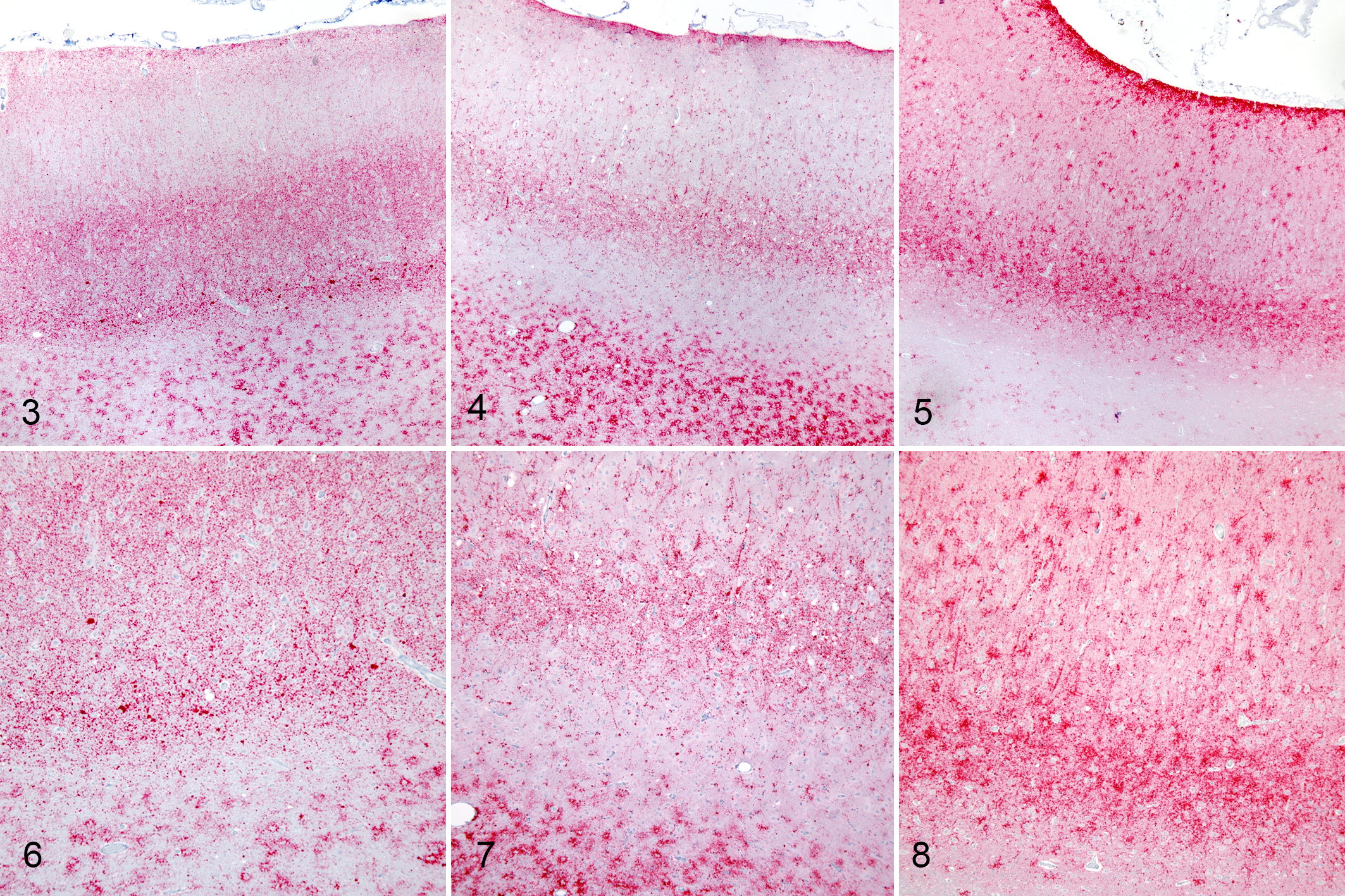
Scrapie, intracranial inoculation, neocortex, sheep. Immunohistochemistry for PrPSc, monoclonal antibodies F89/160.1.5 and F99/97.6.1.
In the neocortex of sheep with the KK and QK genotype (Figs. 3–4, 6–7), PrPSc was found primarily within the white matter and associated with extracellular accumulation of PrPSc in glial cells (stellate and perivacuolar immunolabeling types). In sheep with the KK genotype, PrPSc accumulation in neocortical gray matter was characterized by moderately dense aggregated, granular, and plaque-like deposits that were most commonly present in the deeper cortical layers (external pyramidal through the polymorphic layer). In contrast, in sheep with the QQ genotype (Figs. 5, 8), most of the PrPSc was present in the gray matter with stellate, linear, and aggregated deposits, particularly in the molecular and internal pyramidal layers. In the gray matter of sheep with the QK genotype, intraneuronal, intraglial, and granular immunolabeling types were most prominent in the internal pyramidal layer. There was also a patchy distribution of linear-type immunolabeling throughout the neocortex.
The patterns of PrPSc accumulation in the cerebella of sheep with the QK or KK genotypes were similar to each other and different from those of sheep of the QQ genotype. Similar to what was observed in the neocortex, white matter accumulation of PrPSc was rare in the cerebella of sheep with the QQ genotype and common in sheep with the QK and KK genotypes. Patterns of PrPSc accumulation in the granular and molecular layers were similar across all genotypes with moderate but variable amounts of granular, intraneuronal, and intraglial PrPSc in the granular cell layer and scant punctate, granular, and stellate deposits within the molecular layer. There was a small amount of intraneuronal PrPSc within Purkinje cells.
Spinal cord sections (cervical, thoracic, and lumbar) were immunoreactive for PrPSc in all sheep. PrPSc accumulation occurred primarily in the gray matter with increasing white matter involvement in QK and QQ genotypes. Immunolabeling patterns included intraneuronal, granular, and intraglial.
There was limited peripheral accumulation of PrPSc in intracranially inoculated sheep as evidenced by IHC. Only 3 of 7 sheep with the QK genotype had detectable PrPSc within the retropharyngeal lymph nodes (RPLNs) compared to 2 of 2 of the QQ genotype sheep (Suppl. Table S1). In sheep with the KK genotype, 1 of 5 sheep accumulated PrPSc in the RPLNs; however, immunoreactivity was present only as a few scattered positive cells in a single follicle. A single sheep from the QK group had PrPSc within a lymphoid follicle in the spleen. None of the IC-inoculated sheep had PrPSc within the palatine tonsil, pharyngeal tonsils, or the rectal mucosal–associated lymphoid tissue by means of IHC; however, 1 sheep with a QQ genotype had detectable PrPSc by EIA in the palatine tonsil.
PrPSc Accumulation in Oronasally Inoculated Sheep
To assess the pathological changes and PrPSc accumulation in sheep inoculated by the ON route, we evaluated HE-stained and immunolabeled sections of brain, spinal cord, and peripheral tissues by light microscopy. At the end of the experiment (70 months), all the sheep with the KK genotype were euthanized without developing clinical signs suggestive of scrapie. No immunoreactivity for PrPSc was detected within the central nervous system (CNS) of any sheep with the KK genotype compared to 2 of 5 of the sheep with the QK genotype and 5 of 5 with the QQ genotype (Suppl. Table S1). These same animals had spongiform change in their brainstems. Overall, the pathologic phenotype (PrPSc pattern and distribution) was similar (Figs. 9, 10), but differences in the amount of PrPSc deposition within genotypes (intragenotypic) and between genotypes (intergenotypic) were observed. This was prominently displayed by more intense stellate labeling in the superficial cortex of sheep with the QQ genotype. The intragenotypic difference was observed in sheep with a QQ genotype; there was a positive correlation between incubation period and amount of PrPSc deposition. The intergenotypic differences were most evident in the neocortex. PrPSc deposition was greater in sheep with the QQ genotype compared to the QK genotype despite a longer incubation period for the QK genotype.
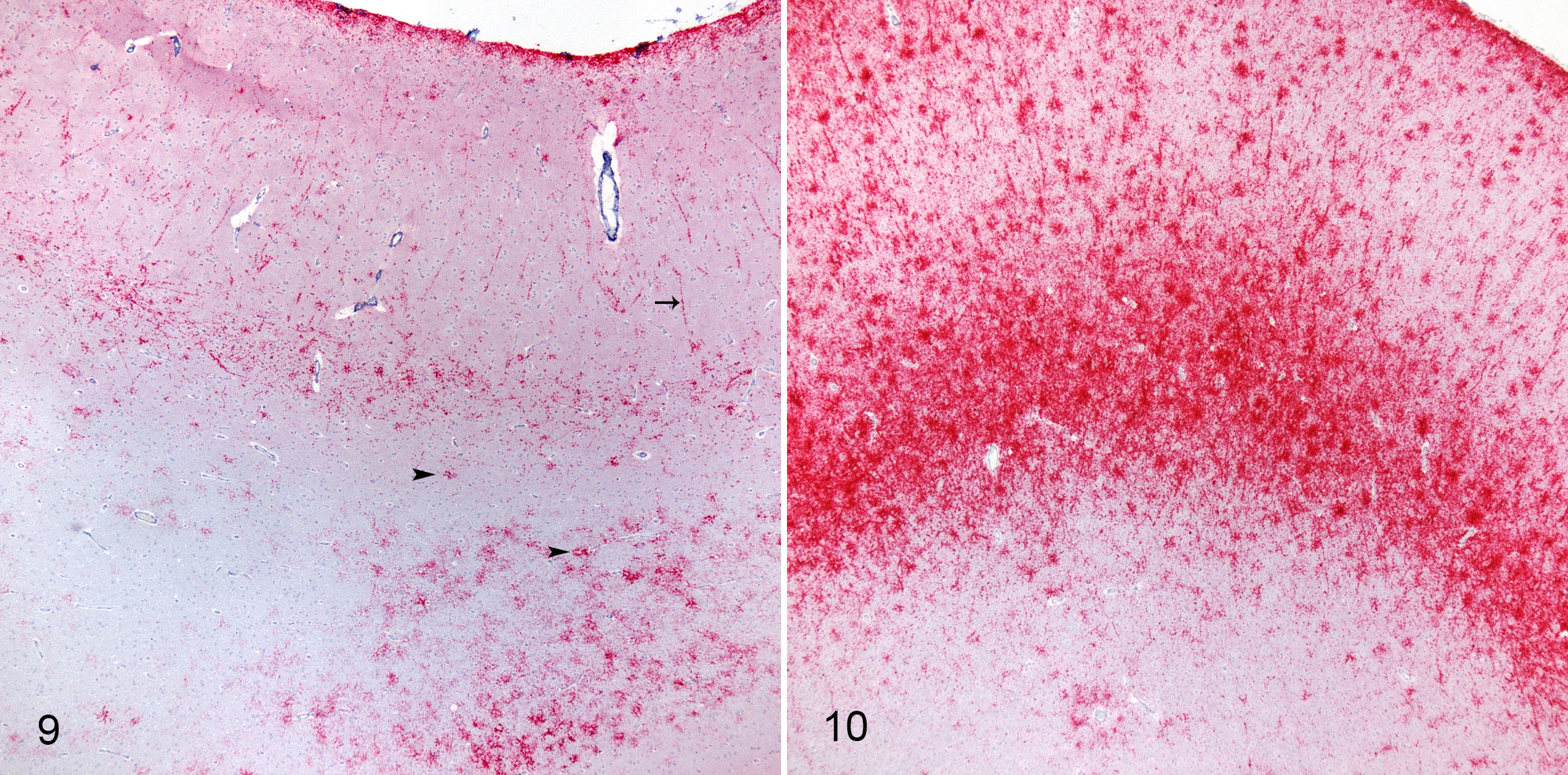
Scrapie, oronasal inoculation, neocortex, sheep. Immunohistochemistry for PrPSc, monoclonal antibodies F89/160.1.5 and F99/97.6.1.
There was no evidence of accumulation of PrPSc in any non-CNS tissues from sheep with the KK genotype. In contrast, QK and QQ genotypes had variably distributed PrPSc within the gastrointestinal (myenteric and submucosal ganglionic plexuses), lymphoreticular, and peripheral nervous systems (Table 2 and Figs. 11–14).
Non–Central Nervous System Accumulation of PrPSc in Oronasally Inoculated Barbado Sheep by Genotype.a
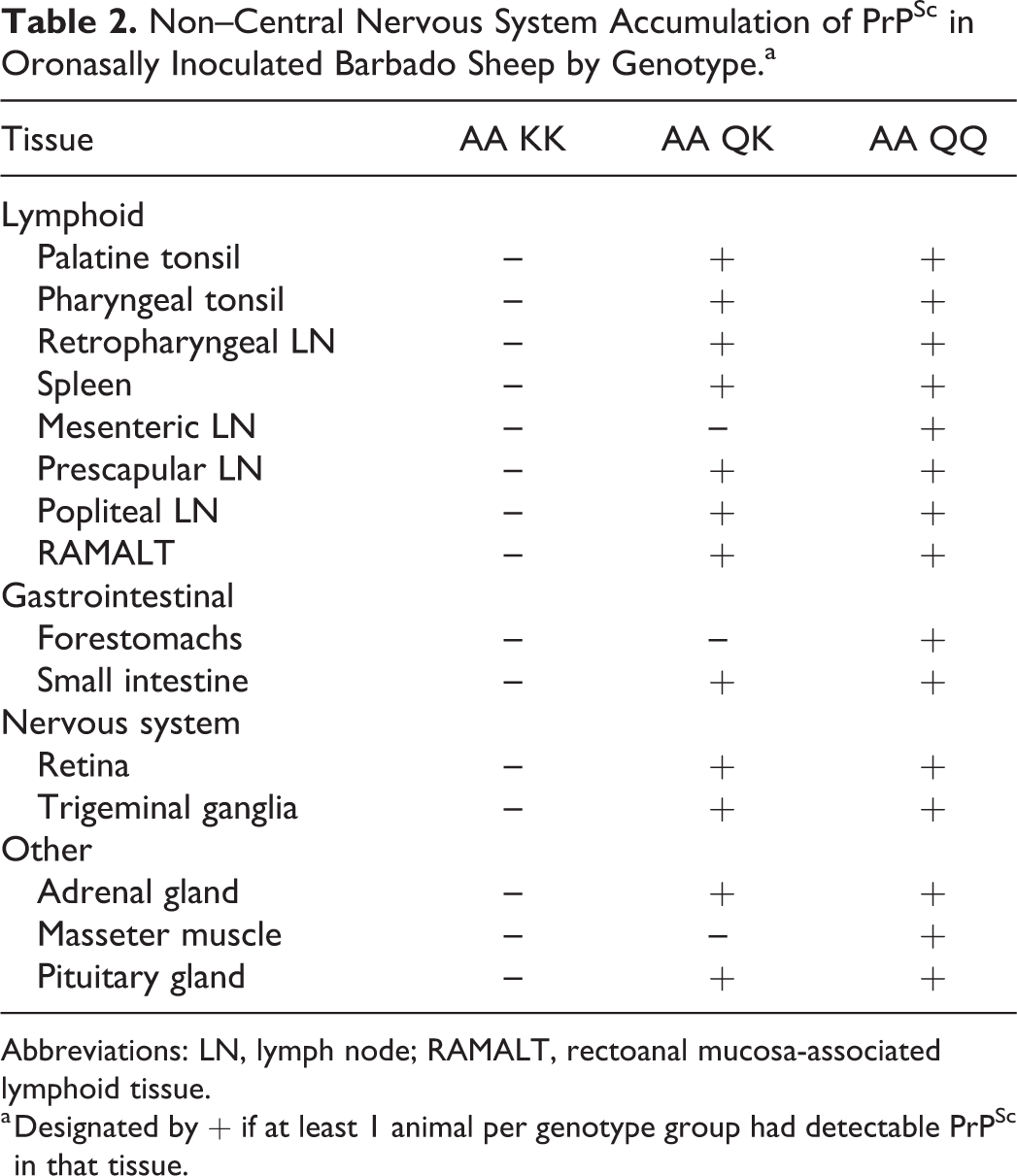
Abbreviations: LN, lymph node; RAMALT, rectoanal mucosa-associated lymphoid tissue.
a Designated by + if at least 1 animal per genotype group had detectable PrPSc in that tissue.
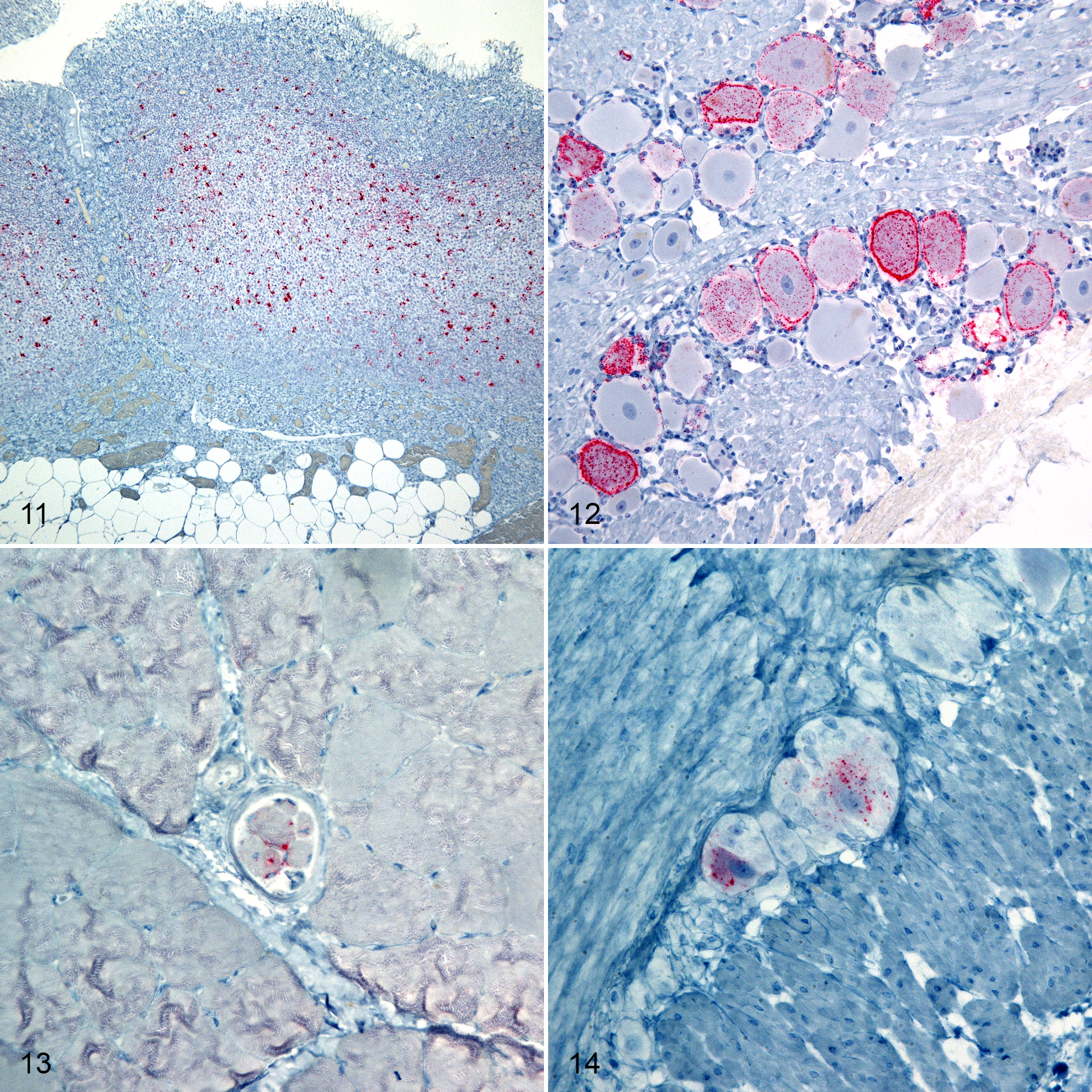
Scrapie, oronasal inoculation, sheep. Immunohistochemistry for PrPSc, monoclonal antibodies F89/160.1.5 and F99/97.6.1.
Western Blot
Molecular profiles obtained from a Western blot of brain homogenates from each sheep were identical to each other and the donor inoculum (Fig. 15). Homogenized brain material from the ON-inoculated sheep with a KK genotype was negative for PrPSc.
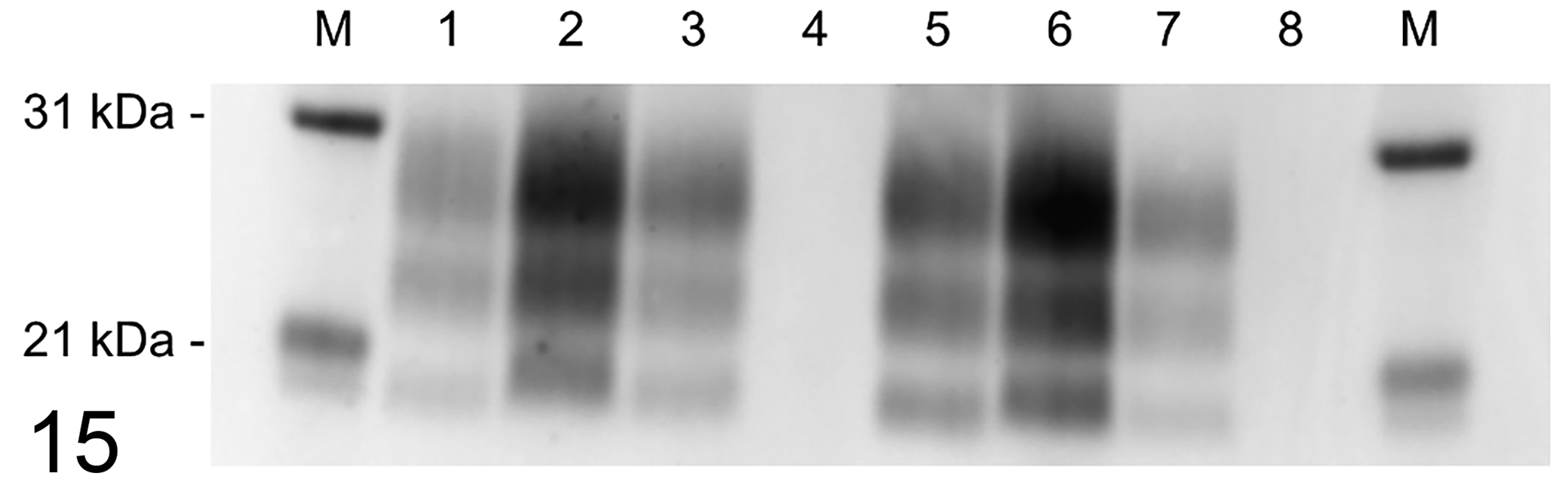
Western blot of brain homogenates from scrapie-inoculated sheep developed with anti-PrPSc monoclonal antibody P4. Lane key: M, molecular mass marker. Lanes 1 to 3, intracranial inoculation: KK (1), QK (2), QQ (3). Lanes 4 to 6, oronasal inoculation: KK (4), QK (5), QQ (6). Lane 7, inoculum scrapie strain 13-7. Lane 8, negative control.
Discussion
In this study, we assessed the effect of lysine at codon 171 of PRNP on the susceptibility of sheep to the scrapie agent. We demonstrated that, in our challenge model, the KK171 genotype confers resistance to the scrapie agent via ON inoculation and prolonged incubation period after IC inoculation. Our results also reveal a prolonged incubation period in sheep with a heterozygous PRNP genotype (QK) as compared to the QQ genotype when inoculated via both the IC and ON routes.
We used ON inoculation to simulate natural exposure to scrapie and found that sheep with the KK genotype are resistant (0% attack rate), similar to orally inoculated sheep with the ARR/ARR genotype. 13,23 The reason for comparable susceptibility between these amino acid polymorphisms, lysine (K171) and arginine (R171), could be attributable to similarities between the functional groups in their side chains. The amino (lysine) and guanidino (arginine) groups are both basic and have similar isoelectric points (9.7 and 10.8). By comparison, glutamine (Q) has a nonbasic nitrogen side chain that contains an amide functional group and has an isoelectric point of 5.7. 33
After ON inoculation, prolonged incubation periods occurred in sheep with the heterozygous QK genotype compared to sheep with the QQ171 genotype. Only 2 of 5 sheep with the QK171 genotype developed neurologic signs and accumulated PrPSc in the brain. The remaining 3 of 5 sheep accumulated PrPSc in peripheral tissues but did not develop neurologic signs before the experimental end point at 70 MPI. The mean incubation period in QK171 genotype sheep with clinical signs (52.7 MPI) was much longer than the mean incubation period observed in sheep with the QQ genotype (21.6 MPI). The presence of PrPSc in peripheral tissues of all ON-inoculated sheep of the QK genotype indicates that they are susceptible to the classical scrapie agent (strain 13-7) despite a prolonged incubation period.
These findings suggest that the resistance imparted by the QK171 genotype is similar but weaker than the QR171 genotype. For example, orally inoculated Cheviot lambs with the ARQ/ARR genotype exhibited prolonged incubation times compared to the ARQ/ARQ genotype. 13 This is similar to the difference between ARQ/ARK and ARK/ARK in the present study; however, differences in experimental design involving scrapie strain (13-7 vs RBP1) and inoculum volume (0.1 g once vs 1 g repeated for 5 days) make direct comparisons between these studies difficult. We previously used the No. 13-7 scrapie agent to conduct an oral challenge of neonatal Suffolk lambs of the QR genotype, but there was no evidence of transmission. 15 The present study used identical methods with respect to inoculum volume and scrapie strain, but we observed a 100% attack rate in the QK genotype. This suggests that heterozygous arginine may have a stronger effect on resistance than lysine. That hypothesis is consistent with recently published investigations of naturally occurring scrapie in herds with relatively high frequencies of ARK haplotypes. The authors reported that K171 was the second most resistant allele after arginine (R > K > H > Q). 6
Evaluation of IHC-stained tissues from IC-inoculated sheep revealed intergenotypic differences in PrPSc phenotypes (pattern and distribution). White matter immunolabeling was largely present in the KK and QK genotype, contrasting a relative absence in sheep with the QQ genotype. In sheep with the QK genotype, neocortical immunolabeling resembled an amalgamation of the KK and QQ phenotypes. It is unclear to what extent serial passage in ARQ/ARQ genotype sheep influenced these differences, and it is possible that a QK genotype-adapted strain would impart a different phenotype. Since PrPSc in scrapie-infected sheep is predominantly composed of the susceptible allelic protein isoform, 22,26 we expected that even in IC-inoculated animals, the QK group’s pathologic phenotype would more closely resemble the QQ phenotype, because Q171 is more susceptible to conversion. Instead, we observed a “combined” phenotype. This finding suggests that a combination of PrPSc containing both K and Q at codon 171 was present. Quantification of the PrPSc allotype composition in these sheep would be useful to further evaluate differential expression of protein isoform fractions between genotype and inoculation groups.
The results of the present study indicate that sheep with a homozygous lysine-171 (KK171) genotype are resistant to scrapie via oronasal exposure in our challenge model and that sheep with the heterozygous genotype (QK171) have prolonged incubation periods. These findings may be useful in designing breeding programs to enhance scrapie resistance in breeds of sheep that carry the K171 prion protein allele.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985818817066 - Sheep With the Homozygous Lysine-171 Prion Protein Genotype Are Resistant to Classical Scrapie After Experimental Oronasal Inoculation
Supplemental Material, DS1_VET_10.1177_0300985818817066 for Sheep With the Homozygous Lysine-171 Prion Protein Genotype Are Resistant to Classical Scrapie After Experimental Oronasal Inoculation by Eric D. Cassmann, Sarah Jo Moore, Jodi D. Smith and Justin J. Greenlee in Veterinary Pathology
Footnotes
Acknowledgements
We thank Martha Church, Kevin Hassall, Joe Lesan, Leisa Mandell, and Trudy Tatum for providing technical support to this project. Mention of trade names or commercial products in this article is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the Department of Agriculture. The Department of Agriculture is an equal-opportunity provider and employer.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded in its entirety by congressionally appropriated funds to the United States Department of Agriculture, Agricultural Research Service. The funders of the work did not influence study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
